Abstract
Study Design:
Cadaver study.
Objective:
The retropharyngeal space’s (RPS’s) clinical relevance is apparent in anterior cervical spine surgery with respect to postoperative hematoma, which can cause life-threatening airway obstruction. This cadaver study aims to establish guidance toward a better understanding of the tolerance of the RPS to accommodate fluid accumulation.
Methods:
Five fresh-frozen cadavers were dissected in the supine position. A digital manometer and a 20 Fr Foley catheter were inserted into the RPS via an anterolateral approach. While inflating the Foley catheter, the position of the esophagus/trachea was documented using fluoroscopy, and the retropharyngeal pressure was measured. We quantified the volume required to deviate the esophagus/trachea >1 cm from its original position using fluoroscopy. We also recorded the volume required to cause a visible change to the normal neck contour.
Results:
A mean volume of 12.5 mL (mean pressure 1.50 mm Hg) was needed to cause >1 cm of esophageal deviation. Tracheal deviation was encountered at a mean volume of 20.0 mL (mean pressure of 2.39 mm Hg). External visible clinical neck contour changes were apparent at a mean volume of 39 mL.
Conclusion:
A relatively small volume of fluid in the RPS can cause the esophagus/trachea to radiographically deviate. The esophagus is the structure in the RPS to be most influenced by mass effect. The mean volume of fluid required to cause clinically identifiable changes to the normal neck contour was nearly double the volume required to cause 1 cm of esophageal/tracheal deviation in a cadaver model.
Keywords
Introduction
Anterior cervical spine surgery (ACSS) routinely involves the retropharyngeal space (RPS). This defined anatomical space extends from the skull base to the superior mediastinum. It is defined by the pharynx and esophagus and anterior to the prevertebral muscles 1 and is enclosed by the prevertebral fascia posteriorly, the carotid space laterally, and the buccopharyngeal fascia anteriorly. As part of the deep cervical fascia, the alar fascia extends bilaterally from the carotid space’s medial border and divides the RPS into an anteriorly positioned true RPS and a posteriorly located RPS that extends to the superior mediastinum. The RPS can be divided further into infrahyoid and suprahyoid sections, the latter of which contains fat and lymph nodes, whereas the former contains fat only. 1
Postoperative fluid accumulation in the RPS is of concern for a number of reasons: airway compromise, dysphagia, dysphonia, and other related symptoms. Dysphagia is the most common risk after ACSS, although the etiology is not fully understood. 2,3 Other less common postoperative complications associated with potential airway compromise occur in up to 14% of patients after a multilevel ACSS. 4,5 The RPS’s clinical relevance in ACSS is associated with postoperative hematoma, which can cause life-threatening airway obstruction. Their reported incidence ranges from 0.2% to 1.9%, with no clear etiology, and appropriate treatment varies considerably. 6,7 Other causes can include cerebrospinal fluid accumulation, abscess formation, and generalized edema.
Although postoperative hematoma accumulation in the RPS is a devastating complication, there is no consensus regarding the tolerable volume in the RPS. The purpose of this study is to establish a better understanding of the volumetric effect caused by fluid accumulation in the RPS. Thus, this study aims to create an anatomical model to simulate a retropharyngeal mass occupying lesion and calculate the volume and pressure required to deviate the trachea and esophagus.
Materials and Methods
Five fresh-frozen Caucasian cadavers (4 male, 1 female) with a mean age at death of 82.4 years (mean body mass index [BMI] of 24.66 kg/m2) were selected for dissection while the specimens were at room temperature. Specimens were placed in the supine position on a radiolucent table. All images were taken in anteroposterior (AP) plane.
The vertebral level of C5/C6 was identified using fluoroscopy. A small transverse incision was made on the right anterolateral neck, and the platysma muscle was dissected carefully parallel to the incision. Using the facial plane between the carotid sheath laterally and the trachea and esophagus medially, the deep cervical fascia was located. The fascial plane was developed superiorly to the C2/3 intervertebral disc space and inferiorly to the anterior border of the sternocleidomastoid muscle. Blunt dissection was carried down to the prevertebral fascia and across to the contralateral uncovertebral joint to replicate surgical dissection in ACSS. We then inserted a digital air pressure manometer (Perfect-Prime AR1890 Professional Digital Air Pressure Meter Manometer) into the RPS, after which a 20 Fr Foley catheter (Bradex Foley catheter, 20 Fr 5-mL balloon) also was inserted. The tip of the catheter was removed beforehand, so that only the balloon was in the RPS. A regular wound closure was performed, and the catheter was kept in its desired position via fluoroscopy (Figure 1). A suction tube (6 mm diameter) was placed in the esophagus, which was flushed with a barium sulfate suspension to measure the distance between the esophagus and the C5 spinous process using ImageJ. 8 To account for magnification, a stainless-steel ruler (diameter 3 cm) was used to calibrate the fluoroscopy images. We inflated the Foley catheter balloon gradually in 5-mL increments to a total of 70 mL. After each 5-mL injection, the position and movement of the esophagus were documented using fluoroscopy, and the pressure was measured in millimeters of mercury after waiting 3 minutes to allow sufficient time for calibration (Figure 2). After a volume of 70 mL was reached, we deflated the balloon in 10-mL increments and documented the deflation using fluoroscopy again. We defined tracheal and esophageal deviation as displacement of the trachea and esophagus >1 cm from its baseline position using the center of the C5 spinous process as a reference point for initial measurement. This was measured using ImageJ software with C5 as a reference point. 8 The entire procedure was repeated with the suction tube in the trachea. Furthermore, we recorded the volume required to cause physical exam changes affecting the normal contour of the neck. The onset of clinically visible changes of neck contour secondary to mass effect of the underlying fluid collection was observed independently by 2 spine fellowship trained spine surgeons blinded to the amount of fluid and pressure. The average volume between the 2 observers was recorded. There is no competing conflict of interest to declare.
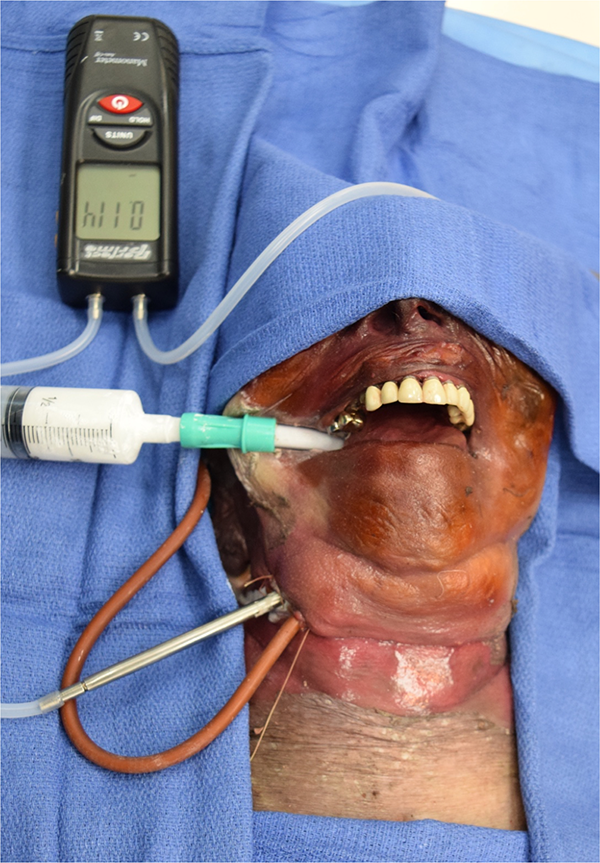
Pressure sensor, 6-mm suction tube, and implanted Foley catheter placed using a small transverse incision that was made on the right anterolateral neck.
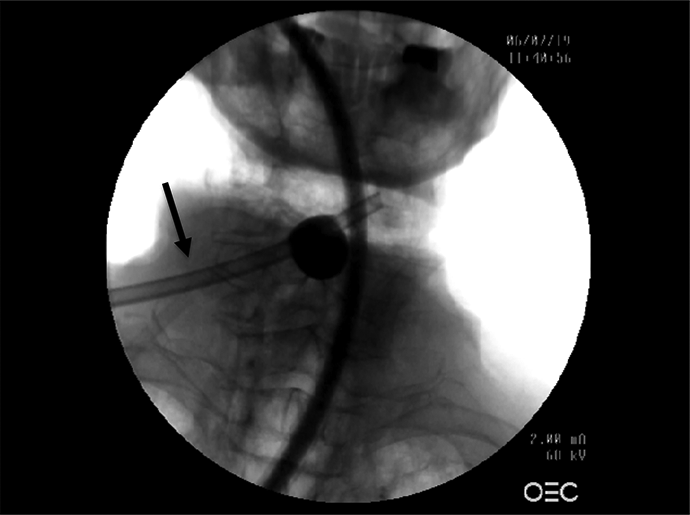
Anterior posterior fluoroscopy image (10-mL injection [Foley]; suction tube in esophagus).
Results
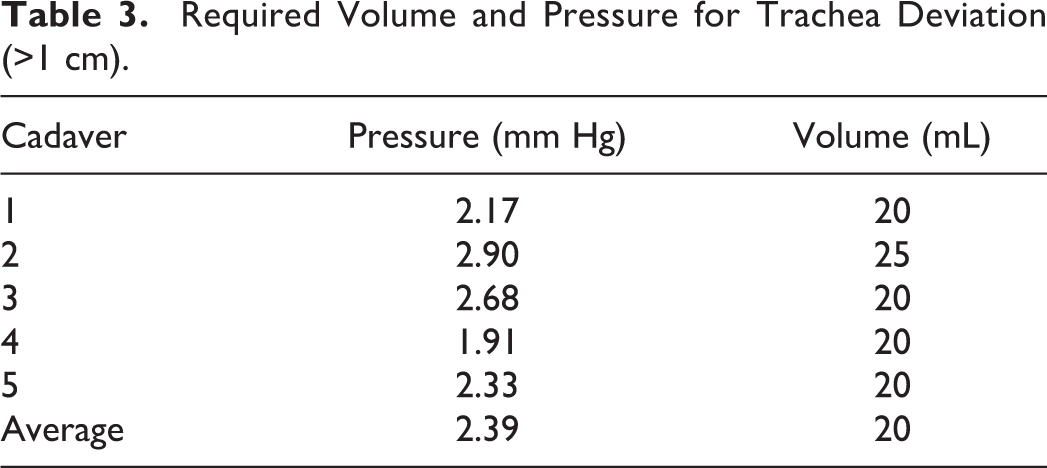
Demographic information regarding demographics of cadavers can be found in Table 1.The right anterior approach to the RPS was carried out without visible damage to regional anatomical structures. The esophagus deviated >1 cm from its baseline position at a mean volume of 12.0 mL; the mean pressure at the time of deviation was 1.50 mm Hg. The minimum volume required to cause tracheal deviation was 10 mL (Table 2). The trachea deviated >1 cm at a mean volume of 20 mL and at a mean pressure of 2.39 mm Hg. The minimum volume required to cause tracheal deviation was 20 mL (Table 3). Cervical soft-tissue changes of the neck were appreciated at a mean volume of 39.0 mL (Table 4). The earliest recognition of neck swelling was at 25 mL in the thinnest specimen (male; BMI = 18.3 kg/m2).
Demographic Data of the Cadavers.
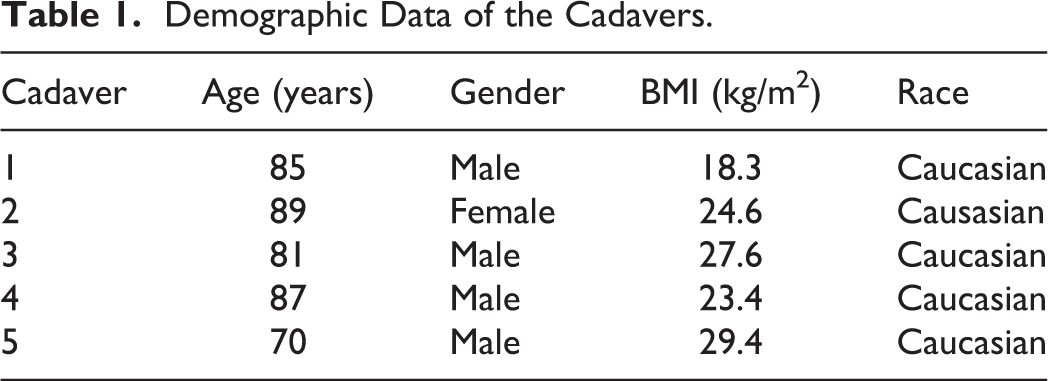
Required Volume and Pressure for Esophagus Deviation (>1 cm).
Required Volume and Pressure for Trachea Deviation (>1 cm).
Volume Required to Induce Cervical Soft-Tissue Changes of the Neck Clinically Noticed by Inspection of the Neck Contour.

Discussion
The anterior approach to the cervical spine is a common approach to many spine pathologies. 9 Although the incidence of complications associated with these procedures is relatively low, 5,6 dysphagia and dysphonia are most frequently encountered (transient 4%-30%, persistent 0.7%-5%). 2,10 Acute airway obstruction (AAO) secondary to postoperative hematoma and soft-tissue edema in the RPS is a devastating, albeit rare, postoperative complication. The RPS is a potential space for a clinically relevant fluid collection as described above. Particularly in the context of increasing numbers of spinal procedures in an outpatient setting like anterior cervical discectomy and fusion (ACDF), 6,11 clinicians should have a focus on the mentioned complications. This study quantifies the volume of fluid in the RPS required to cause radiographic esophageal/tracheal deviation and clinical changes in neck contour.
Anatomical vascular variations relevant to the anterior approach to the cervical spine have been described. However, no direct correlation with anatomical variation and postoperative retropharyngeal hematoma have been identified. 12 -15 The superior and inferior thyroid arteries have been identified as etiology of hemorrhage in some retrospective reviews of postoperative hematoma after ACDF, although most reported cases do not have an identifiable source of hemorrhage. 16,17
Esophageal deviation occurred at a mean volume of 12.5 mL compared with the trachea, at 20 mL, which is likely secondary to its position directly anterior to the RPS. Moreover, the esophagus is more pliable than the trachea and is composed of rigid hyaline cartilage. 18 -20 Therefore, dysphagia may be a more sensitive early sign of retropharyngeal bleeding. 7,21 This also explains why a patient may present with only dysphagia and “collar tightness” with normal oxygen saturation in the early stages of a retropharyngeal hematoma. 22 The clinical progression from dysphagia to airway obstruction is rapid and must, therefore, be identified quickly. 23 The expected clinical presentation of patients with a tense mass under the incision or swelling of the anterior neck may be a late and ominous warning sign. 24,25 Our study validates this given that visible neck contour changes were appreciated at a volume nearly double (39.36 mL) that required to cause tracheal deviation (20 mL). Therefore, it is the authors’ opinion that clinically identifiable changes in neck contour are not suitable early warning signs. The authors recommend close observation in patients with progressive dysphagia and early intervention and workup when a postoperative retropharyngeal hematoma is suspected, even in the absence of visible neck edema or palpable hematoma.
Risk factors identified for postoperative retropharyngeal hematoma include the following: (1) presence of diffuse idiopathic skeletal hyperostosis, (2) presence of ossification of the posterior longitudinal ligament, (3) therapeutic heparin use, (4) longer operative times, and (5) greater number of surgical levels. 17 Although some surgeons advocate for a drain placement to mitigate the incidence of retropharyngeal hematomas, O’Neill et al 17 retrospectively studied 2375 ACSSs and identified 17 patients with postoperative hematoma and found no correlation with drain placement as a preventive measure.
Although AAO after ACSS is a devastating complication and can be caused by a retropharyngeal hematoma, the 2 are not mutually exclusive, and AAO may also be caused by generalized soft-tissue edema. The latter has more of a subacute clinical course, and although both are of great clinical importance, our study focuses particularly on the volumetric effect of fluid in the RPS.
Limitations
As with all studies, ours is not without limitations. Although cadaver models are commonly used in many studies to explain behavior both in vitro and in vivo, the question still remains whether the specimens used reflect the normal in vivo situation. 26 We expect that a cadaver’s hollow organs would demonstrate less physiological characteristics—for example, muscle contraction. Therefore, the results must be interpreted with caution and the actual volume required to cause esophageal and tracheal deviation may have been overestimated in this study. However, we believe that the clinical importance is stressed by the fact that visual appreciation of the fluid collection by spine surgeons blinded to the amount of fluid injected was approximately twice the amount required to cause >1 cm of esophageal and tracheal deviation. This highlights the importance of early diagnosis prior to the presence of clinically identifiable changes to the overlying soft tissues. Another limitation is the demographics of the cadaver models, which do not represent the patient population undergoing ACSS. The lack of clinical symptomatology associated with cadaver studies is also a limitation. Although >1 cm of tracheal and esophageal deviation appears relevant to this study, there are no guidelines of acceptable amounts of deviation after ACSS. Moreover, acceptable values of tracheal and esophageal deviation in conditions such as pneumothorax or hemothorax do not apply to ACSS given the inherent postoperative edema and inflammation associated with ACSS.
The Foley balloon has a defined spherical shape. This may not represent a retropharyngeal hematoma in vivo, which can span several levels and have a more fusiform in shape. The idea of injecting a radiopaque fluid into the RPS was considered; however, the viscosity of the fluid is not analogous to the viscosity of blood. Unlike blood, radiopaque fluid cannot congeal and form a mass occupying lesion and would, therefore, extravasate into the soft-tissue planes. Therefore, the decision was made to use a Foley catheter balloon to resemble the appearance of a mass occupying hematoma.
Conclusion
Postoperative retropharyngeal hematoma after ACSS is a rare but catastrophic complication, which can cause AAO. The mean volume of fluid required to cause clinically identifiable changes to the normal neck contour was nearly double the volume required to cause 1 cm of esophageal and tracheal deviation in a cadaver model. Early recognition of this condition is paramount, and spine surgeons should strive to identify this complication prior to the presence of clinically identifiable changes to the overlying soft tissues on physical exam, which may be an impending late sign.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
