Abstract
Study Design:
Observational cohort study.
Objective:
To compare 1-year perioperative complications between structural allograft (SA) and synthetic cage (SC) for anterior cervical discectomy and fusion (ACDF) using a national database.
Methods:
The TriNetX Research Network was retrospectively queried. Patients undergoing initial single or multilevel ACDF surgery between October 1, 2015 and April 30, 2019 were propensity score matched based on age and comorbidities. The rates of 1-year revision ACDF surgery and reported diagnoses of pseudoarthrosis, surgical site infection (SSI), and dysphagia were compared between structural allograft and synthetic cage techniques.
Results:
A comparison of 1-year outcomes between propensity score matched cohorts was conducted on 3056 patients undergoing single-level ACDF and 3510 patients undergoing multilevel ACDF. In single-level ACDF patients, there was no difference in 1-year revision ACDF surgery (P = .573), reported diagnoses of pseudoarthrosis (P = .413), SSI (P = .620), or dysphagia (P = .529) between SA and SC groups. In multilevel ACDF patients, there was a higher rate of revision surgery (SA 3.8% vs SC 7.3%, odds ratio = 1.982, P < .001) in the SC group, and a higher rate of dysphagia in the SA group (SA 15.9% vs SC 12.9%).
Conclusion:
While the overall revision and complication rate for single-level ACDF remains low despite interbody graft selection, SC implant selection may result in higher rates of revision surgery in multilevel procedures despite yielding lower rates of dysphagia. Further prospective study is warranted.
Keywords
Introduction
Anterior cervical discectomy and fusion (ACDF) has emerged as the most commonly performed surgical treatment for degenerative cervical spine disease. 1 Each year, over 132 000 ACDFs are performed in the United States and volumes have increased 5.7% annually from 2006 to 2013. 2 Since early descriptions of anterior surgical approaches in the 1950’s, advancements in techniques, instrumentation, and implant materials have focused primarily on achieving more reliable fusion of unstable or symptomatic segments.3-5 Options for interbody grafts have evolved from tricortical iliac crest autograft to structural allograft (SA) or synthetic cages (SC) of various materials.
Autograft harvested from the iliac crest is biocompatible and nonimmunogenic and has been reported to have high fusion rates with relatively few incidences of graft complications.5-7 Hence, autograft is considered to be the gold standard for ACDF. However, in addition to a second surgical site with increased blood loss and operative time, the donor site is frequently reported to have complications such as pain, hematomas, seromas, infections, and fractures contributing to potential functional disability.8,9
The potential for graft site complications and additional operative time has led surgeons to increasingly substitute autograft with structural allograft or synthetic cages of various materials such as polyetheretherketone (PEEK) and titanium alloys.10,11 In a retrospective review examining geographic variation in anterior cervical fusion procedures, McGuire et al 12 demonstrated a marked decrease in autograft utilization from 86% in 1999 to 10% in 2008. During the same time period, they reported a significant increase in the use of structural allograft and synthetic cages from 14% to 59% and 0% to 31%, respectively. In a 2017 international survey of spine surgeons, PEEK cages (64.1%) were the most commonly used interbody implant worldwide. 11 While respondents universally agreed that achieving fusion was successful to a good clinical outcome, only half of respondents were satisfied with comparative effectiveness data available on graft materials.
While the use of structural allograft and synthetic cages for single- and multilevel ACDF has been individually reported in multiple studies with favorable outcomes,13-15 there are few direct comparisons of complications and rates of revision surgery between structural allografts and synthetic cages. Krause et al 16 recently reported that the use of PEEK implants in single-level ACDF is associated with significantly higher rate of pseudoarthrosis and need for revision surgery when compared with the use of structural allografts. In their study, 29 of 56 (51.8%) PEEK implants demonstrated radiographic evidence of pseudoarthrosis compared to 7 of 71 (10%) structural allograft. Furthermore, 7 patients from the PEEK group required revision (12.5%) while 1 patient with structural allograft required revision (1.4%). To increase sample size, other authors have utilized a large-scale administrative database (PearlDiver Patient Record Database) to compare outcomes between structural allograft and synthetic cages.17,18 Goz et al 17 included 17 000 patients from 2007 to 2014, while Pirkle et al 18 included 6130 patients from 2007 to 2016. After correcting for age, gender, comorbidity burden, and number of levels fused, Goz et al 17 reported that graft choice was not an independent predictor of complications, with an overall rate of revision surgery of <1% in both groups. In contrast, Pirkle et al 18 found a higher rate of revision surgery for synthetic cages (5.32%) compared with structural allograft (1.97%), and this difference remained significant after controlling for confounding variables such as levels fused and smoking status.
In an era of maximizing value, our objective was to further understand potential differences in one-year clinical outcomes between structural allograft and synthetic cages. We selected the TriNetX platform, a multicenter, longitudinal database, to obtain the most recent comparative data across a large patient sample.
Methods
This research was deemed exempt from institutional review board review by the institution’s clinical research committee. The TriNetX Research database was retrospectively queried as of May 22, 2020 to evaluate all patients undergoing single or multilevel ACDF surgery between October 1, 2015 and April 30, 2019. All patients had a minimum of one-year follow up data available, and the study time period aligned with the implementation of International Classification of Diseases 10th edition (ICD-10) to maintain consistency in code-based definitions. Patients were then grouped by receipt of structural allograft or synthetic cage. Analysis was performed using propensity score matched cohorts. Propensity score matching was performed based on age, race, gender, and the presence or absence of the following diagnoses within the year prior to ACDF surgery: cervical disc disorders; spondylosis; cervical radiculopathy; hypertension; chronic pain; anxiety, dissociative, stress-related, somatoform and other nonpsychotic mental disorders; diabetes mellitus, mood (affective) disorders; overweight or obesity; nicotine dependence; disorders of bone density and structure, and malnutrition. The primary outcome measure was revision ACDF surgery within 1 year of the initial procedure. Revision surgeries included ACDF, posterior decompression and fusion, posterior decompression without fusion and other procedures (osteotomy, posterior open treatment of fracture or dislocation, laminectomy for lesions and neoplasm, anterior decompression). Secondary outcome measures included reported diagnoses of pseudoarthrosis, surgical site infection (SSI), or dysphagia within the 1-year postoperative period. Adjacent segment disease was considered for evaluation as a secondary endpoint but excluded due to the lack of a specific ICD-10 code for this condition. Primary and secondary outcomes were identified by relevant diagnosis or procedure codes. A full list of definitions for the surgical procedures, comorbidities, and outcomes is presented in Table 1. All statistical analysis was performed using TriNetX Analytics. Significance was assessed at an alpha of .05.
Coding Definitions: CPT and ICD-10 Code Definitions Provided for Single-Level Structural Allograft and Synthetic Interbody Spacer, Multilevel Structural Allograft and Synthetic Interbody spacer, Patient Comorbidities, and Procedure Outcomes.
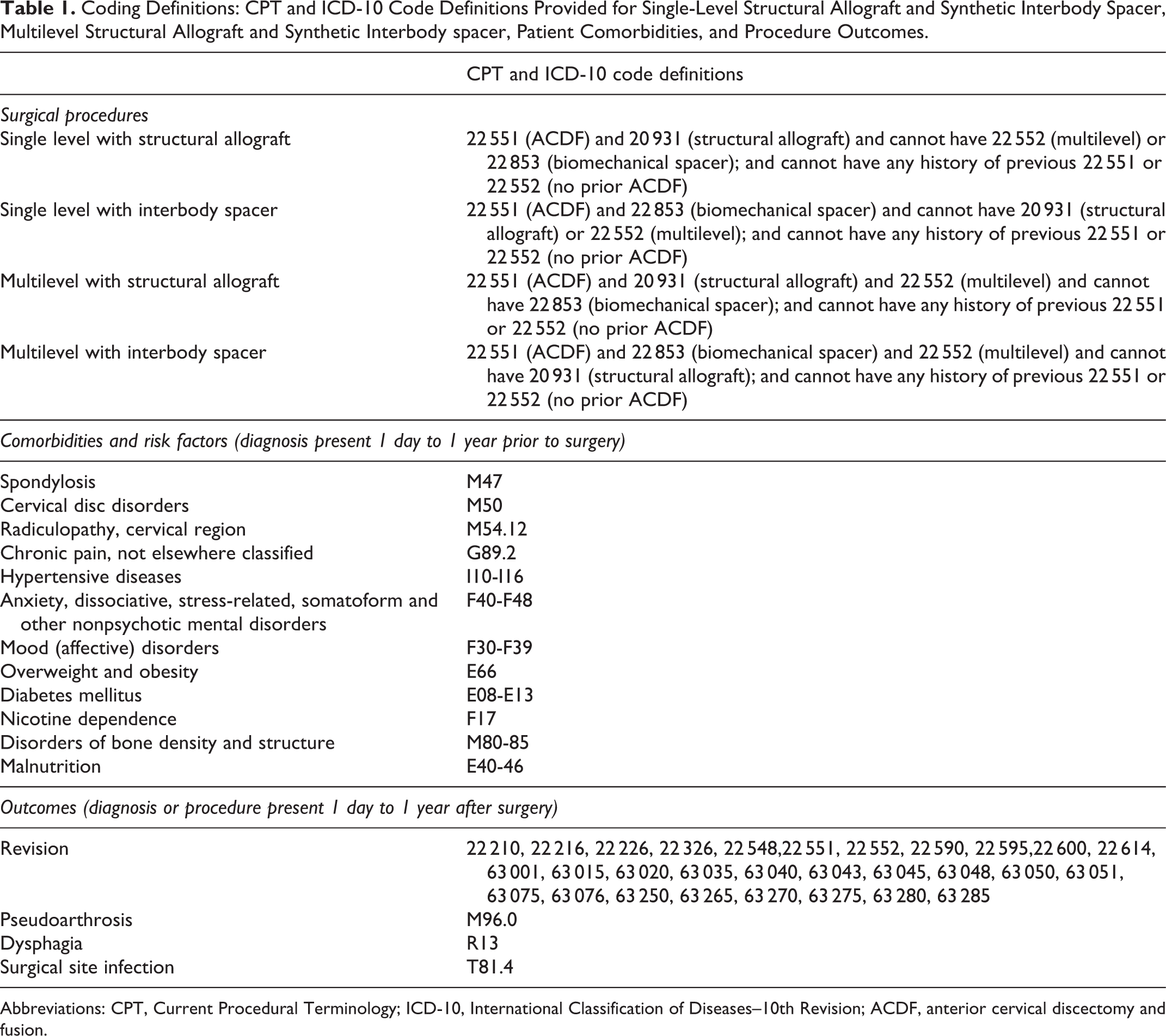
Abbreviations: CPT, Current Procedural Terminology; ICD-10, International Classification of Diseases–10th Revision; ACDF, anterior cervical discectomy and fusion.
About TriNetX
TriNetX is a “global health research network that optimizes clinical research and enables discoveries through the generation of real-world evidence.” 19 The research platform includes longitudinal data from 26 health care organizations and includes over 37 million patients. As a federated network, TriNetX received a waiver from Western Institutional Review Board since only aggregated counts, statistical summaries of de-identified information, but no protected health information is received, and no study-specific activities are performed in retrospective analyses. De-identified, HIPAA (Health Insurance Portability and Accountability Act) compliant electronic health record (EHR) data is collected from participating health care organizations who submit structured and unstructured data elements. On average, participants submit data retrospectively for 7 years, with some providing historical data 13 years or older. Variables captured include demographics, diagnoses (all mapped to ICD-10 coding) procedures (ICD-10 PCS and CPT), medications, lab values, and genomics information. Statistical analysis is performed within the analytics platform using parallel R and Python queries triangulated to maximize test accuracy. 19
Results:
A total of 8103 patients were included in this observational cohort study. Of the 3775 patients undergoing single-level ACDF, structural allograft was used in 2213 (59%) patients and synthetic cages in 1562 (41%) patients. Of the 4328 multilevel ACDF procedures, structural allograft (SA) was used in 2482 (57%) patients and synthetic cages (SC) in 1846 (43%) patients.
To control for differences in comorbidities across the sample, the cohorts were propensity score matched based on age, gender, race, and comorbid conditions. Prior to propensity score matching, patients undergoing single-level ACDF using structural allograft were significantly younger (SA 53.9 ± 12.9 vs SC 55.1 ± 13.0 years, P = .004), had higher rates of cervical disc disorders (SA 62.7% vs SC 59.0%, P = .020), higher rates of cervical radiculopathy (SA 43.6% vs SC 40.2%, P = .040), higher rates of nicotine dependence (SA 17.6% vs SC 10.7%, P < .001), lower rates of spondylosis (SA 49.9% vs SC 55.1%, P = .002), and lower rates of disorders of bone density and structure (SA 4.4% vs SC 6.7%, P = .002). After propensity score matching, no significant differences remained (Table 2). Prior to propensity score matching, patients undergoing multilevel ACDF using synthetic cage were significantly older (SA 55.8 ± 11.0 vs SC 57.8 ± 10.9 years, P < .001), more likely to be White (SA 79.2% vs SC 82.7%, P = .004) and had higher rates of cervical disc disorders (SA 58.2% vs SC 63.7%, P < .001), higher rates of chronic pain (SA 21.4% vs SC 27.2%, P < .001), higher rates of disorders of bone density and structure (SA 5.0% vs SC 7.7%, P < .001), lower rates of cervical radiculopathy (SA 50.7% vs SC 45.9% P = .002), lower rates of nicotine dependence (SA 17.0% vs. SC 12.0%, P < .001), and lower rates of malnutrition (SA 1.3% vs SC 0.5%, P = .018). After propensity score matching none of these differences remained (Table 2).
Patient Characteristics and Comorbidities Before and After Propensity Score Matching: Demographics and Comorbidities Data Provided for Single- and Multilevel ACDF With the Original Cohorts and Propensity Score Matched Cohorts.
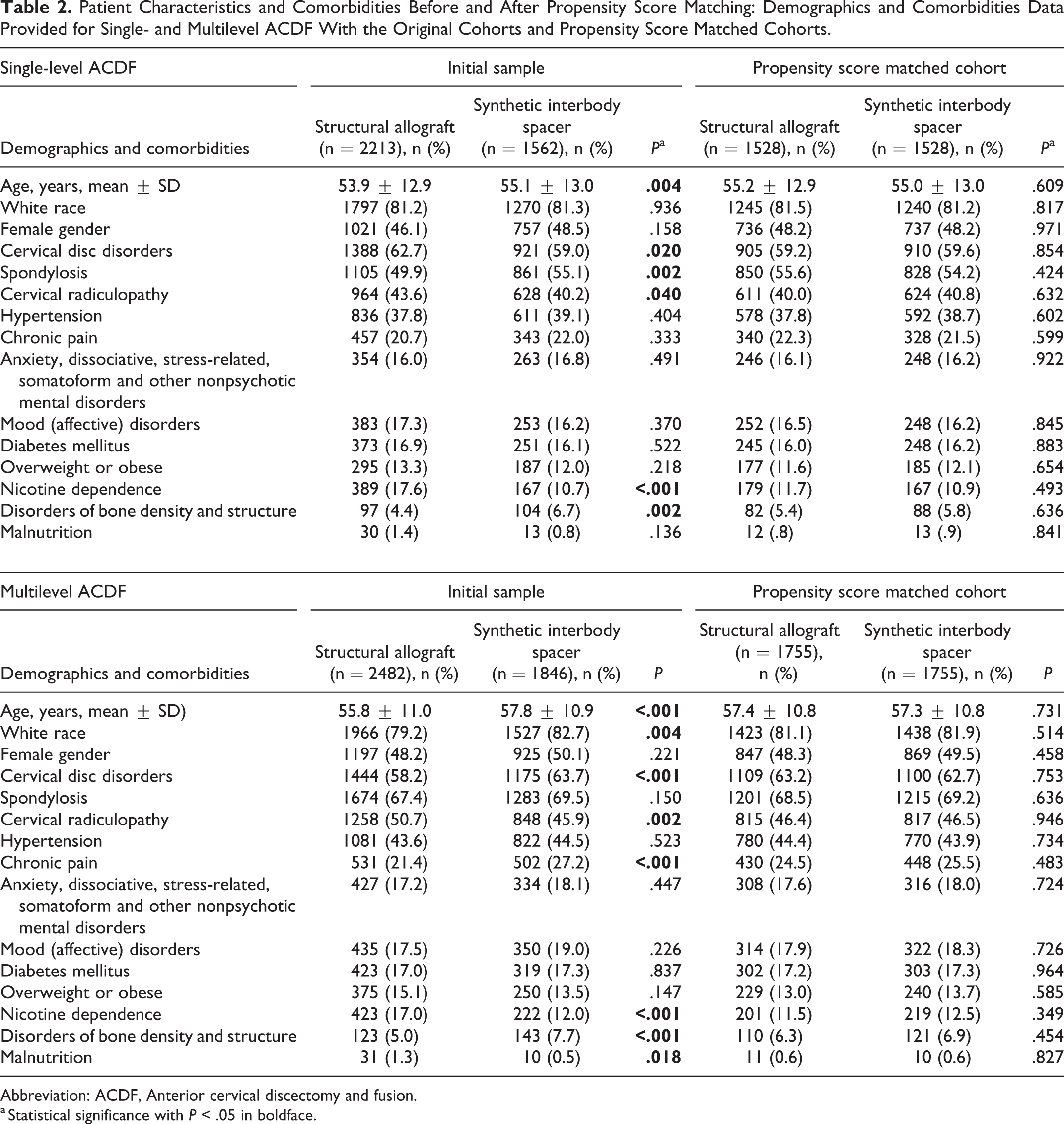
Abbreviation: ACDF, Anterior cervical discectomy and fusion.
a Statistical significance with P < .05 in boldface.
Following propensity score matching, 3056 patients underwent single-level ACDF, with 1528 (50%) patients in the structural allograft group and 1528 (50%) in the synthetic cage group. Comparing 1-year outcomes, there was no difference in the rate of revision surgery (P = .573) or reported diagnoses of pseudoarthrosis (P = .413), SSI (P = .620), or dysphagia (P = .529) (Table 3).
Propensity Score Matched Cohorts, 1-Year Outcomes: Data Provided for Propensity Score Matched Patient Cohorts With Single- and Multilevel ACDF Utilizing Structural Allograft or Synthetic Interbody Spacer.
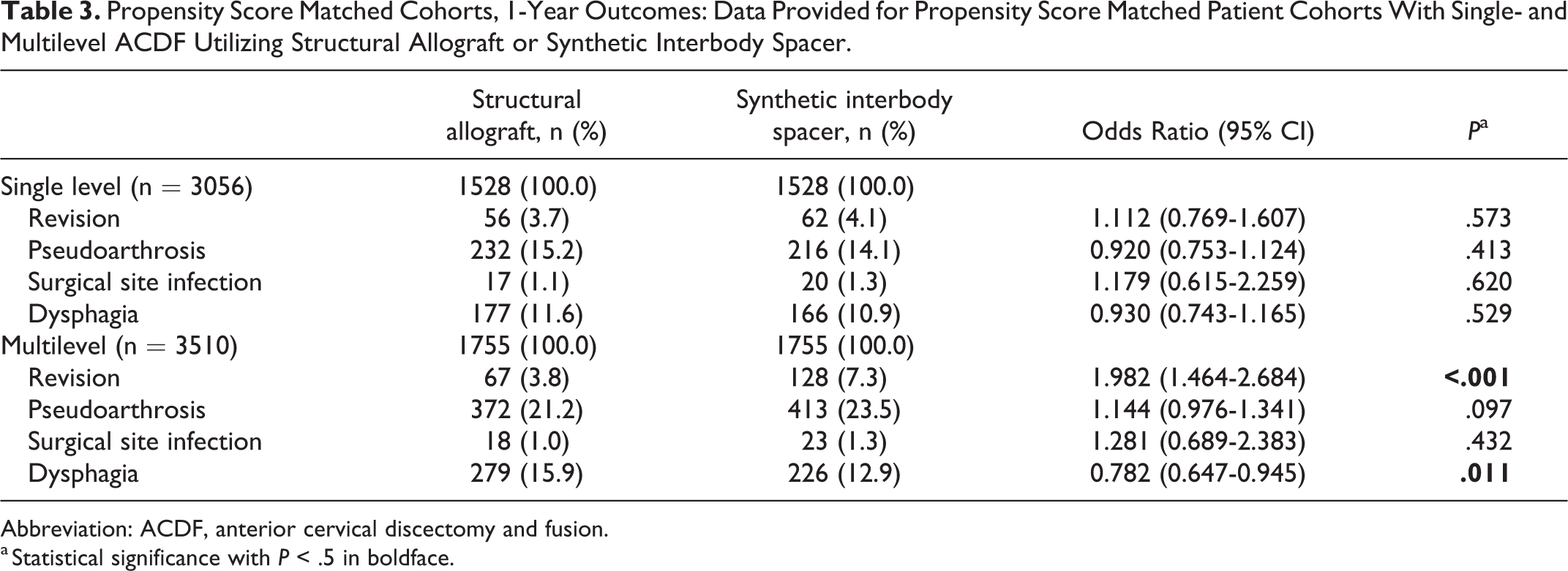
Abbreviation: ACDF, anterior cervical discectomy and fusion.
a Statistical significance with P < .5 in boldface.
A total of 3510 patients remained in the multilevel ACDF cohort following propensity score matching, with equal distribution among the SA and SC groups. For patients in the SC group, there was a higher likelihood of revision surgery (SA 3.8% vs SC 7.3%, OR = 1.982, P < .001) and lower likelihood of dysphagia (SA 15.9% vs SC 12.9%, OR = 0.782, P = .011). No significant differences in SSI (P = .439) or pseudoarthrosis (P = .097) were observed (Table 3).
Discussion
Outcomes following ACDF have been shown to be effective in patients with degenerative cervical disease.5,13,20 With an increasing number of synthetic cage materials and implants available to surgeons, our study represents an analysis of the most recent data available comparing 1-year complications between structural allografts and synthetic cages. Compared with structural allograft, patients undergoing multilevel ACDF with synthetic cage were at increased risk for 1-year revision surgery, while those receiving structural allograft were at increased risk for dysphagia.
For single-level ACDF, our comparison of 1-year outcomes between structural allografts and synthetic cages did not reveal any differences in the risk of revision surgery, pseudoarthrosis, SSI, or dysphagia. In contrast to our results, Krause et al 16 demonstrated radiographically higher rate of pseudoarthrosis (SA 10% vs PEEK 51.8%, P < .001) and greater need for revision surgery (SA 1.4% vs PEEK 12.5%, P = .01) with the use of PEEK in single-level ACDF. Based on these results, Krause and colleagues concluded that PEEK devices are associated with significantly higher rates of pseudoarthrosis and need for revision surgery compared with the use of allograft for single-level ACDF. Similarly, an administrative database study by Pirkle et al 18 suggested the use of structural allograft to be superior over synthetic cages after finding higher pseudoarthrosis rates in patients who underwent single-level ACDF with synthetic cages compared to structural allograft (SA 1.9% vs SC 4.2%, P = .0007) after controlling for number of levels treated, diabetes status, and tobacco use. We posit that our results may have deviated from these findings given the relatively low rates of complications observed in our cohort of single-level ACDFs, especially over the 1-year time horizon. In addition, our study employed more extensive propensity score matching than the Pirkle and Krause studies, suggesting that previously unexamined comorbidities and risk factors beyond smoking status, sex, diabetes, age, and body mass index—which were controlled for in the prior studies—may be important in determining the optimal implant for single-level ACDF.
Previous studies have observed significantly higher rates of pseudoarthrosis and need for revision surgery in multilevel ACDF compared to single-level ACDF utilizing structural allografts and synthetic cages.21-23 In our present comparative study, we observed higher rates of pseudoarthrosis and likelihood of revision surgery in the multilevel ACDF cohort. Our results further show that the patients who underwent multilevel ACDF with synthetic cages have a significantly higher likelihood of revision surgery when compared with structural allografts. Our results are directionally in line with the Pirkle et al 18 comparative review which demonstrated significantly higher pseudoarthrosis rate with 2-level (SA 1.7% vs SC 6.1%, P < .001), and 3+ level (SA 2.9% vs SC 6.3%, P < .001) ACDF. Similar to this study, 18 we included both anterior and posterior procedures in our criteria for revision surgery in order to broadly capture these clinical scenarios.
In another comparison, Goz et al 17 evaluated the rate of perioperative complications in patients undergoing ACDF found a statistically significant correlation with larger overall rate of complications in the synthetic cage (SA 7.76% vs SC 8.71%, P < .01) group and higher likelihood of revision surgery in the structural allograft (SA 0.56% vs SC 0.50%, P = .03) group. However, the authors considered these differences to be clinically insignificant given the low absolute differences observed. Based on these results, they concluded that synthetic cages are associated with a marginally higher overall rate of complications with similar revision rates, but acknowledged that additional large studies are necessary to further elucidate which graft combination provides the ultimate combination of fusion rates, low complications, and low cost. In contrast, our study showed significantly higher revision rate with synthetic cages in multilevel ACDF that may affect clinical practice. Beyond revision rate, our study evaluated the secondary outcomes of SSI, pseudoarthrosis, and dysphagia. While no significant differences between the structural allograft and synthetic cage groups were observed, with the exception of dysphagia in multilevel surgeries, these endpoints are limited in that they are grossly underreported postoperatively. In addition, the broad evaluation of SSI encompasses a wide variety of complications from benign superficial infections to deep infections requiring surgical intervention. Our observation of higher rates of dysphagia in patients undergoing multilevel ACDF with structural allograft is in alignment with Goz et al’s evaluation of dysphagia rates in the single- and multilevel population (SA 0.64% vs SC 0.33%, P < .01), but in contrast to previous studies. 24 This finding must be interpreted cautiously as our study design precludes control for factors that influence dysphagia such as surgical time, number of levels fused, and operative level, especially above C3.25,26
Given our finding of potentially higher one-year revision rates in ACDF with synthetic cages, we suggest further economic investigation of these devices, in parallel with clinical evaluation, is warranted. Limited data comparing the cost-effectiveness of ACDF with structural allograft or synthetic cage is currently available. Using a 10-year-Markov state transition model, Virk et al 27 found that PEEK cages had an incremental cost-effectiveness ratio over $100 000/quality-adjusted life year gained compared with autograft or structural allograft, leading to the conclusion that the technique is not cost effective compared with these alternatives. Beyond the differences in implant costs—structural allograft has been cited to cost up to $2552 and synthetic cages up to $7928 per implant 28 —operative time, hospital length of stay, complication rates, and post-discharge resource utilization are important cost drivers that require further direct comparison.
Synthetic cages that widely exist on the market today are composed of PEEK, titanium alloys, and carbon fiber with numerous studies attesting for their efficacy in spinal fusion. 29 One systematic review assessing clinical and radiographic outcomes of synthetic cages in ACDF found no differences in outcomes between PEEK, titanium, and carbon fiber cages, but titanium and carbon fiber cages were correlated with increased cost and higher subsidence rate in comparison to PEEK cages. 30 Given its radiolucency, low elastic modulus resulting in stress shielding, and similarities to cancellous bone, PEEK has been more widely used as an implant material for spinal fusion.11,29,31 However, PEEK has been demonstrated to have a hydrophobic surface preventing protein absorption and promotion of cell adhesion interfering host-bone integration by the formation of an encapsulating fibrous layer.32,33 While coating PEEK with a bioactive substance such as titanium has shown to be effective in improving its biocompatibility, it is also associated with biological and inflammatory reactions in human and animal studies. 34 In addition to these drawbacks, fusion mass may not form in the space occupied by the synthetic cage with less endplate surface area and intervertebral volume available, as hypothesized in Pirkle et al 18 study. On the contrary, allograft possess osteoinductive properties and provides osteoconductive scaffold for new bone formation with relatively high fusion rates comparable to autografts, especially with plating techniques.29,35 In addition to implant material, the use of an anterior plate and the type of nonstructural biologic material used in conjunction with a synthetic cage are other important aspects of the surgical procedure that influence postoperative outcomes. In a meta-analysis of 19 studies, patients who underwent ACDF with a cage-only technique had significantly lower rates of postoperative dysphagia and adjacent segment disease compared with patients who underwent ACDF with a cage-plate technique, but the cage-plate technique had better radiographic outcomes with significantly less subsidence and better restoration of cervical lordosis. No other significant differences in outcomes or postoperative complications were observed. 36 The choice of nonstructural biologic graft for use with synthetic cages is considered an important factor that influences postoperative fusion rates, but surgeons report there is insufficient comparative data to help select between grafts. 11 Due to small sample sizes and potential for bias, much of the evidence regarding biologic selection is of limited quality, making this an important area of future research. 37 Given the complex interaction between multiple factors that influence ACDF success, our findings are unable to pinpoint a precise reason for the trends observed and highlight the need for a prospective randomized controlled trial comparing structural allograft and synthetic cages before adequate evidence supporting the superiority of either implant is generated.
The analysis of any administrative database comes with several limitations. First, the TriNetX database relies heavily on coded data from multiple institutions yet this data carries the potential lack of fidelity associated with coding and may not be a representative sample of all patients undergoing ACDF surgery. Second, using coded data we were unable to differentiate between various implant materials, important aspects of the surgical procedure performed (such as the type of biologic used in conjunction with a synthetic cage or whether a plate was used), or the number of levels involved in multilevel procedures. The inability to differentiate between the number of segments involved in multilevel fusions is a significant limitation, as previous studies have identified multilevel surgeries as a risk factor for revision,22,23,38,39 and a 27% increased risk of revision has been reported for 3 or more level compared with 2-level ACDF. 23 Third, our data set did not allow for specification of the diagnosis associated with revision surgery, and therefore limits our ability to infer the reason for revision. This is especially important in the context of the postoperative pseudoarthrosis rates presented, as these may or may not have been symptomatic or required further intervention. Fourth, while we feel that the use of propensity score matching provides valuable control for confounding factors that may influence outcomes after surgery, the inherent limitations of observational studies and potential influence of unmeasured confounding covariates remains.
While few studies have directly compared complications and outcomes of structural allografts and synthetic cages, our findings further contribute to a growing body of literature that highlights the apparent increased risk of revision surgery following ACDF with synthetic cages compared with structural allograft bone. Despite the widespread use of synthetic cages, further large-scale, prospective studies that include parallel cost-effectiveness analysis are needed to determine if there are additional clinical benefits that outweigh the additional cost and potential increased risk of complications, including revision surgery.
Conclusion
Compared with structural allograft, patients undergoing multilevel ACDF with synthetic cage may be at increased risk for 1-year revision surgery, while those receiving structural allograft may be at increased risk for dysphagia. No differences in postoperative complication rates between implants were observed in single-level ACDF. Further large-scale, prospective comparison of structural allografts and synthetic cages for ACDF is warranted before the superiority of either implant can be concluded.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
