Abstract
Objective
Design
Single center, retrospective cohort
Methods
Consecutive C2-sacrum PSF patients operated on by 4 surgeons at a single-center from 2015-2020 were reviewed. Demographics, comorbidities, indications, surgical history, and radiographic parameters were collected.
Results
23 patients underwent C2-sacrum PSF. 13 (57%) were male, and 21 (91.3%) were adults. Mean age at time of first spine surgery was 44 years (range 5-71) and 53 years (range 14-72) at the time of C2-sacrum PSF. Six patients (26%) had osteoporosis, and 6 patients (26%) had neurologic comorbidities—including Parkinson’s disease (4), cerebral palsy (1), and Brown Sequard syndrome (1). Four (17%) had connective tissue disease. Two patients underwent C2-sacrum PSF as an index procedure: (1) 67M with myelomatous fractures and 124° of cervicothoracic kyphosis; (2) 28F with severe Marfan syndrome with 140° thoracic scoliosis and 130° thoracic kyphosis. The remaining 21 (91%) underwent C2-sacrum PSF as a revision following prior spinal surgeries on average, 4 previous surgeries (range 1-13) over 10.5 years (range .3-37.4). Indications for the remaining 21 C2-sacrum PSF revision procedures included 17 (81%) for kyphosis (5 of whom also had significant coronal deformity), 1 (5%) for only coronal malalignment, 2 (10%) for instrumentation failure, and 1 (5%) for myelopathy.
Conclusions
91% (21/23) of patients requiring C2-sacrum PSF were treated as revisions of prior fusions, with a mean of 4 prior surgeries over 10 years. Over 80% of these patients underwent C2-sacrum PSF to address kyphosis. 26% had neurologic conditions, and 26% had osteoporosis.
Keywords
Introduction
Spinal deformity burdens both children and adults in myriad forms, including congenital, neuromuscular, adolescent/adult idiopathic, degenerative, and iatrogenic scoliosis.1,2 While nonoperative management is initially prescribed for many patients with spinal deformity, surgical intervention is often needed and can significantly improve clinical outcomes.3-5 Despite advances in surgical techniques, over 1 in 4 patients may require reoperation. 6 The most common mechanical complications, such as proximal junctional kyphosis (PJK), cervicothoracic kyphosis (CT-K), thoracic kyphosis (TK), distal junctional kyphosis (DJK), pseudarthrosis, rod fracture, and implant failure, often require an extension of the prior fusion, either proximally or distally. Among spinal deformity patients undergoing operative intervention, a poorly studied subset of patients requires C2-sacrum instrumented posterior spinal fusion (PSF), a near total spinal column fusion.
Though most deformity surgeons have experience with these constructs, patients with C2-sacrum PSFs are relatively rare; and little is known about this population. Much of the current literature on long-construct fusion focuses on fusion from the thoracic spine to the pelvis.7,8 Several important clinical questions remain regarding the natural history preceding a C2-sacrum PSF as well as patients’ functional status and abilities in the postoperative setting. The current study examines the demographics, comorbidities, and antecedent surgical and clinical histories of patients who have undergone a C2-sacrum PSF. It also explores key radiographic parameters in this population.
Methods
Study Design
This study was a retrospective case series, based on prospectively collected data, conducted from a single institution consisting of multiple spinal surgeons. Following institutional review board approval, spinal deformity patients with C2-sacrum PSFs from 2015-2020 were reviewed.
Patient Population
Enrollment criteria from the prospective ASD database were similar to those of prior studies of ASD patients. 9 Inclusion criteria consisted of all patients, both pediatric and adult, who have undergone a C2-sacrum PSF during the study period and had full-body low-dose stereoradiographs (EOS Imaging, Paris, France) pre- and postoperatively.
Data Collection
To document each patient’s full operative history, several temporal variables were collected before the definitive C2-sacrum PSF. Temporal variables included date of first spine surgery, dates of all subsequent spine surgeries, and date of C2-sacrum PSF. Demographic and comorbidity data included the presence of osteoporosis/osteopenia, neurological diagnoses (Parkinson’s disease, cerebral palsy, Brown Sequard syndrome), Klippel Feil syndrome, and connective tissue diseases (Loeys Deitz, Marfan syndrome, Ehlers Danlos). Smoking status was documented if patients reported smoking at any time after their first spine surgery, including cessation details. The indications for each spine surgery and associated complications were reviewed. When determining the indications for the ultimate C2-sacrum PSF, all spine radiographs following the preceding surgeries were reviewed to determine the presence of any mechanical complications, including PJK, DJK, pseudarthrosis, rod fracture, progressive coronal/sagittal deformity, or additional instrumentation failure, such as screw breakage or pullout.
In addition to a patient’s history, relevant radiographic parameters were recorded. For those who experienced PJK, the proximal junctional angle (PJA) was recorded (when available) immediately following the preceding surgery as well as before revision to C2-sacrum PSF. Kyphosis was likewise measured for those with CT-K, DJK, and TK.
Statistical Analysis
Due to the small sample size, mainly descriptive statistics were utilized. Continuous variables were summarized with mean, standard deviation, and ranges. Count variables were summarized with frequency and total percentage. Differences among patients were evaluated with student’s t-tests. Significance was set at an alpha of 0.05.
Results
Patient Demographics
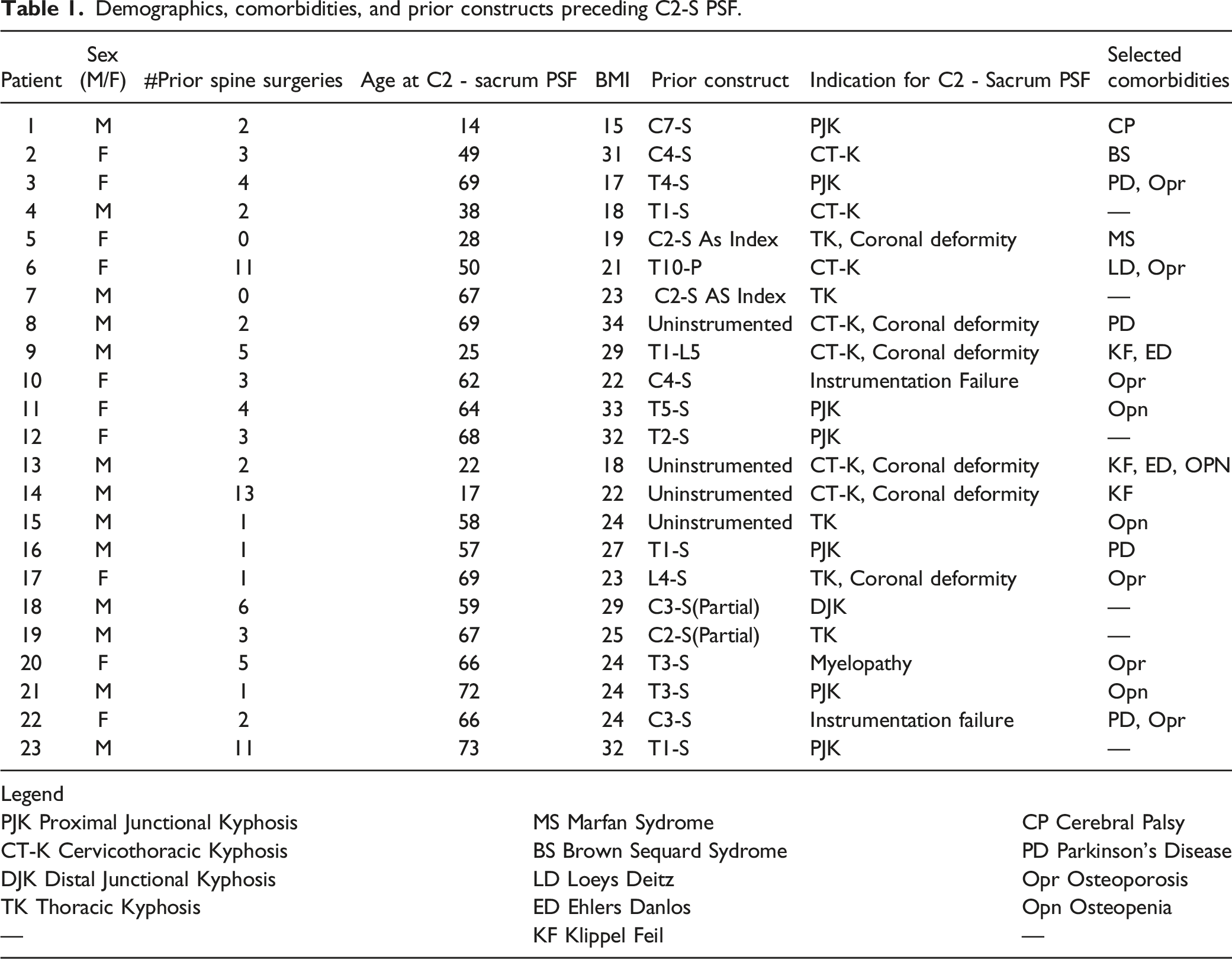
Demographics, comorbidities, and prior constructs preceding C2-S PSF.
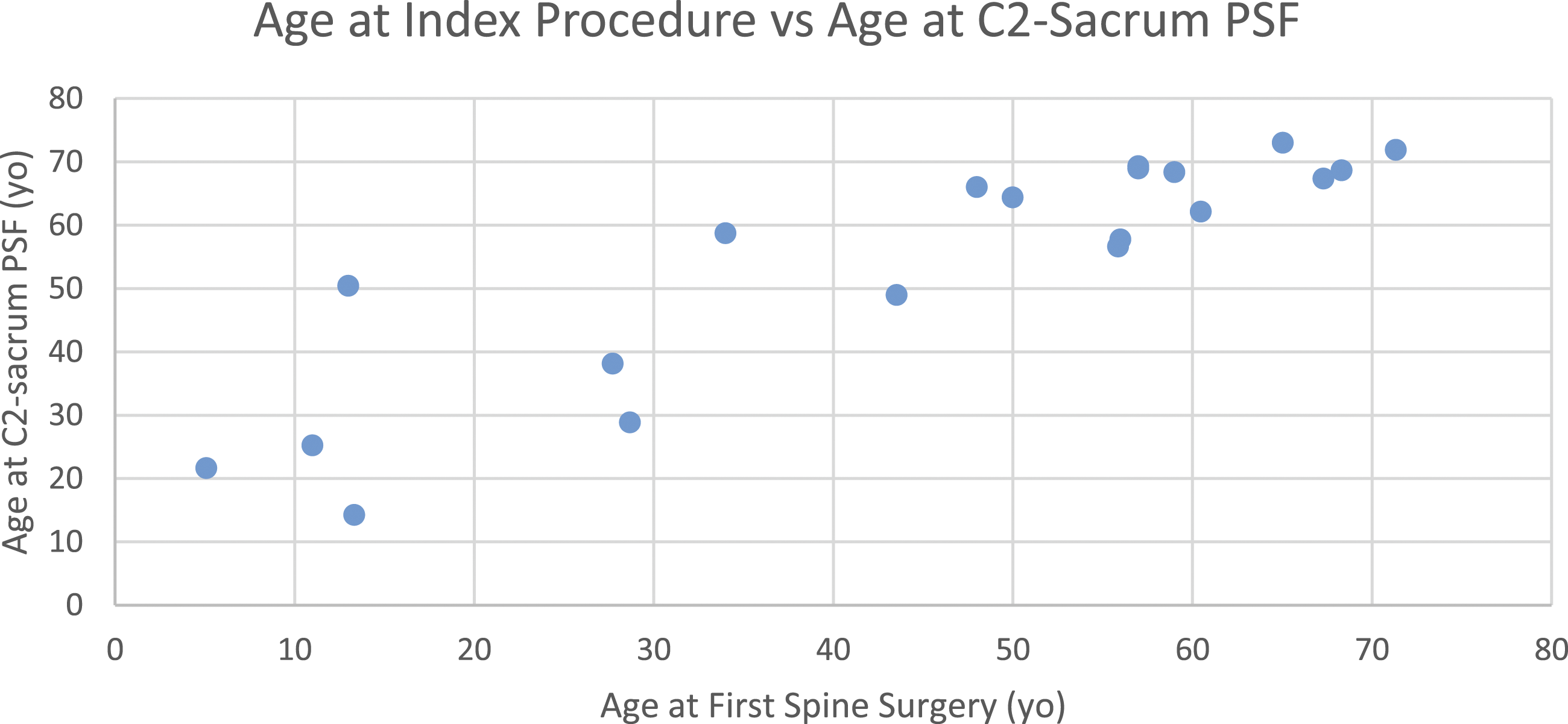
Relationship between age at initial spine surgery and age at C2-sacrum PSF.
Comorbidities
Six patients (26%) had osteoporosis, and 4 (17%) had osteopenia. Six patients (26%) had neurologic comorbidities, including Parkinson’s disease (4), cerebral palsy (1), and Brown Sequard syndrome (1). Four patients (17%) had connective tissue disease (CTD)—two with Ehlers Danlos disease, 1 with Marfan syndrome, and 1 with Loeys-Dietz syndrome. Notably, the 3 patients with Ehlers Danlos and Marfan syndrome all required C2-sacrum PSF in their twenties. Of the patients requiring C2-sacrum PSF before 30 years old, 60% of them had Klippel Feil syndrome. Those with Klippel Feil syndrome were significantly younger than those without it at the time of C2-sacrum PSF (age with Klippel Feil=21 vs age without=58, P < .001). CTD was seen in 60% of patients. Overall, those with CTD were significantly younger at time of C2-sacrum PSF than the other patients undergoing C2-sacrum PSF (age with CTD=32 years vs age without=58 years, P = .008). There was no significant difference in age at time of C2-sacrum PSF among those with neurological comorbidities compared to those who did not have neurological comorbidities (age with neurological comorbidities=55 years vs age without=53 years, P = .810).
Operative History
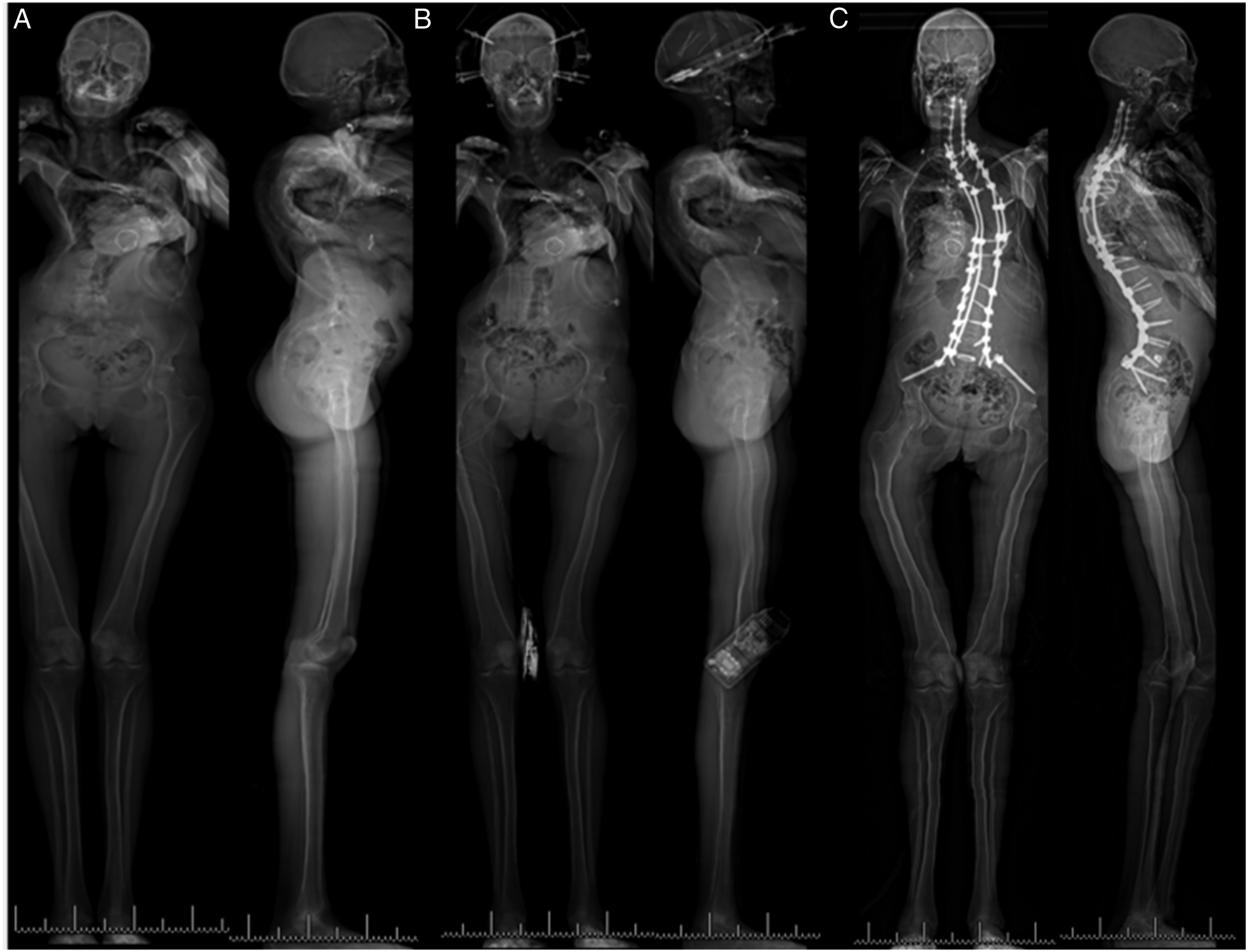
Twenty-one patients (91%) underwent C2-sacrum PSF following complications from prior surgeries—on average, 4 prior surgeries (range 1-13) over 10.5 years (range .3-37.4). Notably, 1 patient had 13 prior procedures: a 17-year-old boy (at the time of C2-sacrum PSF) with Klippel-Feil had 12 prior surgeries for his VEPTR, ultimately producing coronal plane deformity. Nineteen of these patients (81%) underwent C2-sacrum PSF as a revision procedure for kyphosis: 7 (33%) for PJK, 6 (29%) for CT-K, 5 (24%) for TK, and 1 (5%) for DJK. Of the 6 patients with CT-K, half had significant coronal deformity. Of the patients with thoracic kyphosis, 2 (40%) had significant coronal deformity. Just 1 patient (5%) had coronal malalignment as the main reason for revision, and another 2 (10%) had implant failure. Of those with implant failure, 1 had a loss of fixation from pullout of cervical spine lateral mass screws, leading to a positive sagittal balance (Figure 2). Similarly, the other patient with implant failure had lateral mass screw pullout at C3-C5, 4 months after a revision C3-sacrum surgery. One patient (5%) had myelopathy: a 67-year-old female status post T3-sacrum PSF 1-year prior developed myelopathy, with hyperreflexia, impaired tandem gait, and left upper extremity pain. AP and lateral radiographs of a 62-year-old woman who presented with screw pullout. Preoperatively, she had a C4-sacrum PSF (done for PJK following a prior surgery), with cervical spine screw pullout at multiple levels: a. | She underwent revision with C2-sacrum PSF: b.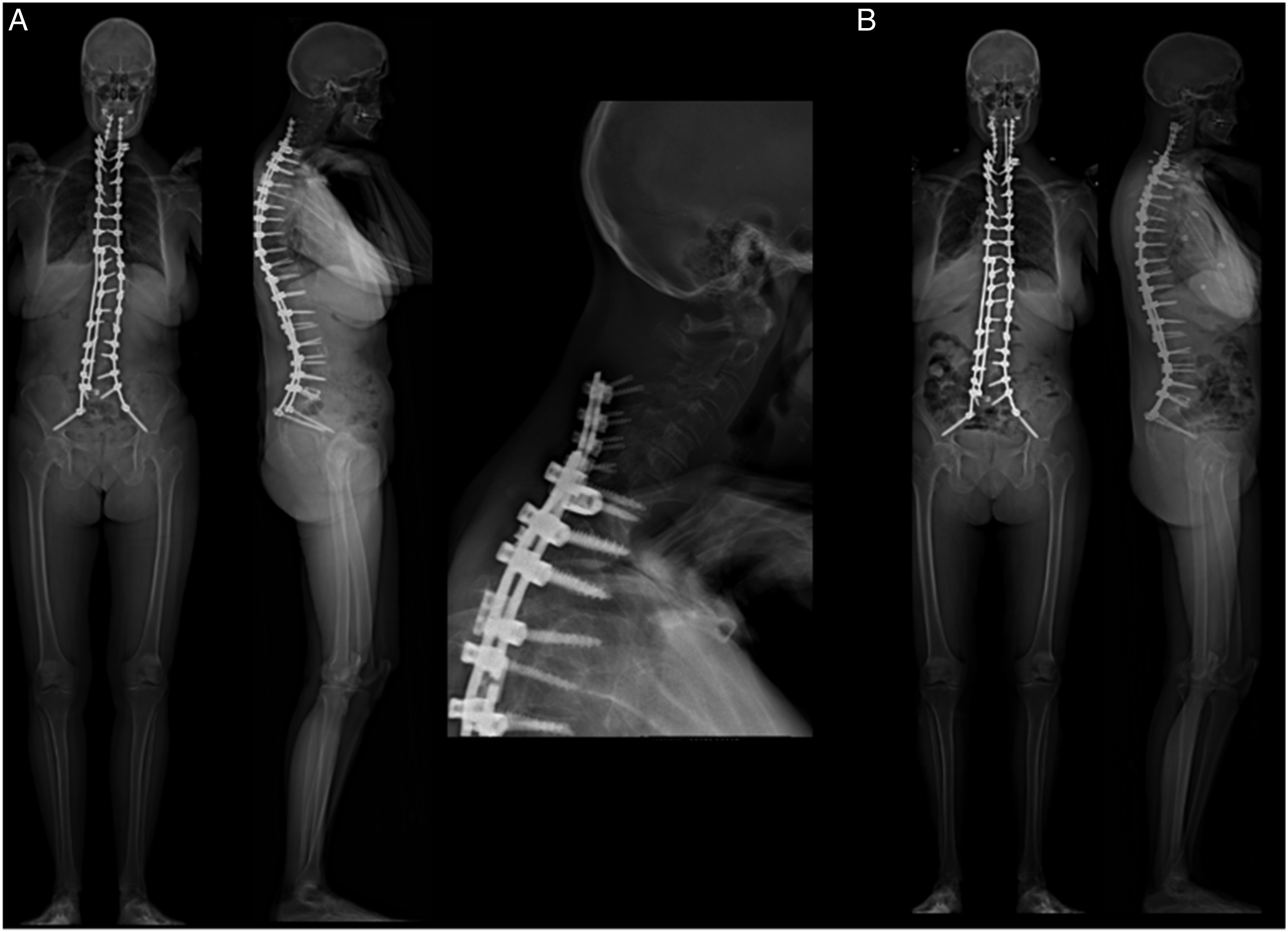
The remaining 2 patients (9%) underwent C2-sacrum PSF as their index procedure. One patient, a 67-year-old male with multiple myeloma, had sustained contiguous pathologic compression fractures, resulting in 124° of cervicothoracic kyphosis. He underwent C2-sacrum PSF in a single stage (Figure 3). The other patient was a 28-year-old female with restrictive lung disease and Marfan’s that resulted in severe scoliotic spinal deformity. She had 140° of thoracic scoliosis and over 130° of thoracic kyphosis, requiring a staged procedure. After halo placement, she required C6-sacrum PSF before the second stage of C2-sacrum PSF (Figure 4). Anteroposterior and lateral radiographs of a 67-year-old man who required C2-sacrum PSF for pathologic compression fractures. Preoperative radiographs at presentation with 124° of TK: a | Preoperative CT scan: b | Radiographs after halo traction: c | Radiographs after C2-sacrum PSF, with 67° of TK: d. Anteroposterior and lateral radiographs of a 28-year-old woman with severe thoracic kyphoscoliosis and restrictive lung disease, who required C2-sacrum PSF. Preoperative radiographs demonstrate over 140° of thoracic scoliosis and over 130° of thoracic kyphosis: a. | Prior to her first stage, she was placed in 20 pounds of halo gravity traction for 3 weeks: b. | In 2 stages, she underwent a C2-sacrum PSF: c. In the first stage, she underwent C6-sacrum instrumentation with L5/S1 TLIF. In the second stage, she underwent a two-level VCR (T8/9) and instrumentation up to C2.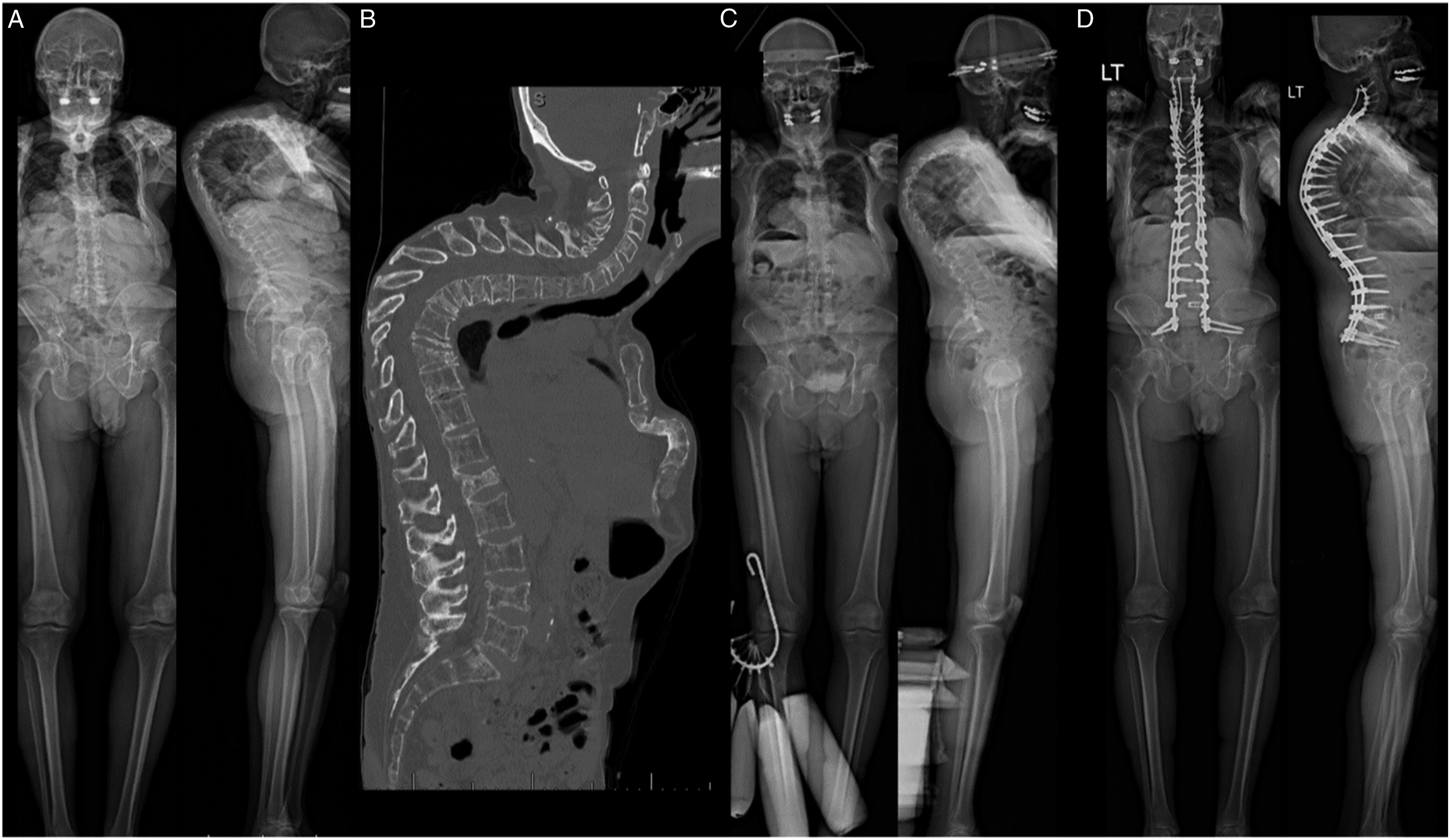
Congenital Scoliosis
In our sample cohort, there were 3 patients (patients 9, 13, and 14 in Table 1) with congenital scoliosis. All had Klippel Feil syndrome. These 3 patients were significantly younger at the time of their C2-S PSF procedure than the remainder of the cohort (mean age 21 with congenital scoliosis vs mean age 58, without). Two of the patients required C2-S PSF for cervicothoracic kyphosis. All 3 had a significant coronal deformity; 1 patient’s main indication for surgery was to address the coronal deformity (the only such patient in our cohort).
Patient 9 was a 25-year-old male with a history of several prior surgeries. His prior construct was a T1-L5 construct. At the time of his presentation, he had lumbosacral pain as well as coronal imbalance resulting in head tilt. Patient 13 was a 21-year-old male with severe cervicothoracic deformity. He required C2-S PSF in 2 stages, with the second stage involving a VCR to correct his severe ear on shoulder deformity. Patient 14 was a 17-year-old male with Klippel Feil syndrome whose congenital scoliosis required multiple prior VEPTR rod placements and lengthenings. He decompensated in the coronal plane, leaning towards the right with his ribs below his pelvis. After several weeks of halo gravity traction, he too underwent C2-S PSF.
Radiographic Parameters
Proximal junctional angles in those with PJK before C2-S PSF.
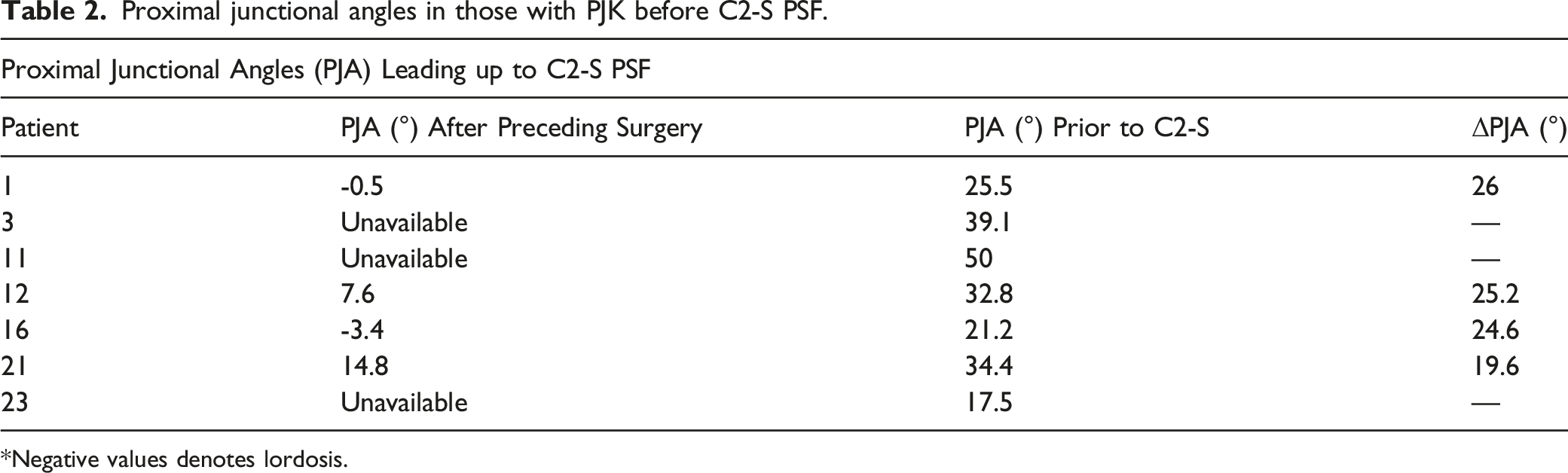
*Negative values denotes lordosis.
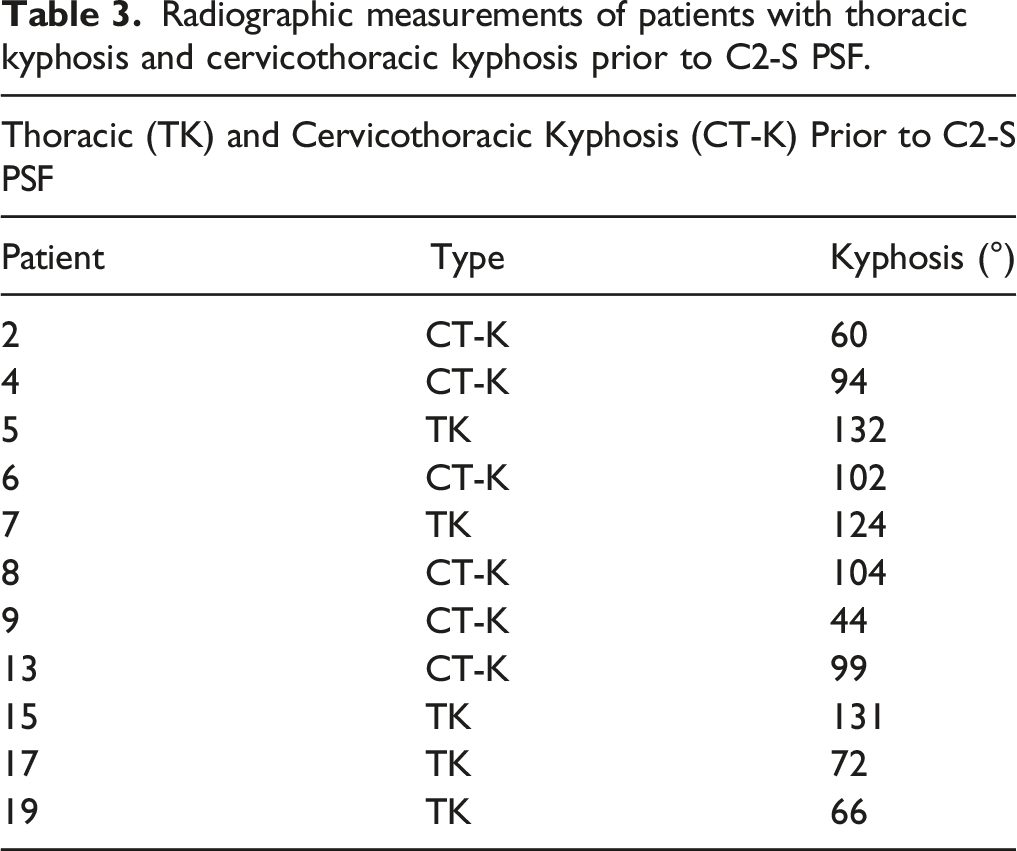
Radiographic measurements of patients with thoracic kyphosis and cervicothoracic kyphosis prior to C2-S PSF.
Illustrative Cases of Patients Revised to C2-Sacrum PSF for Kyphosis
Seven patients required a revision to C2-sacrum PSF for PJK. One representative case is illustrated in Figure 5. A 69-year-old female presented to our institution following an extensive history of procedures at an outside hospital. 12 years prior, she had an L5-S1 PSF. However, this was complicated by adjacent segment degeneration, which required revision to L3-S1 PSF. This, in turn, was complicated by pseudarthrosis, adjacent segment degeneration, and severe stenosis, requiring revision from T11-S1, along with ALIF L2/3 and L3/4. Six weeks after this procedure (1 year prior to C2-sacrum PSF), she sustained a burst fracture with stenosis at T11, requiring revision T4-T11 PSF with segmental instrumentation to the previous construct. This construct was then complicated by PJK. At this time, the patient presented to our institution, where she underwent revision with a C2-sacrum PSF procedure. Anteroposterior and lateral radiographs of a “typical” patient who required revision to C2-sacrum PSF following multiple prior spine procedures, most recently T4-S1 that was complicated by PJK. Preoperative radiographs, with 96° of TK and a proximal junctional angle of approximately 40°: a | Preoperative CT demonstrating PJK: b | Postoperative radiographs, with 40° of TK: c.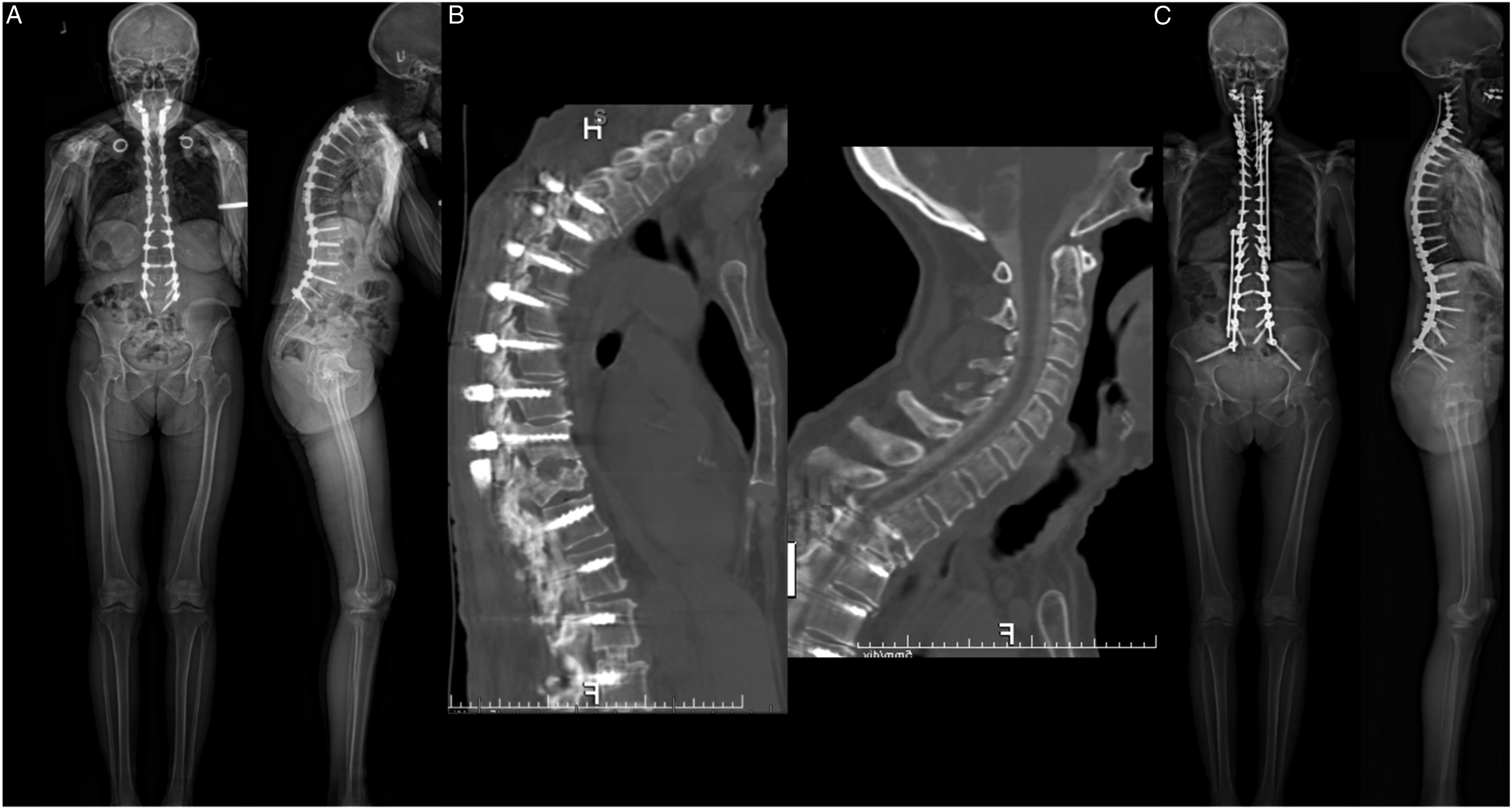
Similarly, several patients experienced CT-K, requiring revision to C2-sacrum PSF. One such patient was a 68-year-old male with a history of Parkinson’s disease and 2 prior surgeries: 11 years prior, he underwent a lumbar decompression; 4 years prior, he underwent an ACDF C5-C7. Over time, he developed a neurogenic sagittal and coronal deformity (Figure 6). He had an approximately 50° right lumbar curve and over 100° of cervicothoracic kyphosis from C2 to T12. His sagittal imbalance was over 20cm. Though he was a practicing lawyer, he was severely debilitated by this deformity, unable to walk for more than 10 minutes without pain. Having exhausted nonoperative management, he underwent C2-sacrum PSF. AP and lateral radiographs of a 68-year-old male, who presented with approximately 104° of cervicothoracic kyphosis and near chin-on-chest deformity, preoperatively: a. | C2-sacrum PSF was performed with multiple interbody fusions: b.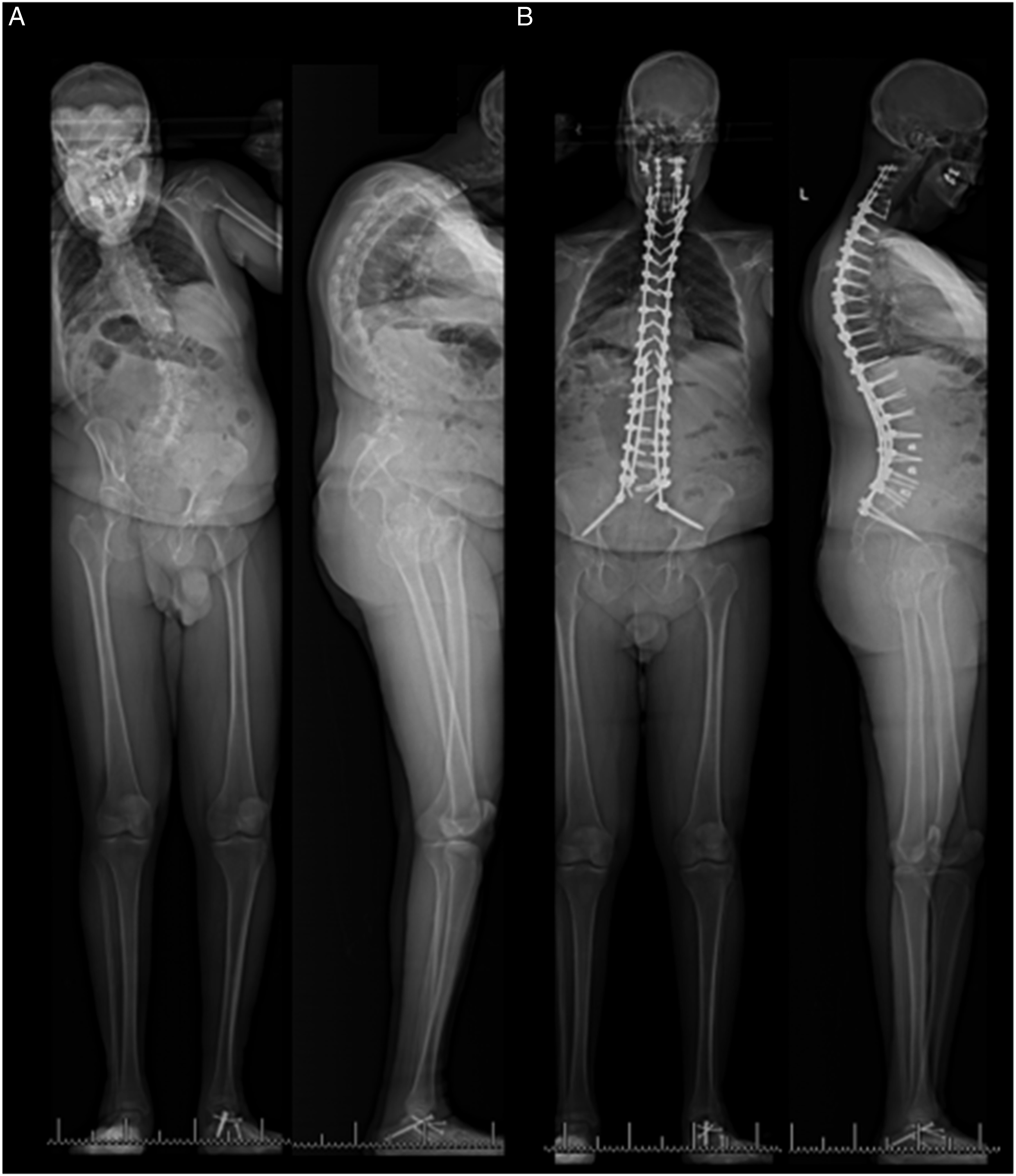
Discussion
The current single-center case series describes the largest cohort to date of patients who underwent a C2-sacrum PSF. The mean age was 53 years, and patients had an average of 4 prior surgeries over a mean of 10.5 years. Comorbidities were common and included osteoporosis (26%), neurological conditions (26%), and CTD (17%). Over 90% of patients undergoing C2-sacrum PSF underwent prior spine surgery, and the most common indications among this group for C2-sacrum PSF were kyphosis (81%), and instrumentation failure (10%). Two patients had a C2-sacrum PSF as index procedures to address severe deformity.
Notable Comorbidities
While the ultimate reason for C2-sacrum PSF is multi-factorial, certain comorbidities likely played an integral role in the failure of prior surgeries. In particular, osteoporosis, CTD, Parkinson’s disease, and Klippel Feil syndrome were common in our sample population, especially among those undergoing revision procedures.
Osteoporosis was present in over a quarter of our sample. Both patients who had lateral mass screw pullout (comprising all instrumentation failures in our sample) also had osteoporosis—which may warn against relying on lateral mass screws in long constructs like C2-sacrum PSF for osteoporotic patients. This is consistent with prior recommendations for longer fusions to avoid proximal failure in osteoporotic patients. 10 Moreover, only one third of osteoporotic ASD patients are treated for their underlying osteoporosis, most commonly with bisphosphonates or teriparatide, 11 underscoring the need for a holistic approach to these patients that incorporates appropriate perioperative pharmacologic management in addition to meticulous surgical planning and technique.
Given the close relationship between neuromuscular comorbidities, degeneration, and spinal deformity, it is unsurprising that this cohort had a high frequency (17%) of Parkinson’s disease. Bourghli et al reviewed a series of 12 patients with Parkinson’s who required T2-pelvis PSF, either as revision (5 patients) or index (7 patients) procedures; 6 required revision after T2-pelvis PSF. Two of these patients had PJK, and 3 had instrumentation failure (rod fracture). Of note, only 1 patient with PJK required C2-sacrum PSF, highlighting the rarity of C2-sacrum PSF patients. The findings in this review, and in our sample, have been substantiated by more recent findings that patients with Parkinson’s undergoing deformity correction are more likely to experience complications including nonunion and loss of correction, 12 prompting some to call for longer-fusion constructs and additional rods to prevent these complications. 13 In addition to the higher prevalence of osteoporosis in this population, changes in muscle characteristics and tone 14 may have adverse effects on posture and spinal alignment. The advent of novel therapeutics including deep brain stimulation may mitigate the risks of progressive deformity, surgical complication, and, ultimately, the need for revision surgery. 15 Nevertheless, operative candidates with PD and spinal deformity must be followed closely and counseled that their condition predisposes them to increased risks of surgical complication, requiring longer-construct fusions, potentially a C2-sacrum PSF, with modifications in surgical technique.
One in 5 patients in our sample had CTD, which, like neurologic comorbidities, may contribute to poor muscle tone and postural reasons for continued deformity. On average, our patients with CTD were 26 years younger at time of C2-sacrum PSF than those without CTD. McMaster reviewed 5 patients with Ehlers Danlos who had spinal deformity first recognized before 4 years of age. Though attempts at casting were made in 3 patients, all required posterior fusion, at a mean age of 11 years, for severe deformity. 16 Similarly, Akpinar et al 17 reviewed 5 patients with Ehlers Danlos who required thoracolumbar corrective fusion and highlighted the surgical risks inherent to spinal fusion in this population earmarked by vascular fragility. Notably, neither of these series involved patients with fusion to the cervical spine. Patients with Marfan syndrome likewise have an increased rate of neurologic complications, postoperative infection, and curve decompensation 18 —perhaps due to underlying issues such as dural ectasia, as well as altered soft tissue and bone morphology. These physiologic variations in patients with CTD underscore the importance of extensive preoperative workups to assess for neurologic, bone density, and other abnormalities.
Similarly, those with Klippel Feil syndrome in our sample were nearly 40 years younger at time of C2-sacrum PSF than the remainder of the cohort. While patients in our sample underwent C2-sacrum PSF at a mean of 53 years, over 1 in 5 required the procedure before 30 years old. Of those who had the procedure before 30, over half also had Klippel Feil syndrome. Hensinger et al 19 reviewed patients with Klippel Feil and found that 60% of patients had significant scoliosis and that 75% of those patients required posterior fusion to address the deformity. Their findings likewise indicated that patients with Klippel Feil demonstrate deformity at an earlier age than those without the syndrome. The congenital fusion of multiple cervical and thoracic vertebra, combined with adjacent level degenerative changes and other congenital aberrations such as hemivertebrae, likely serve as deforming, unnatural forces on the spine. Two of the 3 patients in this sample with Klippel Feil also had Ehlers Danlos syndrome, complicating these patient profiles with the aforementioned risks of CTD. Thus, the constellation of patient features, including neurologic, cardiovascular, and ocular, among other abnormalities, 20 demands vigilance and multidisciplinary management.
Focused Literature Review on C2-Sacrum PSF
Studies of patients with C2-sacrum PSF are few, and our cohort bears similarities to prior series of these patients. One prior study by Iyer et al investigated the outcomes of 55 patients with fusions from the cervical spine to the pelvis, though UIVs ranged from C2 to C7. 21 The most common UIV in their study was C2, with 16 patients undergoing fusion at that level. The most common indication for fusion from the cervical spine to pelvis was PJK. Apart from select case reports, the remaining literature focuses on patients with long fusion constructs with a UIV in the cervical spine or the upper thoracic spine. Kelly et al described a patient undergoing C2-sacrum PSF for deformity due to osteoporotic vertebral compression fractures. 22 Chen et al 23 reported on the case of a multiply-operated patient who initially underwent PSF for idiopathic scoliosis, which was complicated by PJK at multiple levels, ultimately necessitating C2-sacrum PSF. Thus, it is our hope that the current study helps fill the empirical vacuum on C2-sacrum PSF patients.
Limitations and Future Directions
The current study is not without limitation. The limited size of this study may affect the ability to extrapolate the results to all patients undergoing C2-sacrum PSF. However, the limited size also reflects the infrequency of this intervention. Moreover, the retrospective nature of this study complicates attempts to draw causal relationships between patient demographics, comorbidities, and outcome metrics.
Future studies are needed on this population of patients undergoing C2-sacrum PSF, mainly with control groups. Though the current case series sheds light on a rarely studied patient population, risk factors cannot be studied in the absence of a control group. A case-control study would be ideal to elucidate predictors of revision to a C2-sacrum PSF. Additionally, examining clinical outcomes and complications in this population will greatly aid patient counseling for those undergoing long fusions, more generally. Furthermore, our study was limited by the number of surgeons performing this procedure at a single institution. Future studies with a larger number of surgeons, from multiple centers, would make these results more generalizable. Moreover, as many of these patients arrived from other institutions and providers, detailed information regarding prior operations at outside institutions and corresponding clinical courses was somewhat limited. Finally, this study does not cover patient courses after C2-sacrum PSF, which would offer additional data on the outcomes of these individuals following this long fusion construct.
Conclusions
Most patients requiring C2-sacrum PSF were treated as revisions of prior fusions due to sagittal malalignment, with a mean of 4 prior surgeries over 10 years. Almost all patients required C2-sacrum PSF after a previous surgery (90%), though 2 patients (10%) underwent C2-sacrum PSF as the index procedure. A total of 26% had neurologic conditions, and 26% had osteoporosis. Revision to C2-sacrum PSF was most often for kyphosis (81%), followed by implant failure (10%).
Supplemental Material
Supplemental Material - What Radiographic and Clinical Factors Ultimately Necessitate a C2-Sacrum Instrumented Posterior Spinal Fusion?
Supplemental Material for What Radiographic and Clinical Factors Ultimately Necessitate a C2-Sacrum Instrumented Posterior Spinal Fusion? by Justin Mathew, Scott L Zuckerman, Hannah Lin, Matthew Simhon, Gerard Marciano, Meghan Cerpa, Nathan J Lee, Venkat Boddapat, Ronald A Lehman, Zeeshan M Sardar, Marc D Dyrszka, Joseph M Lombardi, and Lawrence G Lenke by Global Spine Journal
Supplemental Material
Supplemental Material - What Radiographic and Clinical Factors Ultimately Necessitate a C2-Sacrum Instrumented Posterior Spinal Fusion?
Supplemental Material for What Radiographic and Clinical Factors Ultimately Necessitate a C2-Sacrum Instrumented Posterior Spinal Fusion? by Justin Mathew, Scott L Zuckerman, Hannah Lin, Matthew Simhon, Gerard Marciano, Meghan Cerpa, Nathan J Lee, Venkat Boddapat, Ronald A Lehman, Zeeshan M Sardar, Marc D Dyrszka, Joseph M Lombardi, and Lawrence G Lenke by Global Spine Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Compliance with Ethical Standards
Columbia University Institutional Review Board Approved Protocol # AAAT2038 for HIPAA form B, waiver for Informed Consent.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
