Abstract
Study Design
Observational Database Study.
Objectives
Prospective clinical trials in spinal surgery are expensive to conduct, especially when randomized, appropriately powered, and/or multicentered. Industry collaborations generate symbiotic relationships promoting technological advancement; however, they also allow for bias. To the authors’ knowledge, there is no known analysis of correlations between industry sponsorship and publication rates of spine-related clinical trials. This observational work evaluates such potential associations.
Methods
The ClinicalTrials.gov database was queried with terms spine, spinal, spondylosis, spondylolysis, cervical, lumbar, and compression fracture over an 11-year period. Design characteristics and outcomes were recorded from 822 spine surgery-related trials. Trials were stratified based on funding source and intervention class. Groups were compared via two-tailed chi-square test of independence or Fisher’s exact test (α = .05), based on completion status and publication rates of positive vs negative results.
Results
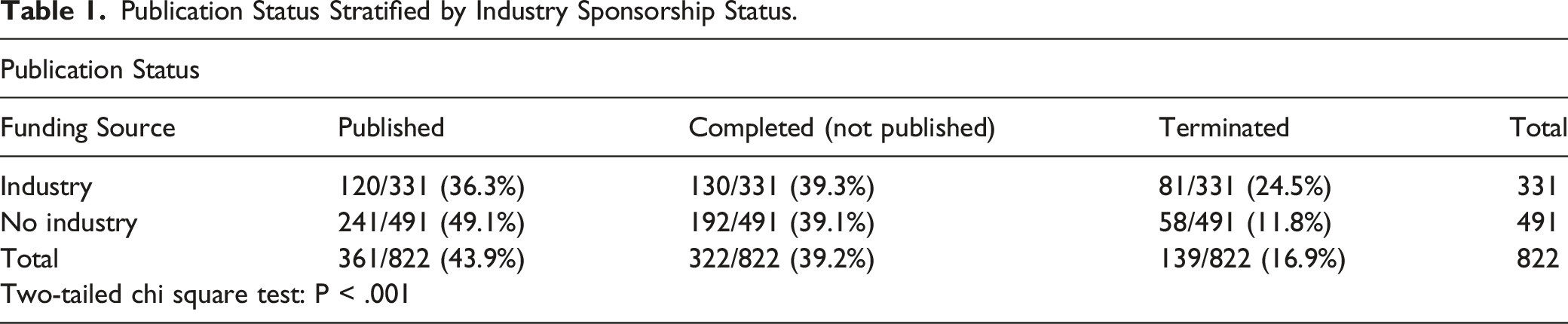
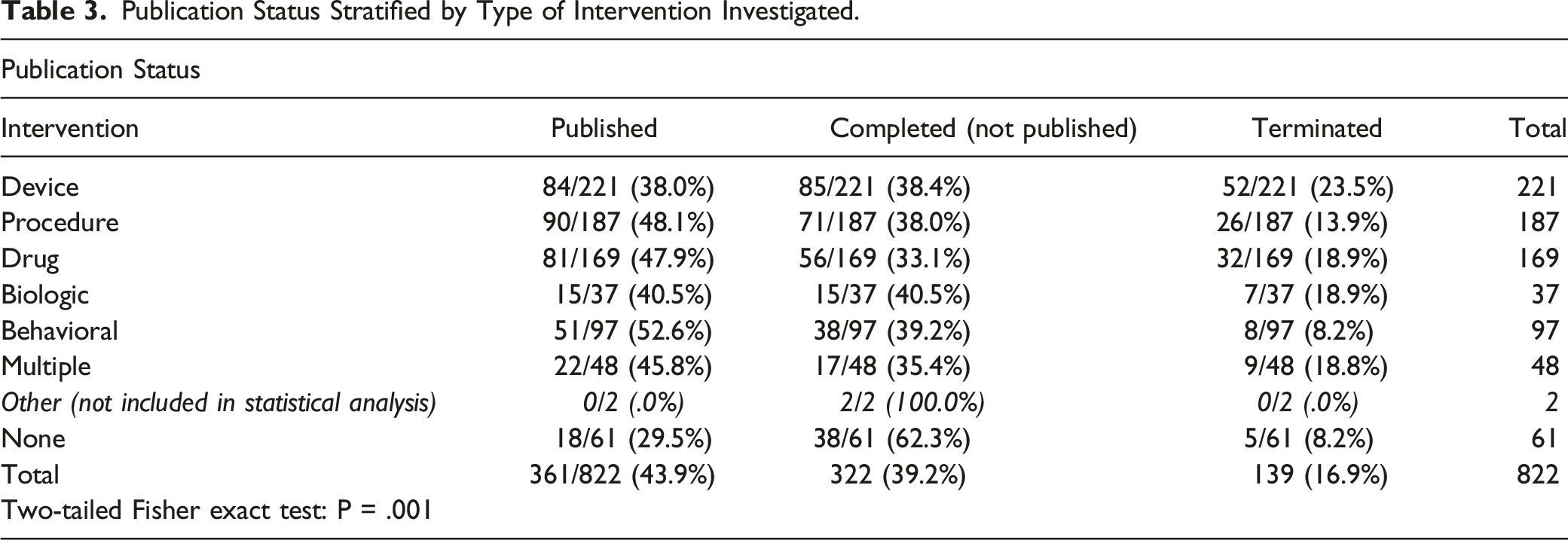
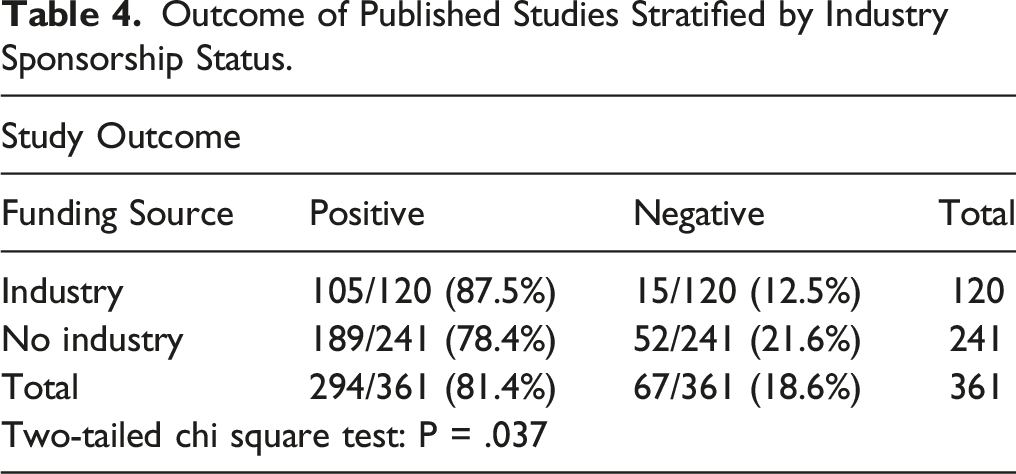
Industry-sponsored spine-related clinical trials were more likely to be terminated than their non-industry-sponsored counterparts (P < .001). Of the trials achieving publication, industry-sponsored trials reported positive results at a higher rate than did trials without industry funding (P = .037). Clinical trials examining devices were more likely to be terminated than those studying other intervention classes (P = .001).
Conclusions
High termination rates and positive result publication rates among industry-sponsored clinical trials in spinal surgery likely reflect industry’s influence on the research community. Such partnership alleviates financial burden and provides accessibility to cutting-edge innovation. It is essential that all parties remain mindful of the significant bias that funding source may impart on study outcome.
Introduction
For approximately four decades, the complex environment surrounding industry-sponsored research in the medical field has garnered both interest and scrutiny. 1 The potential introduction of bias and conflicts of interest offsets the attractive combination of funding and access to cutting-edge technology.2,3 This general trend is of particular relevance in the field of spine surgery, which continues to progress due to the national focus on healthcare reform and the emphasis on evidence-based medicine. 4 A recent study reported that clinical trials of spine devices were substantially more likely to be industry-sponsored, and that those device trials were more likely multi-centered and included significantly more participants. 5 Clinical trials, which are defined as research studies that prospectively evaluate a medical/surgical intervention, are not only of particular importance to the progression of evidence-based medicine, but also create an atmosphere that favors collaboration between industry and surgeons.6,7 This is due to both the high cost and time-consuming nature of implementation of clinical trials, which often create challenges in obtaining and maintaining funding. 8 Although the relationship between industry and clinicians is frequently mutually beneficial and crucial for technological advancement and the progression of medicine and surgery, these ties can potentially breed bias in research publication. 9 This risk has been investigated by several past studies of clinical trials across various medical disciplines, demonstrating a significant association between industry sponsorship and the publication of positive findings; prior work also indicates that industry sponsorship may influence researchers to emphasize conclusions in favor of their products.9-12
While prior works have widely recognized the collaborative efforts and influences between industry and the medical community with regards to publication status and positive result reporting, there is a paucity of evidence regarding the specific effect of industry on spinal surgery clinical trials. A recent study from the United Kingdom of all spinal surgery research publications from 2010 reported that industry sponsorship is significantly associated with both a lower level of evidence of study, as well as a higher likelihood of reporting favorable outcomes. 13 This work, however, was not focused on clinical trials and only represented a single year of publication data. Additionally, a recent work that did look at clinical trials in the context of spine surgery reported a lower likelihood of publication when associated with industry sponsorship, but did not examine whether or not this correlated with increased reporting of positive results. 14
With this in mind, the primary objective of the current database search is to investigate whether any relationship exists between funding source and completion status of spine-related clinical trials. Secondary objectives were used to evaluate published studies, which included examining any relationships between type of intervention studied and completion status, or between funding sources and whether conclusions support or refute the stated hypotheses. As defined by standard field entries on ClinicalTrials.gov, intervention types include devices, procedures, drugs, biologics, behavioral changes, and “other.” Accordingly, funding sources include industry, the NIH, non-NIH US Federal Government, medical centers/universities, private foundations, and “other.” This study is not intended to judge the merit of industry sponsorship in spine research, but rather to provide data-driven context for analysis of the results of prospective clinical trials.
Materials and Methods
This database review did not directly study human participants, and therefore informed consent and IRB approval were not warranted. The National Institute of Health Clinical Trials Database at ClinicalTrials.gov was queried for titles including the keywords spine OR spinal OR spondylosis OR spondylolysis OR cervical OR lumbar OR compression fracture, between 2008 and 2019. These keywords were chosen with the intention of identifying the greatest number of spine-related studies; in an effort to limit search results to these studies, broader terms such as “stenosis” were not included. Studies with no participating site in the United States and those not pertaining to the spine were excluded. Using information extracted from the Clinical Trials Database as well as PubMed searches, completion and publication status, type of intervention tested, funding source, study design, and reported outcomes were recorded. PubMed queries to determine the publication status of the clinical trials included searches by National Clinical Trial number, title, all listed authors, and a minimum of two keywords or phrases from the study description. On ClinicalTrials.gov, all studies are designated as “completed” or “terminated.” Because many trials reach completion but not publication, a literature search was done to identify all manuscripts resulting from trials designated as “completed.” Those trials that did progress to publication in manuscript form were instead designated as “published.” Conversely, trials that had been published in abstract form only were not categorized as “published.” If multiple publications resulted from the same clinical trial, the publication date of the earliest published manuscript was documented. For each published study, a single reviewer documented the outcome of each study as “positive” or “negative,” depending on whether any conclusion was drawn in support of the primary tested hypothesis. It was also noted whether the study completed tests of statistical significance or published only descriptive statistics and/or qualitative measures.
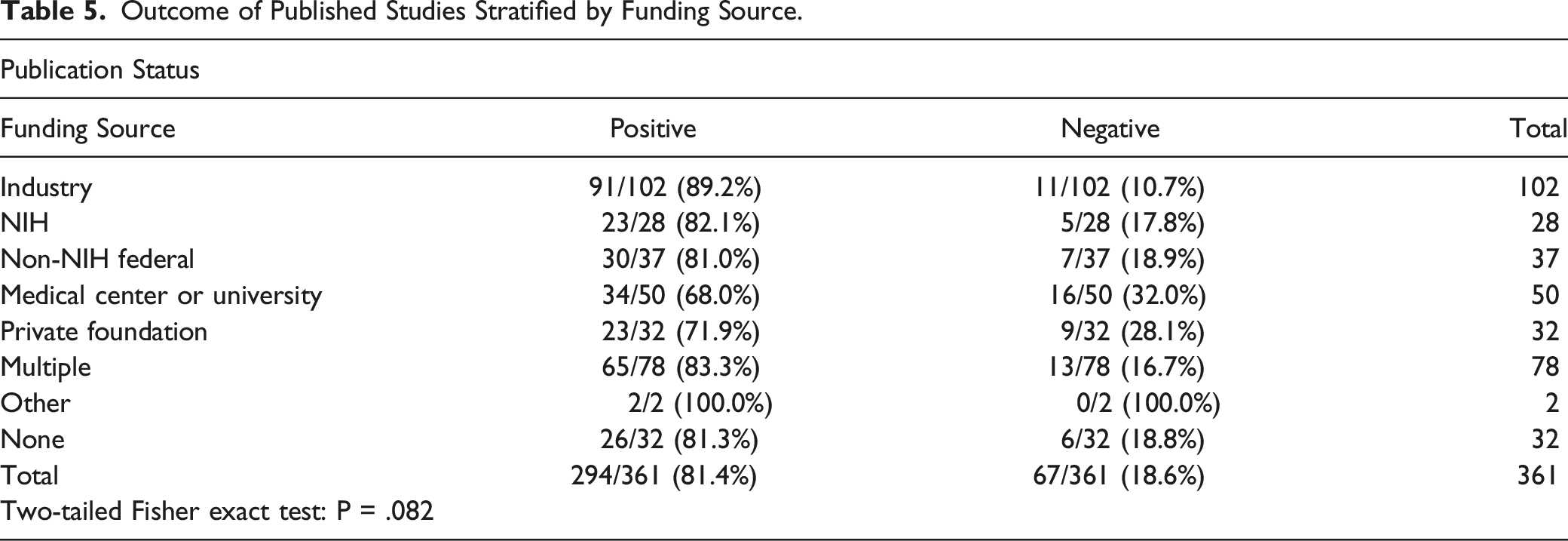
The trials were stratified based on the following standard fields as logged in ClinicalTrials.gov entries: industry-sponsorship status, other reported funding sources, and type of investigated intervention (“device,” “procedure,” “drug,” “biologic,” “behavioral,” “other,” “multiple,” or “none”). For published studies, all manuscripts were examined for disclosure of additional funding sources. The publication status and outcomes of published studies were then compared between these groups using either two-tailed chi square tests or Fisher exact tests, with α = .05 for all statistical measures.
Results
The ClinicalTrials.gov database returned 3639 studies from the initial search. There were 363 trials excluded for having no study sites in the United States, and 2454 trials were excluded for being unrelated to the spine. Remaining were 822 spine-related clinical trials that were included in this review (Figure 1). Flow chart illustrating application of inclusion and exclusion criteria.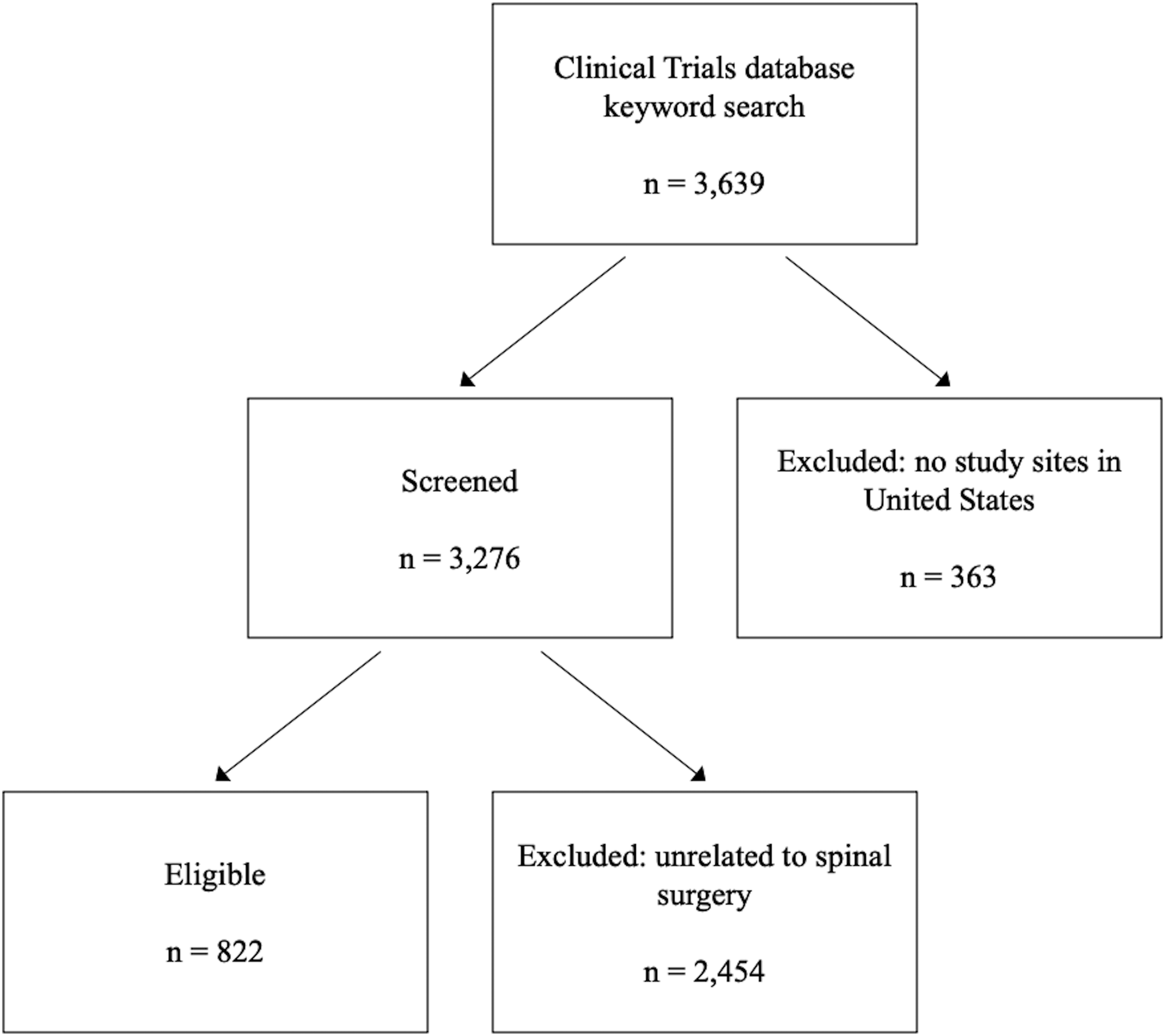
Publication Status Stratified by Industry Sponsorship Status.
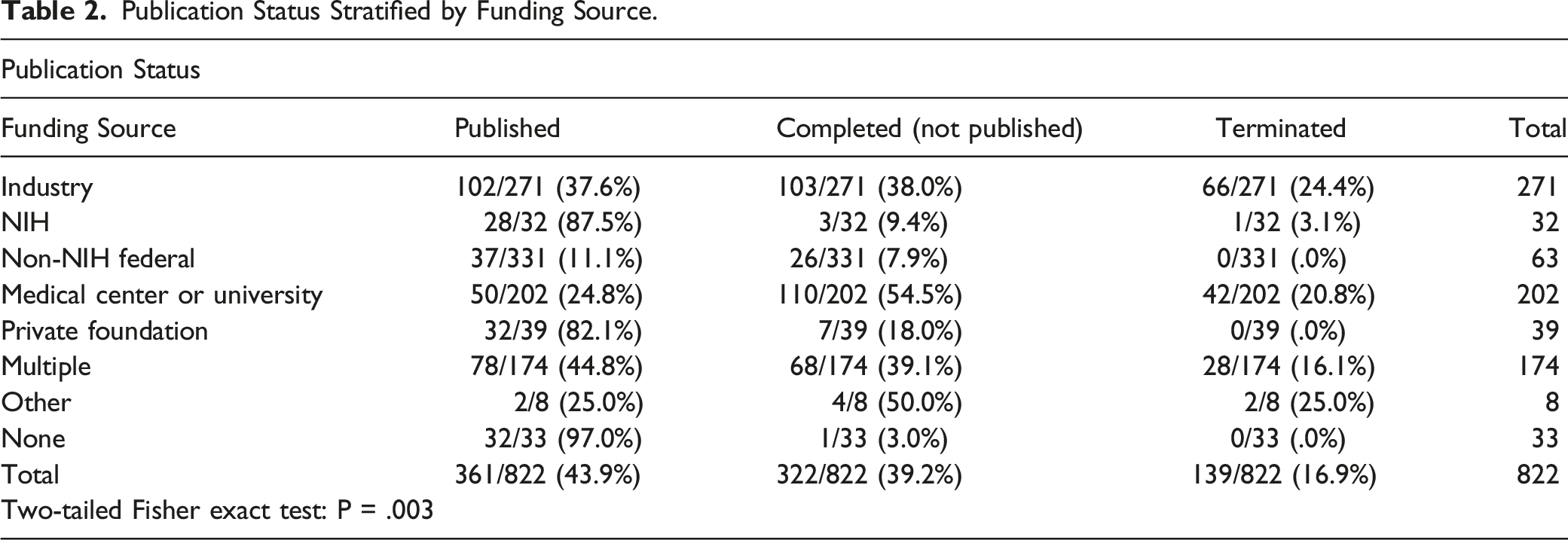
Publication Status Stratified by Funding Source.
Publication Status Stratified by Type of Intervention Investigated.
Outcome of Published Studies Stratified by Industry Sponsorship Status.
Outcome of Published Studies Stratified by Funding Source.
Discussion
Prospective clinical trials are essential to the advancement of spine surgery, especially with the advent of healthcare reform and an ever-increasing demand for evidence-based medicine to support the value of various medical and surgical interventions. 4 Disorders of the spine are increasingly associated with substantial disability, decreased productivity, and diminished health-related quality of life, with spine-related medical expenditures escalating significantly over the past two decades. 15 Thus, it is imperative to continue to pursue high quality clinical and surgical advancements via spine research. Prospective clinical trials generally constitute a lengthy, costly process; partnering with industry not only offsets these factors but also establishes a direct pipeline for new technologies to reach clinicians and patients. Therefore, prospective clinical trials remain an attractive target for industry sponsorships, which creates the risk of significant industry-centric bias.
In 2014, Amiri et al considered industry sponsorship of spine-related research studies of various levels of evidence published in five major medical and surgical journals over the course of a single year (2010), which demonstrated a significant association between industry funding and reporting of favorable outcomes in published manuscripts. 13 This work did not focus on prospective clinical trials and represents only a single year of publication. Ohnmeiss in 2015 reported on the fate of spine-related clinical trials, revealing a 38.9% publication rate across all spine-related clinical trials and noted a significant association between funding source and publication rate, with industry sponsorship being associated with decreased rate of publication. 14 However, there was no focus on the potential influence that funding source may have on the reporting and publication of positive or negative results. This current query expands on these important prior works13,14 by incorporating prospective clinical trials from an 11-year period and publications across all PubMed-indexed journals. Most notably, this effort considers trends not only among published studies, but also examines those that were terminated or completed yet unpublished, while remaining indexed on the Clinical Trials database.
Overall, clinical trials that obtained at least partial industry-sponsorship were less likely to be published and more likely to be terminated than their non-industry funded counterparts. Of those industry-sponsored trials that did achieve publication, an overwhelming majority published results in favor of their studied intervention. The influences leading to this outcome are likely multifactorial. These findings likely reflect some degree of positive results bias. However, may also indicate the rigorous review and refinement that companies impart upon their medical devices. If a single company develops a product and subsequently funds a clinical trial testing it, that company finds itself in a unique position to receive immediate feedback concerning the product’s performance. When shortcomings are found, the company then has the opportunity to pull the product and its related trials, implement improvements, and conduct a new trial. This ability to improve devices soon after their introduction to the spinal surgery community likely contributes to the higher termination rates and publication of positive findings among industry-sponsored clinical trials, as well as the higher termination rates among trials investigating medical devices.
While termination of clinical trials does present industry and surgeons alike with the ability to focus on implementation of the most cutting-edge, effective devices, it remains a controversial practice. Factors such as futility or ethical concerns can dictate the termination of clinical trials, but those that are terminated for other reasons can make it more difficult for subsequent trials on similar topics to obtain funding. 16 In addition, including data from a previously terminated trial in the literature can lead to overestimation of treatment effects, potentially prompting investigators to draw false conclusions. 16 Finally, discoveries that lead to termination of trials could hide important conclusions from the global research community. This potential source of waste may fuel the financial burden associated with the execution of high-quality clinical trials.
As with any study, this review does have associated limitations. Published studies that are not PubMed indexed have not been identified in this review. In addition, the inclusion of funding sources was limited to those disclosed within the Clinical Trials database and within any subsequent publications, and there may have been additional funding sources that were not disclosed. Furthermore, “completion” or “termination” status was not defined or assessed by any author, but rather was defined by the designated status of each study as assigned within the Clinical Trials database.
Conclusion
Industry sponsorship of spinal surgery-related clinical trials unequivocally correlates with higher termination rates, as well as greater likelihood to publish positive results. The various influences that may guide this trend include positive results bias and conflicts of interest, as well as pre-publication feedback and development. Further exploration is needed to further examine the motivations behind trial termination and arrested publication of industry-sponsored clinical trials in spinal surgery. However, the findings outlined in this study magnify the unique environment posed by industry sponsorships; prior to embarking on an industry-sponsored prospective clinical trial, spinal surgeons should carefully weigh both their positive and negative implications on the spinal research community.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
