Abstract
Study Design
Literature Review (Narrative)
Objective
To propose a new framework, to support the investigation and understanding of the pathobiology of DCM, AO Spine RECODE-DCM research priority number 5.
Methods
Degenerative cervical myelopathy is a common and disabling spinal cord disorder. In this perspective, we review key knowledge gaps between the clinical phenotype and our biological models. We then propose a reappraisal of the key driving forces behind DCM and an individual’s susceptibility, including the proposal of a new framework.
Results
Present pathobiological and mechanistic knowledge does not adequately explain the disease phenotype; why only a subset of patients with visualized cord compression show clinical myelopathy, and the amount of cord compression only weakly correlates with disability. We propose that DCM is better represented as a function of several interacting mechanical forces, such as shear, tension and compression, alongside an individual’s vulnerability to spinal cord injury, influenced by factors such as age, genetics, their cardiovascular, gastrointestinal and nervous system status, and time.
Conclusion
Understanding the disease pathobiology is a fundamental research priority. We believe a framework of mechanical stress, vulnerability, and time may better represent the disease as a whole. Whilst this remains theoretical, we hope that at the very least it will inspire new avenues of research that better encapsulate the full spectrum of disease.
Keywords
Introduction
Degenerative Cervical Myelopathy [DCM] is an all-encompassing term for cervical spinal cord dysfunction as a result of degenerative, arthritic, and/or congenital changes to the cervical spine. 1 DCM causes a wide range of symptoms, including pain, imbalance and difficulty walking, loss of dexterity, sensory loss, bowel or bladder dysfunction, and in extreme circumstances total paralysis. DCM is estimated to affect 1.4% to 3.1% of adults 2 and today is often associated with significant disability, despite treatment. 3 This has life-long implications, with dependency, unemployment, and mental health difficulties prevalent.4-8
AO Spine RECODE-DCM (aospine.org/recode) [REsearch objectives and COmmon Data Elements for DCM] is an international consensus project which aims to accelerate knowledge discovery to improve outcomes, by developing a set of research tools. 9 This process included a James Lind Alliance research priority setting partnership, which brought together both individuals living and working with DCM, to establish the most important unanswered questions. Research prioritization aims to catalyze progress by consolidating resources on key knowledge gaps. The Number five priority identified was improving our understanding of the pathophysiology—that is, the biological basis of DCM.
Amongst the first descriptions of DCM was a surgical case series, published in 1928 by Byron Stookey, 10 which provided a detailed description of symptoms related to ventral compression of the spinal cord. Macroscopically, he reported indentation and displacement of the spinal cord and correlated it with the patient’s neurological symptoms, including motor and sensory disturbances. Initially mistaken for chondromas, Peet end Echols, clarified that the “tumours” are most likely “intervertebral disc protrusions” 11 and thus have a degenerative etiology. Presently, it is recognized that a variety of degenerative changes in the spine can trigger cervical myelopathy.1,12,13 This led Nouri et al. (2015) 13 to propose “Degenerative Cervical Myelopathy” as a new umbrella term, subsequently endorsed through consensus by the AO Spine RECODE-DCM initiative (aospine.org/recode). 9
There are very few human post-mortem studies of DCM. 12 Histologically, Brain et al. noted significant deformation of the spinal cord 14 extensive white and gray matter degeneration, cavity formation, and loss of neurons and axons. In addition, a unique series of ultramicroscopic studies demonstrated evidence of demyelination and incomplete remyelination indicating simultaneous injury and repair. 15 Fehlings et al. suggested that the extensive loss of cells, at least in part, may be related to widespread cellular apoptosis in the spinal cord. 16 Our recent unpublished study investigating markers of macro-autophagy in post-mortem tissue has demonstrated a correlation between symptom severity and dysregulation of autophagy, providing the first human evidence for a mechanistic explanation for the apoptotic cell death observed in DCM tissue.
Together, these studies highlight the intricate relationship between structural changes in the spinal column and the functional consequences that occur in the spinal cord, which form the basis of DCM.
Pre-clinical studies to model DCM pathogenesis are also limited.
17
Compression-based studies include genetic models, such as the
The longstanding view has been that chronic tissue compression secondary to spinal canal narrowing from degenerative and/or congenital changes is the direct cause of the spinal cord injury in DCM. This concept is reflected in current surgical practice, where decisions are often based on the extent of cord compression visualized by MRI, 28 and less, as our recent clinical guidelines recommend, 29 on the severity of symptoms.
However, the chronic compression paradigm fails to account for the full spectrum of clinical disease (Figure 1), namely: (1) Spinal cord compression is common and most frequently incidental and asymptomatic, with approximately 10% of individuals developing symptoms.
2
(2) The extent of static spinal cord compression does not correlate well with the severity of symptoms, clinical phenotype, or disease trajectory.30-36 (3) The functional decline in DCM is rarely linear; it can be stable, step wise, or particularly in advanced stages the decline appears to accelerate.37-39 (4) Microstructural MRI has demonstrated that cord damage precedes the loss of spinal cord function and is not restricted to the area of compression.40-47
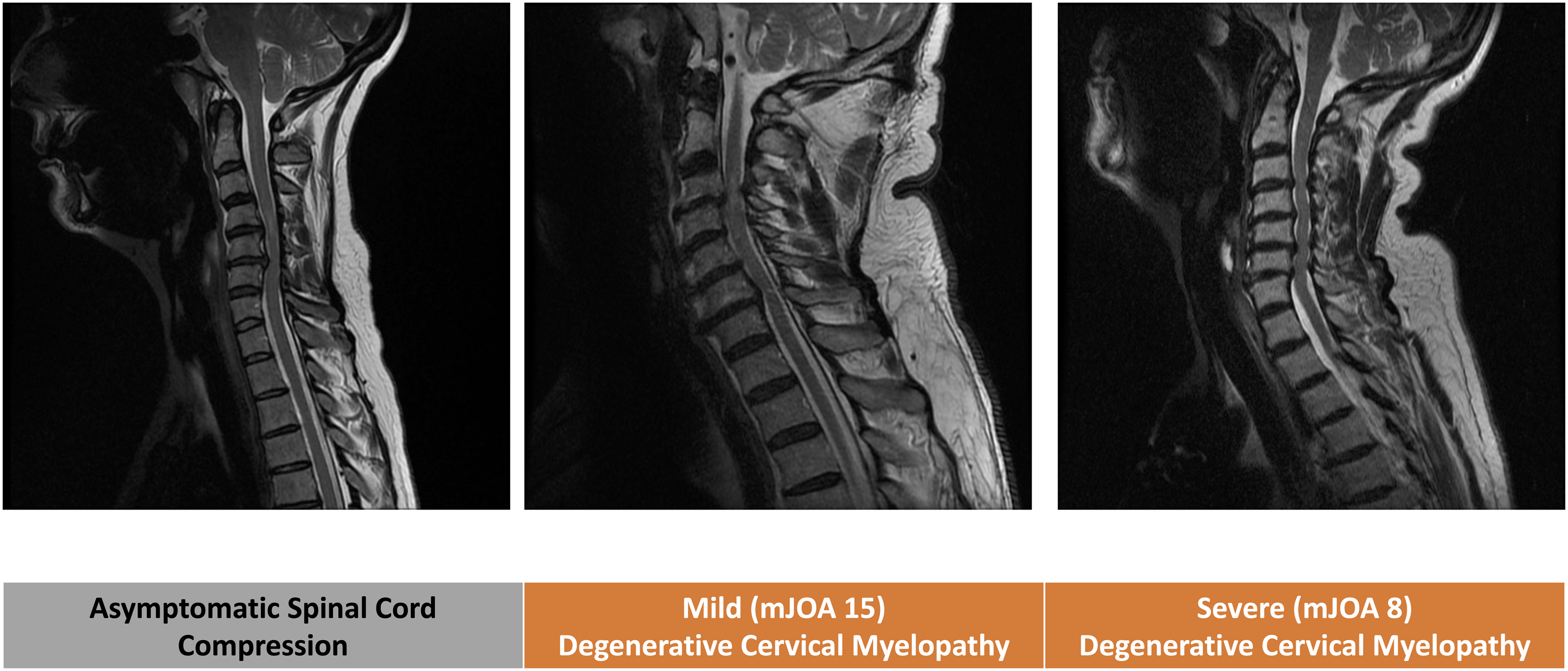
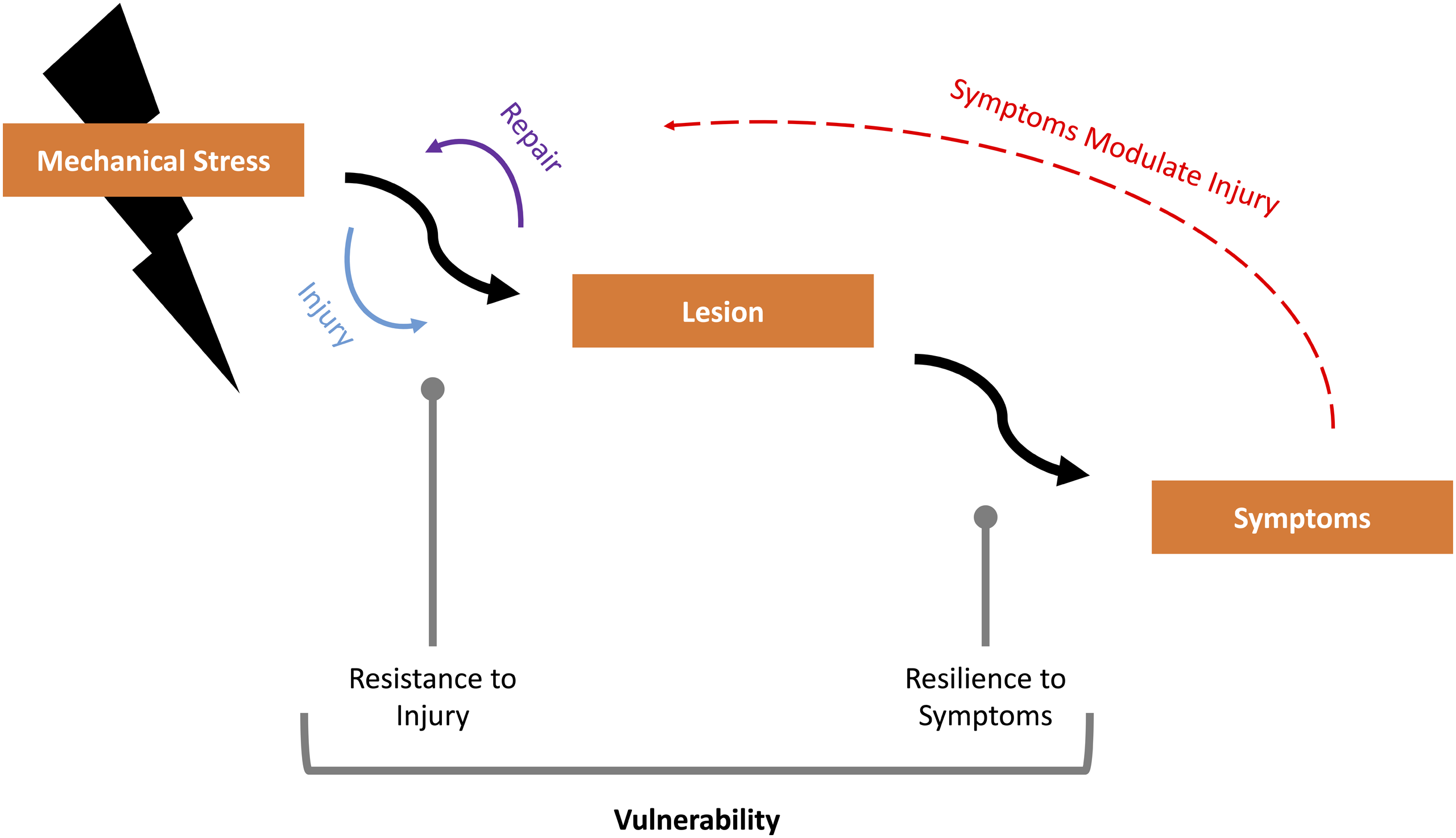
Whilst explanations for these observations have been advanced, including that dynamic spinal cord compression is not captured by routine, supine MRI,13,48 we propose a broader paradigm, to unify these and other critical and emerging observations in DCM. Specifically, we propose that DCM is a function of mechanical stress, duration of injury and an individual’s intrinsic vulnerability to central nervous system injury (Figure 2)
This article will focus on these unifying concepts, that may explain the discrepancies between radiological and clinical findings. Pragmatically therefore, we will not cover in detail the downstream molecular mechanisms demonstrated in DCM research that are well described elsewhere.12,17
Mechanical Stress
While conceptually, the driver of myelopathic progression has been considered progressively worsening compression, this has not been confirmed by empirical data30-32,34-36,49 and this view oversimplifies the dynamic situation where several forces will distort not only parenchymal tissue but also blood vessels and nerve roots. 50
A force is any interaction, that when unopposed, will change the motion of an object. The application of a set of forces to an object give rise to 5 principal mechanisms of loading: compression, tension (i.e., stretch), bending, torsion, and shear (Figure 3).
51
An object subject to a constant force, is referred to as having a static load, whereas a time-varying force(s) a dynamic load. There are 5 principal types of mechanical loading; Compression [A], Tension (or Stretch) [B], Shear [C], Bending [D], and Torsion [E]. At a small element level, compression, tension, and shear are the local stresses [Orange] but at a structural level, these can act in combination, to give rise to bending or torsional loads [Gray]. Compression is the application of an inward force. Tension is the application of a force which elongates a material. Shear forces result from sliding contact between two parallel surfaces. Loading methods are figuratively represented in row 1, and how these might apply to DCM in vivo illustrated in row 2.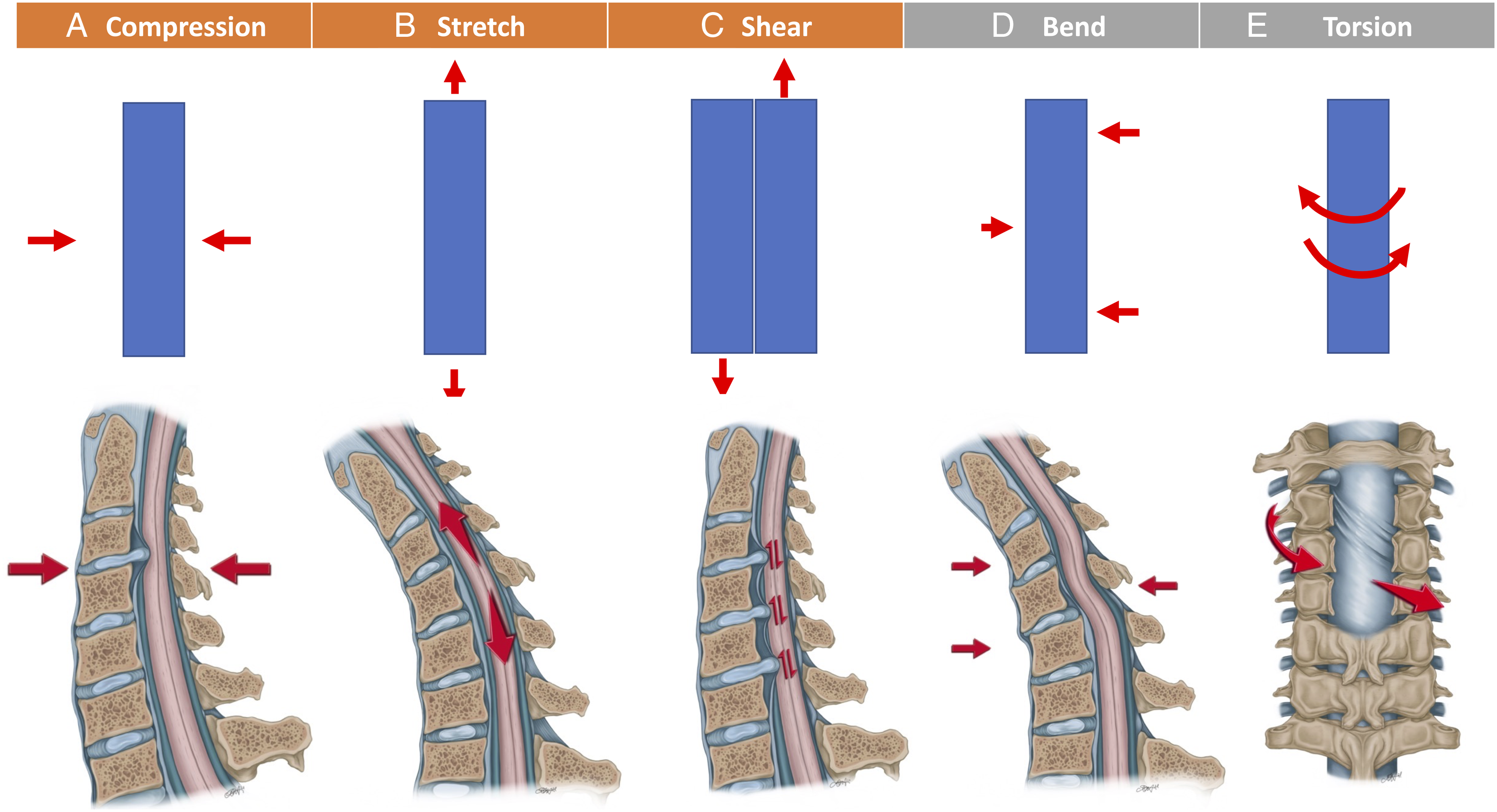
The local loading in a material element is expressed by the mechanical stress defined as the net force acting on a cross-sectional area (
The spinal cord is a viscoelastic material, meaning that it has both viscous (stress and strain are dependent on rate) and elastic (material will return to original form once loading has stopped) properties.
54
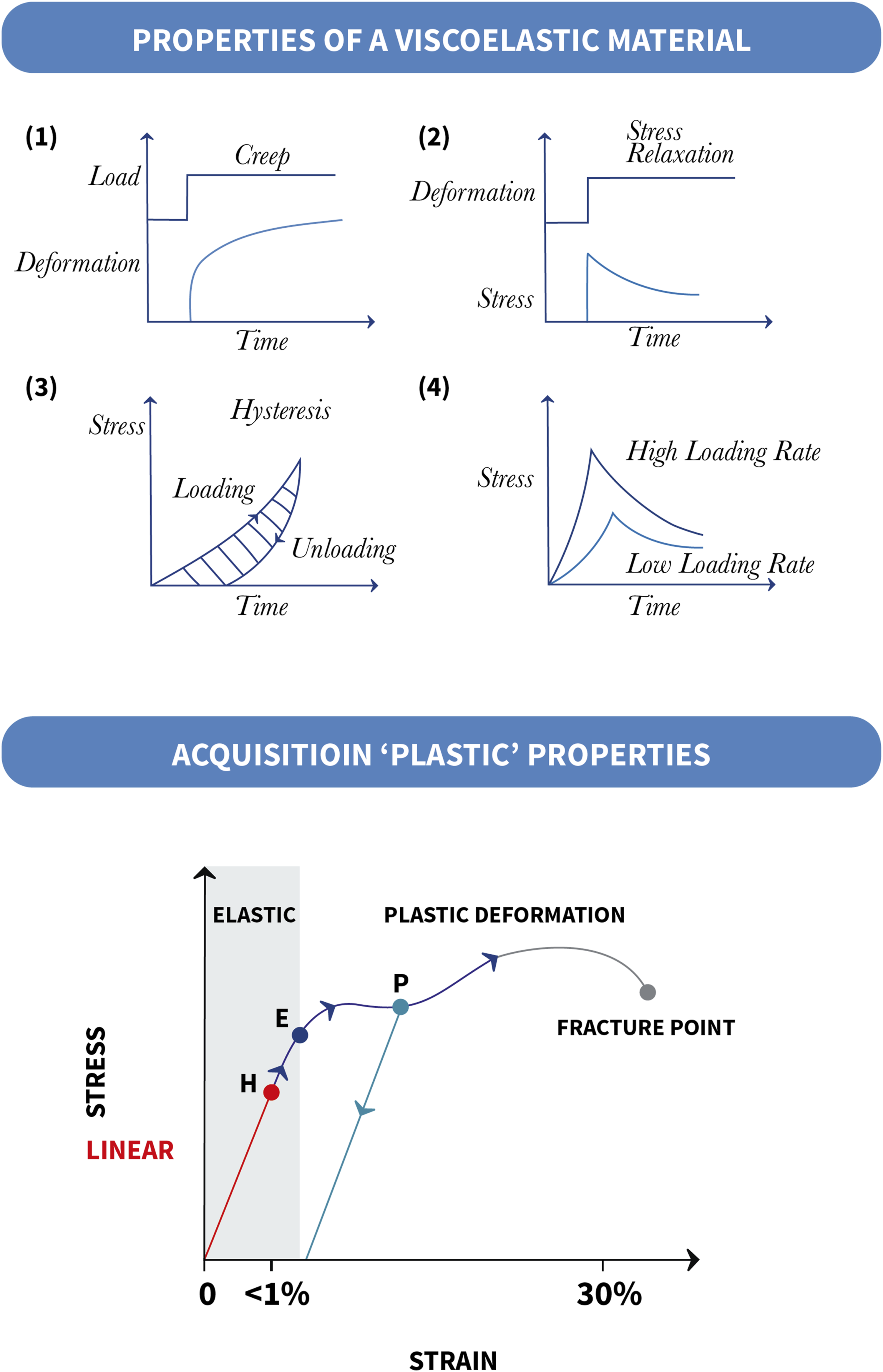
Combining these components gives rise to three main characteristics of viscoelastic material behavior: creep, stress relaxation, and hysteresis (Figure 4). Creep refers to the continued deformation of the material after a load has reached a constant state. Stress relaxation refers to the reduction in stress when a material is held at a constant strain or deformation. Hysteresis refers to differences in the loading and unloading response, as represented by a stress–strain graph. The area between the two curves represents the energy dissipated. For this reason, viscoelastic materials are used as shock absorbers. The peak stress in viscoelastic materials is also dependent on the rate of loading, due to a rate-dependent stiffening of the material. Consequently, a faster loading rate leads to a greater peak stress.
55
Any elastic properties have a threshold beyond which they will exhibit irreversible or “plastic deformation.” (Figure 4) This is best appreciated with an elastic band; stretch up to a limit will enable an elastic band to return to its original shape but stretch beyond this and the band will be permanently elongate, and further increases eventually cause it to snap. A viscoelastic material which acquires plastic properties, becomes a viscoplastic material.
56
Whilst plastic deformation of the spinal cord occurs in DCM, it is not a determinant of it. 49 Harada et al. (1992) demonstrated a subset of patients recovered normal spinal cord shape following surgical decompression 57 and Martin et al. 40 (2018) observed plastic deformation in a cohort of asymptomatic spinal cord compression. In DCM therefore, as static compression is chronic and the spinal cord remains viscoelastic, stress from compression specifically would be limited. 58
However, the response of the spinal cord to loading is complicated by its normal mobility. The spinal cord is suspended (therefore under modest tension) within cerebrospinal fluid (CSF) contained by the dural sac, and only partially anchored by the dentate ligaments and to a lesser extent connected vasculature. The spinal cord continuously moves in all three directions, cranial-caudal, anterior-posterior and right-left.59,60 This movement is now well demonstrated using MRI techniques,61-63 and thought to be principally related to the cardiac cycle, due to local pulsatile vascular changes and/or similar displacement of the brain from arterial in and venous outflow, but also the respiratory cycle. 64 Contrary to previous belief, the denticulate ligaments limit cranial-caudal movements of the SC, being especially resistant to caudal stress, but have minimal or no significant contribution to limiting anterior-posterior motion.65,66 This is now used diagnostically, for example, to identify a tethered spinal cord using MRI in the prone position.67,68 Further, the material properties of the spinal cord change with normal movement of the spine.69,70 This is largely in flexion, where the spinal cord lengthens,71,72 and the cross-sectional area of the spinal cord reduces,73,74 estimated to exhibit a strain of 12% in normal physiological conditions.75-77 Movement of the spine can be abnormally increased in DCM, 78 with spondylolisthesis radiologically prevalent, 27 and an association with laxity disorders such as Ehlers Danlos Syndrome. 79 Such findings have been associated with differing clinical manifestations of DCM.78,80,81
On a biomechanical level then, compression is not the only mechanical force involved, nor likely dominant force in all cases. For example, as illustrated in Figure 5, if one considers a single level disc prolapse (the cause of 10% of DCM cases with in the AO Spine Series
27
), then a variety of different loading mechanisms may occur; compression (due to perpendicular contact), tension (second to displacement and elongation of the spinal cord, and/or the effect of tethering), and shear (due to oscillation of the spinal cord over the surface of the disc prolapse).
82
Compression loading is defined as applying force inwards that is balanced by an at least equal opposing force.
51
As a soft mobile structure therefore, unilateral compression of the spinal cord, for example, may displace and not necessarily compress the spinal cord. Finite-element modeling has replicated this concept, identifying thresholds for ventral compression to exceed before mechanical stress is detected.83,84 This is also supported by histological studies indicating the spinal cord tolerance of some compression,85,86 with a re-alignment of spinal pathways, and cadaveric models demonstrating tension-related deformation before compression-related deformation.82,87,88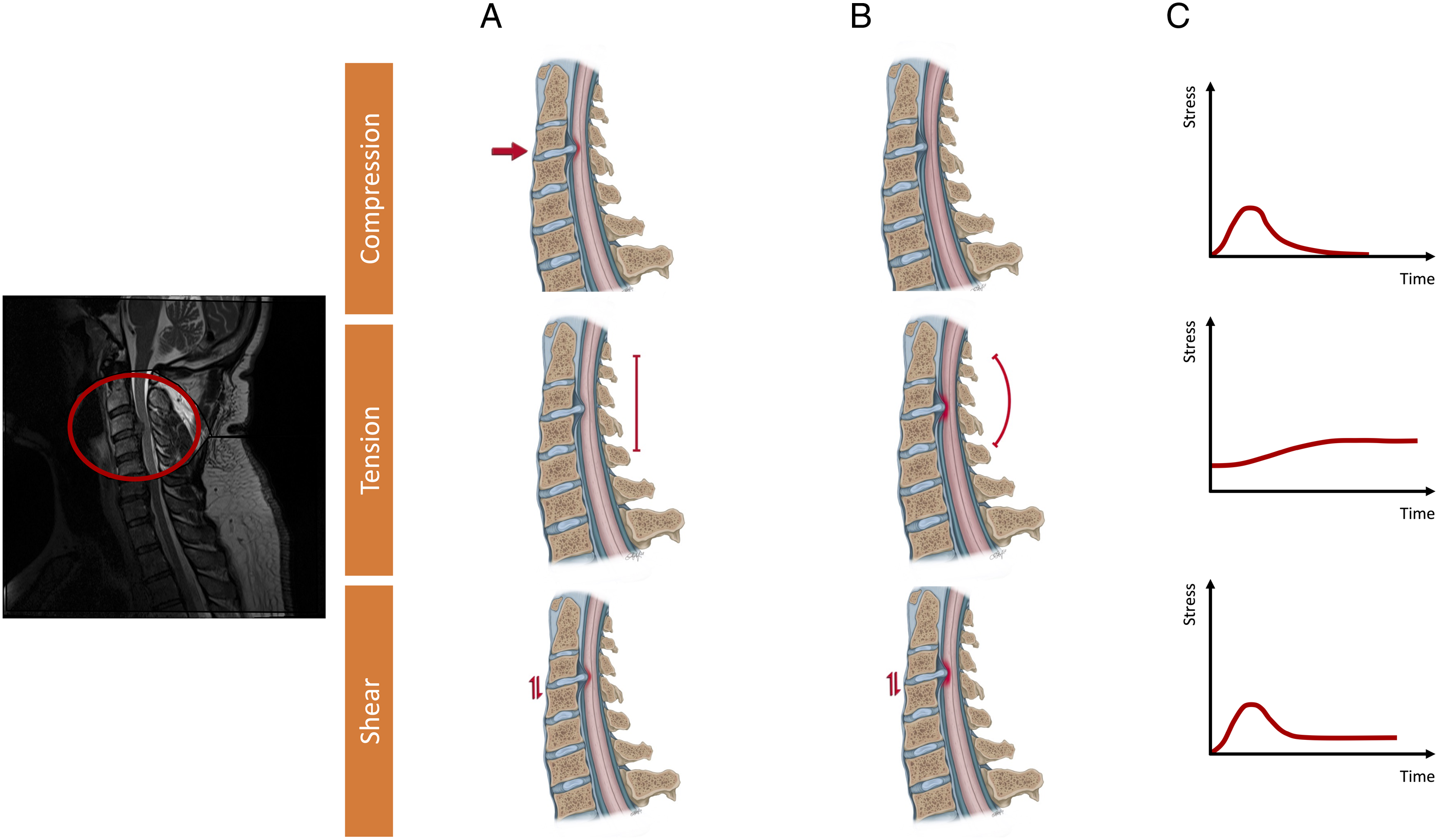
A strong argument for tension (or stretch) as a dominant mechanism of injury in DCM was advanced by Henderson et al. (2005), 76 combining pre-clinical, clinical, and mathematical data sources. This is further supported now by microstructural imaging studies, which are demonstrating structural change throughout the spinal cord,89-91 well beyond the proposed level of stenosis and poses interesting questions for a significant relationship with tandem stenosis. 92
The significance of shear forces however is also likely to be more significant than currently understood. Vavasour et al. 93 (2014) used phase-contrast MRI to evaluate the significance of CSF flow in 13 DCM patients and age-matched healthy controls. Based on their results, whilst CSF flow dynamics were associated with the compression ratio, they were not related to clinical function (as measured using somatosensory evoked potentials or the Japanese Orthopaedic Association scale [JOA]). However, their analysis also measured spinal cord oscillation (cranio-caudal), calculating mean velocity and absolute and maximum displacement, based on integrating the velocity time curve. These measures did correlate with clinical function. These results have been subsequently external validated by Wolf et al. (2018), and the subject of a follow-up, and ongoing observational study.64,94 Wolf et al. (2018) hypothesize that the reduction in CSF volume at levels of stenosis, reduces the ability of CSF to absorb mechanical stress and contributes to increased oscillation. Whilst only case reports, there are clinical indications of the significance of CSF to DCM, 95 for example, where CSF diversion (e.g., a lumbar puncture) has led to a rapidly worsening clinical picture.96-98 The measurement of CSF pressure in DCM, and its response to dynamic manipulation (e.g., head inclination or reclination) is the subject of an ongoing observational study [NCT04345822]. 99
In spinal practice, the pathological significance of non-compressive mechanical loading and spinal cord injury is recognized. For example, tethered cord syndrome arises due to stretch,100,101 and bending considered the principal loading mechanism in cervical flexion myelopathy (Hiramaya’s disease), due to flexion of the spinal cord within a shorter and/or non-elastic dural membrane. 102 At a cellular level, these mechanical forces can change neuronal behavior, for example, neural transmission,103,104 but also cause cellular injury, with evidence focused in particular on stretch105,106 and shear 107 forces, as well as compression.108,109 Of note, much of the more recent evidence evaluating mechanical forces on cell function has originated from within developmental neurobiology, where, for example, axon growth cones have been demonstrated to be modulated by mechanical forces such as stretch. 110 This could have particular relevance to DCM, where due to the chronicity, repair coincides with injury. 15 Russel et al. (2012) using a rat model and Galle et al. (2007) a guinea pig model of spinal cord injury, demonstrated that the location of maximal tissue injury correlated to the area of maximal strain.111,112
In DCM, the mechanical stress experienced will be complicated by how these forces interact, 84 for example, in a finite-element model of static compression of the spinal cord, elasticity measured using ultrasound reduced when the spinal cord was compressed. 113 These forces too, will have been exhibited in combination, within the existing pre-clinical models that underpin our current investigations of DCM.17,114 Mechanical stress will also be influenced by the different mechanical properties and tolerance of microstructures within the spinal cord, for example, the white vs gray matter, or relative myelin content of spinal cord pathways 115 which are also subject to change, for example, with age and evolution of injury.50,116
Consequently, the forces driving spinal cord pathology in DCM are likely to be a combination of loading forces and not simply compression, and we propose would be better represented by an umbrella term of “mechanical stress.” They are also likely to be highly individualized and change over “time.” Much of the literature exploring the biomechanical properties of the spinal cord has been conducted with a view to application in traumatic spinal cord injury.117,118 Whilst the clinical significance of mechanical stress in such an acute, and often devastating injury, has so far provided few practical applications,
119
in DCM, the chronicity of the condition may place greater relevance on this, in particular, because removing
Vulnerability
Whilst a clearer characterization of mechanical stress and its impact may therefore account for some current inconsistencies, it is unlikely to provide a full explanation for the onset and progression of DCM, and in particular that 1) only a subset of individuals with spinal cord compression develop symptoms related to DCM1,2 and that 2) symptom severity and progression does not correlate with spinal cord compression.28,30
Hence, an additional factor accounting for the development and progression of DCM is required. We propose this is an individual’s vulnerability to DCM, a combination of the intrinsic vulnerability of the spinal cord to resist damage from mechanical stress but also the human body to maintain normal homeostatic function; their resilience to symptoms 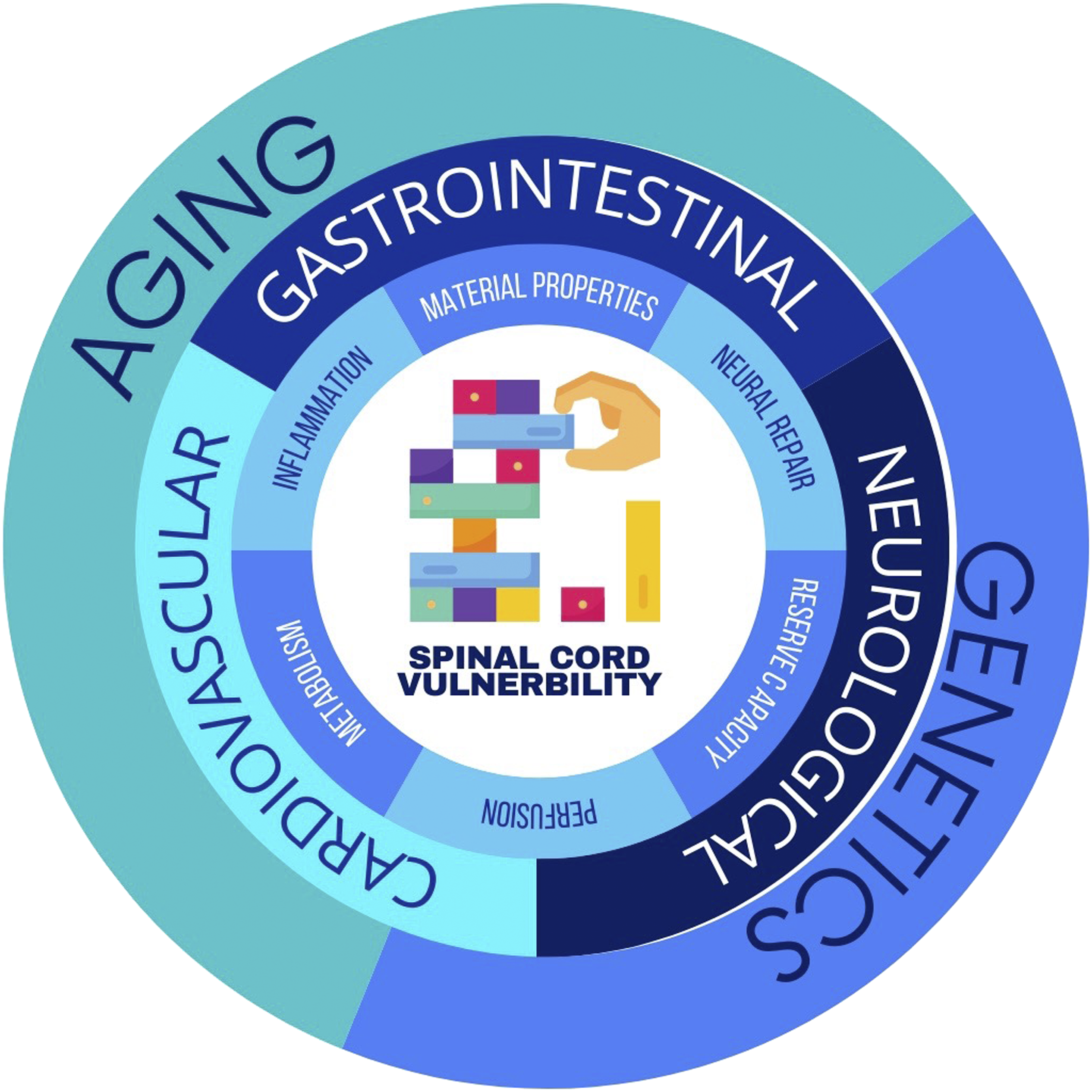
Genetic Effects on the Spinal Cord
Genetic studies support the concept that intrinsic factors have a role in determining DCM. In a case-controlled study of Utah residents, Patel et al.
120
(2012) demonstrated that first and third degree relatives of individuals with DCM had a 5 (CI = 2.1-13.2,
Age Effects on the Spinal Cord
Aging has broad biological implications.135,136 In clinical practice, older age is associated with increased disease severity (as quantified based on neuromuscular function) at presentation, however, this does not appear a consequence of a greater length of time with symptoms, for example, due to delayed diagnosis. 137 Further, whilst operative treatment on average involves more levels and has an increased occurrence of adverse events, 137 when these factors and baseline severity were controlled for using a propensity matched analysis in the AO Spine observational data, age alone still reduced the extent of recovery.138,139
The significance of age is likely to be multifactorial, contributing both to resilience to symptoms but also resistance to injury. 136 Age is recognized to interact with neural repair mechanisms, for example, affecting axonal outgrowth140,141 and remyelination.142,143 Imaging144-147 and autopsy studies148-150 have also demonstrated that the volume and properties of the spinal cord alter with age in the absence of disease. This occurs both within the gray and white matter, albeit is markedly less (approximately 1/3) of what is observed within the brain. 144 Exploring this using a mouse model, Piekarz et al. 151 (2020) demonstrated that age was associated with axon loss and demyelination, in particular a loss of and altered morphology of alpha-motor neurons, and an altered extracellular matrix. Further, biological processes changed, with evidence of increased apoptosis, low levels of inflammation and increased vascular permeability, and different genetic expression patterns with a reduction in mitochondrial protein synthesis. Of note, the increased vascular permeability, as measured using contrast MRI specifically, was only altered for the cervical spinal cord. Morales et al. 152 (1987) using electrophysiology in young and aged cats, identified increased resistance and reduced conduction velocity in alpha-motor neurons.
On a biomechanical level, these changes will alter the material properties and the response to loading of the spinal cord. Sparrey et al. (2009) explored this using a finite-element model of thoracic spinal cord compression. They demonstrated that changes to both white and gray matter structures increased measured mechanical stress, with changes to the white matter most significant. 153 Okazaki et al. 116 (2018) comparing young and old bovine spinal cord specimen demonstrated different tensile and compression stress patterns, in particular, during compression modeling within young specimen, stress was largely confined to white matter structures, whereas in older specimen distributed throughout the spinal cord.
Understanding the significance of age is complicated by the emerging distinction between chronological, that is, the passing of time, and what is termed biological age, that is, the presence of specific aging hallmarks within cells. 154 A number of markers of biological age, including epigenetic expression patterns, have been determined. 155 Whilst their assessment is further complicated by the observation that biological age can differ between tissue or organ systems, 156 accelerated biological aging is now linked to numerous diseases of the central nervous system and a potential therapeutic target. 157 This distinction perhaps aligns with clinical practice, where, for example, frailty is a better indicator of morbidity than age in DCM. 158
Cardiovascular System Effects on the Spinal Cord
Ischemia and/or microvascular changes within the spinal cord are a common feature in pre-clinical models and autopsy specimen of DCM. Ischemia due to a perfusion deficit 74 possibly as a result of microvascular changes 22 has been proposed as a common final pathway of critical compression. 23 Ellingson et al. 159 (2019) using MRI to evaluate spinal cord perfusion in a cohort of cervical spondylosis with and without myelopathy, demonstrated that neurological function using the modified JOA (mJOA) was inversely correlated with oxygen extraction.
The importance of maintaining perfusion is better demonstrated in traumatic spinal cord injury, where it is considered an important neuro-protective strategy.160,161 Spinal cord perfusion is physiologically maintained through autoregulation; however, spinal cord damage can cause its dysregulation.162,163 This has been demonstrated in pre- or non-operative 164 and peri-operative DCM patients.165-167 Hukude et al. (1988) using a canine model of DCM, demonstrated differing features of spinal cord injury in the context of high, normal, and low systemic blood pressure, across a range of compression thresholds. 168 This is supported clinically more recently in studies, which have demonstrated that the presence of cardiovascular disease was negatively associated with functional outcomes following surgery,33,169 and one study demonstrating different signal intensity patterns within the spinal cord on MRI. 170 A prospective observational study of cord perfusion, in patients undergoing surgery for DCM is currently underway [NCT04345822]. 99
Cardiovascular disease, including atherosclerosis, acute coronary syndromes and hypertension, are common co-morbidities amongst patients with DCM. 171 The observed ischemia in the spinal cord however is unlikely a direct consequence of plaque or emboli; the incidence of spinal cord infarction is extremely low, 172 and in an autopsy study of 603 aged spinal cord specimen, Wang et al. 173 (1996) observed that atherosclerotic changes were extremely rare within the spinal cord or its radicular arteries, even in the presence of systemic atherosclerosis. Instead, a recent population study from Taiwan identified DCM as an independent risk factor for the occurrence of acute coronary syndromes, including myocardial infarction 174 suggesting a potentially consequential relationship of DCM on the cardiovascular system. This is supported by a series of observational studies in patients undergoing decompressive surgery, in which surgery led to a reduction in blood pressure, with some able to stop prior anti-hypertensive medication.175-177 Autonomic dysfunction is also demonstrated in cervical spondylosis without myelopathy and asymptomatic spinal cord compression,167,178,179 including a further population study from Taiwan reporting an increased incidence of arrythmia amongst those with cervical spondylosis. 180 Whilst studies have not conducted longitudinal observation in these cohorts, to explore any relationship to the subsequent onset of DCM, this would align with the idea of compensation and a vulnerability threshold, for which an already impaired and/or medicated cardiovascular system may have implications.
Gastro-Intestinal System Effects on the Spinal Cord
Biological processes and pathways are can be influenced by nutrition. In extreme circumstances, these can cause neurological disease directly, such as in the case of B12 or Folate deficiencies. However, this is likely to represent one end of a spectrum, with less significant deficiencies a contributory factor for example with implications for neural repair. 181 In clinical studies, there are increasing indicators for this. Nouri et al. 182 (2020) in a retrospective analysis of 725 patients undergoing surgery for DCM or degenerative cervical radiculopathy, demonstrated macrocytic anemia was more common in DCM, and its presence associated with poorer pre-operative neurological status . Further, Allam et al. 183 (2017) (61) conducted a randomized controlled study (N = 192), in patients declining surgery for DCM of Cerebrolysin, a mixture of amino-acids and peptides given via intramuscular injection, demonstrating neurological benefit for the treatment arm.
Related to nutrition, is the increasing interest within Neuroscience of the gut-brain axis, a system with bidirectional (central nervous system on gut, and gut on central nervous system [CNS]) significance on human function and disease. 184 This includes increasing interest in the role of the microbiota (the organisms that reside within the gut) and the microbiome (the genetic material of the microbiota) in a range of neurological diseases, including Parkinson’s Disease, Autism Spectrum Disorder, Multiple Sclerosis, Stroke 185 but also traumatic Spinal Cord Injury. 186 Mechanistically changes in the gut microbiota are principally hypothesized to influence the CNS as consequence of a differential production, expression and turnover of neural transmitters, or via modulation of inflammatory response. 184 Conversely modulation of the brain to gut function is partly a cause of the autonomic system which is frequently disrupted in disease of the spinal cord. Longitudinal studies in traumatic spinal cord injury demonstrate a significant change in the microbiota following injury. 186 This autonomic disruption may also have wider implications for the gastro-intestinal system, including organs that direct metabolism (such as the liver, adrenal glands, and adipose tissue) but also mediate the immune response (such as the spleen). 187 Altered metabolism itself is linked to an altered immune response; with so called meta-inflammation, a pro-inflammatory state. 188 Inflammation is emerging as a key mediator of disease and disability in DCM.189-192 In a chronic disease such as DCM, one could therefore speculate that there is a further area of evolving interaction; that gastro-intestinal dysfunction due to DCM could modulate their spinal cord biology. In support of this hypothesis, Nouri et al. 193 (2020) in a secondary analysis of the AO Spine series observed that patients with gastro-intestinal co-morbidities had equivalent disease severity as measured using the mJOA, but different patterns of disease on conventional MRI, including the frequency and nature of signal intensity changes.
Neurological System—Adaptive Neuroplasticity and Redundancy to Increase Resilience
Injury to the cervical spinal cord causes structural and functional connectivity changes within the brainstem and cerebrum that may include adaptive neuroplasticity.90,194,195 Studies in DCM, principally using advanced MRI techniques,196-202 but also transcranial magnetic stimulation, 203 have demonstrated that the presence and nature of these changes can represent clinical symptoms, including pain and neuromuscular function, and predict response to surgery. Principal regions of interest have been the visual pathways (hypothesized to represent an increase reliance on vision for balance)201,204 and supplementary motor areas.195,203,205 For example, Zdunczyk et al. 203 (2017) using transcranial magnetic stimulation of the motor cortex, demonstrated that a greater neuromuscular function (defined using the JOA) was associated with a steeper recruitment curve and activation of supplementary motor areas, and proposed the concept of corticospinal reserve capacity.
This concept has been proposed across a range of neurological diseases to account for disconnects between histological lesion burden and clinical phenotype. 206 Whilst a number of different terms have been used to describe this,187,203 it is increasingly talked of as the “reserve capacity.” 207
This mirrors what is observed in DCM; cervical spinal cord tissue injury in DCM is initially asymptomatic2,40 and the radiological changes affecting the spinal cord, at best offer a partial surrogate for the disease phenotype.31,208,209
Conclusion
There is a clear need to improve understanding of the pathophysiological mechanisms and their clinical correlates for DCM. Resolution of such uncertainty is key to optimizing both management using current therapies and to inform the development of new ones. Based on the current evidence we propose DCM should be considered a function of mechanical stress, vulnerability, and time. Mechanical stress encompasses multiple mechanisms of loading, and not simply compression, whilst vulnerability relates to primary protective mechanisms such as genetic makeup and age, as well as adaptive protective mechanisms, such as autoregulation, functional reserve capacity, and nutritional status [Figure 7]. These concepts remain theoretical at this stage, and areas for further investigation. While our model is an oversimplification of a complex and multifactorial problem,
33
we hope that it will inspire a fresh view on DCM and encourage new avenues of research to better encapsulate the full spectrum of clinical disease. This is critical to optimizing both current management and informing the development of new therapies.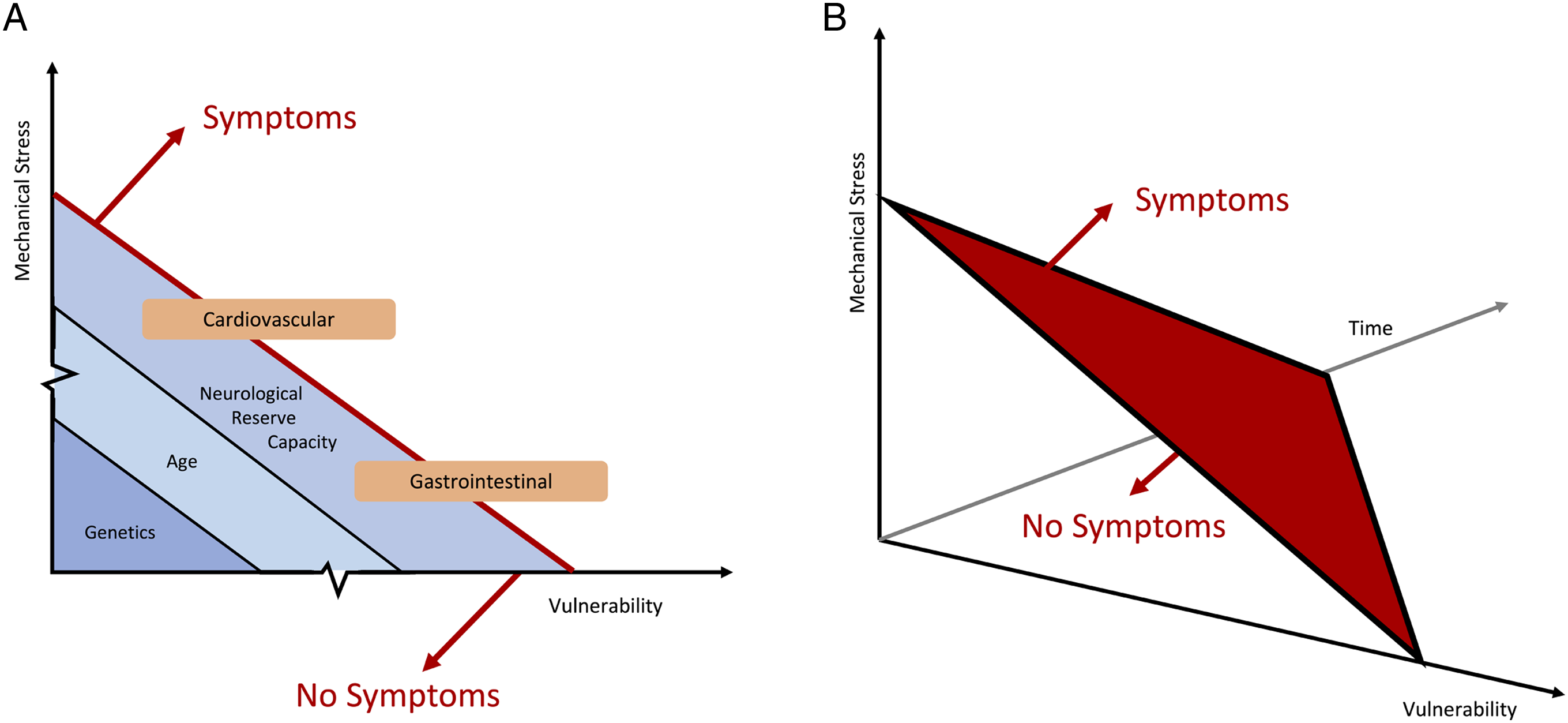
Footnotes
Acknowledgments
Further details on this priority, including how it was prioritized, why it was prioritized, and on-going research activity can be found at ![]() .
.
We thank Dr Robert Farr (London Institute, UK) and Prof. Michael Sutcliffe (University of Cambridge) for support with regards to the physical models. We thank Aimee Hutchinson [www.aimeehutchinson.com], Medial Illustrator, for the anatomical drawings of the cervical spine, and Iwan Sadler (![]() ) for assistance with producing the figures.
) for assistance with producing the figures.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research priorities were organized and funded by AO Spine through the AO Spine Knowledge Forum Spinal Cord Injury, a focused group of international Spinal Cord Injury experts. AO Spine is a clinical division of the AO Foundation, which is an independent medically-guided not-for-profit organization. Study support was provided directly through the AO Spine Research Department.MRNK is supported by a NIHR Clinician Scientist Award and BMD a NIHR Clinical Doctoral Research Fellowship. P.F. is funded by a SNF Eccellenza Professorial Fellowship grant (PCEFP3_181362/1)VFJN is supported by an Academy of Medical Sciences / The Health Foundation Clinician Scientist Fellowship. This report is independent research arising from a Clinician Scientist Award, CS-2015-15-023, supported by the National Institute for Health Research. The views expressed in this publication are those of the authors and not necessarily those of the NHS, the National Institute for Health Research or the Department of Health and Social Care. JB was supported by the Czech Health Research Council grants NV18-04-00159 and by the Ministry of Health of the Czech Republic project for conceptual development in research organizations, ref. 65269705.
