Abstract
Study Design
Retrospective Cohort Study
Objectives
To explore whether classification of the increased signal intensity (ISI) on magnetic resonance imaging (MRI) correlates with clinical presentations and outcomes in symptomatic thoracic ossification of ligamentum flavum (T-OLF) patients.
Methods
All patients with symptomatic T-OLF who underwent laminectomy at four institutions were reviewed. The ISI on preoperative T2-weighted MRI was divided into 3 groups, Grade 0, none; Grade 1, light (obscure); and Grade 2, intense (bright). Neurological function before surgery and at follow-up was evaluated by the revised Japanese Orthopedic Association (JOA) score. Patients’ demographics, clinical manifestations, and surgical outcomes were compared.
Results
A total of 94 patients were involved. Preoperative MRI showed 32 patients in Grade 0, 39 patients in Grade 1, and 23 patients in Grade 2. Low extremities numbness, weakness, and clinical signs were less frequent in Grade 0 patients. The grade of ISI was correlated with the duration of symptoms and cord compression. Grade 0 patients had a better preoperative JOA score than those with ISI changes, while Grade 2 patients showed worse neurological recovery, longer duration of operation, more intraoperative blood loss, and a higher incidence of perioperative complications.
Conclusion
The classification of ISI is an effective parameter for preoperatively assessing cord compression, clinical severity, and surgical outcomes in T-OLF patients. Grade 0 patients have relatively mild neurological impairment but are more likely to be misdiagnosed. Grade 2 indicates the worst clinical impairment and neurological recovery, and implies a risky and challenging surgery.
Keywords
Introduction
Thoracic ossification of ligamentum flavum (T-OLF) is a rare spinal disease that results in slowly progressive spinal cord dysfunction. Patients with thoracic myelopathy caused by T-OLF present with various symptoms, such as sensory and motor disturbances of the lower limbs. 1 Clinical signs such as motor deficits, hyperreflexia, Babinski sign, and clonus are frequently observed. 2 Due to the low prevalence and non-specific clinical manifestations, T-OLF is frequently not recognized or misdiagnosed, resulting in delayed treatment. 3 Surgical decompression is widely acknowledged as the only strategy to provide the potential for the recovery of the spinal cord for symptomatic T-OLF patients. 4 Nevertheless, surgery does not always achieve satisfactory outcomes, and perioperative complications are not uncommon. 5 Although great efforts have been made to investigate the clinical characteristics and surgical interventions of T-OLF patients, it remains unclear whether there is a parameter that is capable of helping researchers and health care providers estimate the disability of patients and suggest possible clinical outcomes.
Magnetic resonance imaging (MRI) is essential to confirm the diagnosis and provide an anatomical description of the underlying pathology.6,7 Increased signal intensity (ISI) of the spinal cord on T2-weighted imaging (T2WI) MRI is widely regarded as a reflection of intramedullary lesions. 8 With the improvement of MRI techniques, the type of ISI has been demonstrated to correlate with different degrees of spinal cord neuropathological changes. 9 However, the clinical relevance of types of ISI in T-OLF patients has never been evaluated. We performed a multicenter study to assess whether types of ISI changes could be used to assess clinical severity and outcomes in T-OLF patients.
Material and Methods
This study reviewed all patients with symptomatic T-OLF who underwent laminectomy between January 2014 and March 2020 at four institutions after being approved by the ethics committee with the waiver of retrospective consent. The four institutions are all university-affiliated hospitals and key medical centers in southwest China. Patients received at least a 12-month follow-up following laminectomy were involved. Other involved spinal regions or concurrent with other pathologies such as ossification of the posterior longitudinal ligament, thoracic disc herniation, infection, or tumors were excluded. All procedures were performed by experienced spine surgeons. Surgical indications, level of decompression, and fusion were determined by surgeons at each institution.
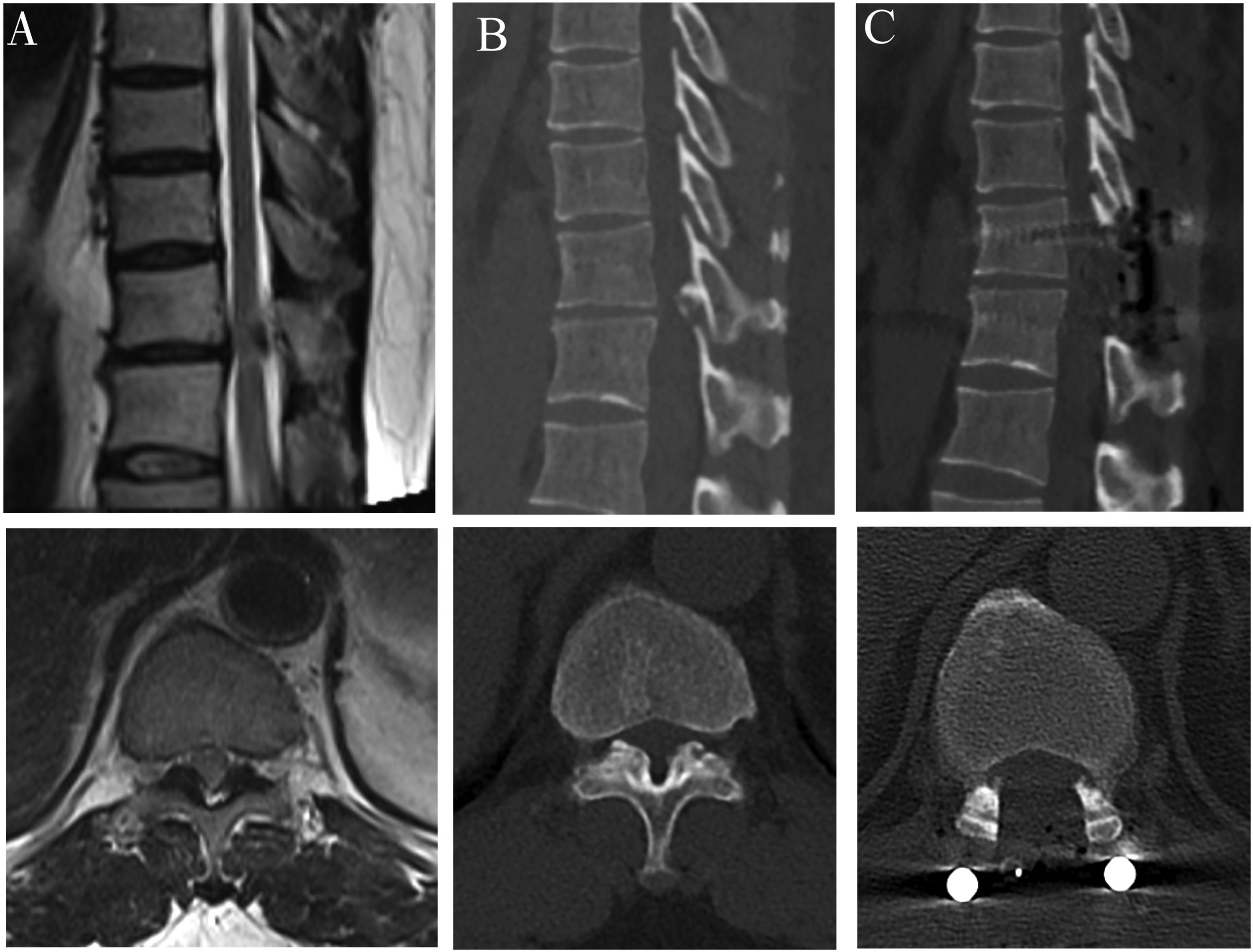
All patients underwent 1.5-T or 3.0-T high-resolution MRI before surgery. The ISI at the narrowest level in sagittal T2WI was divided into 3 groups as described by Yukawa et al.,
9
Grade 0, none; Grade 1, light (obscure); and Grade 2, intense (bright) (Figure 1). Intense ISI refers to the similar signal of cerebrospinal fluid. Two experienced spinal surgeons made the classification independently. The concordance between the two researchers in the assessment of the signal change on T2WI was .91 (kappa = .87; P < .001). The two researchers determined the final classification by consensus. In addition, the low intensity on T1WI was also observed. Three grades of increased signal intensity (ISI) on sagittal T2WI in patients with T-OLF: (A) Grade 0, none; (B) Grade 1, light (obscure); (C) Grade 2, intense (bright).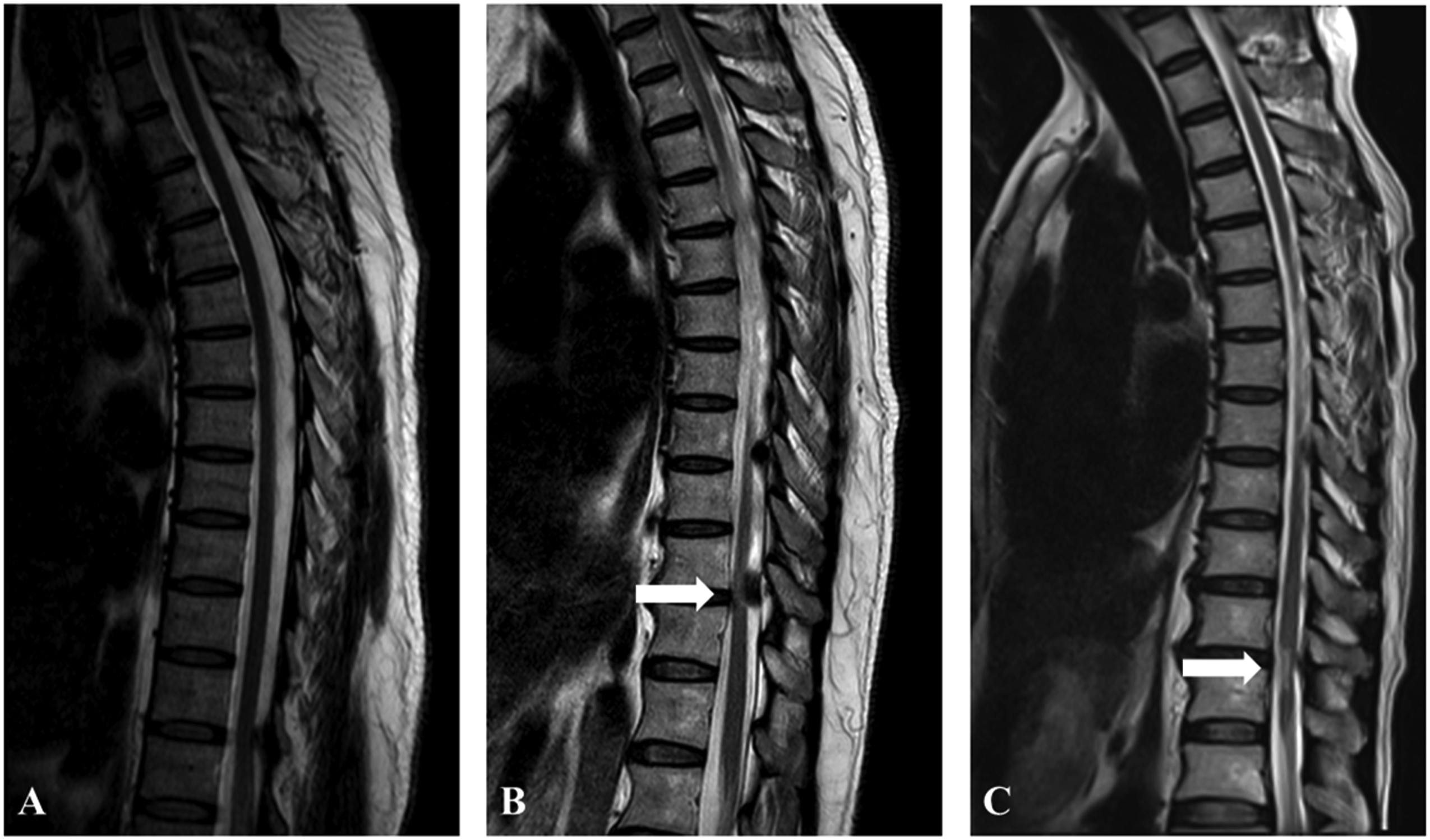
The patients’ demographics, clinical and surgical data, pre-and postoperative neurological function, and recovery rate were compared between the three groups. The demographic information included age, sex, duration of symptoms, smoking history, the prevalence of diabetes mellitus, and hypertension. Numbness, weakness, or pain in lower extremities, back pain, girdle pain, claudication, and sphincter dysfunction were recorded symptoms. Clinical signs included motor deficit, hyperreflexia, Babinski sign, and clonus. Surgical data such as operation time, intraoperative blood loss, and dural ossification (DO) was recorded. Perioperative complications referred to adverse events recorded within 30 days of the procedure.
The level of maximum compression (upper thoracic (T1-T4), middle thoracic (T5-T8), and lower thoracic (T9-T12)) was confirmed on preoperative CT scans. As described by Kuh et al.,
10
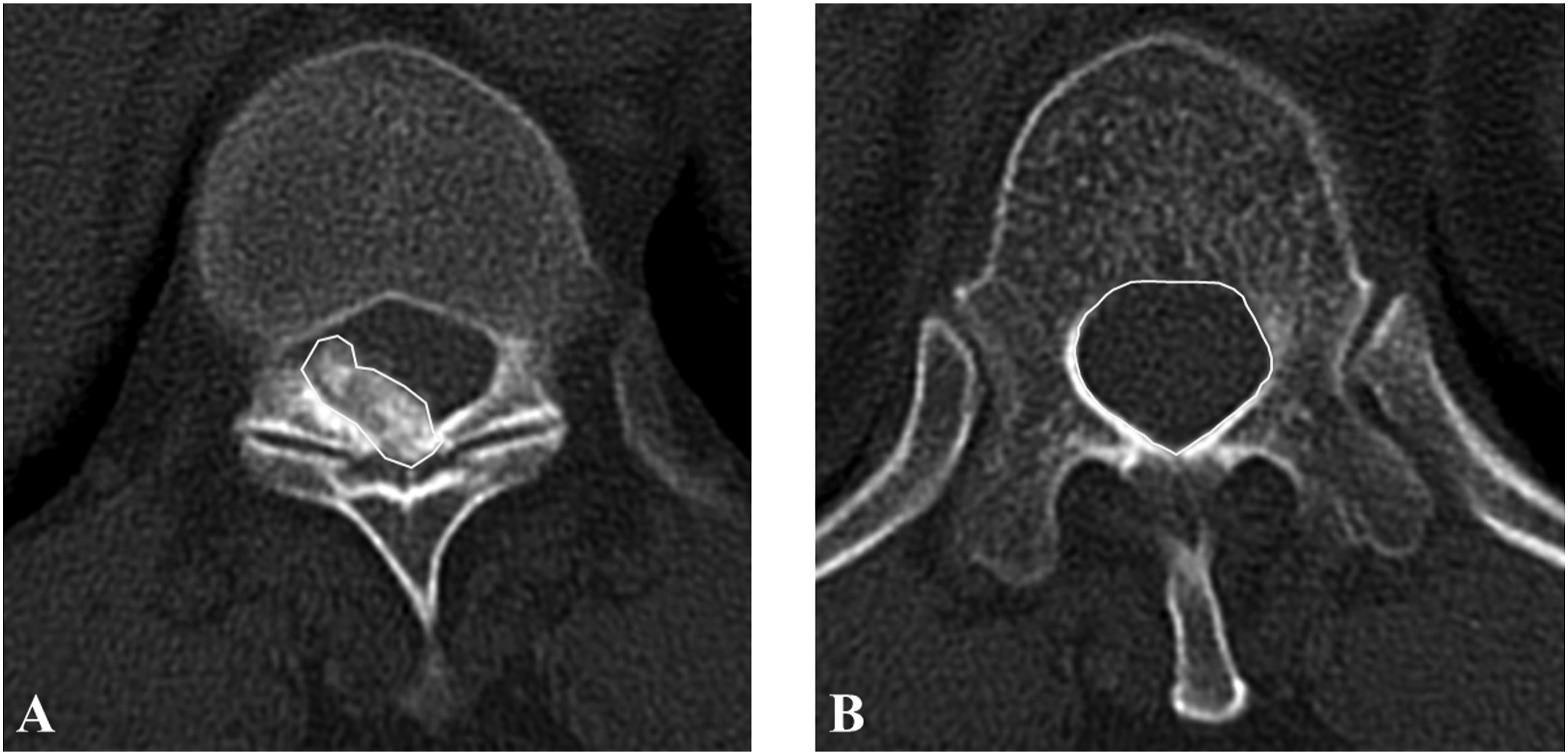
the axial configuration of OLF was classified into three types (unilateral, bilateral, and bridged) on CT, the sagittal type of OLF was divided into two types (beak and round) on MRI. (Figure 2). Ossification occupation rate was measured using Image
11
J as (axial ossified mass area/actual canal area) × 100% at the level of maximum spinal cord compression on axial CT (Figure 3). Three types of axial configuration of OLF on CT: (A) unilateral, (B) bilateral, (C) bridged. Two types of sagittal configuration on MRI: (D) round, (E) beak. The measurement of occupying ratio on axial CT. (A) Axial ossified mass area, (B) actual canal area. Ossification occupation rate = (axial ossified mass area/actual canal area) × 100%.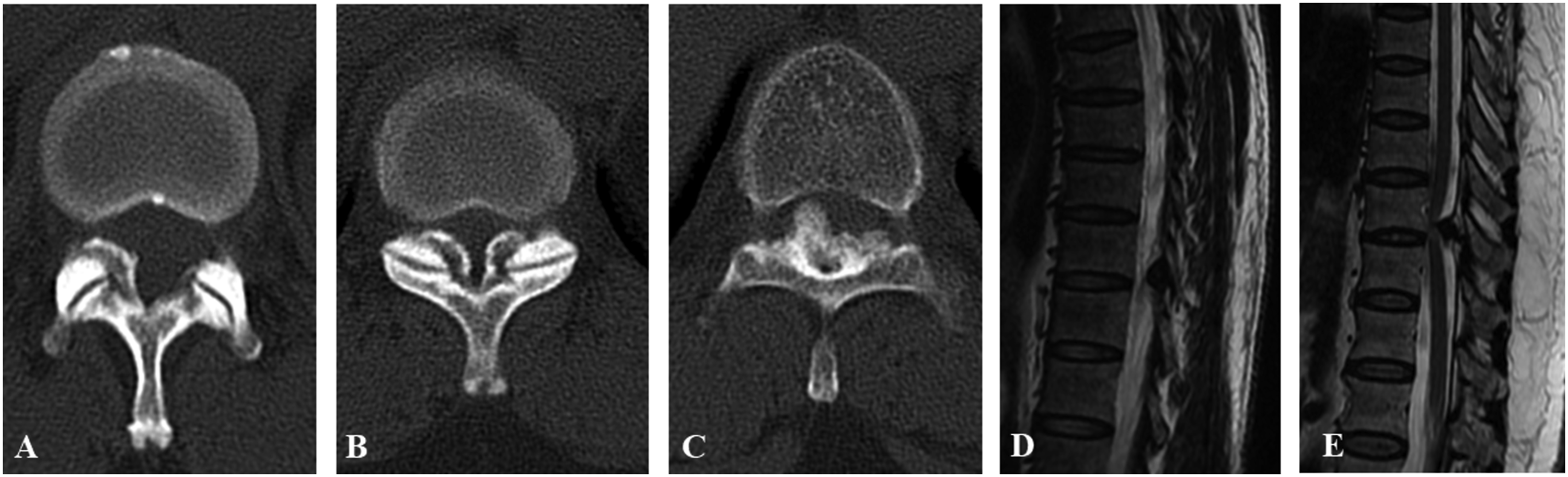
The neurologic function before surgery and at the follow-up was assessed using the revised Japanese Orthopedic Association (JOA) score. The scoring system excludes upper limb sensory and motor scores, with a maximum score of 11 points. The recovery rate was calculated as follows: (postoperative JOA score-preoperative JOA score)/(11-preoperative JOA score) × 100%. 12
All statistical analyses were completed using SPSS statistics software, version 25.0 (SPSS, Inc, Chicago, IL, USA). All values are represented as mean ± standard deviation, and categorical variables are described as numbers and percentages. The
Results
Patients' demographics, clinical information, and surgical outcomes.
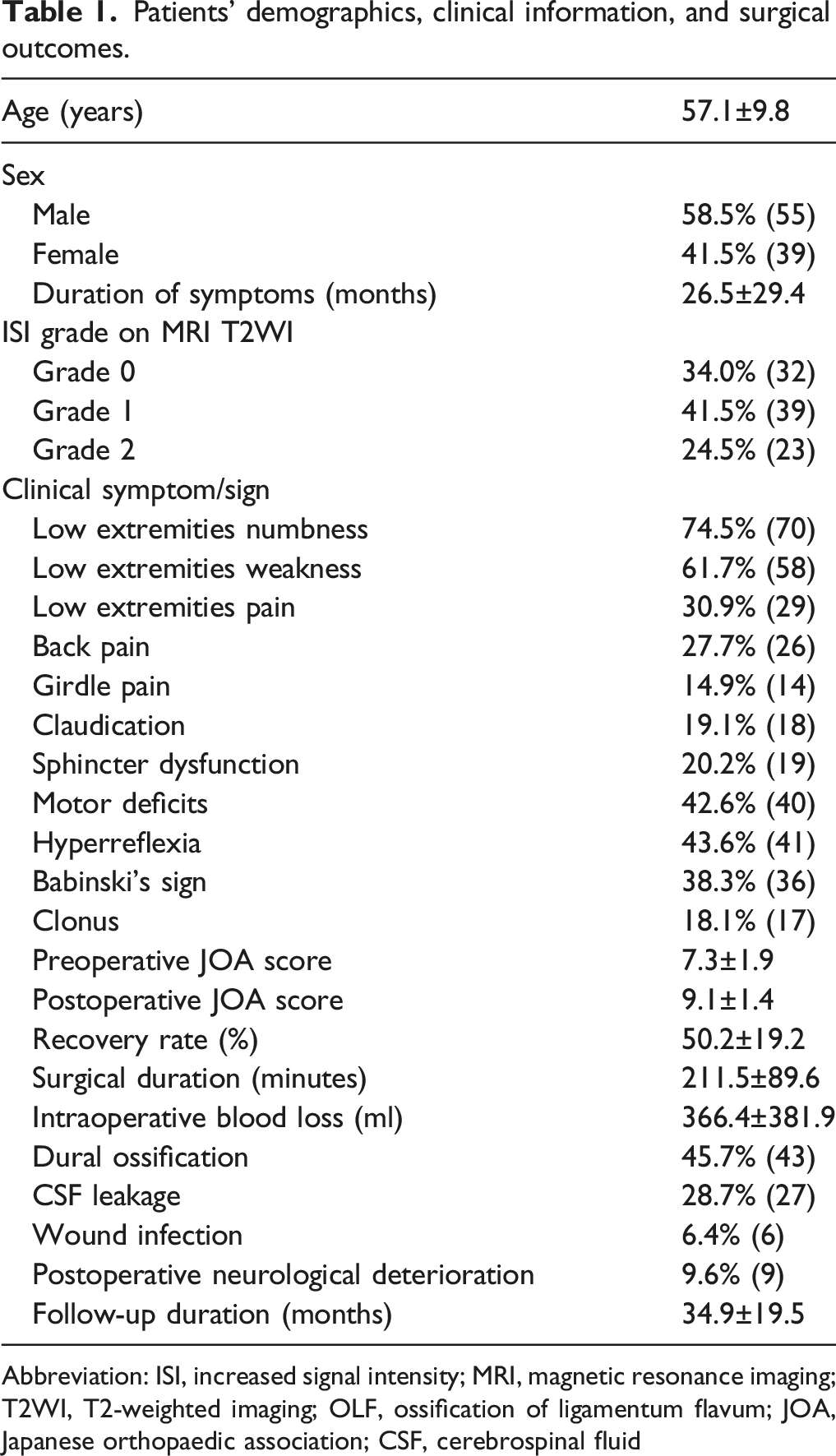
Abbreviation: ISI, increased signal intensity; MRI, magnetic resonance imaging; T2WI, T2-weighted imaging; OLF, ossification of ligamentum flavum; JOA, Japanese orthopaedic association; CSF, cerebrospinal fluid
Relationship between ISI classification and clinical symptom/sign.
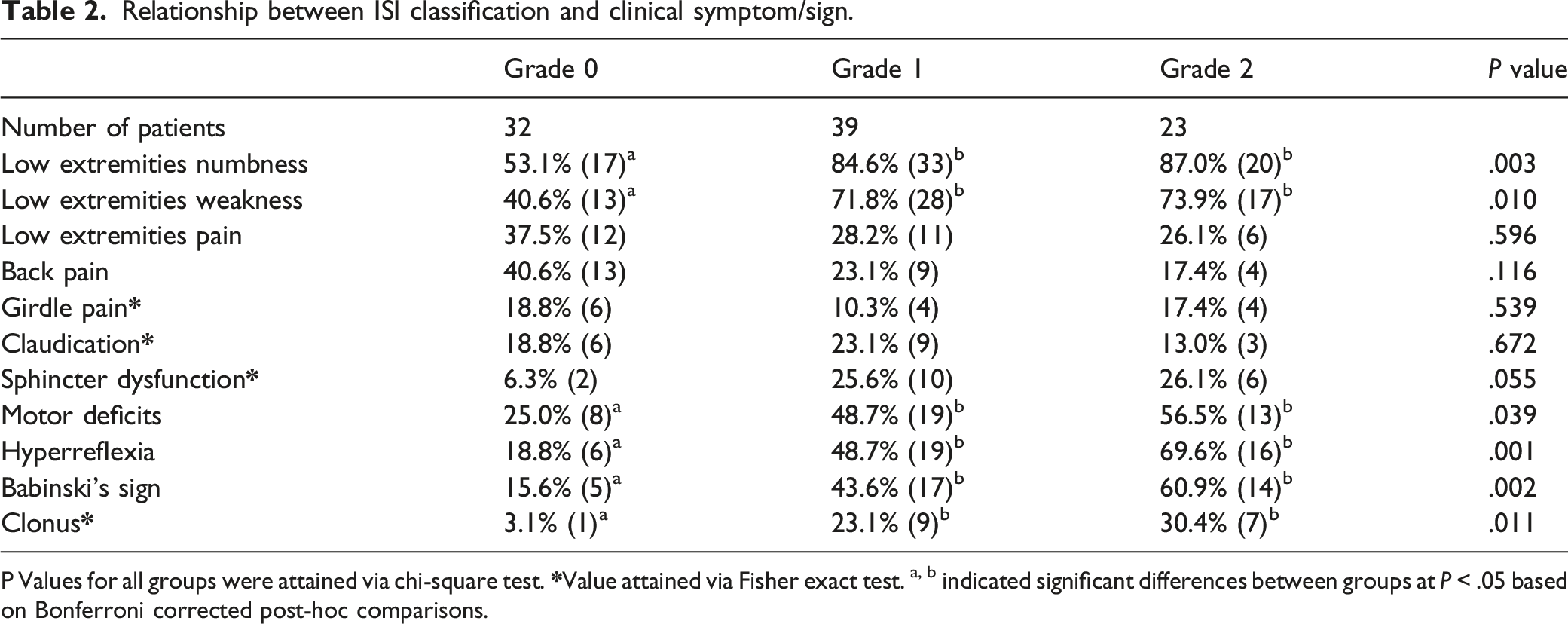
P Values for all groups were attained via chi-square test.
Clinical and radiographic characteristic in each grade of increased signal intensity.
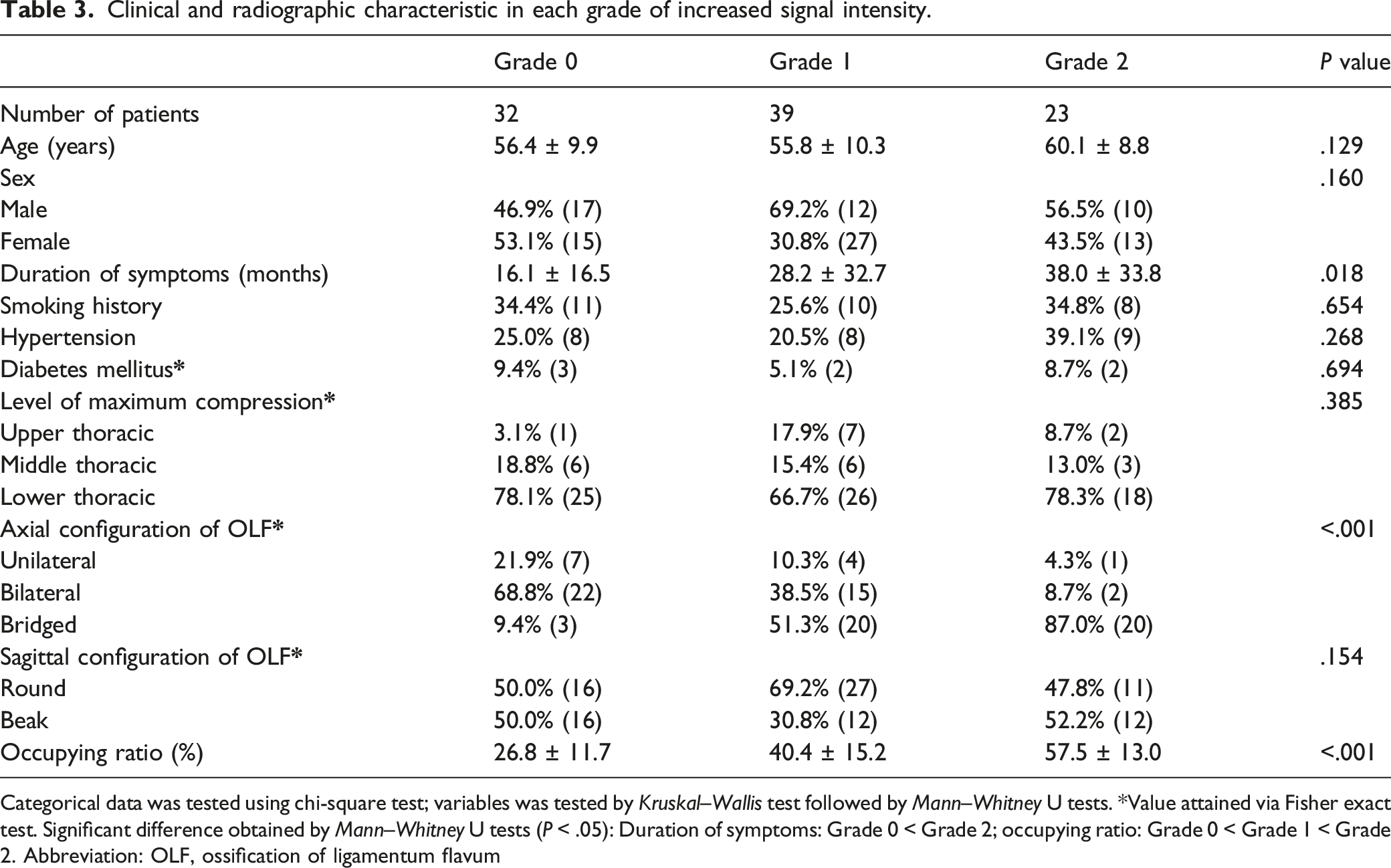
Categorical data was tested using chi-square test; variables was tested by
Surgical data and outcomes in each grade of increased signal intensity.
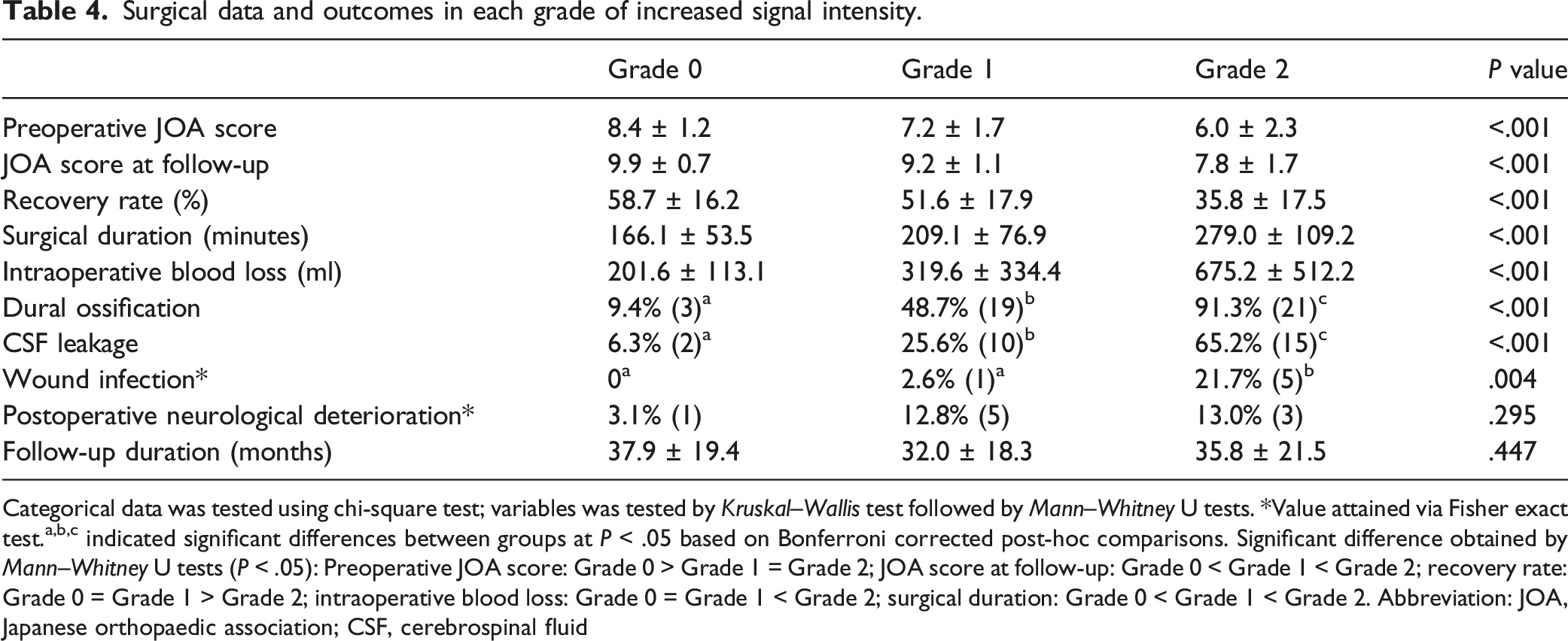
Categorical data was tested using chi-square test; variables was tested by
Illustrative Cases
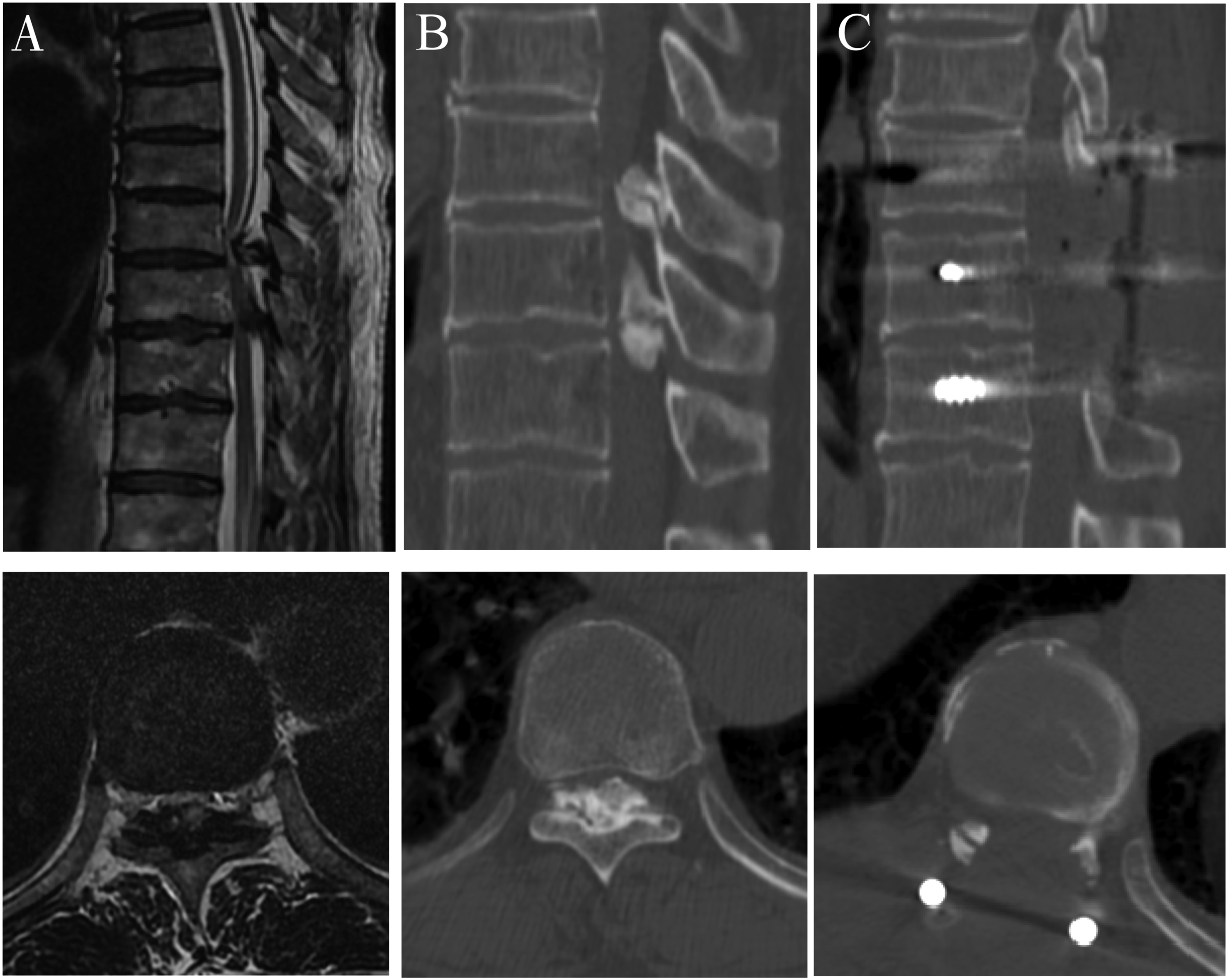
Case 1. A 64-year-old male was diagnosed with severe thoracic myelopathy caused by T-OLF at T9-10 and T10-11. Preoperative MRI T2WI showed Grade 2 ISI change and beak type of the OLF on sagittal view (Figure 4A). Preoperative axial CT scan showed the bridged type of OLF (Figure 4B). Posterior laminectomy and OLF resection with fusion was performed (Figure 4C). Dural ossification was found during the operation, and the dura mater was incised with the neuro-patch sutured. Postoperative CSF leakage occurred but healed in 5 days post-procedure without infection. The JOA score improved from 3 points preoperative to 5.5 at follow-up (recovery rate, 31.2%). Case 2. A 45-year-old male with progressive lower-limb numbness and weakness was diagnosed with thoracic myelopathy caused by OLF at T11-12. Preoperative MRI T2WI showed Grade 1 ISI change and round type of the OLF on sagittal view (Figure 5A). The preoperative axial CT scan revealed the bilateral type of OLF (Figure 5B). We performed laminectomy and OLF resection with fusion (Figure 5C). The JOA score improved from 8 points preoperatively to 9.5 at follow-up (recovery rate, 50%). A 64-year-old male was diagnosed with severe thoracic myelopathy caused by T-OLF at T9-10 and T10-11. Preoperative MRI T2WI showed Grade 2 ISI change and beak type of the OLF on sagittal view (Figure 4A). Preoperative axial CT scan showed the bridged type of OLF (Figure 4B). Posterior laminectomy and OLF resection with fusion was performed (Figure 4C). Dural ossification was found during the operation, and the dura mater was incised with the neuro-patch sutured. Postoperative CSF leakage occurred but healed in 5 days post-procedure without infection. The JOA score improved from 3 points preoperative to 5.5 at follow-up (recovery rate, 31.2%). A 45-year-old male with progressive lower-limb numbness and weakness was diagnosed with thoracic myelopathy caused by OLF at T11-12. Preoperative MRI T2WI showed Grade 1 ISI change and round type of the OLF on sagittal view (Figure 5A). The preoperative axial CT scan revealed the bilateral type of OLF (Figure 5B). We performed laminectomy and OLF resection with fusion (Figure 5C). The JOA score improved from 8 points preoperatively to 9.5 at follow-up (recovery rate, 50%).
Discussion
T-OLF has been regarded as a cause of thoracic myelopathy. A prospective multicenter study conducted by Ando et al. 5 showed that the clinical manifestations of T-OLF were complex, and the postoperative recovery was relatively poor. However, there is still no strategy for assessing the clinical severity and suggesting surgical outcomes before surgery for patients with T-OLF. Recently, a popular ISI classification proposed by Yukawa et al. 9 was reported to be associated with different degrees of pathology in the spinal cord. We conducted a multicenter study to clarify whether such spinal cord pathological changes had an impact on the clinical severity and outcomes of thoracic myelopathy caused by OLF. Our results indicated that the preoperative ISI classification correlated with clinical presentations, duration of disease, the degree of spinal cord compression, and surgical outcomes in patients with T-OLF.
The T2WI hyperintensity and T1WI low signal of the spinal cord on MRI were widely thought to be the reflection of spinal cord lesions.13,14 Although the prognostic significance of ISI on MRI T2WI has previously been evaluated in T-OLF patients, its relationship with clinical presentations, radiologic characteristics, and surgical complications has not been thoroughly elucidated.2,10,15 The ISI changes on T2WI represented a wide range of spinal cord pathological changes from mild to severe. 16 However, most studies investigated the presence of ISI on MRI T2WI but did not mention the grade of ISI. Although the quantitative signal intensity changes on T2WI were reported to be associated with T-OLF patient prognosis, whereas it relies on specific computer software or radiologic workstation. 17 The MRI T1WI low signal of the spinal cord was pathologically indicated as necrosis or secondary syrinx of the spinal cord. 18 However, few studies have evaluated its prognostic significance in T-OLF patients for its several disadvantages. Previous studies have pointed out that the T1WI low signal has a low frequency and poor interobserver agreement in patients with cervical compression myelopathy. 19 Therefore, it was suggested that T1WI low signal was less helpful in clinical practice. In our study, there is a large discrepancy in the recognition of T1WI low signal between the two skilled spinal surgeons. Only two patients out of 62 with ISI changes were eventually determined to have T1WI low signal. Several possible reasons might account for the result. Unlike the spinal signal changes on T2WI, T1WI low signal change has no strong contrast with normal spinal cord signal. In addition, the low signal intensity OLF directly compressed the spinal cord, making it challenging to identify the T1WI low signal in the narrowed spinal cord on sagittal view. The classification of ISI proposed by Yukawa et al. 9 was found to be a simple qualitative method with high interobserver agreement to evaluate the ISI changes. From the view of neuropathology, the less intensity ISI represented milder lesions, whereas the intense ISI indicated more severe histologic lesions. 20 It implied that the ISI grade was correlated with the severity of the spinal cord lesion.
T-OLF is often overlooked or misdiagnosed due to its rarity and non-specific symptoms. 3 As previously reported, the most common clinical symptoms in patients with T-OLF were lower extremities numbness and weakness. 1 However, the prevalence of clinical symptoms and signs was significantly different between patients with and without ISI changes. The symptoms of Grade 0 patients were more non-specific, and the majority lacked pyramidal signs, which made them more vulnerable to being misdiagnosed with lumbar disorders or missing diagnosis. The missed diagnosis of T-OLF in patients concurrent with lumbar disorders might lead to severe surgical outcomes. 3 In addition, the relative mild spinal occupation and no signal changes on MRI also disturb the diagnosis and the choice of surgical decompression. The classification of spinal canal degree proposed by Feng et al. 21 was a helpful modality for the diagnosis of myelopathy caused by T-OLF. Conservative treatment is recommended for patients without neurological deficit manifestations, and ossification does not touch the spinal cord. Some quantitative radiologic measurements can also be used to definite the diagnosis and responsible level.21,22 Furthermore, ISI changes were related with more severe clinical manifestations and worse baseline neurological function. Pyramidal signs such as Babinski’s sign, hyperreflexia, and clonus are regarded as the reflection of the severity of myelopathy. 23 These signs were more frequent in patients with ISI changes and should be noted as indirectly indicating pathological changes in the spinal cord and therefore implying timely surgical decompression.
Surgical decompression of the stenotic spinal canal is the only effective modality for the thoracic myelopathy caused by T-OLF. 4 However, complete neurological recovery is often not achieved after surgery. 2 Many studies have pointed out that ISI on T2WI is associated with a poor postoperative outcome, while others have not found any such association.2,10,15 This contradiction may lie in the fact that ISI on T2WI implies a variety of non-specific spinal cord pathological alterations and spinal cord recuperative capacity. 14 Previous studies focused on the presence of ISI on T2WI but did not refer to the type of ISI. Here, patients with Grade 2 ISI had worse neurological recovery than the other two groups, while there was no significant difference between Grades 0 and 1. According to our results and previous research,9,24 we can speculate that the less intense ISI (Grade 1) on T2WI represents relatively mild and reversible pathological changes, whereas the intense ISI (Grade 2) on T2WI represents more severe and irreversible pathological changes. In addition, the ISI classification was associated with the cord compression and the duration of symptoms. This result was not unexpected. Previous research found that persistent mechanical compression may result in neuronal structural changes in the spinal cord. 25 Static compression is the determinant factor in spinal cord dysfunction caused by OLF since it diminishes canal size and compresses the cord. Long-term mechanical stress causes spinal cord ischemia, which results in spinal cord damage. 26
A variety of posterior procedures have been developed to treat thoracic myelopathy caused by OLF. Percutaneous endoscopic decompression (PED) has been recently applied to the decompression of T-OLF.27-29 Under local anesthesia, PED provides safe and satisfactory decompression while minimizing trauma and postoperative kyphosis for single-level, non-fused, or tuberous type OLF patients without DO. 27 It appears to be appropriate for most Grade 0 patients with single-level OLF. However, further research is required to ascertain the postoperative recurrence rate. In practice, laminectomy is still the most common procedure for T-OLF. Based on the image characteristics of T-OLF, various laminectomy approaches have developed.30-32 Although these approaches could achieve direct decompression, they were challenging and accompanied by many complications.33-35 Consistent with previous studies, the most common complication was CSF leakage. 34 DO was the leading cause of dural tears and CSF leakage. And the incidence was reported as high as 62%. 36 In our research, the incidence of DO was 45.7% of all cases and was 91.3% in Grade 2 patients. Besides, patients in Grade 2 had the highest proportion of bridged OLF and the highest occupying ratio. Due to the presence of DO and severe spinal stenosis, the procedures of Grade 2 patients were more complicated and difficult. Here, Grade 2 patients had significantly increased operation time, blood loss, and the prevalence of complications. Luckily, several procedures for these patients with severe T-OLF and DO have been proposed. Miyashita et al. 37 proposed that floating ossification following laminectomy could achieve neurologic improvement and floating ossification reduced by time. Sun et al. 38 found no significant difference in postoperative neurological improvement comparing thinning and floating ossification with direct ossification resection. Recently, indirect decompression approaches were developed for severe T-OLF patients accompanied by DO. The bridge crane technique was a practical and feasible decompression technique by en-bloc isolating the laminae-OLF complex (LOC) and suspending it to the transverse connectors. 39 Thoracic extended laminoplasty was also a new indirect decompression approach, which achieved better mJOA and HRQOL scores than laminectomy without complications.40,41 Although indirect decompression approaches avoided the risk of dual sac and nerve root injury, the inability to observe directly the border of lesion complicated the evaluation of the decompression effect. It is still necessary to pay more attention to the management of patients with Grade 2 ISI.
Early diagnosis is vital for T-OLF patients because patients with greater ISI grade have severe clinical manifestations, complicated procedures, and worse neurological recovery. Surgeons should be aware of the increased surgical challenges in patients with Grade 2 ISI, be adequately prepared before surgery to decrease blood loss, operation time, and inform patients of possible surgical risks and related complications.
Some limitations need to be noted in the present study. Although this is a multicenter study, the patient population is still small due to the rarity of T-OLF, particularly in Grade 2 ISI patients, and the follow-up duration may be insufficient. In addition, our assessment of imaging parameters was influenced by the variance in CT and MRI systems from different institutions involved in the studies. A further prospective study with large samples is still needed to assess the impact of ISI changes on clinical severity and surgical outcome in patients with T-OLF.
Conclusion
The ISI classification is a reliable parameter for assessing clinical severity and suggesting possible surgical outcomes in patients with T-OLF. Grade 0 patients were more likely to be not recognized or misdiagnosed due to non-specific symptoms and the absence of lower extremity signs. Grade 2 represents prolonged and severe spinal canal compression, which not only indicates the worst potential for postoperative recovery, but also represents a more difficult and dangerous procedure. Early diagnosis and treatment are important for T-OLF patients to obtain better surgical outcomes and fewer complications.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
