Abstract
Study Design
Basic research
Objectives
Secreted protein acidic and rich in cysteine (SPARC) is a critical pro-fibrotic mediator. This study aims to characterize the role of SPARC in hypertrophic ligamentum flavum (LF) and fibrosis.
Methods
Hypertrophic LF samples were obtained from 8 patients with L4/5 lumbar spinal stenosis (LSS) during the decompressive laminectomy. Non-hypertrophic LF from age- and sex-matched 8 patients with L4/5 lumbar disc herniation was selected as control. An in vitro model of fibrosis in human LF cells was established by interleukin 6 (IL-6) to assess SPARC expression.
Results
Hypertrophic LF samples had higher fibrosis scores than control samples by Masson's trichrome staining (3.6 vs. 1.3, P < .001). Hypertrophic LF samples had significantly more positive staining for collagen and SPARC. Collagen III (Col3), α smooth muscle actin (α-SMA), and SPARC mRNA expression levels were significantly higher in hypertrophic LF samples than in control samples by qPCR. SPARC expression and fibrotic and inflammatory makers (collagen I, Col3, IL-6, interleukin 1β) were significantly upregulated in IL-6 stimulation of normal LF in vitro.
Conclusion
SPARC was detected in human LF and significantly upregulated in the clinical samples of hypertrophic LF compared to their normal counterparts. We also demonstrated an increased level of SPARC in an in vitro fibrosis model of LF. Thus, SPARC could be a crucial biomarker for the pathogenesis of hypertrophic LF and a therapeutic target for LSS.
Keywords
Background
Lumbar spinal stenosis (LSS) is one of the most common degenerative spinal disorders. Neurogenic claudication is the main symptom of LSS, characterized by painful cramping or weakness in the legs and causing a patient to sustain a decreased walking capacity. The pathophysiology is thought to be ischemia of the dura sac and nerve roots secondary to compression from surrounding hypertrophic ligamentum flavum (LF). Fibrosis is the main cause of hypertrophic LF. LF will become hypertrophic in the pathologic fibrosis process with increased collagen fibers and elastin degradation.1,2 The fibrosis process has resulted from the chronic inflammation in degenerative osteoarthritis and instability of adjacent facet joints. Although the morphologic or histologic changes of the hypertrophic LF were thoroughly discovered, the biochemical mechanisms for hypertrophic LF in LSS are still not clear.
Secreted protein acidic and rich in cysteine (SPARC) is a matricellular, collagen-binding protein that regulates cell shape, adhesion, migration, and proliferation. It also has complex effects on extracellular matrix (ECM) synthesis. Studies in skin, liver, lung, intestines, and heart have shown that the absence of global SPARC expression in mice significantly attenuates fibrosis.3-6 SPARC expression is closely related to collagen expression and deposition in human fibrotic diseases such as scleroderma, 7 liver fibrosis, 8 and glaucoma. 9 Furthermore, several studies have shown that the presence of SPARC in disc cells of aged subjects with disc degeneration is reduced and that deletion of the SPARC gene alters the disc ECM associated with accelerated disc degeneration in aging mice.10,11 Currently, there is limited or no study to explore the relationship between the expression of SPARC and hypertrophic LF.
In this study, we hypothesized that hypertrophic LF in lumbar spinal stenosis is associated with the increased expression of SPARC and SPARC as a potential biomarker. The result of the proposed research will provide pre-clinical evidence of the therapeutic target for the treatment of lumbar spinal stenosis. Thus, this study aims to investigate the role of SPARC in human LF.
Methods
Specimen
In the LSS group, hypertrophic LF samples were obtained during decompression laminectomy in 8 patients with L4/5 spinal stenosis, all LF samples having a thickness greater than 4 mm on axial T2 magnetic resonance imaging (MRI) measurements.12,13 To avoid bias, age- and sex-matched patients with L4/5 lumbar disc herniation and patients with LF thickness measured by MRI less than 2 mm were selected as controls.12,14 Half of each sample was fixed in 4% buffered formalin, dehydrated in graded ethanol concentrations, and embedded in paraffin for Masson's trichrome staining and immunohistochemical study. The other half of each sample was immediately frozen in liquid nitrogen for quantitative polymerase chain reaction (qPCR). Informed consent was obtained from each patient, and the ethics committee approved this protocol of Taipei Medical University (TMU-JIRB No.202004026)
Histologic Examination
Hematoxylin, eosin, and Masson's trichrome stain were used to detect collagen and elastin and evaluate LF fibrosis. The paraffin-embedded tissue samples were cut in 4-µm-thick sections on a microtome. Sections were stained by hematoxylin, eosin, and Masson's trichrome stain according to the manufacturer's instructions. The blue color represents collagen fibers. In each section, ten randomly selected fields were examined under ×200 magnification to assess the degree of LF fibrosis (the semiquantitative score for LF fibrosis). The degree of LF fibrosis was scored. 15 Grade 0 indicates normal LF showing collagen in 20% or less of the entire area. Grade 1 indicates between 20% and 40%. Grade 2 indicates between 40% and 60%. Grade 3 indicates between 60% and 80%. Grade 4 indicates over 80% of the entire area.
Determination of SPARC Expression in the LF
Immunohistochemical analysis was performed to determine the location of the SPARC protein in the LF tissue. Paraffin-embedded tissue samples were cut into 4-µm-thick sections on a microtome. After deparaffinization and rehydration, sections were pretreated with .01 mol/L phosphate-buffered saline (PBS) for 15 min in a microwave oven, followed by treatment with .3 % hydrogen peroxide for 15 min. Sections were treated for 2 hours at room temperature under a humidity chamber with rabbit polyclonal to SPARC antibody (Elabscience, Houston, USA) after incubation with normal blocking serum. Immunoreactivity was visualized following the manufacturer's instructions. Finally, the sections were counterstained with hematoxylin. After each step, the buffer solution used for rinsing consisted of .01 mol/L PBS. Negative controls were obtained by substituting the primary antibody with non-immune rabbit Ig G. Normal human skin as the positive control was also stained according to manufacturers' recommendations.
LF Cells Isolation, Culture, and Treatment
The hypertrophic LF cells originated from the LF samples from 8 patients undergoing spinal surgery after LSS diagnosis, and the non-hypertrophic LF cells were obtained from LF tissue during discectomy operation in 8 patients with lumbar disc herniation. The ligaments were washed with PBS and minced into pieces of approximately .5 mm2. The LF tissues were then digested for 1 hour with .2% type I collagenase (Sigma–Aldrich, St. Louis, MO), followed by washing with Dulbecco's Modified Eagle's medium (DMEM; Gibco BRL, Melbourne, Australia). The collagenase-treated ligament chips were finally placed in six-well plates (Corning- Costar, Oneonta, NY) in DMEM supplemented with 10% fetal bovine serum (Gibco BRL), 100 U/ml penicillin and 100 pg/ml streptomycin, and incubated in a 5%CO2 humidified incubator at 37C. Cells migrating from the explants and becoming confluent were harvested by the treatment with .2% trypsin and .02% ethylenediamine tetraacetic acid and passaged. Characterization of LF cells was demonstrated by cell morphology under a phase contrast microscope and expression of type I collagen under immunofluorescence microscopy. The LF cells were treated with Interleukin-6 for 24 h.
Statistical Analysis
Data will be expressed as the mean ± standard deviation (SD) and analyzed using the SPSS version 12 statistical analysis package (SPSS Inc., Chicago, IL, USA). Statistical analysis will be performed using one-way analysis of variance (ANOVA), Kruskal–Wallis test, and the student's t-test. The value of P < .05 is considered statistically significant.
Results
The thickness and fibrosis scores were higher in the LSS group than in the control group.
Comparison of the LF thickness and fibrosis scores a between the LSS Group and control Group.

LF: ligamentum flavum; LSS: lumbar spinal stenosis.
aThe severity of fibrosis was scored based on the guidelines by Sairyo et al15.
bthe difference is significant (P < .05).
The presence of SPARC and fibrotic markers in LF was highly associated with the LSS group.
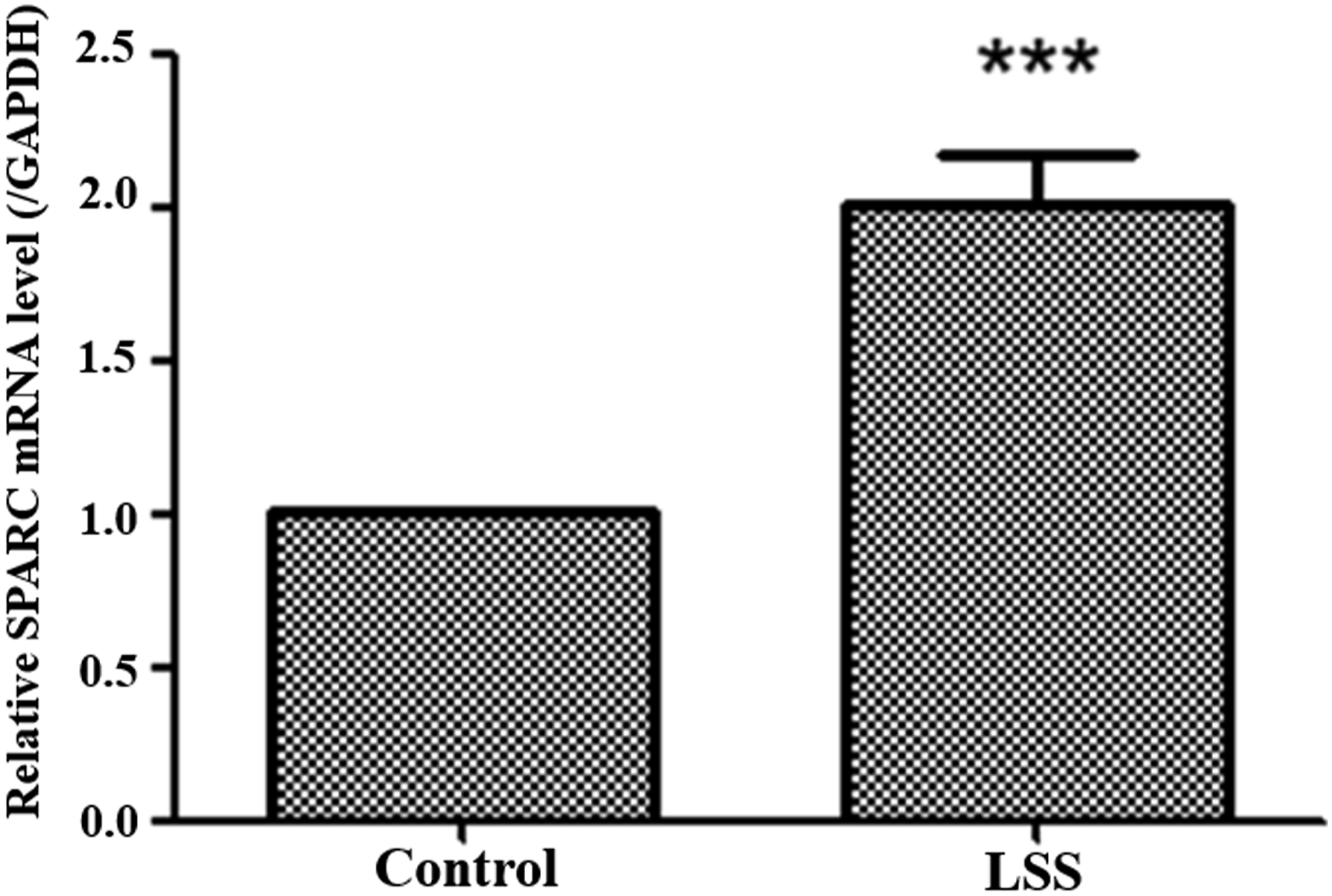
In the immunohistochemical study, SPARC expression was detected in the extracellular matrix of all ligament samples (Figure 1-1), and LF showed significantly more positive staining for collagen and significantly more positive staining with anti-SPARC antibody in the LSS group (Figures 1-2). Features and quantification of collagen fibers and SPARC protein in human LF by immunohistochemical analysis. (A): LF sample showed more collagen fibers and decreased elastin fibers in the LSS group. SPARC could be detected in human LF. (B): LF sample demonstrated significantly more intense staining for collagen and anti-SPARC antibodies in the LSS group than in the control group. Abbreviations: SPARC: secreted protein acidic and rich in cysteine; LF: ligamentum flavum; LSS: lumbar spinal stenosis; *: the difference is significant (P < .05). Comparative qPCR profiling between control and LSS LF samples. Col3, α-SMA, and SPARC mRNA expression were significantly higher in the LSS LF than in the control LF. Abbreviations: SPARC: secreted protein acidic and rich in cysteine; LF: ligamentum flavum; LSS: lumbar spinal stenosis; Col3: collagen III; α-SMA: α smooth muscle actin; *: the difference is significant (P < .05).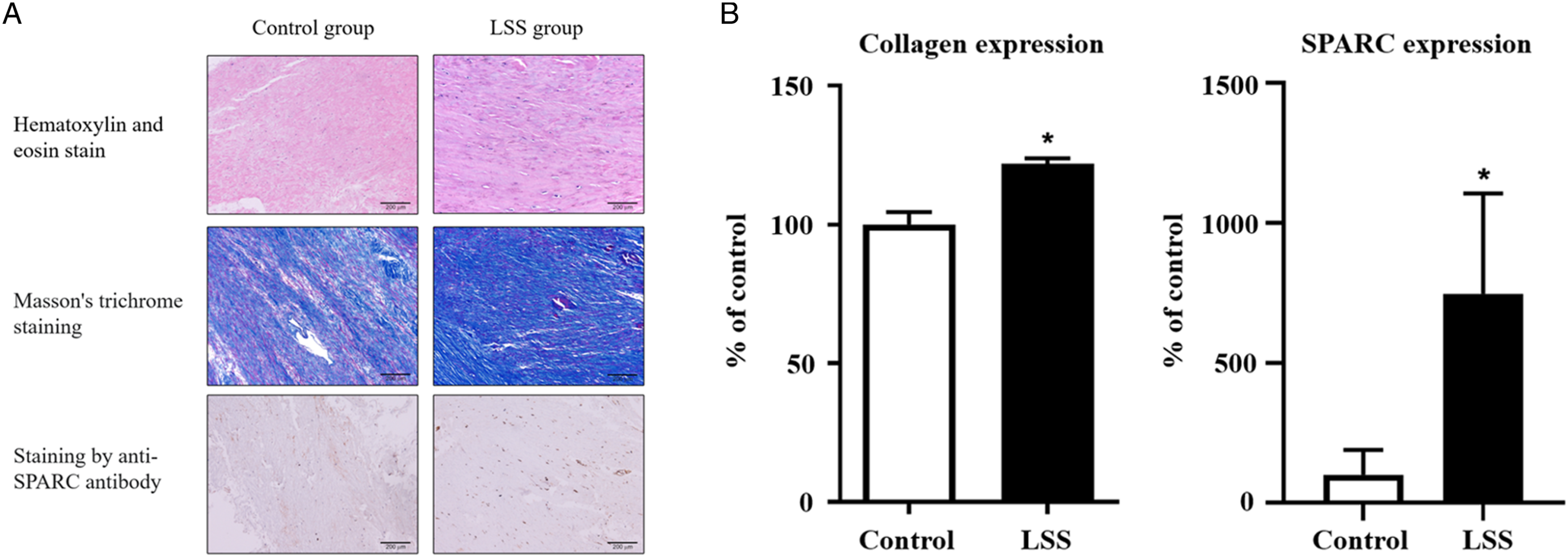
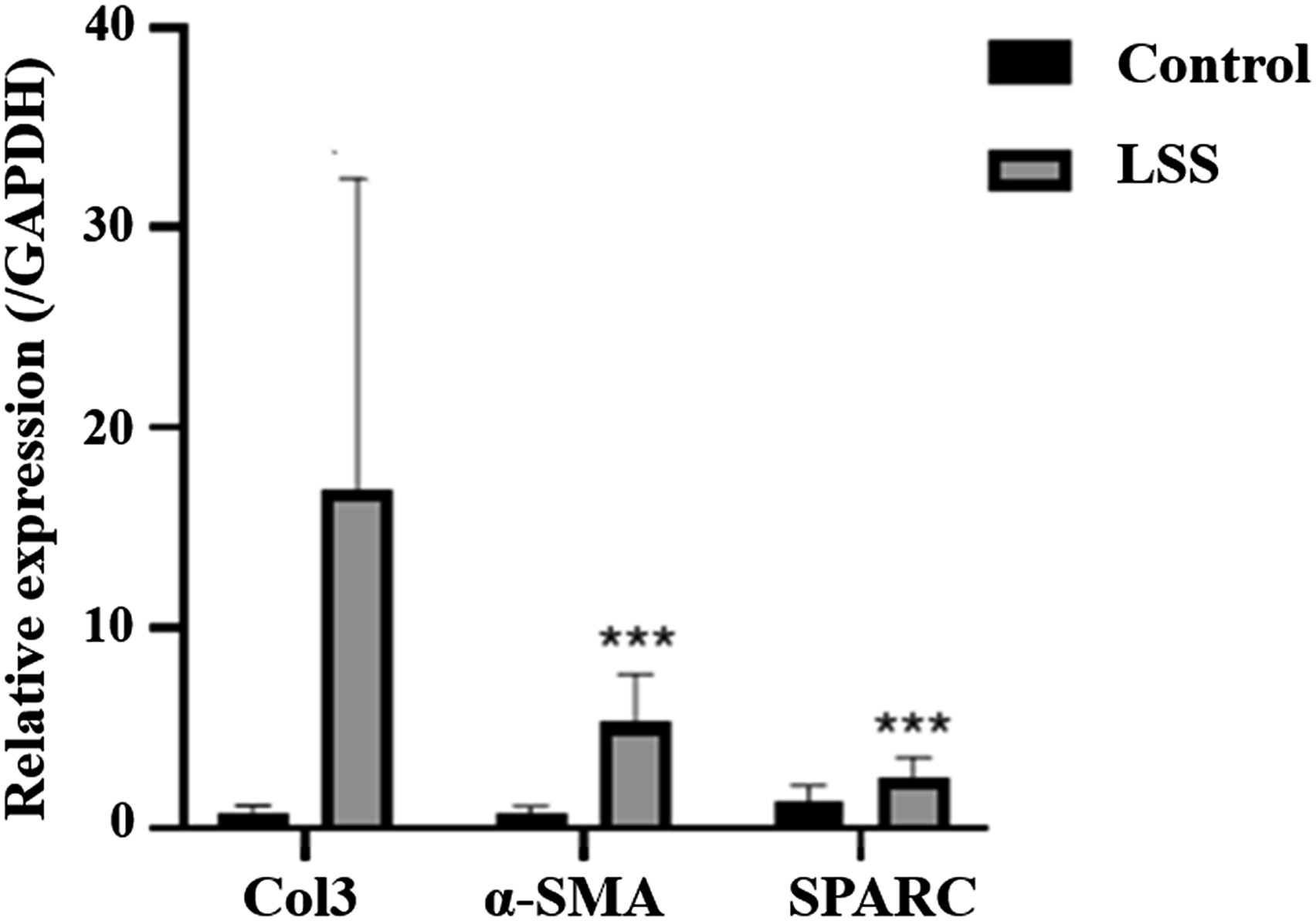
Quantitative polymerase chain reaction (qPCR) was performed to determine mRNA expression of type III collagen (Col3), alpha-smooth muscle actin (α-SMA), and SPARC. Col3, α-SMA, and SPARC mRNA expression was significantly higher in the LSS LF than in the control LF (Figure 2).
SPARC expression was upregulated in the hypertrophic LF cells compared to the non-hypertrophic LF cells
We next evaluated the fibrosis markers between hypertrophic LF cells and non-hypertrophic LF cells. Compared to non-hypertrophic LF cells, hypertrophic LF cells revealed more mRNA expression of Col3 and α-SMA and less mRNA expression of elastin (Figure 3), which indicated that hypertrophic LF cells had more fibrosis process than non-hypertrophic LF cells. Comparison of gene expression of α-SMA, Col3, and elastin between the hypertrophic LF cells and non-hypertrophic LF cells by qPCR. (A, B): The gene expression of α-SMA and Col3 were significantly higher in the hypertrophic LF cells. (C): gene expression of elastin was significantly lower in the hypertrophic LF cells. Abbreviations: LF: ligamentum flavum; Col3: collagen III; α-SMA: α smooth muscle actin; *: the difference is significant (P < .05).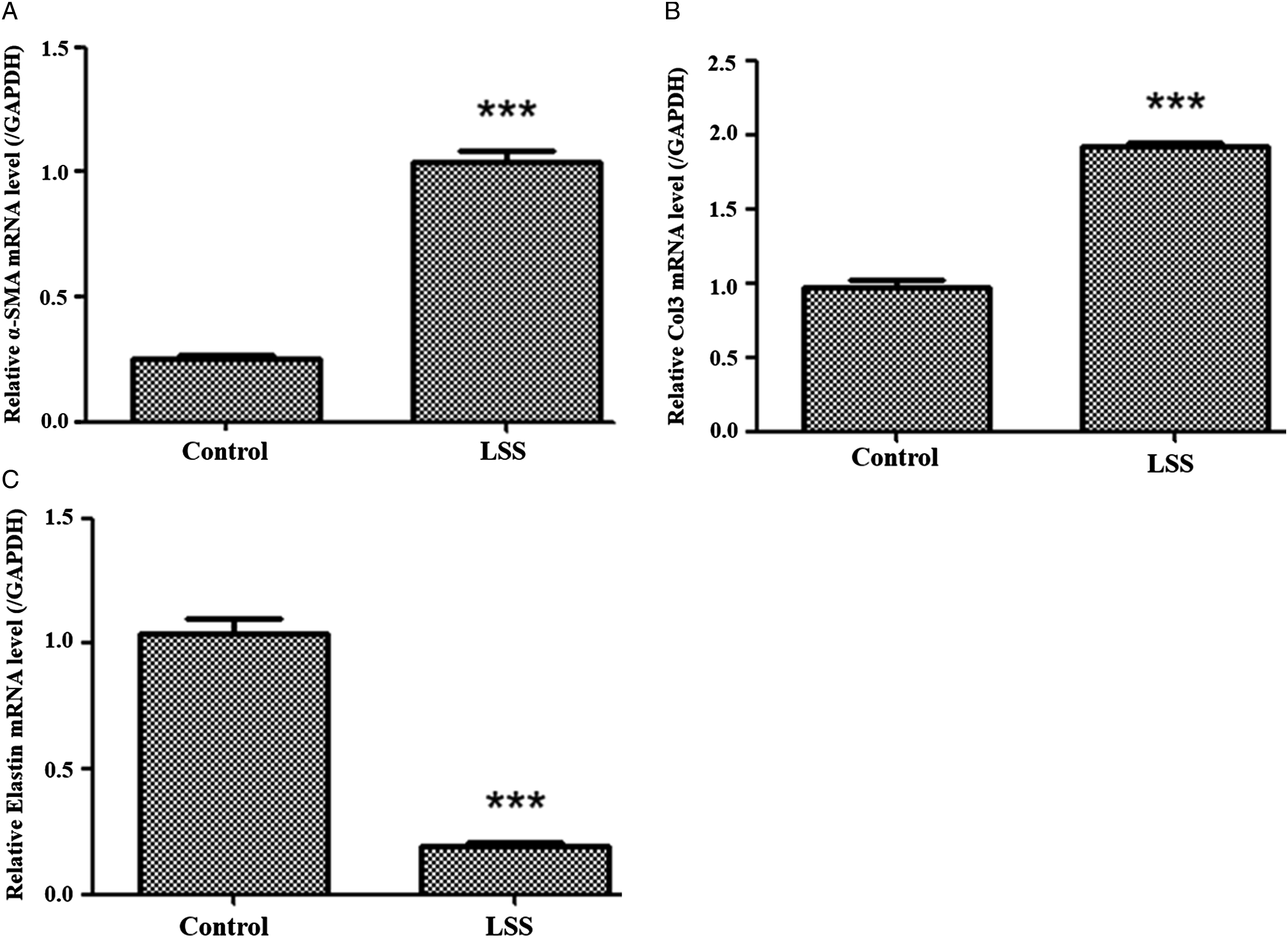
Furthermore, we evaluated the inflammatory process between hypertrophic cells and non-hypertrophic cells. Hypertrophic LF cells possessed higher inflammatory markers such as IL-6, IL-1β, and TGF-β (Figure 4). Meanwhile, we found that the mRNA expression of SPARC was significantly elevated in the fibrotic LF cells (Figure 5). Comparison of gene expression of inflammatory cytokines between the hypertrophic LF cells and non-hypertrophic LF cells by qPCR. (A), (B), and (C): The gene expression of IL-6, IL-1β, and TGF-β were significantly higher in the hypertrophic LF cells. Abbreviations: LF: ligamentum flavum; IL-6: interleukin 6; IL-1β: interleukin 1β; TGF-β: transforming growth factor- β; *: the difference is significant (P < .05). Comparative qPCR SPARC expression analysis between hypertrophic LF cells and non-hypertrophic LF counterparts. The gene expression of SPARC was significantly higher in the hypertrophic LF cells. Abbreviations: SPARC: secreted protein acidic and rich in cysteine; LF: ligamentum flavum; *: the difference is significant (P < .05).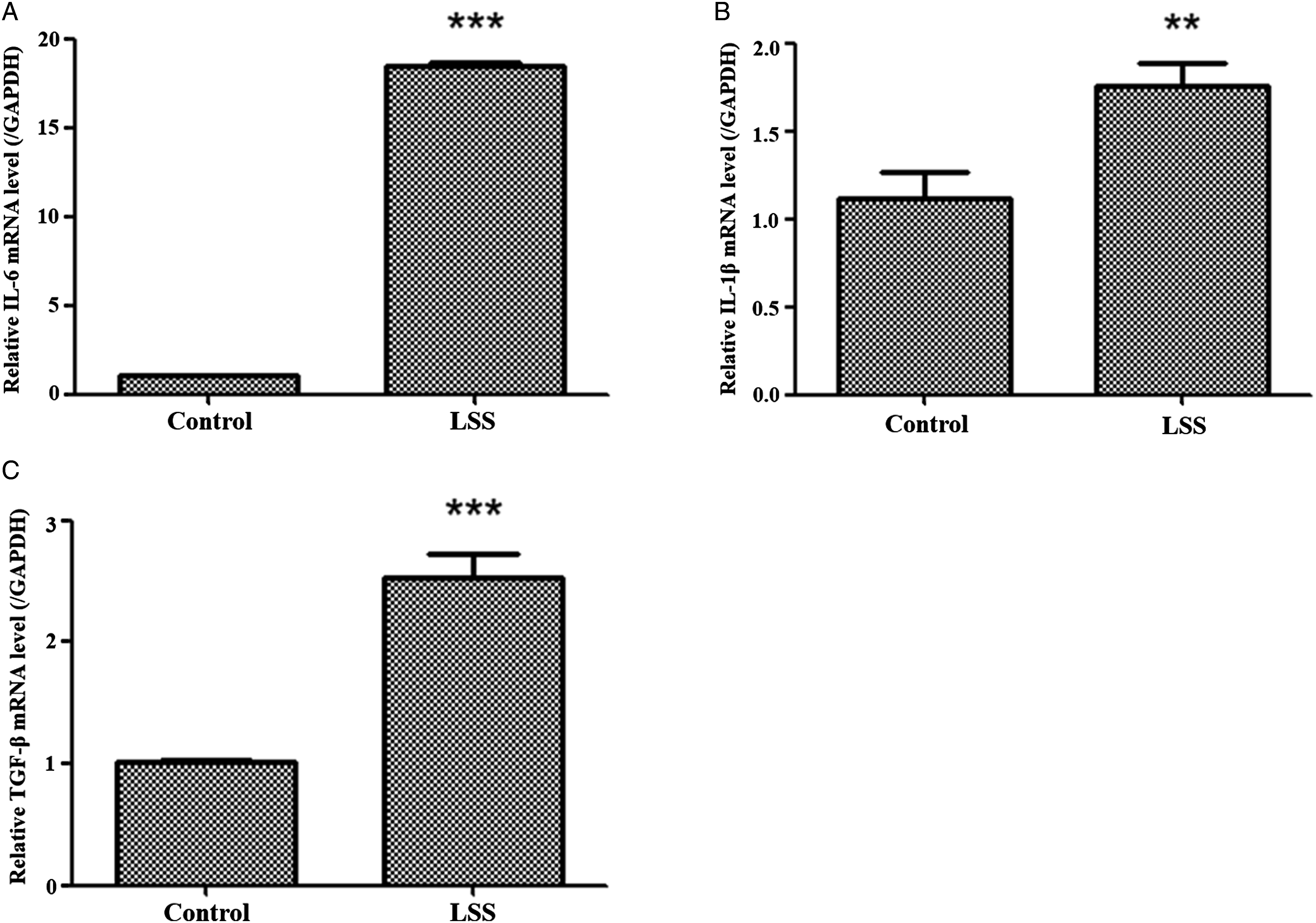
An in vitro fibrosis model of human LF cells was created by stimulation of interleukin 6
The pro-inflammatory cytokine interleukin 6 (IL-6) makes an ideal candidate for the induction of HYPERTROPHIC LF-like biological changes in LF cells or tissues in culture, in which temporal and concentration effects can be explored. The model is usually inexpensive and is very easily manipulated. The ability to expand cells in vitro also means that many replicates are possible, allowing multiple hypotheses to be tested from single tissue sources. We found that IL-6 could induce fibrosis of LF cells obtained in normal and non-hypertrophic LF tissue from patients with HIVD. The expression of collagen I (Col1), Col3, and connective tissue growth factor (CTGF) was significantly elevated after IL-6 was administrated in non-hypertrophic LF cells (Figure 6). The inflammatory cytokine expression, including IL-1β and IL-6, was also upregulated in IL-6-induced fibrosis LF cells (Figure 6). In vitro IL-6-induced fibrosis model of LF cells. The gene expression of SPARC, fibrotic markers (Col1, Col3, and CTGF), and inflammatory cytokines (IL-1β and IL-6) were significantly up-regulated in an in vitro IL-6-induced LF cell fibrosis model. Abbreviations: IL-6: interleukin 6; LF: ligamentum flavum; Col1: collagen I; Col3: collagen III; CTGF: connective tissue growth factor; IL-1β; Interleukin 1β; SPARC: secreted protein acidic and rich in cysteine; *: the difference is significant (P < .05).
SPARC Expression Elevated in the in Vitro Fibrosis Model of Human LF Cells
We investigated the SPARC expression in the fibrosis model of human LF cells. By qPCR, mRNA expression of SPARC was significantly increased in the IL-6-treated human LF cells (Figure 6).
Discussion
The hypertrophy of the LF is a cause of LSS, resulting in the compression of cauda equina or nerve roots. The current study measured LF thickness with a T2-weighted axial MRI. The mean thickness in the LSS group was significantly thicker than that in the control group. The LF specimen showed a high fibrosis score in the LSS group using Masson trichrome staining, more fibrosis markers, such as Col3 and α-SMA, and less elastin expression by qPCR, which was consistent with the previous studies in which hypertrophied LF shows an increase in collagen content and a decrease of elastin to collagen ratio.1,2 Fibrosis has been reported to be the main cause of hypertrophic LF. To realize the nature of hypertrophied LF has highlighted the importance of investigating its pathogenesis. As far as we know, this is the first study to investigate SPARC expression in LF and to illustrate the involvement of SPARC in hypertrophic LF in patients with LSS.
Fibrosis is characterized by excessive extracellular matrix deposition that interferes with normal tissue architecture and function. Increased expression of SPARC in fibrotic diseases has been reported in numerous studies, including pulmonary fibrosis,5,16 Crohn's disease, 17 liver cirrhosis, 8 renal fibrosis,18,19 and scleroderma. 20 Similar to the mentioned-above studies about fibrotic disease, SPARC was detected in human LF, and SPARC expression was upregulated in the hypertrophic and fibrotic LF tissue/ cells compared to non-hypertrophic LF in the current study. In the literature review, there was little research to investigate the relationship between SPARC and ligament. Kurihara and colleagues 21 examined the changes in the expression of SPARC and their proliferative ability according to aging in cultured cells may elucidate characteristic changes in the human periodontal ligament (HPL) induced by aging. Cumulative population doubling levels and cell population doubling time of HPL cells from young donors were greater and shorter than those of HPL cells from senior donors. Levels of SPARC mRNA in HPL cells increased with cellular aging in vivo and in vitro. They concluded that the increase in SPARC with aging may be related to changes in the periodontal ligament's metabolism. Although we made an age-matched selection for the collection of LF specimens, LF hypertrophy was also related to aging. We should investigate the association between LF hypertrophy and aging further. Traweger and colleagues found that loss of SPARC resulted in tendon collagen fibrillogenesis defects in SPARC-/- mice. 22 In 2021, Traweger and colleagues reported a study about load-induced regulation of tendon homeostasis by SPARC. 23 They found tissue hypotrophy and impaired maturation of Achilles tendon enthesis in Sparc−/− mice. Treadmill training revealed a higher prevalence of spontaneous tendon ruptures and a net catabolic adaptation in Sparc−/− mice. Tendon hypoplasia was attenuated in Sparc−/− mice in response to muscle unloading with botulinum toxin A. 23 Although ligaments and tendons are both made up of fibrous connective tissue, both have different functions: Tendons serve to move the bone or structure, and ligaments serve to hold structures together and provide stability for a joint. In Traweger's studies, SPARC is essential for the maturation and maintenance of the Achilles tendon, and lack of SPARC will reduce collagen production, resulting in tendon hypotrophy and tendon rupture. However, the presence of SPARC in ligamentum flavum increases collagen production and decreases elastin production, leading to LF hypertrophy and causing cauda equina compression. Overall, an increase in SPARC expression upregulates collagen formation and results in hypertrophy of fibrous connective tissue.
The current study was the first to establish an in vitro LF fibrosis model with IL-6-induced LF cells. Biologically, many cytokines such as inflammatory cytokines, transforming growth factor β1 (TGF-β1), 24 platelet-derived growth factor-BB (PDGF-BB), 25 and fibroblast growth factor(bFGF) 14 were reported to participate in the pathologic mechanism of LF fibrosis and hypertrophy in a patient with LSS. Previously, several studies showed that progressive LF fibrosis and hypertrophy result from an ongoing chronic inflammatory response. 26 It has been shown that IL-6 plays an essential role in tissue fibrosis as a key player in chronic inflammation. 27 Importantly, a recent study showed that IL-6 was significantly increased in LF tissues, which could elevate collagen expression in LF cells, indicating the critical role of IL-6 in LF degeneration and hypertrophy. 28 In the current study, IL-6-induced fibrosis of LF cells was obtained from the normal and non-hypertrophic LF tissues of patients with HIVD. The expression of Col3 and α-SMA were significantly elevated after IL-6 was administrated in normal LF cells. Therefore, we could investigate the biomolecular expression using the in vitro LF fibrosis model.
In the current study, gene expression of SPARC was increased within in vitro IL-6-induced fibrosis of human LF cells. SPARC is highly expressed as cells establishing the surrounding extracellular matrix (ECM) architecture or tissue remodeling such as wound healing and fibrosis. Associated with fibrosis, SPARC, a collagen-binding protein, has been demonstrated to affect collagen ECM assembly. 6 In a study to investigate the role of SPARC in scleroderma conducted by Zhou, 7 SPARC was overexpressed in the fibroblasts of skin biopsy specimens from patients with scleroderma and overproduction of collagen in scleroderma skin fibroblasts could be attenuated through SPARC silencing. Rentz and colleagues 29 found that the addition of recombinant SPARC reversed collagen I formation in SPARC-null fibroblasts and concluded that SPARC regulated the process of collagen fibrillogenesis in dermal fibroblasts. Taken together, SPARC participated in the fibrosis process of human LF fibroblast-like cells.
In addition to the involvement of SPARC in hypertrophic LF, several studies have shown that reduction or inactivation of the SPARC gene can lead to disc degeneration and low back pain (LBP). In 2004, Gruber and colleagues first reported the detection of SPARC in human intervertebral discs and that SPARC levels were reduced in the disc cells of subjects with aging and disc degeneration. 10 The following year, they published that the radiological features of disc degeneration and histological evidence of disc herniation found in SPARC-null mice were significantly higher than in wild-type mice. 11 In addition, alterations in the disc ECM were associated with accelerated disc degeneration in aging SPARC-null mice. 11 Since 2011, Millecamps and colleagues have shown that SPARC-null mice exhibit signs of LBP behavior caused by disc degeneration.30,31 The intervertebral disc ECM is mainly composed of proteoglycan and collagen. Proteoglycan is responsible for water retention and is entrapped by collagen fibrils. SPARC is a matricellular glycoprotein that modulates interactions between cells and ECM. Based on these studies, deletion of the SPARC gene leads to disc degeneration and LBP through irregular collagen fibrils and reduced proteoglycan aggregates. Therefore, SPARC plays a critical role in the pathogenesis of intervertebral disc degeneration and hypertrophic LF.
There were some limitations in this study. First, cultured LF cells contained various types of cells rather than only fibroblasts, although the microscopic characteristics of LF cells were spindle-shaped. Similar to our findings, many studies have also pointed out that cultured LF cells consisted of various types of cell performance, including chondrocytes, osteoblasts, myofibroblast, and mesenchymal stem cells.32-34 Hence, we should further explore the presence of different characteristics and functions among cultured cell LF. Second, several studies have reported that the thickness of the LF correlates significantly with aging.35,36 Therefore, LF obtained from an elderly patient with a herniated disc is not necessarily non-hypertrophic. Most studies on hypertrophic LF have shown that the mean thickness of the LF in LSS is greater than 4 mm.12,25,37-40 Furthermore, Saito et al defined the thickness of severely hypertrophic LF greater than 4mm and the thickness of non-hypertrophic LF less than 2mm. 12 Therefore, in this study, we defined hypertrophic LF as LF greater than 4 mm in patients with LSS and non-hypertrophic LF as LF less than 2 mm in patients with lumbar disc herniation. Third, the number of patients was small because criteria for collecting LF samples were established to avoid the influence of age, gender, and spine level on LF thickness. Fourth, this study lacked the effect on LF fibrosis without SPARC. We would use Sparc−/− mice for further investigation. Finally, further studies with an animal model of hypertrophic LF are needed to analyze the change in SPARC expression for further investigation and treatment.
In conclusion, Hypertrophic LF was characterized by increased collagen fibers and decreased elastic fiber. Upregulated gene expression of α-SMA, TGF-β, IL-1, and IL-6 was considered as bio-characteristics of hypertrophic LF. SPARC expression was identified in human LF, and its expression was upregulated in the clinical samples of hypertrophic LF compared to their normal counterparts. In addition, we also demonstrated that IL-6 treatment led to an increased level of SPARC in normal LF in vitro. SPARC was crucial in the pathogenesis of hypertrophic LF and could serve as a therapeutic target for LSS.
Footnotes
Author Contributions
Ching-Yu Lee conceived this study, collected the raw data, participated in its design, and drafted the manuscript. Alexander T.H Wu contributed to experimental research and participated in drafting this article. Meng-Huang Wu helped to collect the data and manuscript preparation. Tsung-Yu Huang helped to collect the data. Po-Yao Wang participated in the study design. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Ministry of Science and Technology, Taiwan (grant number: 111-2314-B-038-095) and by Research Project Subsidies for Newly Appointed Faculty Members from Taipei Medical University (grant number TMU110-AE1-B31).
Ethics Approval
Informed consent was obtained from each patient and this protocol was approved by ethics committee of Taipei Medical University (TMU-JIRB No.202004026).
