Abstract
Study Design:
Prospective cohort study.
Objective:
To evaluate whether pre-existing adjacent spinal canal stenosis (SCS) is associated with short-term outcomes after lumbar fusion surgery.
Methods:
We included patients with lumbar spinal stenosis treated surgically between July 2015 and December 2017 at 4 centers. All patients had the same pathology, with L4-S1 as the culprit sections. Patients were divided into 2 groups based on the cerebrospinal fluid occlusion sign on MRI at the adjacent L3/4 level. Patients without SCS (grade 0) and with mild SCS (grade 1) were classified into the non-stenosis (NS) and mild stenosis (MS) groups, respectively. All patients underwent PLIF and completed at least 1-year follow-up. The incidence of adjacent segment degeneration (ASDeg) and clinical outcomes were compared between the 2 groups.
Results:
A total of 308 patients (NS, 156; MS, 152) met the inclusion criteria. The incidence of ASDeg in the NS group (n = 40, 25.6%) was significantly lower than that in the MS group (n = 74, 48.7%; P < .001). The most frequent type of ASDeg in the 2 groups was the SCS-aggravated type. No significant difference was observed in adjacent segment disease incidence between the 2 groups (P = .243). The NS group had better outcomes according to the clinical function scores (P < .05).
Conclusions:
The cerebrospinal fluid occlusion sign on MRI is valuable for evaluating the adjacent segment with pre-existing degeneration. Patients with mild SCS in adjacent segments were more likely to have ASDeg, and the most frequent type of ASDeg was the SCS-aggravated type at early follow-up.
Keywords
Introduction
Adjacent segment pathologies, including radiological adjacent segment degeneration (ASDeg) and adjacent segment disease (ASDis), have become the most vexatious complications after lumbar fusion surgeries.1,2 Several risk factors for adjacent segment pathologies, such as age,1,3,4 sex,1,5 pre-existing adjacent disc degeneration, 5 multilevel fusions,3,5 sagittal imbalance, 6 types of fusion, 4 facet tropism, and laminar inclination, 7 have been reported. As multilevel degeneration is common in lumbar degenerative disease, pre-existing adjacent degeneration makes the development of surgical strategy challenging, and determining the level where the decompression or fusion ends is often cumbersome. Important adjacent pre-existing pathological factors include disc degeneration, facets, and ligamentum flavum tropism (adjacent segment instability does not usually occur before surgery). Few studies have confirmed that pre-existing adjacent disc degeneration is not a risk factor for long-term ASDis8,9; pre-existing facet joint degeneration is associated with a risk of ASDis. 10 However, preoperative facet joint degeneration classification alone on CT could not affect the surgical strategy. Degenerative facet joints and hypertrophy of the ligamentum flavum can cause spinal canal stenosis (SCS). Narrowed adjacent spinal canal makes neural tissues more prone to compression and is a key concern for a spinal surgeon. Few studies have focused on the effect of asymptomatic pre-existing SCS on ASDis that requires additional surgery at an adjacent segment.11-13 However, these studies only used the sagittal diameter or ratio to evaluate the degree of adjacent SCS, and the status of neural tissues in the canal was neglected. There is no consensus on the correct strategy to evaluate the degree of spinal stenosis in adjacent segments using cross-sectional MR images. Moreover, the effect of the degree of spinal stenosis on the imaging and clinical manifestations during early follow-up is unclear.
The present multicenter prospective study was limited to patients with the same preoperative pathology, the same fusion segments, and the same fusion technique. We used cerebrospinal fluid (CSF) occlusion signs to evaluate the degree of adjacent central canal stenosis, 14 describe the short-term radiological alterations in the adjacent segment and evaluate the effects of pre-existing SCS on early postoperative outcomes.
Methods
This study was approved by the relevant institutional ethics committee and was performed in accordance with the principles of the Declaration of Helsinki (IRB00006761-2015150). All volunteers were fully informed about the methods, purposes, and risks involved, and all patients provided signed informed consent. This was a prospective study of patients who underwent posterior lumbar interbody fusion (PLIF) for lumbar spinal stenosis from July 2015 to December 2017 at 4 centers. The inclusion criteria were (1) a clear diagnosis of lumbar spinal stenosis and surgical levels of L4-5 and L5-S1 (the levels to be operated on were decided by matching the clinical symptomatology with the radiological findings of the spinal levels that needed decompression) and (2) unsuccessful conservative treatment for at least 8 weeks. The exclusion criteria were (1) unstable factors at adjacent segment L3/4 (vertebral slippage, rotation, intervertebral angulation, etc.); (2) coronal deformity >10°, sagittal vertebral axis >50 mm; (3) lumbar infection and/or tumor; and (4) a history of lumbar fusion surgery.
Imaging Evaluation and Patient Groups
Dynamic radiography and MRI of the lumbar spine were performed to evaluate the imaging manifestations of the culprit and adjacent segments before surgery and at the final follow-up. The T2-weighted sagittal and axial MRI images were studied, and the following parameters were recorded: (1) grade of CSF occlusion, as described by Lee, 14 in the L3/4 spinal canal at the disc level (the narrowest axial plane was used for grading) (Figure 1); (2) L3/4 disc degeneration by Pfirrmann grade 15 ; (3) high-intensity zone (Hiz) of L3/4 disc; and (4) L3/4 disc herniation as quantified by MSU classification, 16 and the range of motion of the L3/4 segment was defined as the sum of the intervertebral disc angles in the flexion/extension view. 12 All measurements were performed using Centricity RIS/PACS software (GE Healthcare, Mount Prospect, IL).
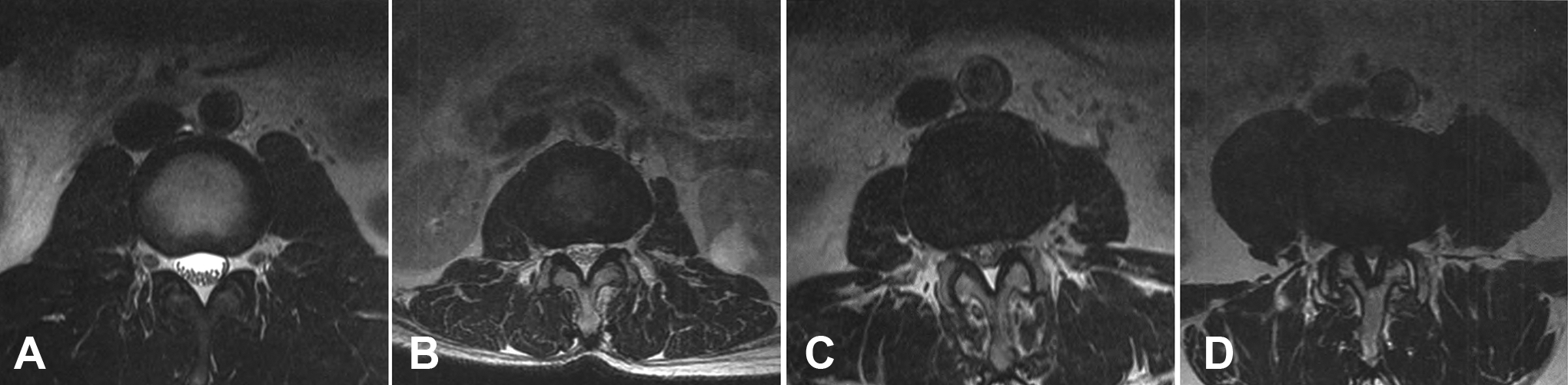
Schematic and axial T2-weighted MRI demonstrating the grading scale for the adjacent segment of lumbar central stenosis according to the obliteration of the anterior CSF space. (A) Stenosis that does not eliminate the CSF anterior to the cauda equina on axial T2-weighted images is graded as 0; (B) stenosis that results in the absence of CSF anterior to the cauda equina but still allows the separation of the cauda equina is considered mild or grade 1; (C) stenosis that leads to some cauda equina bunching is considered moderate or grade 2; (D) stenosis that leads to the absence of space between the elements of the cauda equina is considered severe or grade 3.
Patients were divided into groups according to the preoperative degree of L3/4 SCS, represented by CSF occlusion. To avoid intraobserver bias, images were reviewed by 2 senior spine surgeons at each center. If they disagreed, a third was invited to make a final decision. Patients without SCS (grade 0) at the L3/4 segment were added to the non-stenosis group (NS group), and patients with mild SCS (grade 1) at the L3/4 segment were added to the mild stenosis group (MS group). The imaging manifestations of the L3/4 disc on MRI were not used to group the patients.
Surgical Indications and Procedures
Surgical indications for fusion included segmental instability, such as spondylolisthesis, or predictable instability due to excessive removal of the facet joints. We used the posterior midline approach uniformly for all patients. All patients underwent internal pedicle screw fixation and a decompressive laminectomy. The lateral extent of decompression was considered complete when the traversing nerve roots were observed. During decompression, the lamina and spinous processes of the fused cranial vertebra were partially retained to preserve the connection between the posterior ligament complex and the neighboring spinous process. The capsule of the cranial facet joint was also protected during the surgery. An autologous graft with a PEEK cage was obtained from the decompressed lamina and processed. Mobility was restored in all patients within 3 to 5 days after surgery. The patients could exercise the back muscles 3 weeks after surgery and wore a lumbar protective band for 3 months after surgery.
Data Collection and Follow-Up
The patients’ age, sex, preoperative BMI, and American Society of Anesthesiologists classification of anesthesia (ASA grade) were recorded during the hospital stay. Surgical data, including operation time, blood loss, perioperative complications (including CSF leakage, wound infection, postoperative neurological dysfunction, perioperative secondary surgery, cardiopulmonary complications, cerebral infarction/hemorrhage, etc.), and length of hospital stay was also recorded. Preoperative clinical function questionnaires, including the visual analog scale (VAS) of the lower back, VAS of the legs, and Japanese Orthopedic Association (JOA) and Oswestry Disability Index (ODI) scores for patients were completed on admission for surgery without assistance. Between October and December 2018, all enrolled patients were followed-up for at least 1 year from the date of surgery. Questionnaires were completed in each hospital’s outpatient room at the final follow-up.
Definition of ASDeg and ASDis
ASDeg, a radiological finding, was defined as follows according to manifestations on plain radiography and MRI: (1) progression ≥1 grade in SCS; (2) alteration of L3/4 disc degeneration, herniation, or Hiz sign; and (3) progression of ≥3 mm in vertebral slippage in comparison with preoperative lateral radiographs, a change of ≥10° in the intervertebral angle on flexion and extension lateral radiograph, or scoliosis deformity in the coronal plane. ASDis, considered as a clinical finding, was diagnosed when clinical symptoms such as leg pain or intermittent claudication were found to have deteriorated during a postoperative follow-up, and the culprit lesion was confirmed to be adjacent to the fused level on MRI and radiography.
Statistical Analysis
A modified Kolmogorov-Smirnov test was used to test for normally distributed data. Descriptive statistics of the study population, including the mean, standard deviation, median, interquartile range (IQR), and percentage, were determined. The interobserver reliability of the preoperative degree of L3/4 SCS was calculated using the intraclass correlation coefficient (ICC). The chi-square test and Fisher’s exact test were used for group comparisons. The VAS, ODI, and JOA scores between the NS and MS groups were compared using the Mann-Whitney U test. Within each group, the type of ASDeg was analyzed, including the adjacent SCS-aggravated type, disc factors (herniation or newly presented Hiz), adjacent segment instability, and mixed type. Statistical significance was set at P < .05.
Results
Between July 2015 to December 2017, 308 patients (144 men and 164 women) met the inclusion criteria, with a mean age of 57.4 ± 11.2 years. The L3/4 canal stenosis grade was identified with good reproducibility. The interobserver mean ICC for each center was 0.85, 0.89, 0.92, and 0.88, respectively. There were 156 patients in the NS group and 152 in the MS group. No significant differences were observed in sex, BMI, and ASA grade between the 2 groups before surgery. The median age of the NS group was 54.5 (IQR 15.8) years, and that of the MS group was 62 (IQR 12) years, and the differences were statistically significant (P < .001). With respect to the preoperative Pfirrmann grade, the ratio of ≤2 to >2 was not significantly different between the NS group (53:103) and MS group (46:106; P = .486). Patients in the MS group had longer hospital stays than those in the NS group. No statistically significant differences were observed in preoperative clinical function scores, intraoperative blood loss, operation time, operative complication rate, and follow-up time between the 2 groups (Tables 1 and 2).
Comparisons of Demographic and Preoperative Data.
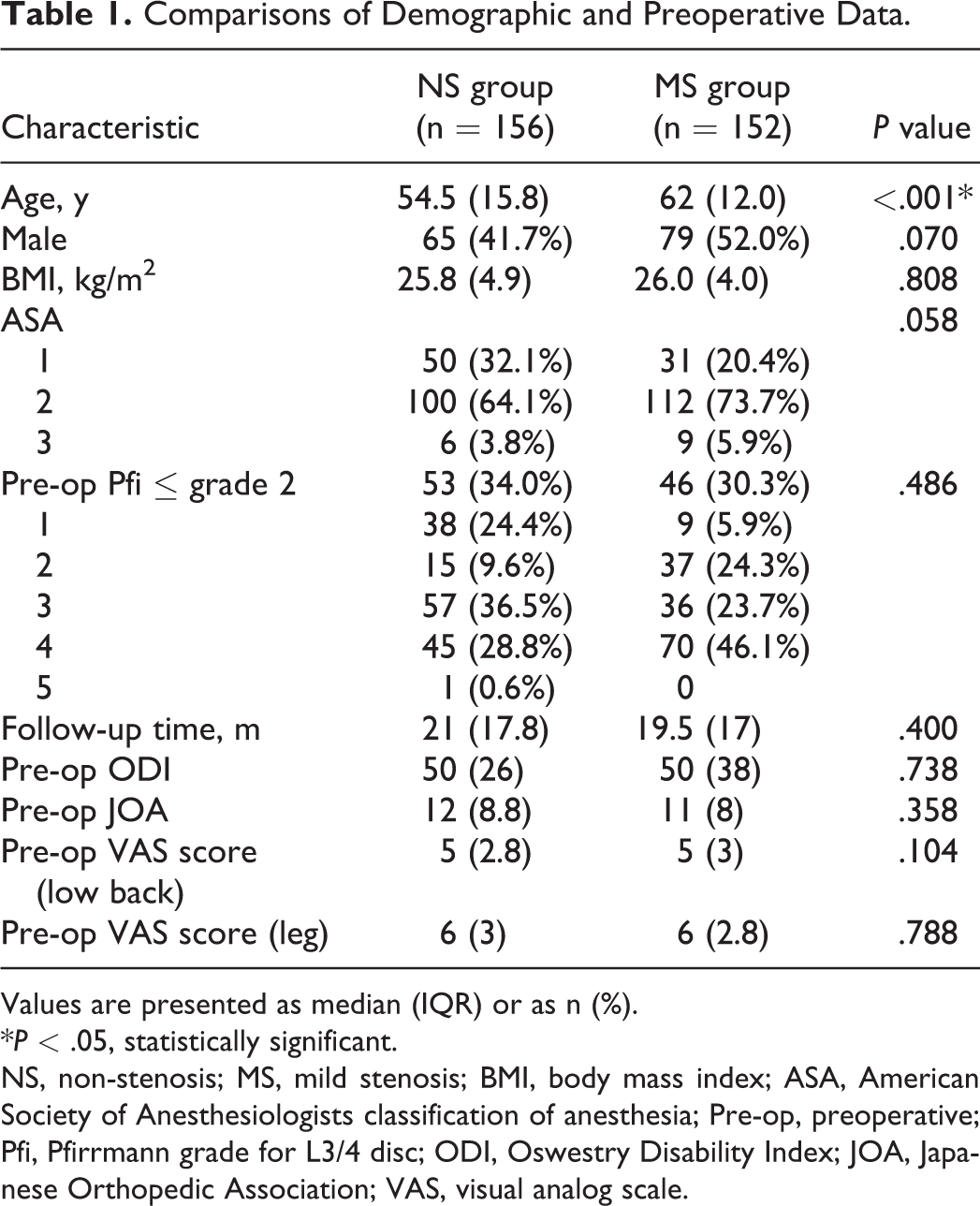
Values are presented as median (IQR) or as n (%).
*P < .05, statistically significant.
NS, non-stenosis; MS, mild stenosis; BMI, body mass index; ASA, American Society of Anesthesiologists classification of anesthesia; Pre-op, preoperative; Pfi, Pfirrmann grade for L3/4 disc; ODI, Oswestry Disability Index; JOA, Japanese Orthopedic Association; VAS, visual analog scale.
Comparisons of Perioperative Data Between the 2 Groups.
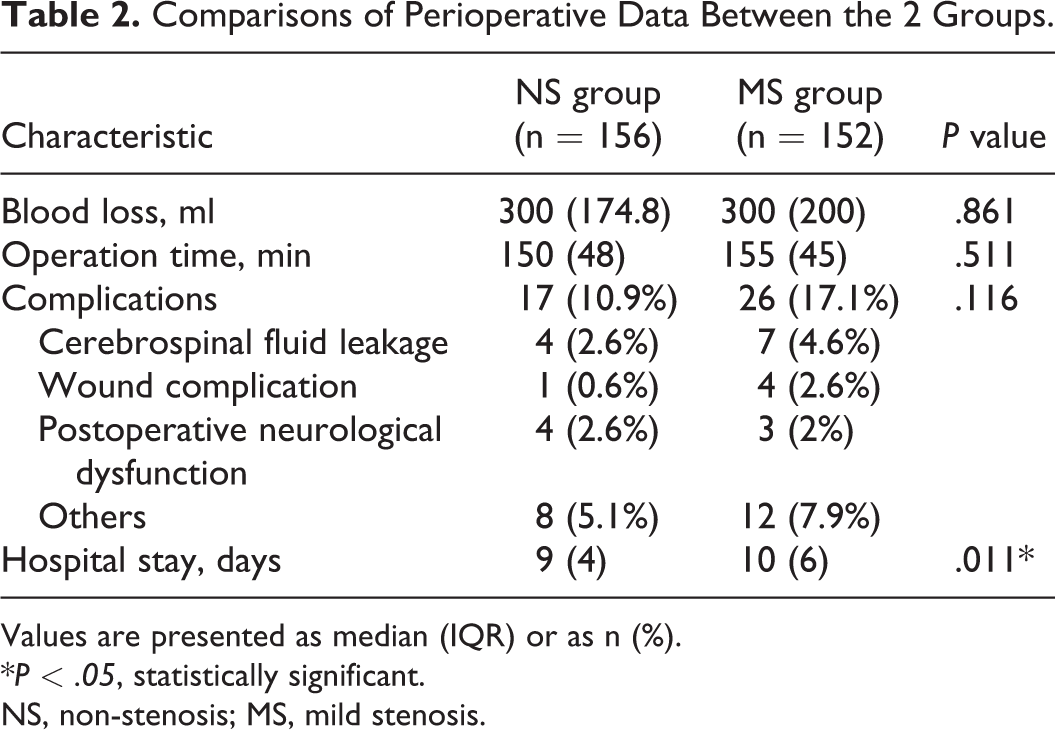
Values are presented as median (IQR) or as n (%).
*P < .05, statistically significant.
NS, non-stenosis; MS, mild stenosis.
At the final follow-up, pseudarthrosis and internal fixation breakage were absent in all cases. There were 40 cases of ASDeg in the NS group (25.6%) and 74 in the MS group (48.7%). The incidence of ASDeg in the NS group was significantly lower than that in the MS group (P < .001). Among the patients who developed ASDeg, adjacent SCS deterioration was the most common type, which accounted for 30 (19.2%) cases in the NS group and 63 (41.4%) cases in the MS group (Table 3, Figures 2 and 3). As for the incidence of ASDis, there were no cases of ASDis in the NS group and 2 (1.3%) in the MS group, and the difference was not significant (p = 0.243). No patients underwent reoperations at the adjacent level. The median ODI and VAS scores of the lower back in the NS group were 6 (IQR 10.8) and 1 (IQR 2), respectively, and were significantly less than those in the MS group (6.7 [IQR 22] and 1 [IQR 3], respectively) at the final follow-up (P < .05). The JOA score in the NS group was significantly higher than that in the MS group (P = .015). Overall, the NS group had better outcomes, as evident from the clinical function scores.
Comparisons of Follow-Up Data Between the 2 Groups.
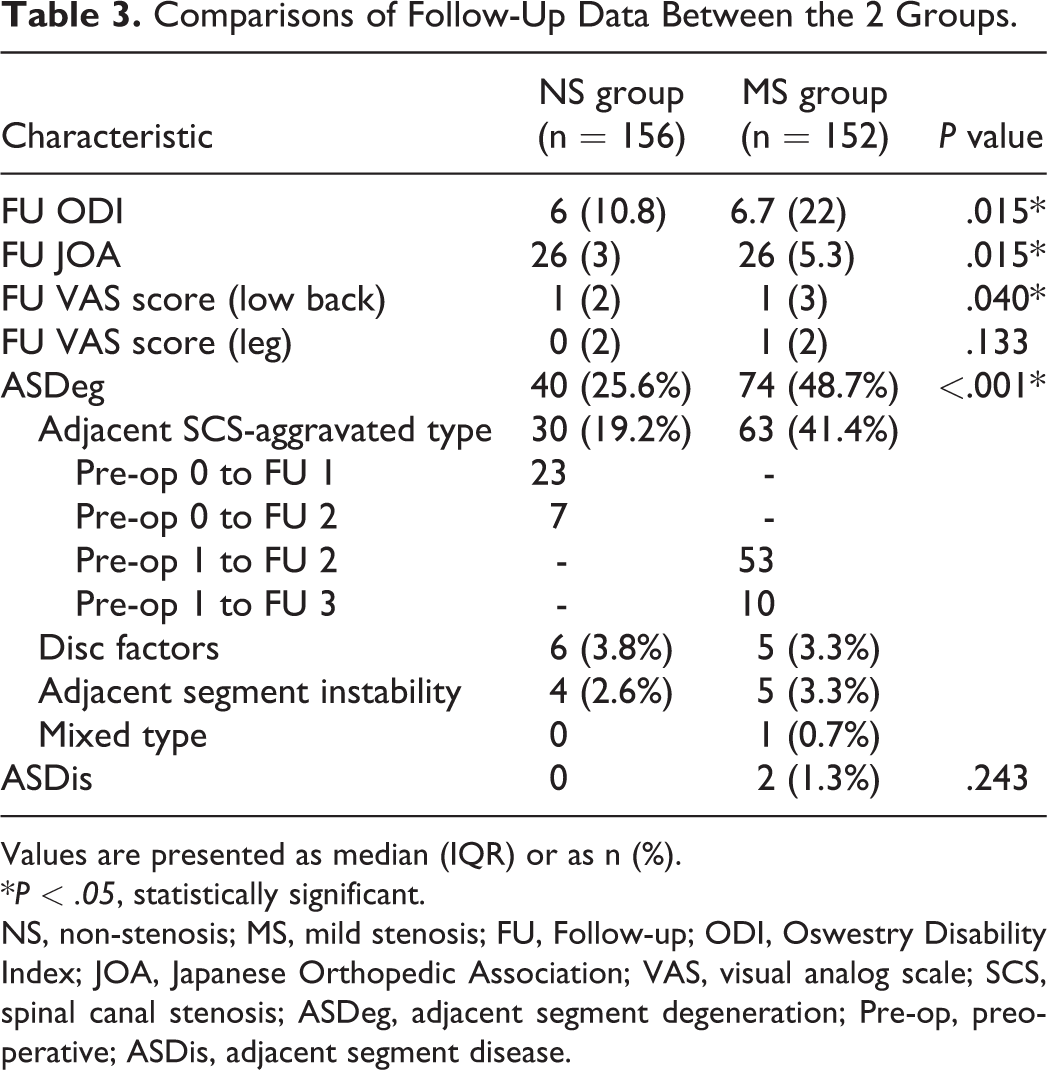
Values are presented as median (IQR) or as n (%).
*P < .05, statistically significant.
NS, non-stenosis; MS, mild stenosis; FU, Follow-up; ODI, Oswestry Disability Index; JOA, Japanese Orthopedic Association; VAS, visual analog scale; SCS, spinal canal stenosis; ASDeg, adjacent segment degeneration; Pre-op, preoperative; ASDis, adjacent segment disease.
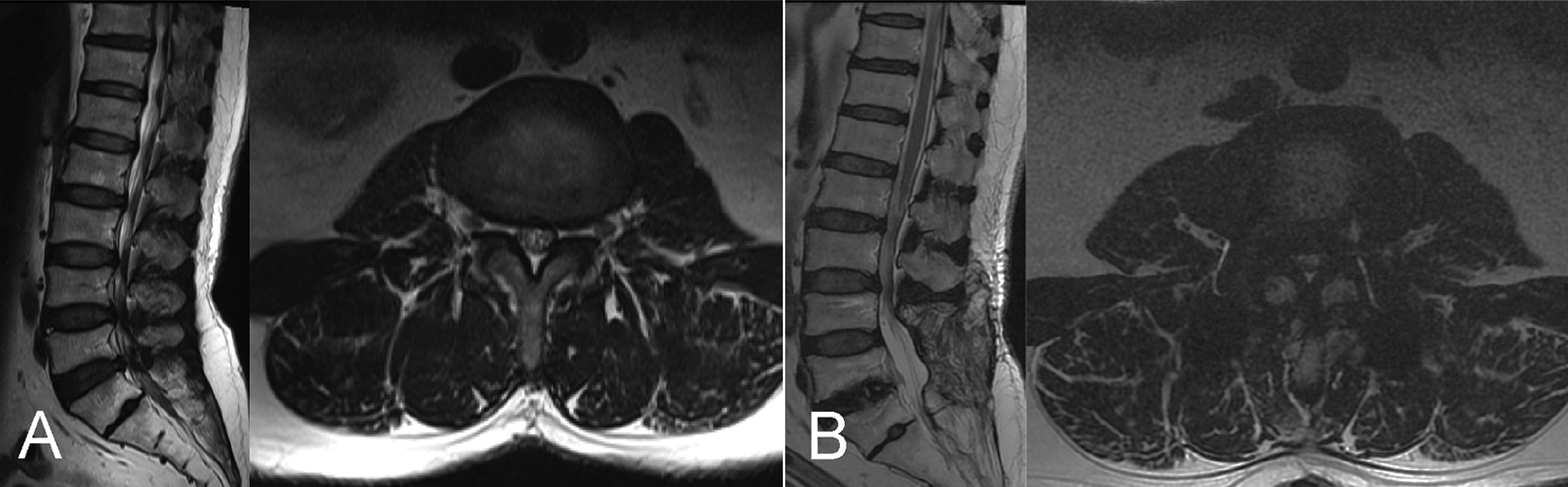
Preoperative and final follow-up sagittal MRI and adjacent L3/4 segment axial MRI of a 56-year-old male patient with L4/S1 segment lumbar spinal stenosis in the MS group. The preoperative SCS was grade 1 (A); SCS aggravation led to grade 3 at the 24-month follow-up, and no ASDis was noted (B).
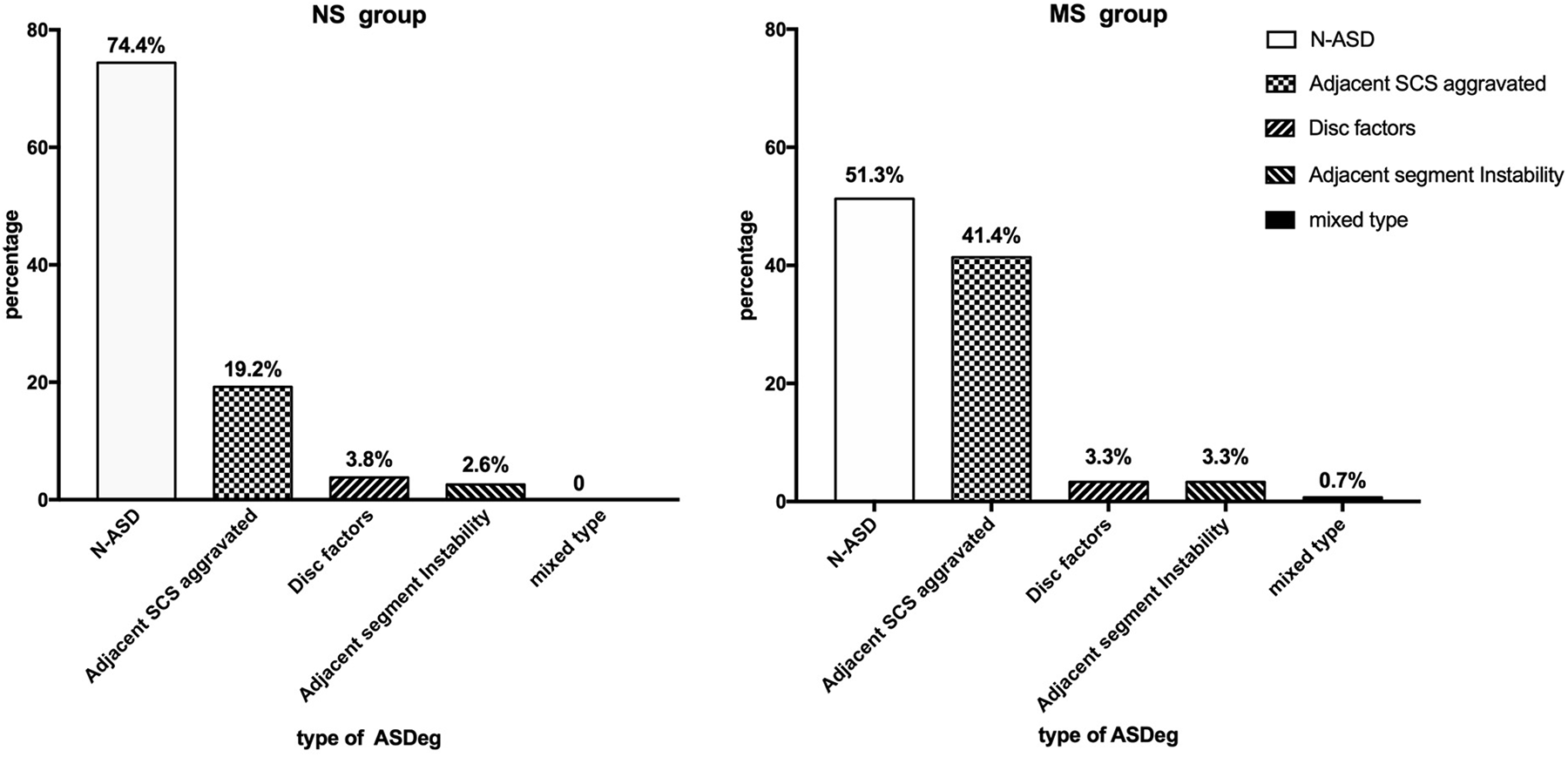
The types of ASDeg in the NS and MS groups.
Discussion
The influence of pre-existing adjacent SCS on clinical efficacy has been rarely mentioned in previous studies. Because the pathological factors of adjacent segment revision surgery were mostly due to adjacent SCS aggravation,17-19 it was necessary to explore the influence of pre-existing adjacent SCS. Even in decompression-only and non-fusion lumbar surgeries, the presence of pre-existing adjacent SCS significantly increased the risk of reoperation. 13 Therefore, it is necessary to develop a grading system to evaluate pre-existing adjacent SCS in lumbar fusion surgeries. As evident from the literature, Cho et al 11 and Zhong et al 20 reached this same conclusion. Although these studies assessed the adjacent SCS by MRI, they did not mention specific methods and did not use any grading system. With the limitations of using the dural sac cross-sectional area or anteroposterior diameter to grade lumbar SCS,14,21 some previous studies mentioned the use of methods in the sagittal plane to evaluate pre-existing adjacent SCS. Yugué et al 12 found that pre-existing SCS at the adjacent segment on myelography was a significant risk factor, while such pathology observed by MRI was not a risk factor in another study. 22 We believe that this was because they measured the anteroposterior diameter or ratio of the dural sac in the sagittal plane to determine the presence of SCS of the adjacent segment. The effect of stenosis on neural tissues was prone to underestimation. In this study, we used the degree of CSF occlusion observed in the axial plane of the MRI to determine the degree of SCS. Because the narrowest axial plane was used for classification, the underestimation of stenosis in the sagittal plane could be effectively avoided. The results of our study showed that patients with mild SCS at the adjacent segment before the index surgery were more likely to develop ASDeg than those without this condition in the short-term follow-up. If the adjacent spinal canal is narrowed, neural tissues are more prone to compression, which is of serious concern for a spinal surgeon. We believe that using this method to evaluate the adjacent segments has great clinical value.
The imaging findings of ASDeg at the early follow-up were inconclusive and were closely related to the evaluation methods of ASDeg. In a retrospective study, 50 patients undergoing PLIF were followed up for at least 2 years. Of these, 24 patients developed ASDeg, 11 of whom had disc-space collapse as determined by Pfirrmann grade V on MRI, which was the most common type of ASDeg. Although the authors of the above study performed MRI at the final follow-up, the SCS at adjacent segments on MRI was not evaluated. 23 Hikata et al 24 retrospectively analyzed 54 patients who underwent PLIF with or without simultaneous decompression at the adjacent segment. Early follow-up (at least 2 years) results showed that ASDeg occurred in 31 patients; among these patients, 16 had disc-space narrowing of more than 3 mm as determined by radiography, and this was the most common type of ASDeg. However, Lee et al 25 followed 115 patients who underwent L5 floating fusion for at least 2 years and noted that the cephalad segment ASDeg was related to spondylolisthesis (n = 14, 56%), and caudal segment ASDeg mainly occurred due to disc degeneration (n = 6, 75%). As a consequence of the above findings, the most common type of ASDeg remains controversial. It is worth noting that none of the above studies evaluated the SCS of adjacent segments. This is because the definition and diagnosis of ASDeg have advanced beyond simple radiographic indicators to comprehensive indicators from radiography, CT, and MRI. If radiography alone is used for diagnosis, disc degeneration can be indirectly judged by the narrowing of the disc space or by the formation of bone spurs (UCLA or Weiner grading system). However, since radiography cannot allow estimation of the degree of SCS of adjacent segments, the incidence of ASDeg can easily be underestimated. 26
Our study also focused on early follow-up results, and we adopted MRI indicators, especially SCS, to diagnose ASDeg. We classified ASDeg into 4 types: adjacent SCS-aggravated type, disc factors (herniation or Hiz sign), adjacent segment instability, and mixed type. The most frequent type of ASDeg was the SCS-aggravated type, which accounted for 75.0% of cases in the NS group and 85.1% of cases in the MS group. A previous study found that SCS was a pathology that tended to deteriorate naturally over time. 27 We believe that the high proportion of the cases with SCS-aggravated type in the MS group might be attributed to the natural progression of the disease. However, patients in the NS group had no SCS in the adjacent segments before surgery, and the proportion was also high. This led us to believe that SCS may aggravate at adjacent segments even in the early postoperative period, although it may not show any symptoms. Cheh et al, 26 in their study with a follow-up of at least 5 years, reported that “SCS-aggravated” was the most common type of ASDis accounting for 82.4% of cases; this was similar to that observed in the present study. Therefore, further studies are needed to determine whether such SCS aggravation might continue in the long term, or whether it could lead to ASDis. We believe the adjacent SCS-aggravated type is the most common type of ASDeg, which was overlooked in early ASD reports, however, requires more attention in future studies.
This study was a prospective multicenter cohort study that aimed to investigate whether pre-existing mild SCS of the adjacent segment before PLIF was a risk factor for ASDeg. Moreover, when designing this study, we limited the surgical method and the fusion segments, which also meant limiting the adjacent segment (L3/4) to be studied, and as a result, we excluded several other possible risk factors. This made the results of this study more reliable.
However, there are some limitations to our study. First, the patients in the NS group in this study were younger than those in the MS group, which introduced the confounding factor of age. However, this phenomenon may be explained by the fact that degenerative factors of adjacent segments are related to age. 5 Second, the follow-up time was relatively short. ASDis is a long-term complication and is time-dependent. 28 In our study, the difference in the incidence of ASDis between the groups was not statistically significant. This was thought to be due to the short follow-up period. We recommend that the effect of aggravation due to adjacent SCS on clinical symptoms should be closely monitored.
Conclusion
The CSF occlusion sign on MRI is a valuable method for evaluating the adjacent segment with pre-existing degeneration. Patients with mild SCS in adjacent segments before surgery are more likely to have ASDeg in the short-term follow-up, and “SCS-aggravated type” was the most frequent type of ASDeg.
Footnotes
Authors’ Note
Zhuoran Sun and Zhuofu Li contributed equally to this work. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee (Peking University Third Hospital Medical Science Research Ethics Committee, reference number: IRB00006761-2 015 150) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. All volunteers were fully informed about the methods, purposes and risks involved in the study protocol and provided their signed informed consent.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Capital’s Funds for Health Improvement and Research (2016-1-4096), National Natural Science Foundation of China (81 871 807).
