Abstract
Objective
The Hospital Frailty Risk Score (HFRS) is a metric that measures frailty among patients in large national datasets using ICD-10 codes. While other metrics have been utilized to demonstrate the association between frailty and poor outcomes in spine oncology, none have examined the HFRS. The aim of this study was to investigate the impact of frailty using the HFRS on complications, length of stay, cost of admission, and discharge disposition in patients undergoing surgery for primary tumors of the spinal cord and meninges.
Methods
A retrospective cohort study was performed using the Nationwide Inpatient Sample database from 2016 to 2018. Adult patients undergoing surgery for primary tumors of the spinal cord and meninges were identified using ICD-10-CM codes. Patients were categorized into 2 cohorts based on HFRS score: Non-Frail (HFRS<5) and Frail (HFRS≥5). Patient characteristics, treatment, perioperative complications, LOS, discharge disposition, and cost of admission were assessed.
Results
Of the 5955 patients identified, 1260 (21.2%) were Frail. On average, the Frail cohort was nearly 8 years older (
Conclusion
Our study is the first to use the HFRS to assess the impact of frailty on patients with primary spinal tumors. We found that frailty was associated with prolonged LOS, non-routine discharge, and increased hospital costs.
Introduction
Over the past decade, American healthcare spending has continued to rise, increasing the incentive for payors to identify mechanisms of reducing costs.1-6 However, in the effort to minimize spending, it is also crucial to ensure the same quality of care is provided. In national payor-level datasets, proxies commonly used to indicate quality include hospital length of stay (LOS), postoperative complications, and non-routine discharge.7-9 Optimization of these outcomes also appears to correlate with lower costs and higher-quality care in spinal surgery, where extended LOS and non-routine discharge have been identified as both drivers of high costs7-9 and inferior surgical outcomes, including increased rates of complication and morality.8,9 However, most of these investigations have been in those patients treated for degenerative pathologies vs spinal tumors. Intradural tumors of the spine comprise 40–45% of primary spinal neoplasms, commonly presenting with pain and progressive sensory or motor deficits.10,11 Surgical resection is the gold standard for most intradural tumors,12,13 but can be associated with high costs and rates of complication. 10 Therefore, further investigation into patient risk-factors associated with inferior outcomes and increased resource utilization is warranted to better patient care.
One such factor that has garnered increased attention of late is frailty, a common condition among the elderly that is defined by decreased physical reserve and which may affect up to 30% of patients with primary spinal tumors.14-16 Prior investigations of frailty in spine oncology have used validated measures such as the modified 5-item frailty index (mFI-5), modified 11-item frailty index (mFI-11), and metastatic spinal tumor frailty index (MSTFI).17,18 However, most of these have been deployed in single-institution datasets and there is increased interest in studying frailty utilizing large, national databases.19,20 One metric of frailty designed specifically for use in these datasets is the hospital frailty risk score (HFRS), which is calculated using 109 internationally standardized and easily accessible ICD-10 diagnostic codes for conditions overrepresented in frailty.17,18,20 The HFRS has been validated as a reliable predictor of extended hospital LOS, intensive care unit (ICU) stays, and non-home discharge in patients undergoing surgery for a degenerative spinal condition. 21 However, it has yet to be applied to patients with primary tumors of the spinal cord and meninges.
The aim of this study was to utilize the HFRS to investigate the impact of frailty on complication rates, LOS, discharge disposition, and total cost of hospital admission in patients undergoing surgical intervention for primary spinal intradural/cord tumors.
Methods
Data Source and Patient Population
The Healthcare Cost and Utilization Project’s Nationwide Inpatient Sample (NIS) database is a stratified discharge database representing 20% of all inpatient admissions from community hospitals in the United States. It is the largest all-payer healthcare database in the US, containing over 7 million hospital admissions (approximately 35 million hospitalizations, weighted) per year. A retrospective study was performed using years 2016–2018 of the NIS for all adult (≥ 18 years old) patients undergoing surgical intervention for primary spinal intradural/cord tumor. Institutional Review Board (IRB) was deemed exempted due to the deidentification of patients in the NIS database.
Within the NIS, the International Classification of Diseases, Tenth Revision, Clinical Modification [ICD-10-CM] diagnosis and procedural coding system (PCS) was used to identify patients with spinal intradural/cord tumors, as well as their previously diagnosed comorbidities and the surgical interventions they underwent. The ICD-10-CM codes C72.0 “Malignant neoplasm of spinal cord”, D32.1 “Benign neoplasm of spinal meninges”, D33.4 “Benign neoplasm of spinal cord”, D43.4 “Neoplasm of uncertain behavior of spinal cord” were used to identify our patient population. Patients with ICD-10-CM codes corresponding to metastatic cancer, peripheral nerve sheath tumors and secondary neoplasms of the central nervous system, and patients who underwent excision of vertebrae were excluded from analysis. Identified patients were then cross-matched using ICD-10-CM codes to determine what surgical interventions the patient underwent.
Hospital Frailty Risk Score (HFRS)
Frailty was defined using the HFRS, a validated ICD-10 coding algorithm that was developed by Gilbert et al. 20 and assessed in subsequent work. 22 The coding algorithm is derived from a cluster of frail individuals with a comprehensive 109 ICD-10 codes assigned to a number of points ranging from .1 to 7.1 correlating to the strength of association predicting membership to the frail cluster. 20 These points are added together for a final frailty risk score. 20 All ICD-10 codes for the admission were entered into the calculation. In addition to presenting frailty as a continuous variable, we then dichotomized the frailty score into Non-Frail (HFRS<5) and Frail (HFRS>5).
Data Collection
Patient demographics such as age, sex, race, median household income, and insurance provider were all collected from the NIS database. Additional hospital characteristics such as size by bed volume, region (Northeast, South, Midwest, and West), and type (rural, urban teaching, and urban non-teaching) were also collected. Elixhauser comorbidities were used to evaluate incidence of congestive heart failure (CHF), cardiac arrhythmia, valvular disease, hypertension (HTN), pulmonary circulation disorders, peripheral vascular disorders, paralysis, other neurological disorders, chronic pulmonary disease, diabetes, hypothyroidism, renal failure, liver disease, peptic ulcer disease excluding bleeding, AIDS/HIV, lymphoma, metastatic cancers, solid tumor without metastasis, rheumatic disease, coagulopathy, obesity, weight loss, fluid and electrolyte disorders, anemia (blood loss or deficiency), alcohol or drug abuse, and psychoses. In addition, incidences of affective disorders, nicotine dependence, opioid dependence were assessed. Data on intraoperative variables such as procedural type (e.g., excision of the meninges/cord, release of spinal meninges/cord, cervical fusion, cervicothoracic, thoracic fusion, thoracolumbar fusion, lumbar fusion, and lumbosacral fusion), whether blood transfusion was required, and postoperative complications were also collected. Of note, HFRS utilizes only diagnostic coding; no procedural coding was identified in making the HFRS algorithm.
Data regarding postoperative complications for each patient was collected by indexing additional diagnoses from the NIS database. Complications included in the analysis were urinary tract infection (UTI), post-procedural fever, pressure ulcer, acute post-hemorrhagic anemia, displacement of internal fixation device of vertebrae, wound disruption, nervous system issues, acute deep vein thrombosis, post-procedural shock or infection, mechanical device complication, hematoma, post-procedural digestive ailments, and any anesthesia-related adverse event. In addition, postoperative outcome measures such as hospital LOS, total cost of hospital admission, and discharge disposition were also assessed between the 2 cohorts. Discharge disposition was classified as routine (patient went home), non-routine (patient sent to short-term hospital, skilled nursing facility, intermediate care facility, home with healthcare services), and other (leaving against medical advice, died in hospital, unknown destination). All-payer inpatient cost-to-charge ratios (CCR) were used to convert total hospital charge to total cost of hospital services.
Statistical Analysis
Discharge-level weights provided by the Agency for Healthcare Research and Quality were used to calculate national estimates. Parametric data was expressed as mean ± SD and compared via two-way independent t tests. Nonparametric data was expressed as median (interquartile range) and compared via the Kruskal–Wallis tests. Nominal data was compared with the χ 2 test. For our primary hypothesis, weighted univariate and multivariate logistic regressions were fitted with extended postoperative hospital LOS (as defined by LOS greater than the 75th percentile for the entire cohort), non-routine discharge, and increased hospital costs (as defined by total costs greater than the 75th percentile for the entire cohort) as the dependent variable. There were no patients with missing information on LOS. Patients with missing total cost variable were excluded from this portion of the analysis. Patients with “other” discharge were also excluded from this portion of the analysis to dichotomize routine vs non-routine discharge. Backward stepwise multivariate logistic regression analysis was used to select variables in the final model, using P ≤ .1 as entry and stay criteria. A P-value of less than .05 was determined to be statistically significant. Statistical analysis was performed using R Studio, Version 3.6.2, RStudio Inc, Boston, MA.
Results
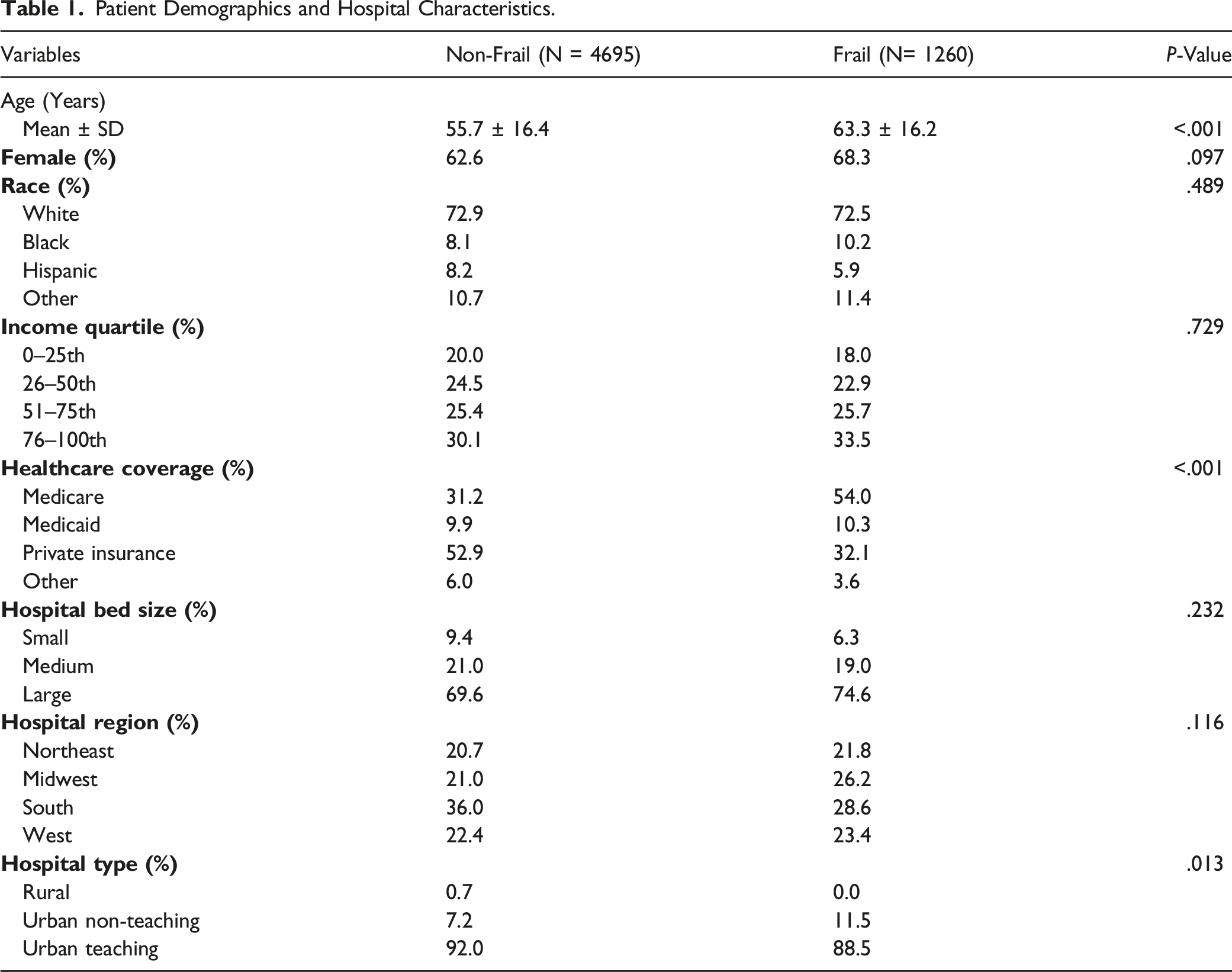
Patient Demographics and Hospital Characteristics
Patient Demographics and Hospital Characteristics.
Patient Comorbidities
Admission and Patient Comorbidities.
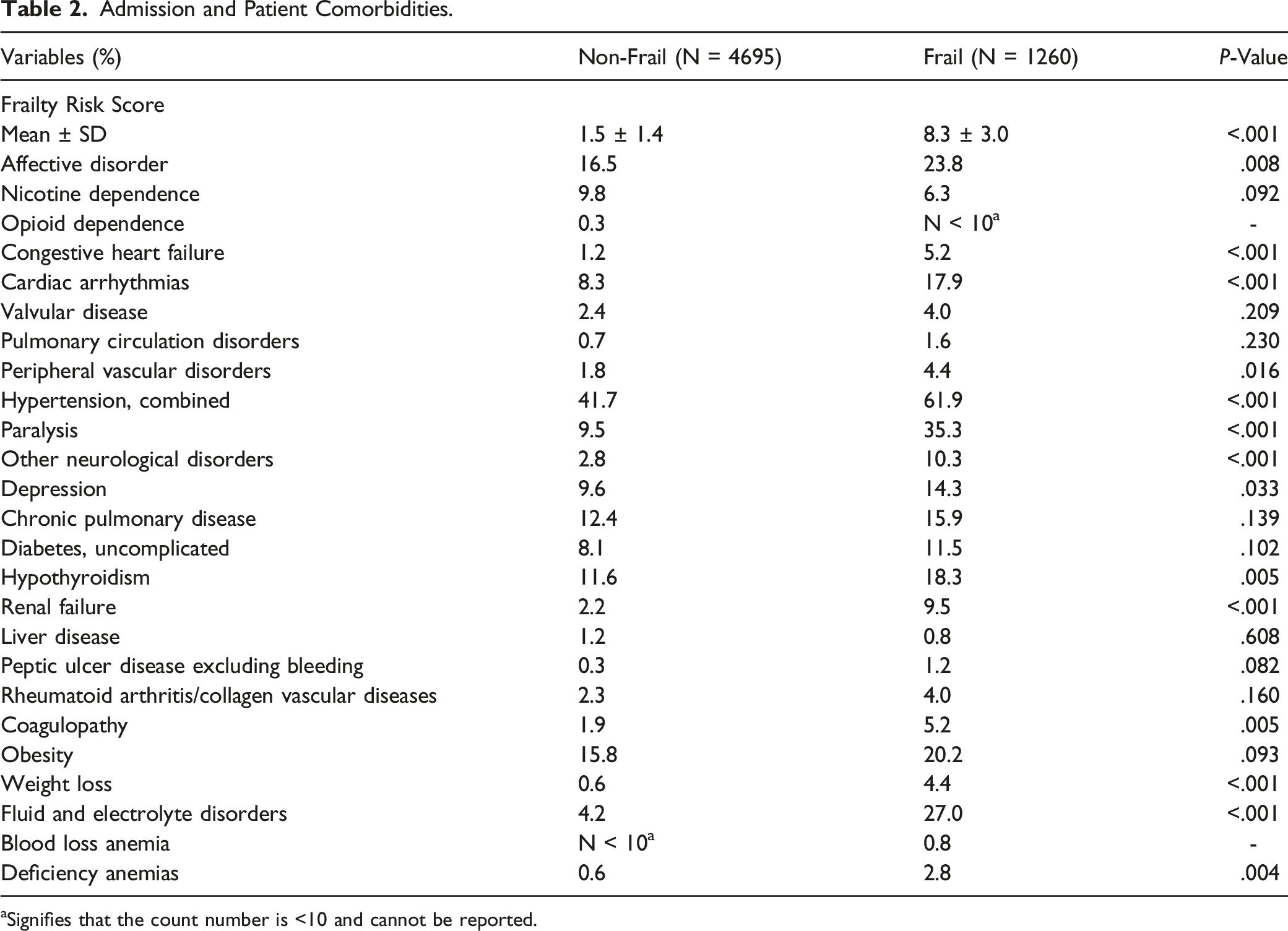
aSignifies that the count number is <10 and cannot be reported.
Intraoperative Variables
Intraoperative Variables.
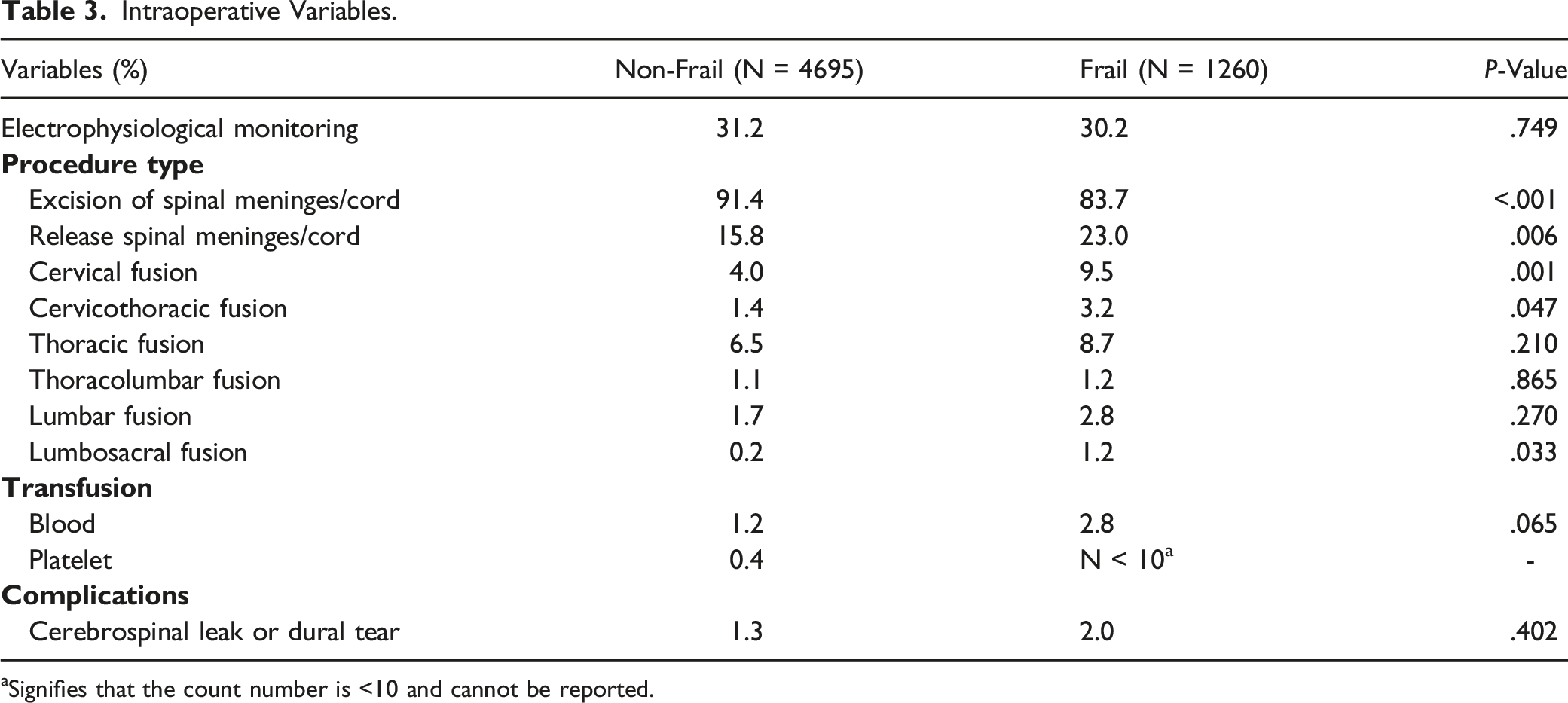
aSignifies that the count number is <10 and cannot be reported.
Postoperative Complications
Postoperative Complications.
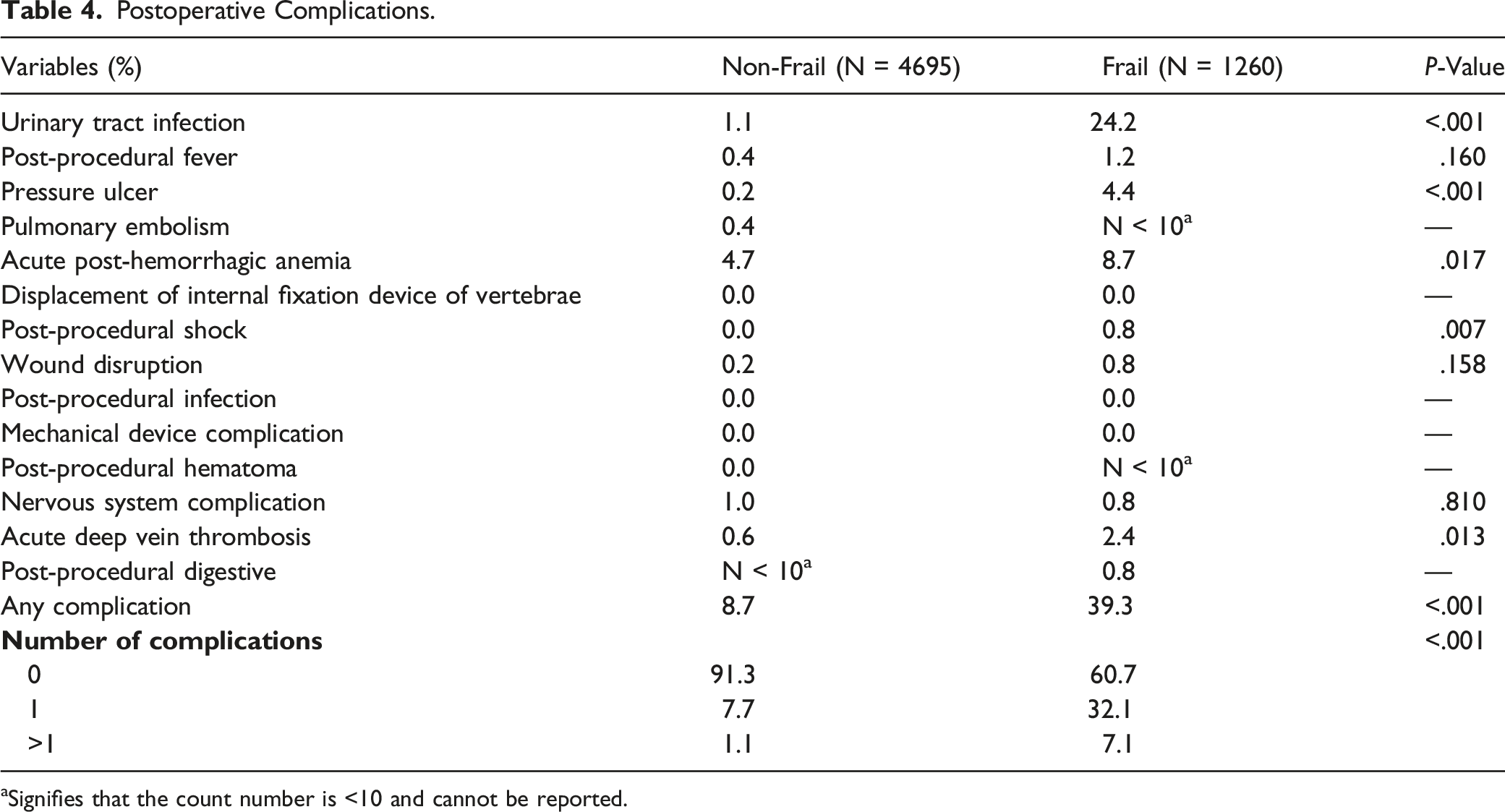
aSignifies that the count number is <10 and cannot be reported.
Hospital Outcomes
Postoperative Inpatient Outcomes.

Multivariate Analysis for Extended LOS
Logistic Multivariate Regression Analysis for Extended Length of Stay.
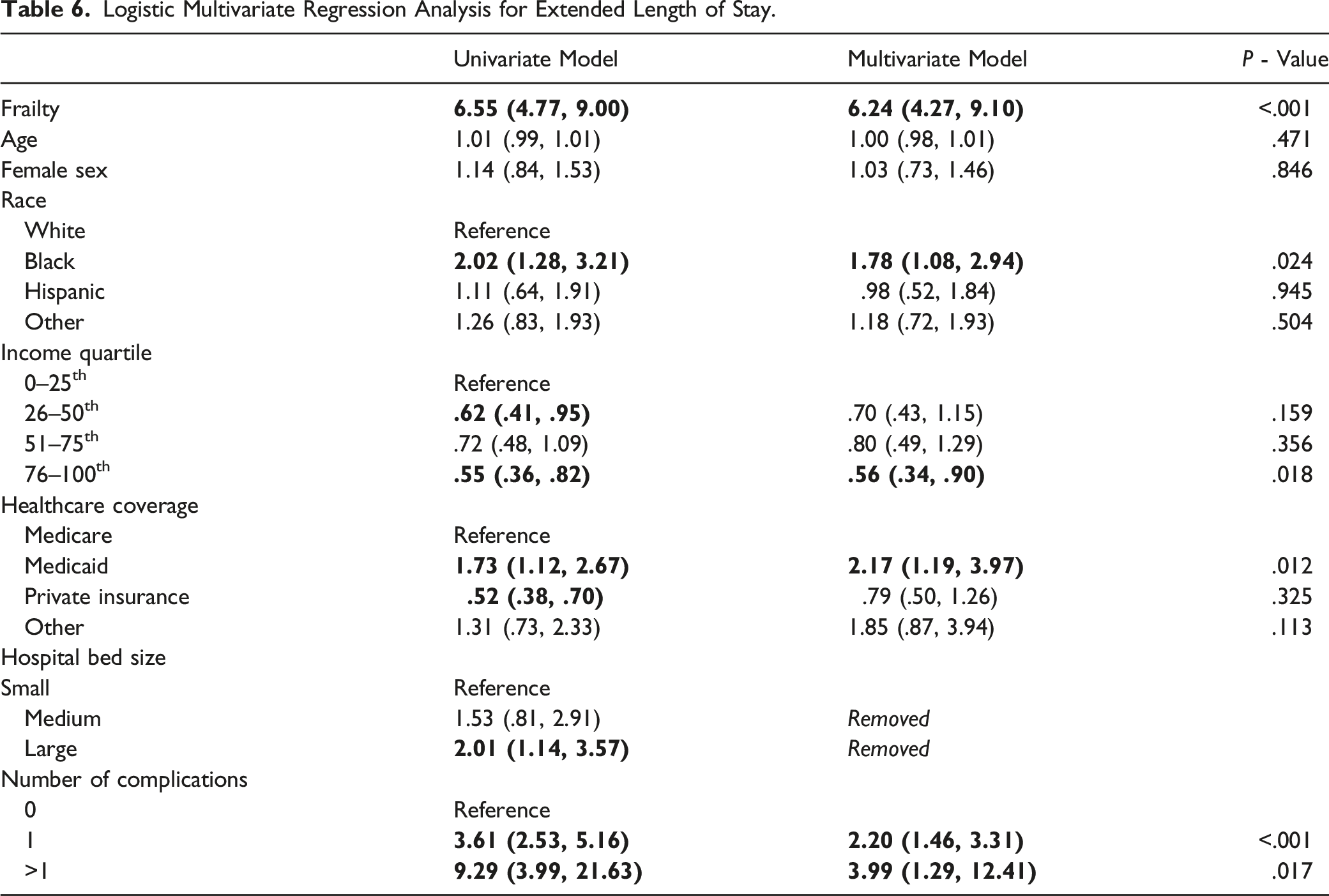
Multivariate Analysis for Non-Routine Discharge
Logistic Multivariate Regression Analysis for Non-Routine Discharge Disposition.
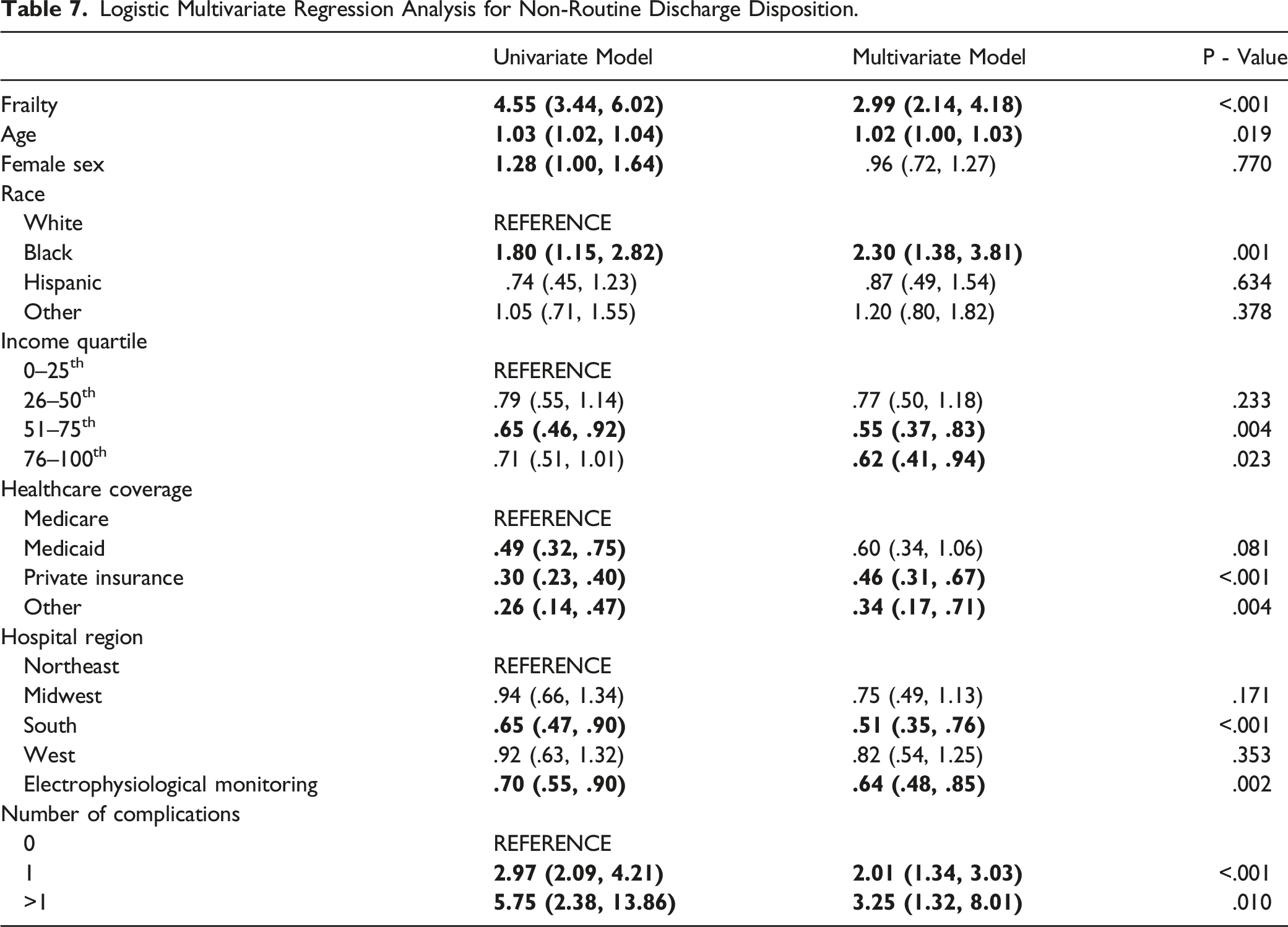
Multivariate Analysis for Increased Cost
Logistic Multivariate Regression Analysis for Increased Cost.
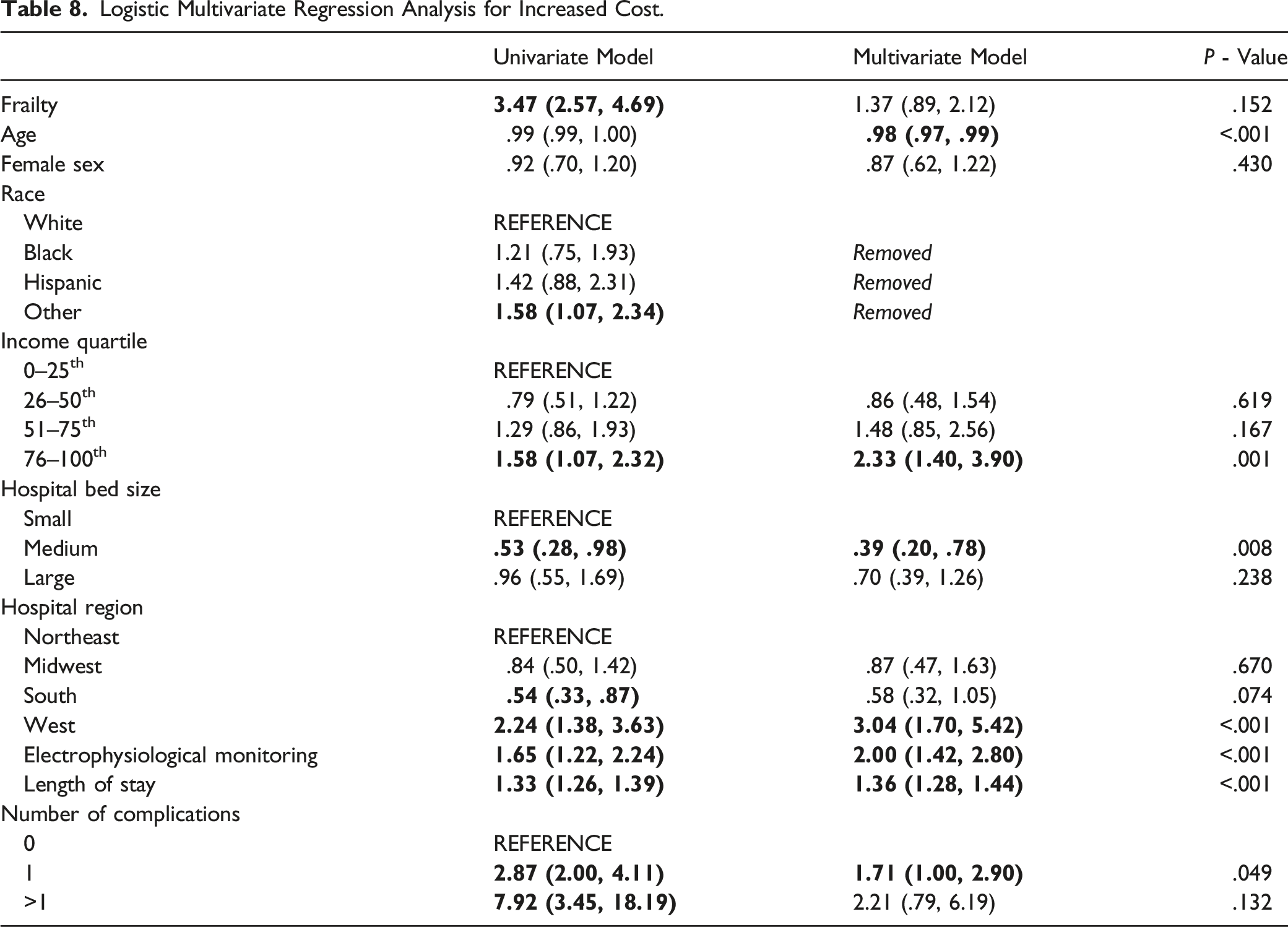
Discussion
In this retrospective, national database study of 5955 patients undergoing surgery for a primary spinal intradural/cord tumor, we found on univariate analysis that frailty was associated with longer LOS, higher rates of non-routine discharges, and increased total costs. Additionally, on multivariate analysis, frailty was found to be an independent predictor of extended LOS and non-routine discharge.
Given the association of frailty with increased vulnerability to adverse healthcare outcomes,14,15 several groups have examined the prevalence of frailty within the primary spinal intradural/cord tumor population. In a retrospective study of 1589 patients from the NIS database surgically treated for primary spinal tumors between 2002 and 2011, Ahmed et al. 16 found using the 9-item spinal tumor frailty index (STFI) that 20.2% of patients were considered mildly frail (STFI = 1), 6.0% were moderately frail (STFI = 2), and 2.2% were severely frail (STFI ≥ 3). 16 In another NIS database study of 18 297 patients surgical treated for benign intradural tumors between 2002 and 2011, Kalakoti et al. showed that many patients presented with frailty-associated comorbidities such as chronic obstructive pulmonary disorder (18.6% of patients), congestive heart failure (2.9%), and hypertension (41.0%). 25 Similarly, in a prospective, single-institution study of 38 patients undergoing surgery for primary intradural extramedullary tumors, Chotai et al. 26 found that frailty-related comorbidities were common: 15% of patients had diabetes, 43% had hypertension, 30% had coronary artery disease, and 5% had a prior myocardial infarction or congestive heart failure. 26 Additionally, the authors showed that 68% of patients had an American Society of Anesthesiologists (ASA) grade of 3 or 4 and 32% had an ASA grade of 2, indicating that the overwhelming majority of patients were living with mild or severe systemic diseases which likely contributes to frailty. 26 In our study, 21.2% of patients undergoing surgery for a primary spinal intradural/cord tumor were considered to be frail by HFRS criteria. Moreover, as the patient population ages and life expectancies increase, the prevalence of frailty will continue to rise. Therefore, continued efforts to identify and better characterize frailty will allow for more targeted preoperative planning and risk stratification.
While the prevalence of frailty continues to rise among patients operated for primary tumors of the spinal cord and meninges, there have been few attempts to characterize the effect of frailty and frailty-associated factors on postoperative complication rates. In the aforementioned study by Ahmed et al., the authors found the odds of experiencing a postoperative complication significantly increased as frailty score progressed from mild (OR: 3.83), to moderate (OR: 6.80), and to severe (OR: 13.05). 16 Similarly, in a retrospective NIS database study of 19017 patients who underwent surgery for a primary tumor of the spine, Patil et al. showed on multivariate analysis that the odds of experiencing a complication were increased for patients with 1 (OR: 1.4), 2 (OR: 1.6), and ≥3 (OR: 1.6) pre-existing comorbidities. 27 Similar to these prior studies, the present study also found frailty to be associated with increased complications. However, what remains unclear is whether the increased risk of complications in frail patients results from the frail phenotype as a whole, or if it is predominately driven by a subset of the comorbidities associated with frailty.
In addition to being associated with increased costs, prolonged hospitalizations following spine surgery are associated with decreased patient satisfaction and increased complication rates. 9 Similar to examinations of frailty and postoperative complications, only a few studies have considered the impact of frailty on LOS following surgery for spine tumors. In the retrospective study of 2170 patients treated surgically for either a primary or secondary spinal tumor, Lakomkin et al. showed that frailty predicted extended LOS, with an OR of 14.91. 28 Interestingly, in the same study, the authors conducted a subgroup analysis that examined the impact of frailty on outcomes just within the 981 patients with a primary tumor and found an even larger impact on prolonged LOS (OR: 323.6 comparing mFI-11 value of 1 vs 0). 28 Similarly, in their study of patients treated for primary tumors, Ahmed et al. 16 showed that mean LOS increased along with advancing frailty score, extending from 6.4 days for non-frail patients to 18.3 days for severely frail patients. 16 With prolonged length of stay, there is an association with increasing costs. Using an alternative definition of frailty—the Charlson Comorbidity Index (CCI)—and 15545 patients from the NIS dataset treated surgically for primary intramedullary tumors, Sharma et al. demonstrated that a high CCI on presentation was predictive of an increased cost of hospital admission (OR: 1.95). 29 These results echoed those of Lakomkin et al., 28 who found each additional point on the CCI score to be associated with a 23% increase in the odds of having a prolonged LOS. Analogously, our study determined that frailty independently predicted prolonged LOS and higher costs. Identifying how frailty impacts LOS, there is opportunities for creating enhanced recovery after surgery (ERAS) pathways. 30 While ERAS protocols are being better understood and implemented into practice, further investigations need to be explored for these observed discrepancies, as doing so may improve patient care and reduce healthcare costs.
The final metric considered here - non-routine discharge—is also associated with increased healthcare resource utilization and poorer patient outcomes. To our knowledge, no previous studies have directly examined the impact of frailty on non-routine discharge following surgery for primary spinal intradural/cord tumors. However, several have found frailty-associated factors to predict this endpoint. In the Sharma et al. study, patients with a high CCI ( ≥ 2) had a nearly 2-fold higher odds of experiencing a non-home discharge relative to those with low CCI (0-1). 29 Analogously, in a study of 18297 patients from the NIS database operated for benign intradural tumors, Kalakoti et al. showed the following frailty-associated comorbidities to independently predict unfavorable discharge: coronary artery disease (OR: 1.47), congestive heart failure (OR: 1.80), coagulopathy (OR: 1.83), and hypertension (OR: 1.23). 25 Moreover, in a retrospective, NSQIP database study of 1232 patients who underwent laminectomy for an intradural extramedullary spinal tumor between 2011 and 2014, Ahn et al. demonstrated on multivariate analysis that specific frailty-associated conditions such as an age of ≥65 years (OR: 2.73) and dependent functional status (OR: 4.30) were associated with non-routine discharge. 31 Likewise, our study found frailty to independently predict non-routine discharge within this patient population. As previously mentioned though, further work is necessary to determine whether the risk is derived from the frail phenotype as a whole, or whether it is predominately driven by a subset of the considered comorbidities. Regardless, it appears that improved preoperative screening that focuses on identifying frail patients may allow for the tailoring of perioperative care pathways so as to reduce adverse events and expedite safe home discharge following surgery for a primary spinal tumor.
This study has several limitations inherent to all studies employing administrative datasets that may impact the generalizability of the findings. First, given that the data was collected by ICD codes, the data may contain coding and reporting biases, misclassified diagnoses and procedures, or incomplete data. Second, as the NIS only has information that pertains to a single inpatient hospital admission, we were unable to assess more long-term outcomes. Third, as the data was analyzed retrospectively, we are unable to assess the impact of potential confounding factors, such as variations in surgical approach and technique which may have significant implications on our results. Lastly, we are comparing outcomes based on frailty. Many different operating definitions have been employed for frailty in the medical literature and it is unclear if any single definition is optimal. Here we used the HFRS and so it is possible that the findings may not directly translate to clinical settings in which an alternate definition, for example, the mFI-11, is employed. Nevertheless, the similarity of the present findings to those in the literature suggests that frailty, more broadly, may be an important predictor of patients at risk for adverse outcomes. Additionally, herein we employed data from the largest inpatient healthcare database in the United States, reducing the risk of sampling bias present in many single-institution investigations.
Conclusion
This study was the first to use the Hospital Frailty Risk Score (HFRS) to assess the impact of frailty on outcomes following surgery for primary tumors of the spinal cord and meninges. We found that in this patient population, Frail patients—those with high HFRS scores—had longer hospitalizations, higher rates of non-routine discharge, and higher total hospitalization costs. Subsequent studies exploring these findings are necessary to further improve quality of patient care and reduce healthcare costs.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
