Abstract
Study Design
Retrospective cohort study.
Objectives
This study aimed to examine whether the use of intravenous TXA in elective spine surgery is associated with reduced perioperative massive hemorrhage requiring transfusion.
Methods
We extracted all patients who underwent decompression with or without fusion surgery for the cervical, thoracic, and lumbar spine between April 2012 and March 2019. The primary outcome was the occurrence of massive hemorrhage requiring transfusion, defined as at least 560 mL of blood transfusion within 2 days of spine surgery or the requirement of additional blood transfusion from 3-7 days postoperatively. Secondary outcomes were the occurrence of thrombotic complications (pulmonary embolism, acute coronary syndrome, and stroke) and postoperative hematoma requiring additional surgery.
Results
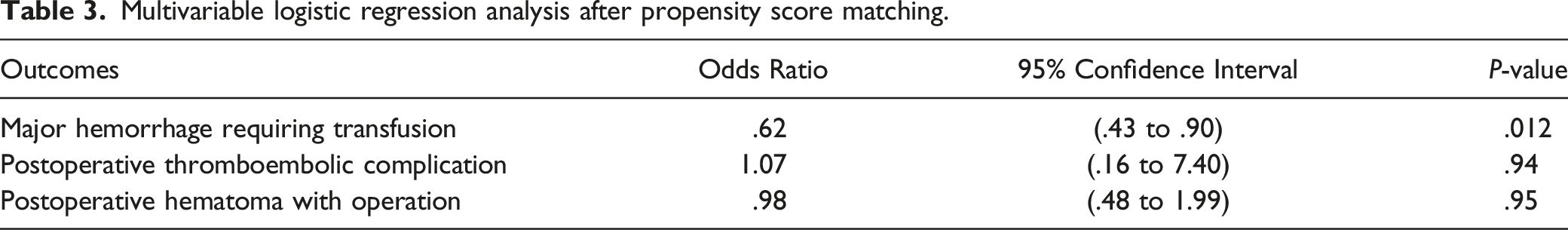
We identified 83,821 eligible patients, with 9747 (12%) patients in the TXA group. Overall, massive hemorrhage requiring transfusion occurred in 781 (.9%) patients. Propensity score matching yielded 8394 pairs. In the matched cohort, the TXA group had a lower proportion of massive hemorrhage requiring transfusion than the control group (.7% vs 1.1%; P = .002). There was no significant difference in the occurrence of thrombotic complications and postoperative hematoma requiring additional surgery between both groups. The multivariable regression analysis also showed that the use of TXA was associated with significantly lower proportions of massive hemorrhage requiring transfusion (odds ratio, .62; 95% confidence interval, .43-.90; P = .012).
Conclusions
In this analysis using real-world data, TXA use in elective spinal surgery was associated with reduced perioperative massive hemorrhage requiring transfusion without increasing thrombotic complications.
Level of evidence
Prognostic Level Ⅲ
Keywords
Introduction
Reducing intraoperative blood loss is one of the most important research topics in spine surgery. It often requires blood transfusions due to perioperative blood loss.1-3 Previous studies have shown that blood transfusion is associated with poor outcomes, including in-hospital mortality, morbidity, and postoperative complications.1,4-6 To reduce perioperative blood loss and blood transfusions, the intraoperative administration of antifibrinolytic agents, including tranexamic acid (TXA), has been widely used.
TXA, a synthetic derivative of the amino acid lysine, is an antifibrinolytic agent that binds to plasminogen and blocks the interaction of plasmin (ogen) with fibrin. 7 Accordingly, TXA prevents the dissolution of blood clots and reduces bleeding. TXA is being increasingly used during surgery because of its hemostatic effect. According to a recent Canadian study, TXA is used with varying frequencies among non-cardiac surgical procedures, with a rate of 18% in spinal fusion surgery. 8 Additionally, a recent randomized controlled trial involving patients undergoing non-cardiac surgery showed reduced incidence of the composite bleeding outcome with tranexamic acid. 9
Although several small randomized controlled trials (RCTs) have been conducted to examine the efficacy of TXA in various spinal procedures, the reported efficacy of TXA in reducing perioperative blood loss is inconsistent.10-16 Despite meta-analyses suggesting the usefulness of TXA in spine surgery,17-19 it remains unclear whether the available evidence on its efficacy from relatively small trials can be directly extrapolated to real-world clinical settings. Moreover, the available evidence is inconclusive regarding the lingering concern of a potential increase in thrombotic complications, mainly because of the small sample size of prior trials. This study aimed to evaluate the effectiveness and safety of intravenous TXA administration in elective spine surgery using a Japanese national inpatient database.
Materials and Methods
This was a retrospective cohort study using the Japanese Diagnosis Procedure Combination database, a national administrative claims database, and discharge data. 20 All academic hospitals were obliged to participate in the database, and over 1000 community hospitals voluntarily contributed to the database. Overall, approximately 50% of all acute-care inpatients in Japan were provided. The database contains encrypted unique identifiers; age and sex; body weight and height; admission and discharge dates; smoking history; institution type; diagnoses coded according to the International Classification of Diseases (ICD) 10th revision; surgical and nonsurgical procedures coded according to Japanese original codes; duration of anesthesia; drugs prescribed; and discharge status. A previous study has shown that the validity of diagnoses and procedure records in the database was high as the sensitivity and specificity of the primary diagnoses were 78.9% and 93.2%, respectively. 21 The database clearly differentiates between comorbidities that were already present at admission and complications that occurred after admission; many studies using the database have been reported elsewhere.22-24 Because all data were de-identified, the requirement for patient informed consent was waived.
From April 2012 to March 2019, we extracted all patients who underwent elective spine surgery with or without 1000 mg of TXA during hospitalization (Figure 1). To ensure the generalizability of spine surgery, we included decompression with or without fusion surgery for the cervical, thoracic, and lumbar spine, and we excluded minimal invasive surgery and long fusion such as corrective fusion surgery. Patients who received intravenous TXA on the day of surgery were defined as the TXA group. Because we could not precisely identify whether the one-shot topical use of TXA was administered intravenously or topically, we excluded patients who were administered less than 1000 mg from the TXA group. We further excluded patients who (i) underwent spine surgery with a main diagnosis of hematoma (ICD-10 code: S141, S241, S341, and T093), infectious disease (tuberculosis A180, B902, osteomyelitis/spondylitis/discitis M46), tumor (C412, C795, D166, and M42), and traumatic disease (M8418, M8448, S12-3, S22-3, S32-3, T02.1, and T03.1); (ii) had thromboembolic/coagulation disorder, renal failure, and hemodialysis on admission; (iii) required blood transfusion and TXA use preoperatively; (iv) had anticoagulation and antiplatelet drug use postoperatively; (v) required the 5% maximum blood transfusion volume outliers; and (vi) underwent the 5% minimum and maximum anesthesia time outliers. The hospital volume of spine surgery was defined as the number of spine surgeries performed annually at each hospital and was categorized into quartiles (very low, low, medium, and high). The hospital volume of TXA use was defined as the number of spine surgeries performed annually at each hospital in which TXA was administered. This volume was categorized into quartiles. To ensure the hospitals that perform a certain number of spine surgeries and hospitals that have a certain number of TXA use cases are only chosen, we also excluded patients who underwent spine surgery at the hospital which was categorized as having a “very low” hospital volume of spine surgery and/or a “very low” hospital volume of TXA use. Flowchart of patient inclusion/exclusion criteria.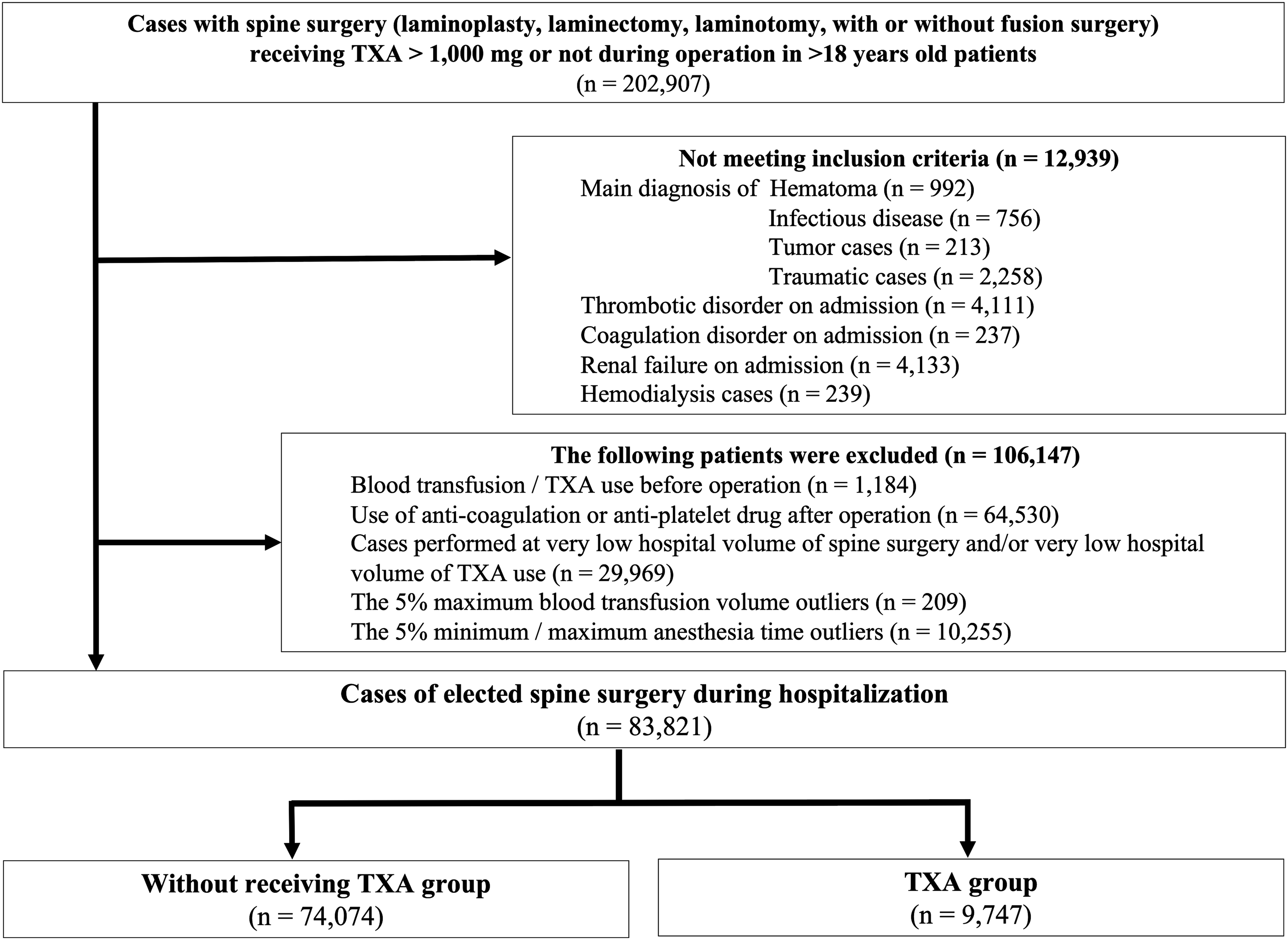
Covariates and Outcomes
The covariates included age; sex; body mass index (BMI) (kg/m2); smoking history (non-smoker, current/past smoker, and missing); academic hospitals; emergency admission; ambulance use; pre-existing comorbidities, including diabetes mellitus (E10-E14), hypertension (I10-I15), and chronic lung disease (J40-J47); history of cerebrovascular disease (I60-I69), cardiac disease (I20-I25, I30-I52), hepatic cirrhosis (K74), and at least one comorbidity; Charlson comorbidity index (CCI) score; 25 Barthel index; hospital volume of elective spine surgery; and hospital volume of TXA use. The following perioperative factors were also included as covariates: surgical site (cervical, thoracic, and lumbar spine), presence of ossification of the posterior longitudinal ligament, cell-saver use during surgery, instrumentation surgery, duration of anesthesia, and total amount of TXA used. Age was classified into 3 groups: 19-49 years, 50-64 years, and ≥65 years. BMI was categorized as underweight (<18.5 kg/m2), normal weight (18.5-24.9 kg/m2), overweight (25.0-29.9 kg/m2), and obese (≥30.0 kg/m2) according to the World Health Organization definition. CCI scores were categorized into 3 groups (≤1, 2, and ≥3).
The primary outcome was the proportion of massive hemorrhage requiring transfusion, defined as at least 560 mL of blood transfusion within 2 days of spine surgery or additional blood transfusion requirements from 3-7 days postoperatively.1,26 Because patients who underwent preoperative autologous donations were more likely to be transfused their own blood during perioperative management, we did not include autologous blood transfusion for calculating the amount of transfusion. Secondary outcomes were serious postoperative thrombotic complications (pulmonary embolism, acute coronary syndrome, and stroke) and postoperative hematoma requiring additional surgery. Complications after admission were identified with the following diagnoses recorded after admission: surgical site infection (T814) along with the text data “surgical site infection” or “deep infection” or “postoperative infection”; postoperative hematoma (S141, S241, S341, T093, and T810) along with the text data “hematoma” or “epidural hematoma”; pulmonary embolism (I26); respiratory complications (pneumonia [J12-J18 and J69], respiratory failure [J96], and respiratory disorders [J95]); acute coronary syndrome (I21-I24); heart failure (I50); stroke (I60–I64); urinary tract infection (N30, N34, N36-N37, and N39); and renal failure (N17–N19). We identified patients with at least one complication during hospitalization.
Statistical Analysis
We performed a propensity score matching analysis to balance the measured potential confounders between groups. 27 We estimated the propensity scores for TXA using a multivariable logistic regression model. All covariates were included as predictive variables in the model. We used the one-to-one nearest neighbor method with a caliper width of .2 of the standard deviation of the estimated propensity scores. Each patient in the TXA group was matched to one patient in the control group without replacement. We calculated the absolute standardized differences for each covariate before and after matching and confirmed the balanced distribution of covariates between the TXA and control groups. To adjust for clustering within hospitals, generalized estimating equations were fitted with multivariable logistic regression models after propensity score matching. Age, sex, BMI category, surgical site, instrumentation surgery, cell-saver use, duration of anesthesia, CCI category, and hospital volume for spinal surgery were included in the model.
We also performed the following sensitivity analyses. First, to assess the robustness of our results in propensity score matching, we conducted propensity score-stabilized inverse probability of treatment weighting (IPTW). 28 This method can maintain the sample size of the original data, yield more precise interval estimations of the variance of the main effect, and control type I error, as compared with the non-stabilized IPTW. Second, to control for unmeasured confounders and confirm our propensity score analyses, we conducted an instrumental variable (IV) analysis.29,30 An IV analysis can adjust for measured and unmeasured confounders, similar to a randomized controlled trial. In this study, IV was defined as the proportion of TXA use in each hospital, that is, the hospitals’ preference regarding TXA. These preference-based variables are obviously associated with the treatment of interest but have no direct association with the outcome. We used a two-stage residual inclusion method for IV analysis. Third, to improve the generalizability of our results, we included patients who were treated with anti-coagulant or anti-platelet therapy after surgery.
Continuous variables are presented as mean (standard deviation) and categorical variables are described as numbers (%). We compared the crude outcomes between the TXA and control groups using the t-test for continuous outcomes and χ 2 test for binary outcomes. A two-sided P-value <.05 was considered statistically significant for all tests. All analyses were performed using Stata/MP version 15 (StataCorp, College Station, TX).
Results
Baseline characteristics before and after propensity score matching.
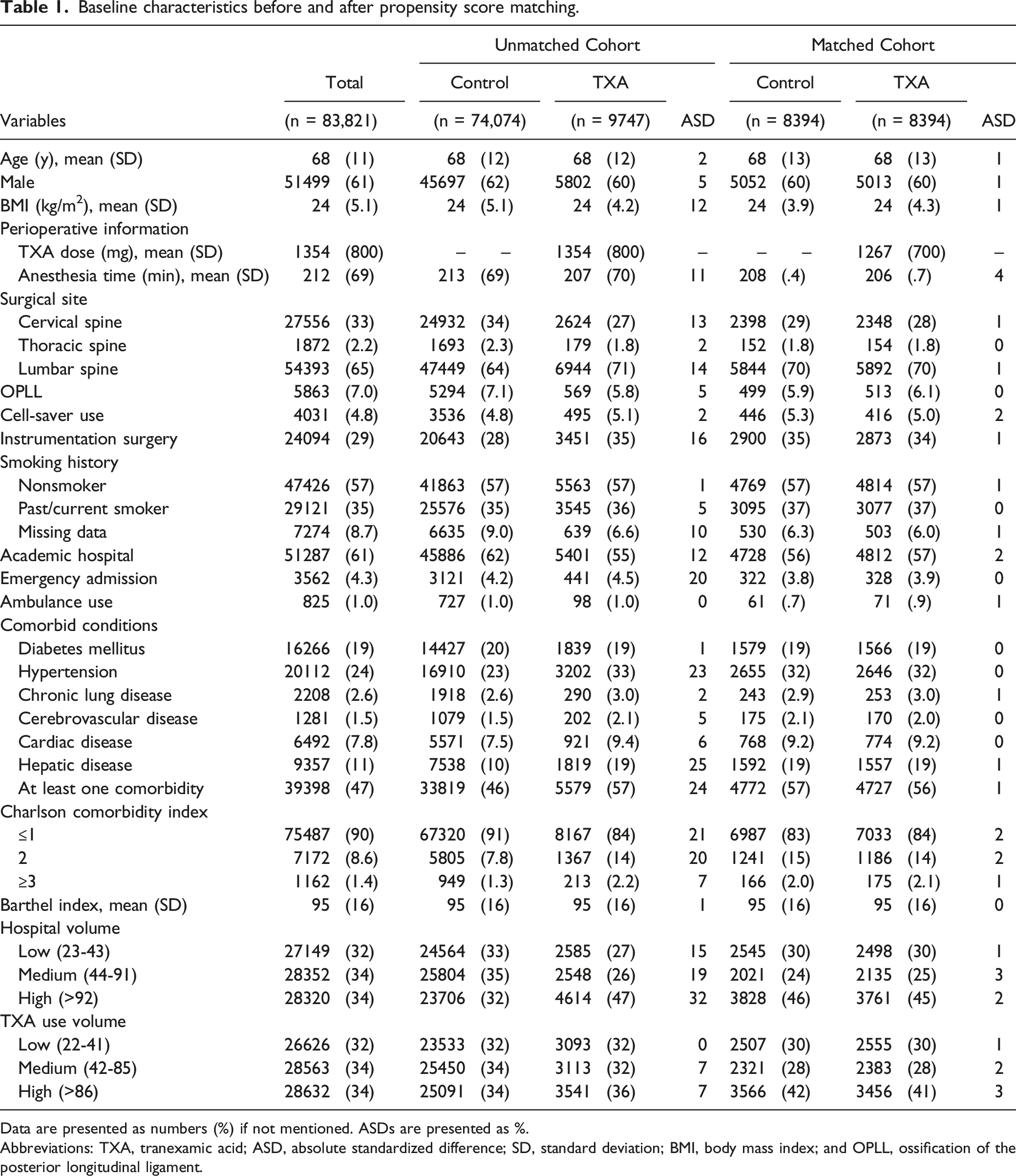
Data are presented as numbers (%) if not mentioned. ASDs are presented as %.
Abbreviations: TXA, tranexamic acid; ASD, absolute standardized difference; SD, standard deviation; BMI, body mass index; and OPLL, ossification of the posterior longitudinal ligament.
Outcomes before and after propensity score matching.
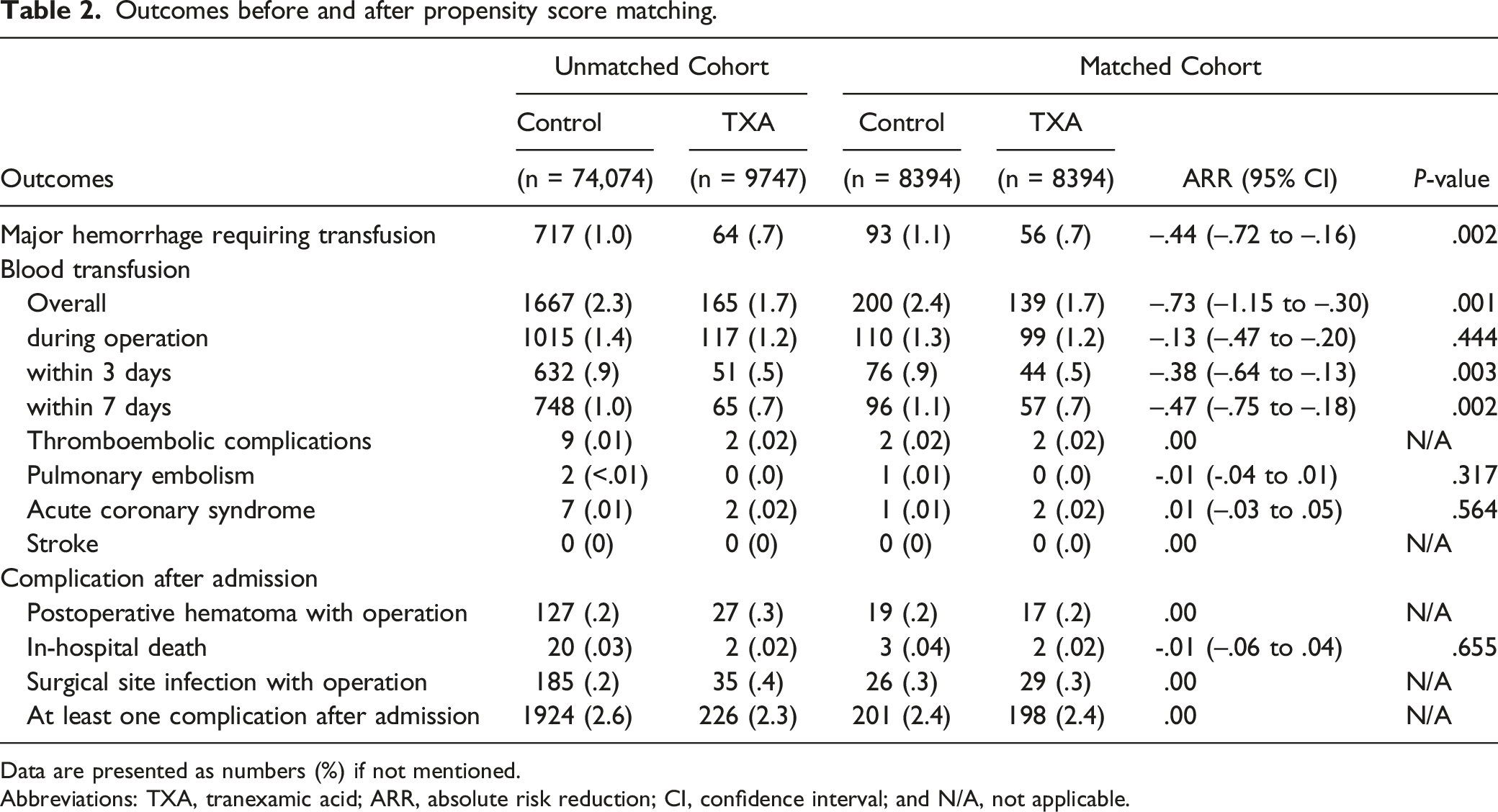
Data are presented as numbers (%) if not mentioned.
Abbreviations: TXA, tranexamic acid; ARR, absolute risk reduction; CI, confidence interval; and N/A, not applicable.
Multivariable logistic regression analysis after propensity score matching.
Discussion
In the present analysis of nationwide real-world data, we found that the intraoperative use of TXA was associated with reduced perioperative massive hemorrhage requiring transfusion during elective spine surgery without increasing thrombotic complications. There was no significant difference in the occurrence of postoperative hematoma that required additional surgery between groups.
Previous RCTs have shown that TXA reduced intraoperative estimated blood loss in spine surgery,11,13-15 whereas several other studies on TXA in spine surgery failed to reveal significant blood loss intraoperatively.10,12,16 The limited settings and small sample sizes of the RCTs may be the reasons for this inconsistency. Furthermore, most RCTs have been conducted at a single institution, which may have led to different results. The present study revealed that the use of intravenous TXA was associated with reduced massive hemorrhage requiring transfusion and perioperative blood transfusion, whereas intraoperative blood transfusion was not different between patients who used TXA and those who did not. These results are similar to those of previous meta-analyses.17-19
Although previous RCTs10-16 and meta-analyses17-19 have studied the efficacy of TXA use in spine surgery, some limitations exist in those RCTs regarding the safety profile in real-world clinical settings. Postoperative serious complications in spinal surgery, including pulmonary embolism, acute coronary syndrome, and stroke, are rare, ranging from .0%-.9%.23,31 Although a previous meta-analysis suggested that intravenous TXA was safe for patients who underwent any medical discipline, no population-based study has evaluated the risk of TXA use during elective spine surgery. 32 The present study is the largest and has enough sample size to estimate rare outcomes with an effect size of .2 and 90% power. Our results indicate that serious thrombotic complications were not associated with the use of TXA in elective spine surgery.
Another concern is whether the use of TXA increases or decreases the formation of postoperative epidural hematoma. Several studies have reported that the incidence of symptomatic postoperative epidural hematoma in spinal surgery ranges from .0%-1.0%.33-35 Studies have also reported that obesity (body mass index ≥35 or higher), multilevel procedures, an American Society of Anesthesiology classification > III, revision surgery, dural repair intraoperatively, and perioperative transfusion were independent risk factors for postoperative hematoma.33-35 However, it is difficult to precisely evaluate the risk of postoperative hematoma because of its low incidence and complex heterogeneity. The present study showed that the use of TXA was not associated with any increase or decrease in postoperative epidural hematoma that required additional surgery.
This study has several limitations. First, we could not obtain precise information on the operation. The American Society of Anesthesiology class and intraoperative complications, including dural tear and uncontrollable bleeding, may affect perioperative bleeding and postoperative hematoma formation and could be unmeasured confounders. However, we used IV analysis as a confirmatory analysis and found similar results to those of the main analysis. Thus, we believe that these unmeasured confounders may have little effect on perioperative blood transfusions and serious postoperative complications. Second, there was no information regarding the timing, dose, and administration route of intraoperative TXA use intraoperatively. Because the common criteria for the timing and dosage of intravenous TXA is a loading dose of 20 mg/kg (maximum 1000 mg) plus a maintenance dose of 1 or 2 mg/kg/h in Japan, 36 we excluded patients who were administered less than 1000 mg of TXA from the TXA group. It is expected that these patients were administered one-shot TXA at the end of surgery. We were also unable to identify whether TXA was used postoperatively, such as for the treatment of cerebral hemorrhage, or as part of the standardized postoperative deep venous thrombosis prophylaxis protocol. However, because no patients experienced stroke, including cerebral hemorrhage, postoperatively, there were no cases of TXA use, at least for cerebral hemorrhage postoperatively. Furthermore, according to the sensitivity analysis, there was little impact of anti-coagulant or anti-platelet therapy on the effect of TXA. Third, the database lacked information on hemoglobin and hematocrit values. However, we used blood transfusions as a surrogate variable for these predictors. Despite these limitations, the present study was a large-population study to reveal the effectiveness and safety of intravenous TXA use in elective spine surgery.
In conclusion, our study showed that the use of intravenous TXA in elective spinal surgery was associated with reduced perioperative massive hemorrhage requiring transfusion without any increase in postoperative adverse events, including thrombotic complications. This study underlines the effectiveness and safety of the intraoperative use of TXA in spine surgery in real-world settings.
Supplemental Material
Supplemental Material - Effect of Intraoperative Tranexamic Acid on Perioperative Major Hemorrhage Requiring Transfusion in Patients Undergoing Elective Spine Surgery: A Propensity Score-Matched Analysis Using a National Inpatient Database
Supplemental Material for Effect of Intraoperative Tranexamic Acid on Perioperative Major Hemorrhage Requiring Transfusion in Patients Undergoing Elective Spine Surgery: A Propensity Score-Matched Analysis Using a National Inpatient Database by Akira Honda, MD, Yoichi Iizuka, MD, PhD, Nobuaki Michihata, MD, PhD, Kazuaki Uda, PT, MPH, Tokue Mieda, MD, PhD, Eiji Takasawa, MD, PhD, Sho Ishiwata, MD, Yohei Kakuta, MD, PhD, Yusuke Tomomatsu, MD, Shunsuke Ito, MD, Kazuhiro Inomata, MD, Hiroki Matsui, PT, MPH, Kiyohide Fushimi, MD, PhD, Hideo Yasunaga, MD, PhD, and Hirotaka Chikuda in Global Spine Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Ministry of Health, Labour and Welfare, Japan (21AA2007 and 20AA2005) and the Ministry of Education, Culture, Sports, Science and Technology, Japan (20H03907).
Research Ethics and Patient Consent
This study was approved by the Institutional Review Board of The University of Tokyo [approval number: 3501-(3) (December 25th, 2017)]. Because all data were de-identified, the requirement for patient informed consent was waived.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
