Abstract
Study Design
Retrospective review of prospective cohort.
Objectives
Reoperation at L3-L4 for adjacent segment disease (ASD) is common after L4-L5 spine fusion. L4-S1 lower lumbar lordosis (LLL) accounts for the majority of global lumbar lordosis (GLL) and is modifiable during surgery. We sought to determine if a reduction in LLL leads to an increase in L3-L4 focal lumbar lordosis (L3-L4 FLL) and resulting risk of ASD at L3-L4.
Methods
We reviewed the records of a prospective cohort with lumbar spinal stenosis who underwent L4-L5 or L4-L5-S1 fusion between 2006 and 2012. Radiographic parameters—GLL, LLL, L3-L4 FLL, upper lumbar lordosis, lordosis distribution index, pelvic tilt, and pelvic incidence—were extracted from preoperative and postoperative lumbar spine radiographs. Statistical comparisons were made between those who underwent revision for post-fusion adjacent level stenosis at L3-L4 (REVISION) and those who did not (NO REVISION).
Results
Inclusion criteria were met by 104 patients. The REVISION cohort included 19 individuals. No significant differences in baseline demographics or operative details for the index procedure were found between groups. Postoperatively, when compared to the NO REVISION cohort, the REVISION cohort had a decrease in LLL (-2.6° vs + 1.5°, P = .011) and LDI (-5.1% vs + 1.3%, P = .039), and an increase in L3-L4 FLL (+2.6° vs -.6°, P = .001).
Conclusions
A reduction in LLL and compensatory increase in L3-L4 FLL after initial lower lumbar fusion surgery resulted in more reoperation at L3-L4 for post-fusion adjacent level spinal stenosis.
Introduction
Instrumented lumbar spine fusion for lumbar spinal stenosis is one of the most common surgical procedures of the lumbar spine in North America,1-3 with the most frequent location of initial lumbar fusion being the L4-L5 level.4,5 Unfortunately, revision surgery after lumbar fusion is common, with reoperation rates ranging between 10-20%.6-8
Reasons for reoperation are diverse, including complications such as infection, hematoma, and implant or screw failure.7,9 However, the most reported reason for revision surgery after initial lumbar fusion is adjacent segment disease.6,9
Adjacent segment disease (ASD) is a general term used to describe the various pathologies that can occur at an adjacent spinal level after spine surgery, including spinal stenosis, disc herniation, disc height loss, spondylolisthesis, osteophyte formation, and scoliosis.10-12 In our practice, post fusion adjacent level spinal stenosis is the most common indication for revision surgery secondary to ASD. 13 Although ASD can affect levels both inferior and superior to a fused segment, the most common scenario in lumbar spine surgery is ASD at L3-L4 after L4-L5 fusion.14,15 ASD is thought to occur as a result of spinal fusion altering the inherit biomechanics of the spine, eliminating mobile segments, which then overloads adjacent levels and accelerates their degeneration.16-21
Given this biomechanical basis for ASD, much has emerged around radiographic parameters and their association with ASD.22-36 Among these parameters, a decreased preoperative22,24,29-32 or postoperative22,24,26,29,30,32-34 global lumbar lordosis angle has been identified as a risk factor for ASD. 37 Global lumbar lordosis, however, is only partially modifiable during short-segment lower lumbar fusion. On the other hand, the L4-S1 lordosis angle is directly modified during short segment lumbar fusion surgery. Since the L4-S1 segments account for the majority of global lumbar lordosis, 38 during short-segment lower lumbar fusion, a loss of L4-S1 lordosis may play a significant role in the development of post-fusion ASD.
The purpose of this study is to compare the pre-operative to post-operative change in L4-S1 lordosis after L4-L5 or L4-L5-S1 fusion in a cohort of patients with and without revision surgery for post-fusion adjacent level spinal stenosis. Our hypothesis is that, compared to the non-revision group, the revision group will have a decreased L4-S1 lordosis and compensatory increased L3-L4 focal lumbar lordosis.
Materials and Methods
Study Design
A consecutive series of 212 patients surgically treated for spinal stenosis between 2006 and 2012 was identified from a prospectively maintained database of patients with symptomatic degenerative lumbar spinal stenosis refractory to a minimum of 3 months of non-operative care. Informed consent was obtained from the patients at the time of enrolment. Approval for this retrospective review was obtained from our Institutional Research Ethics Board (IRB 114542) before data collection and analysis. All patients who had an index decompression surgery with posterior lumbar interbody fusion or posterolateral fusion at the levels of L4-L5 or L4-L5-S1 were included for analysis. Indications for fusion—in addition to decompression—included foraminal stenosis and signs of radiographic instability (greater than 3 mm translation between supine and standing radiographs) at the affected level. These patients were followed 10 years post-operatively, and their records were evaluated retrospectively. Patients with index decompression alone procedures, or index spinal fusion of levels other than L4-L5 or L4-L5-S1 were excluded. Patients with post-operative infection were also excluded.
Patients were categorized into two groups: REVISION or NO REVISION. The indication for revision surgery was symptoms of claudication and/or radiculopathy secondary to spinal stenosis that was resulting from facet hypertrophy, ligament hypertrophy, facet cyst, and/or disc herniation, with or without spondylolisthesis or scoliosis. The reoperation procedure could include decompression with laminectomy and/or discectomy—with or without a fusion extension at the L3-4 level.
Radiographic Evaluation
Lumbar spine radiographs were obtained prior to the index surgery and at the first post-operative follow-up appointment (between 6 and 12 weeks after surgery). Surgimap® (version 2.3.2.1, Nemaris, Inc.) software was used to measure global lumbar lordosis angle (GLL), pelvic incidence (PI), pelvic tilt (PT), sacral slope (SS), pelvic incidence minus lumbar lordosis (PI-LL), lower lumbar lordosis angle (LLL), upper lumbar lordosis angle (ULL), focal lordosis at L3-L4 level (L3-L4 FLL), and the lordosis distribution index (LDI). Cobb’s method was used in all angle measurements. GLL was measured between the L1 and S1 superior endplates, ULL between L1 and L4 superior endplates, LLL between L4 and S1 superior endplates, and L3-L4 FLL between L3 and L4 superior endplates. LDI is a ratio between LLL and GLL and was calculated using the formula: LLL/GLL multiplied by 100 and expressed as a percentage (Figure 1). Normal values of LDI range from 50% to 80%, with LDI lower than 50% or greater than 80% suggesting hypolordotic or hyperlordotic maldistribution, respectively.
39
Lordosis distribution index (LDI) is a ratio of LLL and GLL calculated with the formula α/β x 100%. Normal LDI is generally considered to be between 50% and 80%, with LDI <50% and LDI >80% considered to be hypolordotic and hyperlordotic, respectively.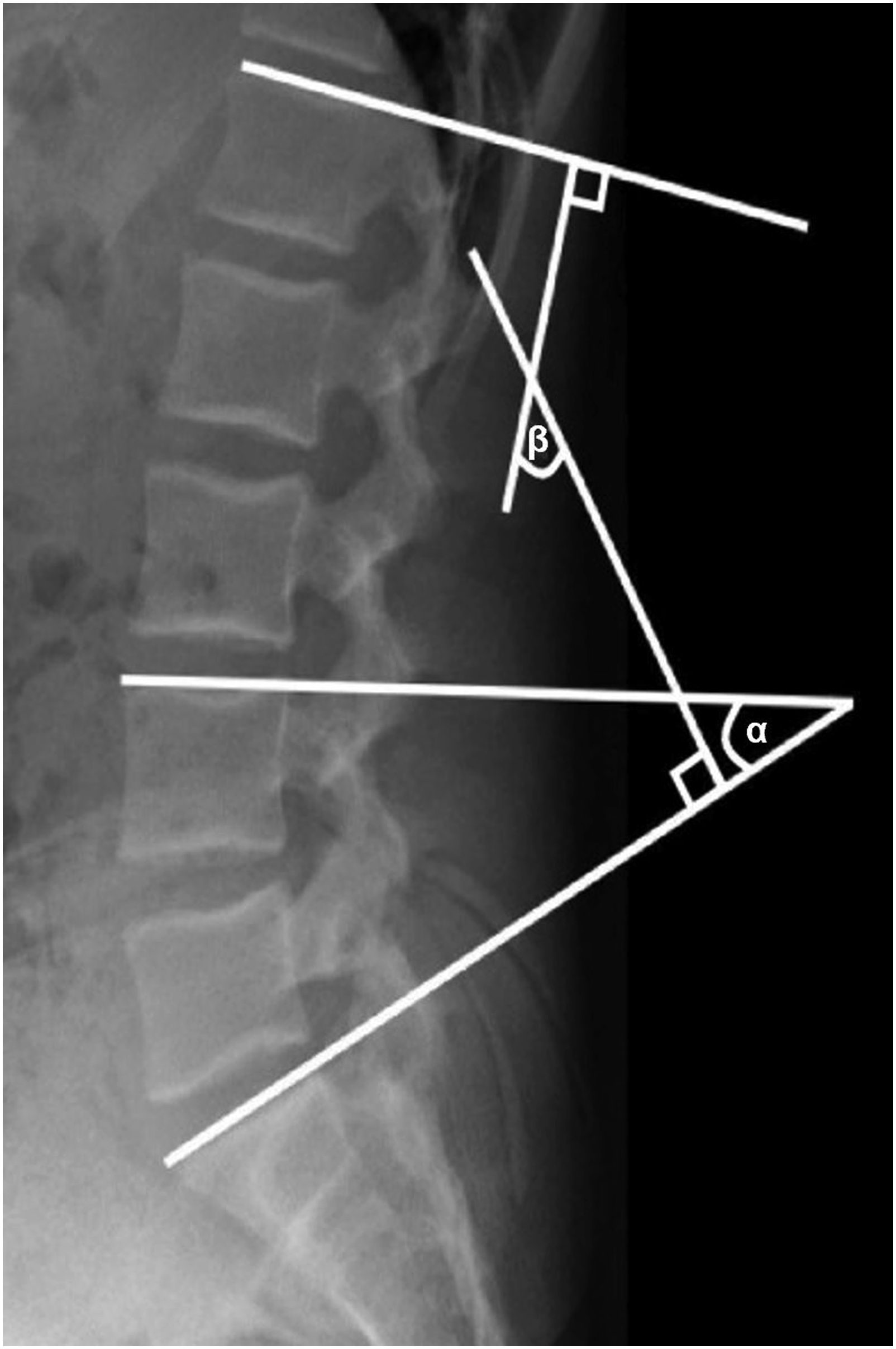
Statistical Analysis
Data was analyzed using SPSS Windows version 27 (SPSS Inc., Chicago, IL, USA). Normality was assessed using the Shapiro-Wilk Test. Chi-square test or Fisher’s exact test (where appropriate) and independent t-tests were used for comparison of between group demographic, operative, and radiographic categorical and continuous variables, respectively. Within group (preoperative vs postoperative) radiographic variables were compared using a paired t test. A mixed model of longitudinal regression for repeated measures accounting for the correlation (assuming compound symmetry) among the outcome score on the same patient pre- and post-operatively. Group (Revision vs No revision), time (categorical), group × time interaction and age and sex were included as fixed variables. Significance was set at P < .05.
Results
Baseline Characteristics.
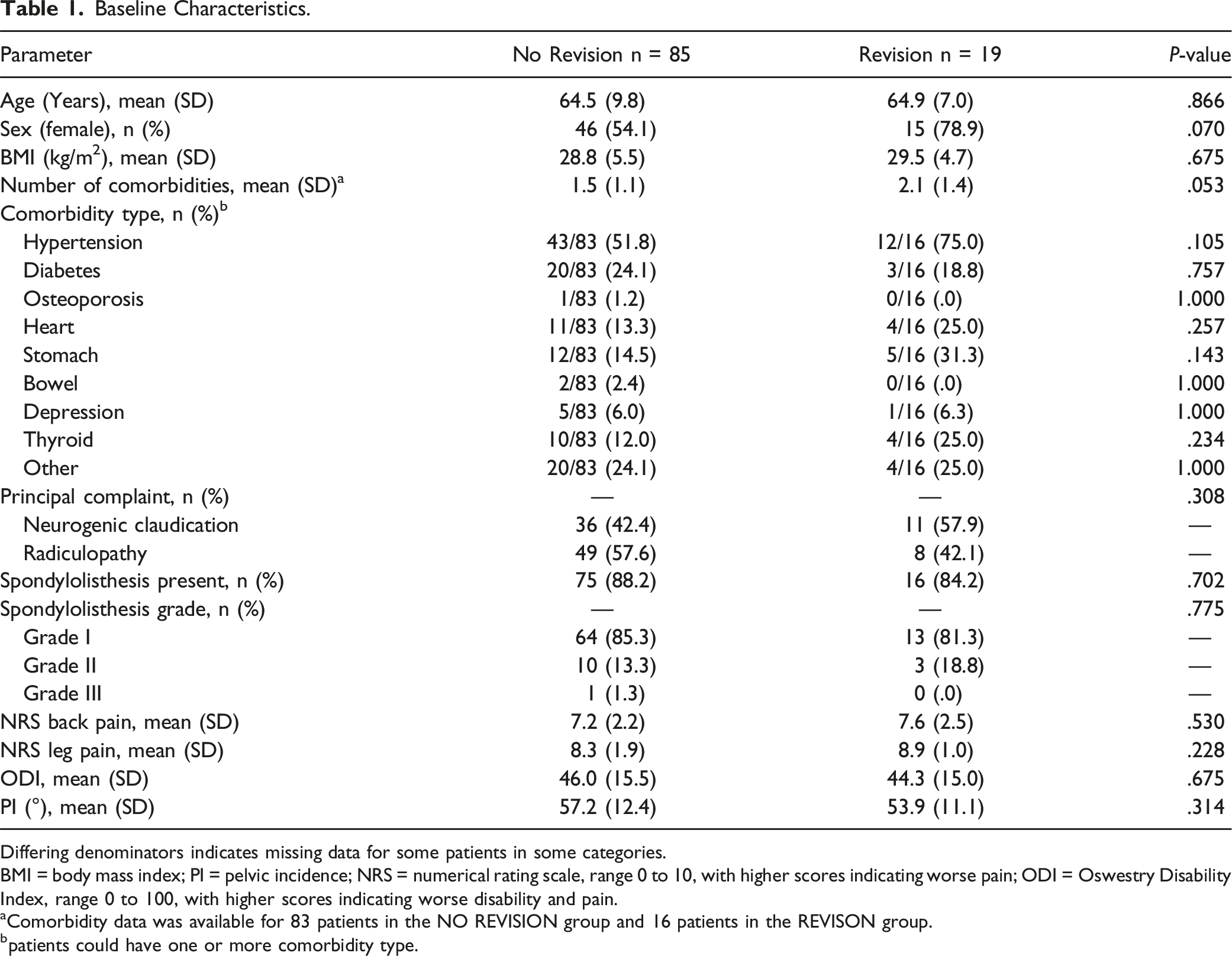
Differing denominators indicates missing data for some patients in some categories.
BMI = body mass index; PI = pelvic incidence; NRS = numerical rating scale, range 0 to 10, with higher scores indicating worse pain; ODI = Oswestry Disability Index, range 0 to 100, with higher scores indicating worse disability and pain.
aComorbidity data was available for 83 patients in the NO REVISION group and 16 patients in the REVISON group.
bpatients could have one or more comorbidity type.
Operative Details of Index Surgery.

Comparison of Preoperative, Postoperative, and Change in LLL, GLL, L3-L4 FLL and PI-LL in Patients That had Revision Surgery (REVISION) and Those That did not Have Revision Surgery (NO REVISION).
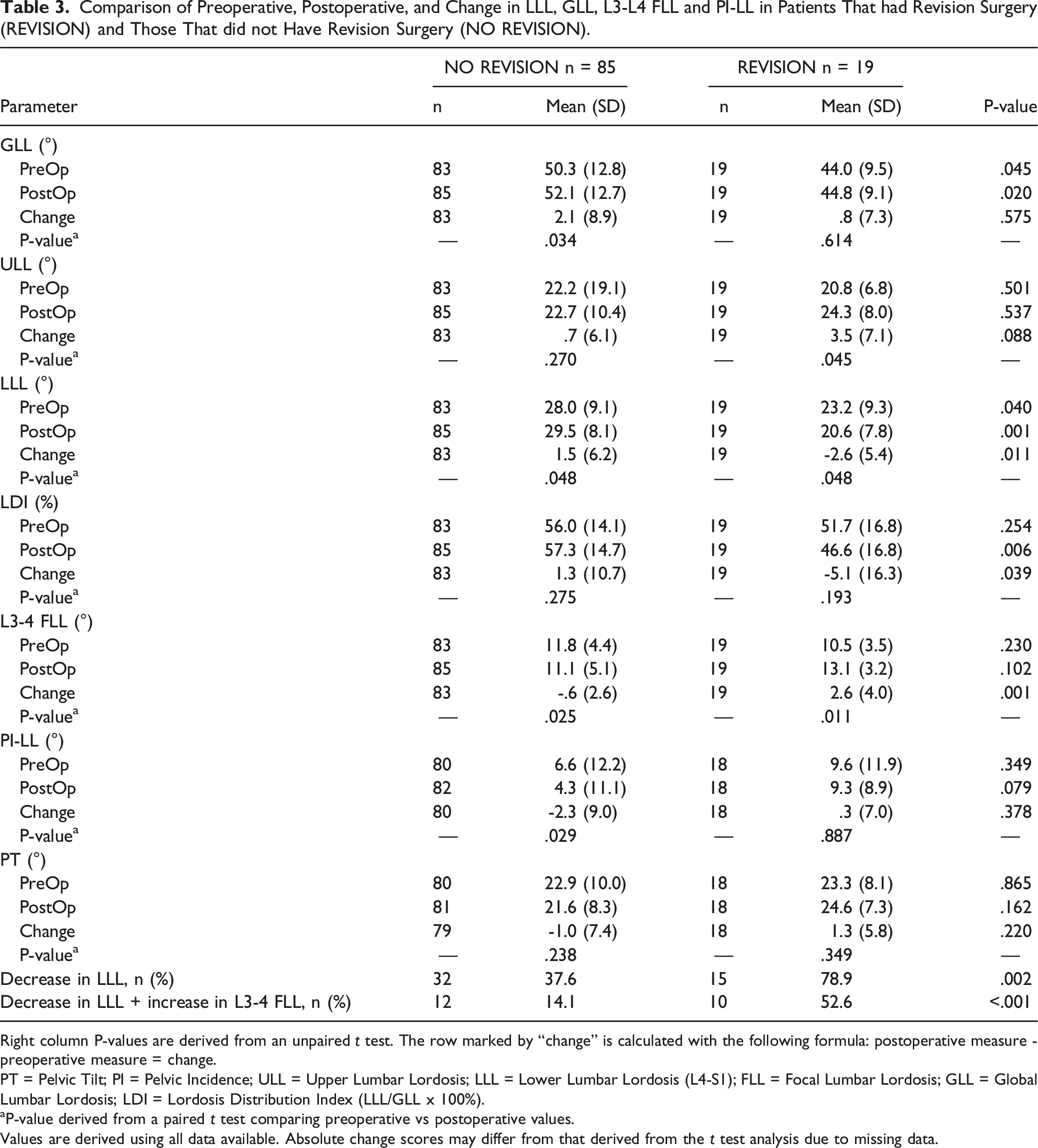
Right column P-values are derived from an unpaired t test. The row marked by “change” is calculated with the following formula: postoperative measure - preoperative measure = change.
PT = Pelvic Tilt; PI = Pelvic Incidence; ULL = Upper Lumbar Lordosis; LLL = Lower Lumbar Lordosis (L4-S1); FLL = Focal Lumbar Lordosis; GLL = Global Lumbar Lordosis; LDI = Lordosis Distribution Index (LLL/GLL x 100%).
aP-value derived from a paired t test comparing preoperative vs postoperative values.
Values are derived using all data available. Absolute change scores may differ from that derived from the t test analysis due to missing data.
Comparison of Preoperative and Postoperative GLL, ULL, LLL, LDI, L3-L4 FLL, PI-LL, and PT in Patients That had Revision Surgery (REVISION) and Those That did not Have Revision Surgery (NO REVISION), Adjusting for Age and Sex.
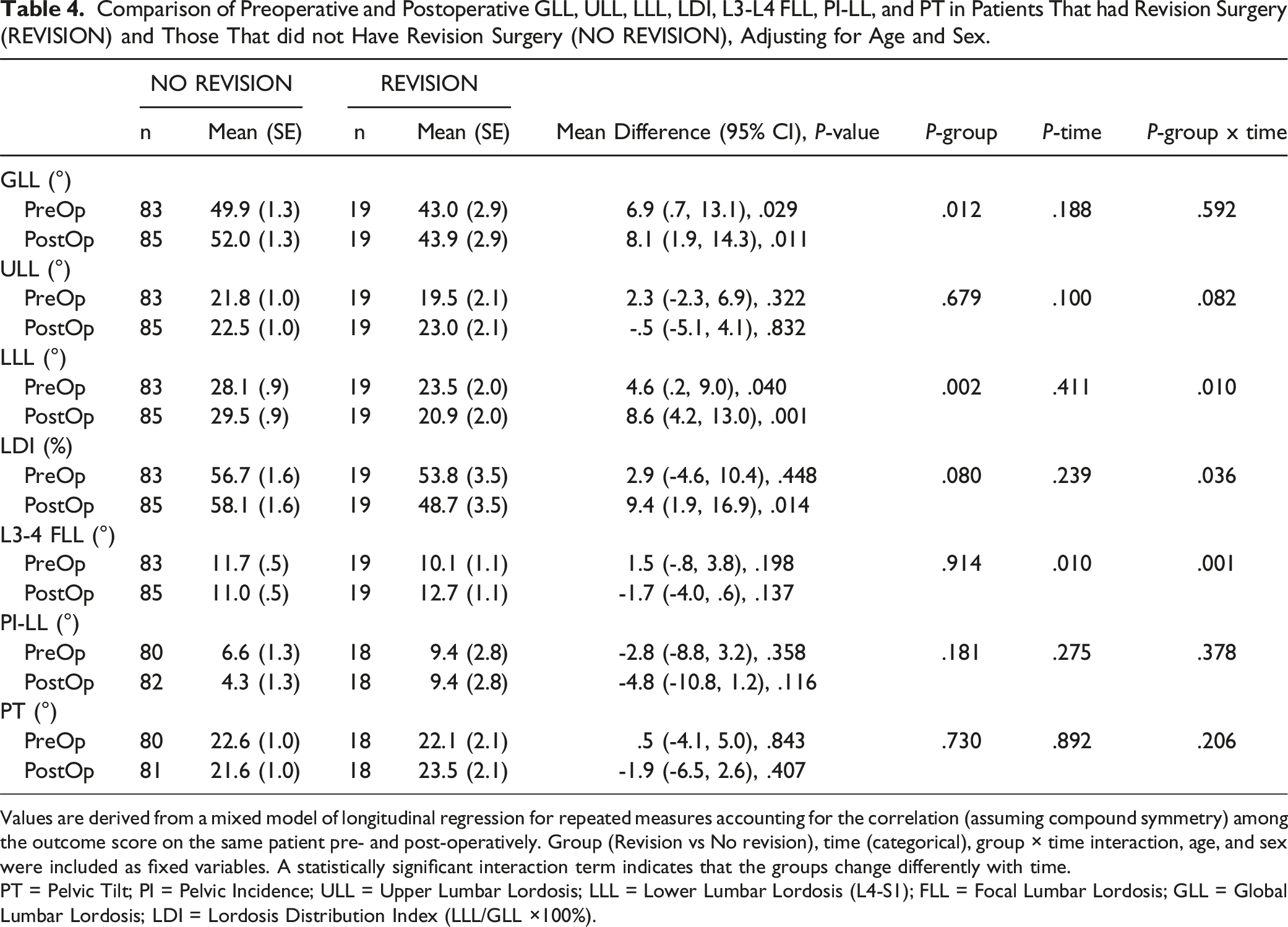
Values are derived from a mixed model of longitudinal regression for repeated measures accounting for the correlation (assuming compound symmetry) among the outcome score on the same patient pre- and post-operatively. Group (Revision vs No revision), time (categorical), group × time interaction, age, and sex were included as fixed variables. A statistically significant interaction term indicates that the groups change differently with time.
PT = Pelvic Tilt; PI = Pelvic Incidence; ULL = Upper Lumbar Lordosis; LLL = Lower Lumbar Lordosis (L4-S1); FLL = Focal Lumbar Lordosis; GLL = Global Lumbar Lordosis; LDI = Lordosis Distribution Index (LLL/GLL ×100%).
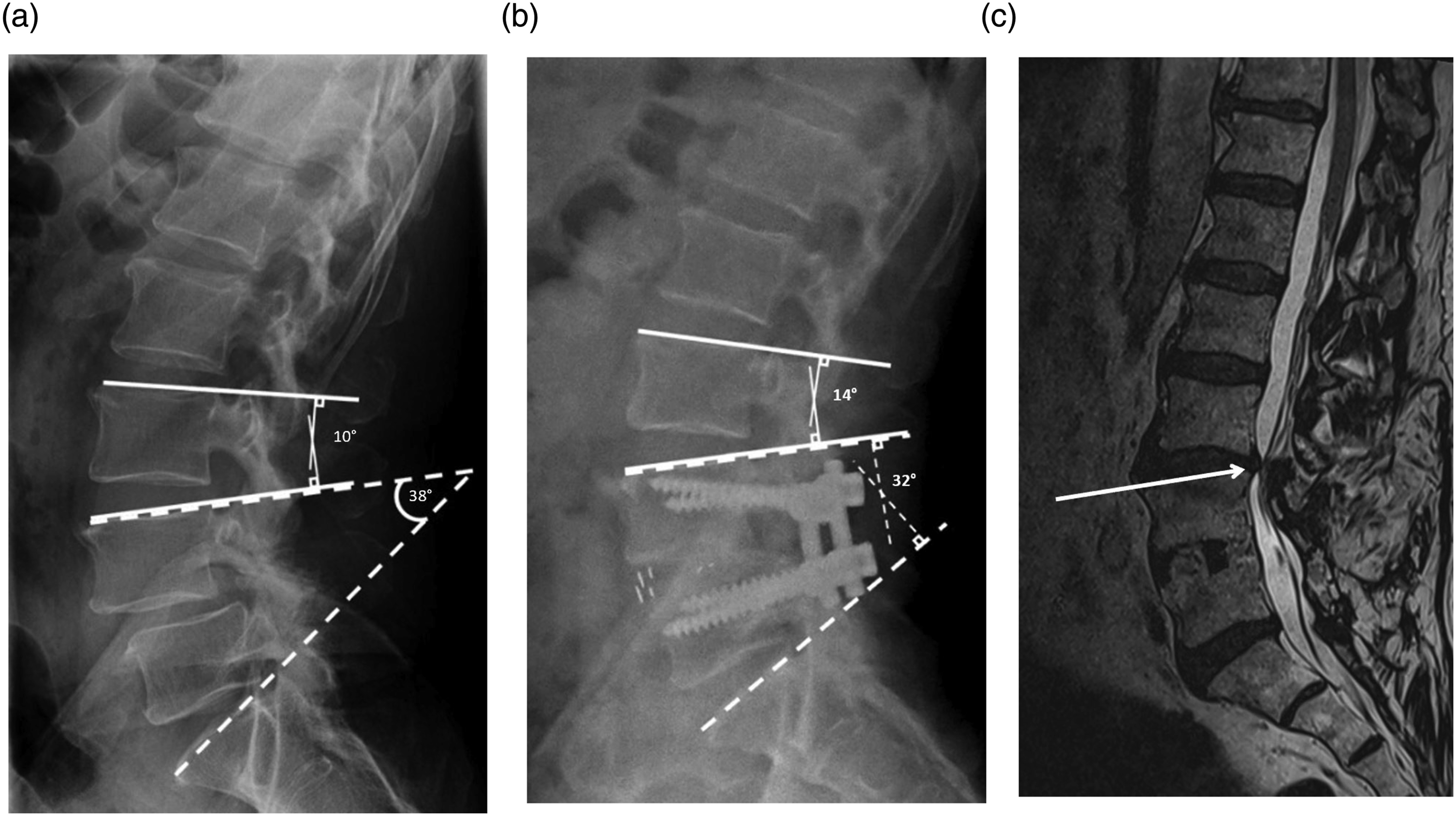
A. Sagittal lumbar spine radiograph before initial L4-L5 fusion with L4-S1 lower lumbar lordosis (dashed line) and L3-L4 focal lumbar lordosis (solid line) angles drawn. B. Sagittal lumbar spine radiograph after L4-L5 fusion in the same patient with L4-S1 lower lumbar lordosis (dashed line) and L3-L4 focal lumbar lordosis (solid line) angles drawn. C. Select sagittal T2 weighted magnetic resonance image of the lumbar spine in the same patient 6 years after initial L4-L5 fusion surgery. Spinal stenosis can be appreciated at the L3-L4 level (arrow), which ultimately required reoperation at that level.
Discussion
In a 2017 study using musculoskeletal models of the spine, the angle of lumbar fusion (or instrumentation angle) was shown to affect adjacent spinal segment joint forces. 40 Specifically, hypolordotic fusions at L4-L5 led to increased shear forces at L3-L4, while hyperlordotic configurations exhibited a reduction in forces at L3-L4. Similarly, in a cadaveric study, 20 decreased lordosis at L4-S1 following fusion resulted in increased load, shearing, and lordosis at the L3-L4 level. This increase in lordosis and resulting forces through L3-L4 after hypolordotic adjustments to L4-S1 has been replicated elsewhere.21,41,42
If biomechanical forces through L3-L4 are increased as a result of hypolordotic fusions at L4-S1, clinically, we would expect the incidence of ASD at L3-L4 to increase in patients whose LLL was decreased as a result of fusion surgery below L4. Indeed, in our cohort of individuals who underwent L4-L5 or L4-L5-S1 fusion for lumbar spinal stenosis, the REVISION group had a mean decrease in LLL compared to the NO REVISION cohort. Additionally, in keeping with previous biomechanical work, 20 the REVISION group had a mean increase in L3-L4 FLL. Importantly, not only were there differences in the magnitude of the mean change between groups, but also the proportion of patients with a loss in LLL and reciprocal increase in L3-L4 FLL was very different between cohorts (52.6% in REVISION vs 14.1% in NO REVISION, P < .001).
One measure closely linked to LLL is the LDI. LDI is the ratio between LLL and GLL, multiplied by 100.43,44 Since LLL normally accounts for approximately 60% of GLL, 38 and GLL is highly variable (ranging from 24-88°),38,45,46 LDI is a beneficial tool that—for any given GLL—can provide a standardized guide for the expected amount of LLL. It’s typically stratified into categories of low LDI (<50%), moderate LDI (50-80%), and high LDI (>80%).26,39,47 Previous work has shown that, as post-operative LDI decreases to <50%, the risk of ASD increases.26,39,47 That is, as the proportion of LLL contributing to GLL decreases, the risk of ASD increases. In the present study, we calculated LDI and found not only that the revision cohort had a smaller post-operative LDI (consistent with the literature as <50%), but also a significant reduction in LDI resulting from surgery. This finding is consistent with our results showing no significant difference in the change between the pre-operative and post-operative GLL for either cohort, while the LLL and L3-L4 FLL in the revision cohort were significantly altered.
Since LLL encompasses both the L4-L5 and L5-S1 angle, one question that arises is how a two-level fusion involving L4-L5-S1 may affect rates of ASD compared to a one-level fusion at L4-L5. With a two-level fusion, the surgeon would theoretically have more control over LLL, but at the sacrifice of increased surgical time and a longer construct. As previous in-vitro work shows increased shear forces at L3-L4 after hypolordotic L4-L5 instrumentation, 40 and the present study suggests an increased risk of adjacent level stenosis at L3-L4 after a reduction in LLL, a surgeon could theoretically increase LLL more with a two-level construct spanning from L4-S1 than with a one-level construct from L4-L5, which could in theory decrease the risk of developing ASD at L3-L4. In the present cohort, an insufficient number of patients underwent initial fusion at L4-L5-S1 to make an adequate statistical comparison to those who underwent L4-L5 fusion.
Lastly, pelvic incidence minus lumbar lordosis (PI-LL) is a radiographic parameter that has previously been implicated in the risk of ASD. 32 It is generally accepted that when PI-LL <10°, a balanced lumbopelvic relationship exists due to an adequate lumbar lordosis, and the risk of ASD decreases.32,39,48 No statistical significance was demonstrated between PI-LL and the risk of revision surgery for adjacent level spinal stenosis in our cohort. The reason for this is potentially 2-fold. First, the change in global lordosis was not different between groups, despite the differences in LLL, and L3-4 FLL. This speaks to the relative importance of LLL and LDI compared to GLL and PI-LL in assessing the risk of clinically significant ASD. Second, the mean PI-LL was <10° in the REVISION and NO REVISION groups. Both speak to the relative importance of LLL and LDI compared to GLL and PI-LL in assessing the risk of clinically significant ASD.
Limitations
At our center, we do not routinely operate on asymptomatic ASD. As this was a retrospective follow-up of a previous prospectively collected cohort, unless new symptoms arose, lumbar spine radiographs were not obtained in patients after 2 years post-surgery. In the 85 patients who did not require revision surgery over the 10-year follow up period, radiographic evidence of ASD may have existed and not have been captured. The focus in this study, however, was on revision surgery for post fusion adjacent level stenosis.
In this prospectively followed cohort, only lumbar spine radiographs were obtained at follow-up visits. Complete spine films, although sometimes performed, were not available in a large percentage of these patients. As such, we could not examine the effect of global spine parameters (such as sagittal vertical axis) on the risk of revision surgery for adjacent level stenosis. We recognize that global spine parameters have been shown to play a role in the development of ASD. 30 However, unlike L4-S1 lordosis, they are not directly modifiable in short segment L4-L5 or L4-S1 fusion, which is the focus of the present article.
As the index surgery in our study group occurred at least 10 years prior to our analysis, we were unable to recover a large percentage of preoperative magnetic resonance images. With previous work demonstrating increased preoperative Pfirrmann grade to be a risk factor for developing ASD, 49 we recognize that preoperative Pfirrmann grade could be a potential confounder contributing to risk of requiring revision surgery between our two cohorts. To the best of our knowledge though, we are unaware of any research linking postoperative LDI and preoperative Pfirrmann grade.
Our inclusion criteria encompassed all individuals with spinal stenosis—we did not delineate patients with associated instability such as spondylolisthesis, lateral listhesis, or short segment degenerative scoliosis. This does make our findings more generalizable, but the effect of decreasing LLL on L3-L4 FLL and ASD may vary between the different causes of degenerative spine disease. Future work could seek to include a larger number of patients and stratify by specific disease pathology to better examine these group differences.
Our study was in part triggered by a clinical observation that, in our mainly posterior approach-based practice, the sizes of the cages and their small lordotic angle were causing flattening of the disc space in patients with more lordotic disc spaces in L4-S1 surgeries, thus raising concerns for causing a loss of the LDI in these patients. Given the limitations of a narrow corridor between the neural elements on a posterior approach, there is a limit to increasing interbody device sizes and angles. In our practice, to try to mitigate the loss of lordosis, we often attempt to use the biggest and most lordotic device possible, pushing it as close as possible to the anterior portion of the vertebral body for anterior column support. In addition to this, compression through the rods before locking the screw-rod constructs is another one of our strategies to increase the lordotic gain—but this is limited by potentially worsening of foraminal stenosis and by the posterior intervertebral body distance of the annulus fibrosus of the disc. At our center during the timeframe of inclusion for this study, anterior lumbar interbody fusion (ALIF) and transforaminal lumbar interbody fusion (TLIF) were not routinely performed by the surgeons contributing to this cohort of patients. As recent evidence emerges around the possible capacity of ALIF to better increase segmental lordosis, 50 future endeavors should seek to compare radiographic changes following that procedure, as this may be one avenue surgeons can pursue to increase LLL and LDI in short segment spine fusion.
The retrospective nature of this single center study increases risk of sampling and selection bias. Due to the lower sample size, even though no statistical significance was observed between certain demographic and radiographic parameters, we cannot conclude no definitive differences for others, as this was a sample of convenience and no a priori power analysis was performed. However, even with a smaller sample size, a significant difference was observed for our primary object, specifically the difference between groups in LLL and L3-L4 FLL.
Conclusion
In a retrospective review of patients with lumbar spinal stenosis who underwent L4-L5 fusion or L4-L5-S1 fusion, a reduction in L4-S1 lumbar lordosis and resulting increase in L3-L4 focal lordosis was found in the cohort that had revision surgery for post-fusion adjacent level stenosis at L3-L4. This has implications for the spine surgeon during short segment lower lumbar surgery, as decreasing the lordosis at L4-S1 during fusion surgery may lead to an increased risk of revision surgery at the L3-L4 level.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
