Abstract
Study Design
Systematic review.
Objective
To determine the frequency of pulmonary effusion, pneumothorax, and hemothorax in adult patients undergoing thoracic corpectomy or osteotomy for any condition and to determine if these frequencies vary by surgical approach (i.e., anterior, posterior, or lateral).
Methods
Electronic databases and reference lists of key articles were searched through September 21, 2015, to identify studies specifically evaluating the frequency of pulmonary effusion, pneumothorax, and hemothorax in patients undergoing thoracic spine surgery.
Results
Fourteen studies, 13 retrospective and 1 prospective, met inclusion criteria. The frequency across studies of pulmonary effusion ranged from 0 to 77%; for hemothorax, 0 to 77%; and for pneumothorax, 0 to 50%. There was no clear pattern of pulmonary complications with respect to surgical approach.
Conclusions
There is insufficient data to determine the risk of pulmonary complications following anterior, posterior, or lateral approaches to the thoracic spine. Methods for assessing pulmonary complications were not well reported, and data is sparse.
Keywords
Introduction
Infections, tumors, and herniated disks can affect the stability of the thoracic spine and cause neurologic dysfunction. A significant proportion of pathology in this region involves the anterior and middle columns of the spine. 1 Different surgical approaches are used to access this area including anterior approaches requiring thoracotomy as well as posterior approaches, which include costotransversectomy and lateral extracavitary approaches; in recent years, direct lateral approaches have become more popular. 2 , 3 , 4 Pulmonary complications can occur given the proximity of the pleural space to the anterior thoracic spine. The true incidences and differences in pulmonary complications between these approaches have not been well delineated in the literature because they are poorly reported. This study will systematically review the frequency of pulmonary complications. Specifically, this systematic review seeks to answer the following questions:
What is the frequency of pulmonary effusion, pneumothorax, and hemothorax in adult patients undergoing corpectomy or osteotomy for any condition?
Does the frequency vary by approach (anterior, posterior, lateral, or combined)?
Materials and Methods
Further details about methods can be found in the online supplementary material.
Results
We identified 14 studies meeting the inclusion criteria. These studies form the basis for this report. Fig. 1 shows the inclusion and exclusion of articles from the search. A list of excluded articles can be found in the online supplementary material.
Among the 14 studies identified, 13 were retrospective studies, 2 , 3 , 4 , 5 , 6 , 7 , 8 , 9 , 10 , 11 , 12 , 13 , 14 and 1 was a prospective study. 15 All but 2 had had a high risk of bias, 4 , 15 class of evidence IV (Table 1). The details on the class of evidence can be found in the online supplementary material. A variety of approaches were used in the studies: 8 reported anterior, 4 , 5 , 6 , 7 , 8 , 9 , 10 , 15 7 posterior, 3 , 4 , 5 , 8 , 9 , 10 , 11 4 lateral, 2 , 7 , 12 , 13 and 3 combined approaches. 9 , 14 , 15 The common indications for surgery were herniated nucleus pulposus, metastatic disease or tumor, or spinal injury (fracture or otherwise), and others. Interventions included decompression, thoracotomy, diskectomy, interbody fusion, reconstruction, and corpectomy and more (Table 1).
The mean age generally ranged from 40 to 60 years, but included some patients as young as 12 and as old as 88 years. There was a higher proportion of males in studies where the indication was spinal injury only.
Flowchart showing results of literature search.
Summary of study characteristics and pulmonary adverse events from thoracic spine surgery stratified by approach
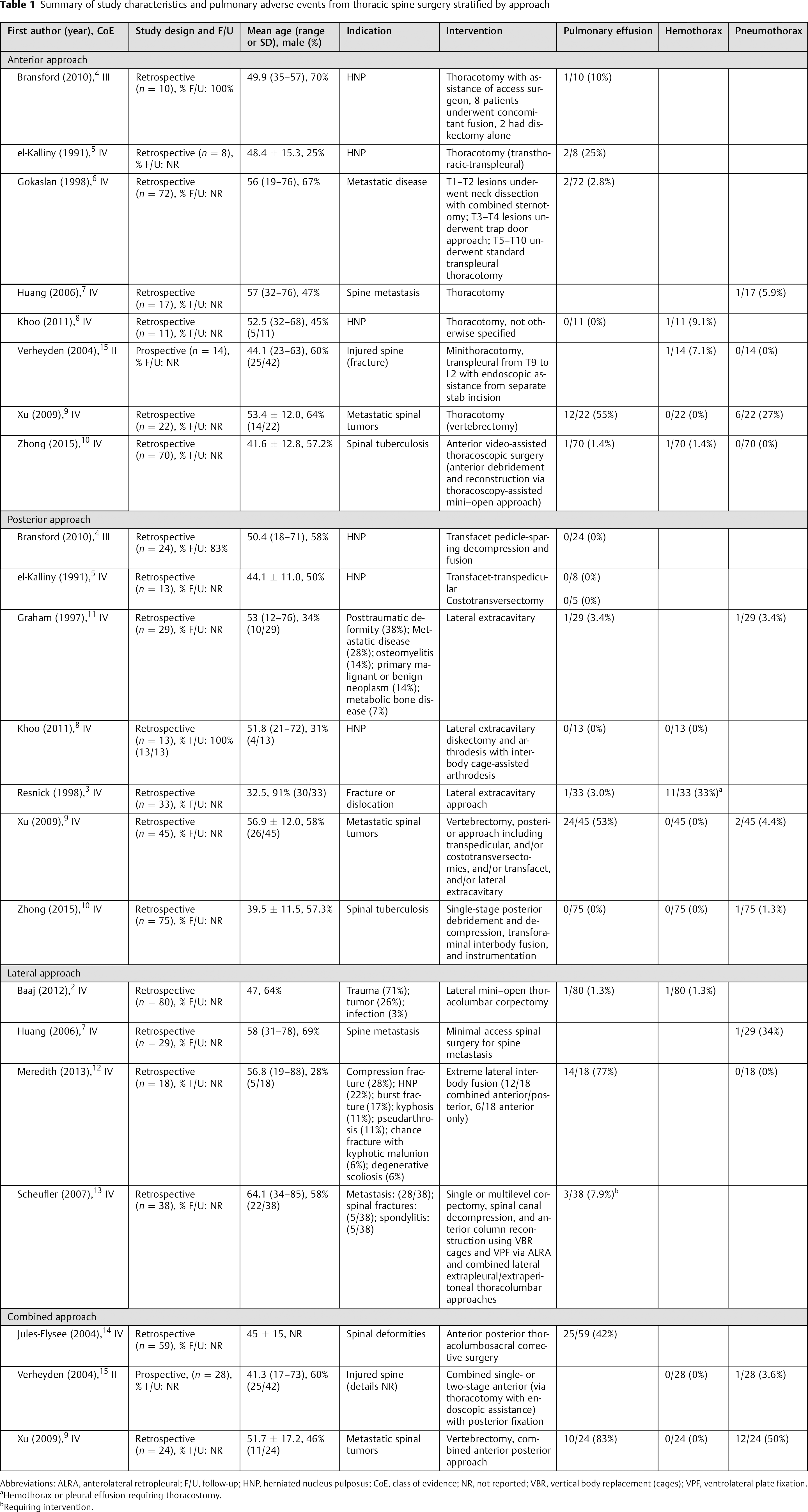
Abbreviations: ALRA, anterolateral retropleural; F/U, follow-up; HNP, herniated nucleus pulposus; CoE, class of evidence; NR, not reported; VBR, vertical body replacement (cages); VPF, ventrolateral plate fixation.
Hemothorax or pleural effusion requiring thoracostomy.
Requiring intervention.
Pulmonary Effusion
The 12 studies reporting on pulmonary effusion used a variety of approaches. The frequency of pulmonary effusion ranged from 0 (0/11) to 55% (12/22) in 6 studies using the anterior approach, 4 , 5 , 6 , 7 , 8 , 9 , 10 , 15 0 (0/75) to 53% (24/45) in 7 studies using the posterior approach, 3 , 4 , 5 , 8 , 9 , 10 , 11 1 (1/80) to 77% (14/18) in 3 studies using the lateral approach, 2 , 7 , 12 , 13 and 42 (25/59) to 83% (10/24) in 3 studies using the combined approach (Table 1). 9 , 14 , 15
Hemothorax
Six studies report on hemothorax using a variety of approaches. 3 , 8 , 9 , 10 , 15 The frequency of hemothorax ranged from 0 (0/22) to 9% (1/11) in 4 studies using the anterior approach, 8 , 9 , 10 , 15 0 (0/75) to 53% (24/45) in 4 studies using the posterior approach, 3 , 8 , 9 , 10 1 (1/80) to 77% (14/18) in 1 study using the lateral approach, 2 and 0 (0/28 and 0/24) in 2 studies using the combined approach (Table 1). 9 , 15
Pneumothorax
Five studies reported pneumothorax using a variety of approaches. The frequency of pneumothorax ranged from 0 (0/70) to 27% (6/22) in 4 studies using the anterior approach, 7 , 9 , 10 , 15 1 (1/75) to 4% (2/45) in 3 studies using the posterior approach, 9 , 10 , 11 0 (0/18) to 34% (1/29) in 2 studies using the lateral approach, 7 , 12 and 4 (1/28) to 50% (12/24) in 2 studies using the combined approach. 9 , 15
Illustrative Case
A 53-year old woman presented to the emergency department for evaluation of high thoracic back pain with radiation into the legs, as well as progressive numbness. The imaging illustrated an infiltrative, extradural mass encompassing the T2 and T3 vertebral bodies, pedicles, and posterior elements causing cord compression with significant bony erosion. After appropriate workup, biopsy, and embolization, she underwent a T2–T3 costotransversectomy and vertebrectomy with cage placement and posterior instrumented fusion from C6 to T9. Once all instrumentation was in place, the patient desaturated and developed hypotension and respiratory acidosis. The patient was emergently closed and turned supine. Decreased breath sounds were noted on the left side, a presumptive diagnosis of pneumothorax was made, and an emergent chest tube was placed with a positive rush of air. She subsequently had improvement in her ventilation and oxygenation. The chest tube was removed after 1 week with daily chest radiographs to ensure no recurrence of the pneumothorax.
Presumptive causes for the pneumothorax included aberrant probe placement during instrumentation, interrupting the parietal and visceral pleura, injury of the pleura during costotransversectomy, or injury to the pleura during tumor debulking.
Discussion
Traditional anterior approaches requiring thoracotomy should theoretically have a higher risk of pulmonary complications compared with posterior or lateral approaches. Gokaslan et al reported on 72 patients who underwent thoracotomy for metastatic disease. Their median chest tube duration was 4 days (range 1 to 26 days) and pleural effusions were seen in only 2 patients. 6 Another study by Khoo assessed 24 patients, of whom 13 underwent a minimally invasive posterior lateral extracavitary approach and 11 underwent standard thoracotomy for thoracic disk herniation. 8 No patients in the posterior group required chest tubes, whereas all patients in the thoracotomy group had a chest tube for an average of 1.5 days (longest 4 days) postoperatively. Additionally, 1 patient in the thoracotomy group developed a delayed hemothorax that required thoracentesis. Zhong et al studied 145 patients with spinal tuberculosis. 10 In their series, 70 patients underwent video-assisted thoracoscopic surgery. Of these 70 patients, 10 had pulmonary complications including 3 lung lacerations, 1 pleural effusion, 1 hemopneumothorax, 2 atelectasis, 2 pneumonias, and 1 empyema. Contrarily, the remaining 75 patients underwent costotransversectomies from a posterior approach and only 1 patient had a pulmonary complication (pneumothorax). Another study by Jules-Elysee found significantly higher rates of abnormal chest X-rays in patients who had thoracic incisions. 14
Perhaps the best study illustrating the differences in pulmonary complications between anterior and posterior approaches to the thoracic spine was done by Xu et al. 9 They reported on 91 patients with metastatic disease: 22 patients underwent anterior-only procedures via thoracotomy, 45 patients underwent posterior-only procedures, and 24 patients underwent combined anterior and posterior procedures. Although not a complication, they found that the rate of chest tube placement during surgery was significantly higher in the anterior (81.8% of cases requiring chest tube) and combined cases (79.2% of cases requiring chest tube) compared with the posterior-only cases (only 20% of cases). Additionally, the placement of postoperative chest tubes was significantly higher in the anterior (4 patients) and combined group (5 patients) compared with the posterior group (0 patients). The duration of chest tube drainage was highest in the combined group (8.8 days on average), which was significantly different from the anterior-only group (4.7 days); interestingly, the duration of chest tube drainage was not statistically different between the combined and posterior-only groups (5.8 days). Furthermore, the rate of perioperative pneumothoraces was highest in the combined group (12 patients) compared with the anterior group (6 patients) and posterior group (2 patients), although statistical significance was only seen between the combined and posterior group. Pleural effusions were common in all groups but were statistically higher in the combined approach group (83.3% compared with 54.4% in the anterior group and 53.3% in the posterior group).
On the other hand, a thoracotomy may not always carry a higher risk of pulmonary complications. For example, Bransford et al reported on 34 patients, of whom 10 underwent thoracotomy and 24 underwent transfacet pedicle-sparing fusion for thoracic disk herniations. 4 They reported only one pulmonary effusion, which led to a subsequent pneumonia. There were no significant differences between the two groups in terms of pulmonary complications. Huang reported on 46 patients with metastatic disease, 17 of whom underwent standard thoracotomy and the remainder underwent what they termed “minimal access spinal surgery.” 7 They found no significant difference in chest tube duration (5.7 days for the minimal access spinal surgery group and 6.4 days for the thoracotomy group) or the number of pneumothoraces (each group had one).
Some authors have seen relatively high rates of pulmonary complications in posterior approaches. Resnick and Benzel reported on 33 patients who underwent lateral extracavitary approaches for trauma. 3 They had 11 patients with pneumothoraces and 1 patient with a pulmonary effusion. Although it is well established that thoracotomy has significant morbidity, the rate of pulmonary complications may not be high as originally believed compared with other surgical approaches.
Direct lateral approaches to the spine have become increasingly popular because they can be performed by most spinal surgeons without the need for an access surgeon. The rate of pulmonary complications after such procedures remains poorly studied. In a study of 80 subjects undergoing lateral interbody fusion for varied pathologies, only two pulmonary complications were noted. 2 The first was a hemothorax that developed after a T9–T10 corpectomy for osteomyelitis that required video-assisted thoracoscopy. The second was a pulmonary effusion that was not further discussed. Another study of 18 patients by Meredith et al undergoing lateral interbody fusion for varied pathologies showed a relatively high rate of pulmonary-related issues. 12 In their series, chest tubes were left in all patients and were discontinued on postoperative day 2 in most cases. Eight patients developed post–chest tube pleural effusions of which 2 required treatment. The first was a 78-year-old woman who underwent a T12 corpectomy for a burst fracture; she had replacement of her chest tube with resolution of the effusion. The second patient was transferred to a higher level of care for monitoring but did not require any invasive procedures. In a third series by Scheufler of 38 patients who underwent lateral approaches for various pathologies, only 3 had chest tubes placed perioperatively. 13 Interestingly, all 3 of these patients underwent lateral approaches for metastatic disease. All of the other patients in Scheufler's series had retropleural drains placed, which were removed on postoperative day 3. It is unclear if these “retropleural” drains may have acted as surrogate chest tubes, but the study specifically delineated between chest tube and retropleural drain placement.
The rate of pulmonary complications for various pathologies remains unclear. Based on our review of the literature, it is not known if certain pathologic processes have a higher rate of postoperative pulmonary problems. In theory, metastatic disease to the spine with pleural invasion would be at high risk; however, it is unknown if this risk is higher than a patient undergoing surgery for a herniated disk or infection. It is also unclear if age, gender, or other medical comorbidities play a role in the development of pulmonary complications after spinal surgery. Jules-Elysee et al assessed pulmonary complications in 59 patients undergoing surgery for “spinal deformity.” 14 They assessed findings on chest X-rays taken on postoperative day 3. They found that there was no differences between those with “abnormal” chest X-rays and those with normal chest X-rays regarding age, levels fused, preoperative pulmonary function testing, blood loss, transfusion, duration of surgery, or patient weight. At this point, there are no clear patient-defined factors that increase the risk of pulmonary complications after spinal surgery.
The method of diagnosing pulmonary complications postoperatively is not frequently discussed. Only four studies we reviewed discussed the imaging that was used to diagnose pulmonary complications. Although it is safe to assume that most pulmonary-related problems in the other studies were likely diagnosed based on clinical exam in combination with chest X-ray or computed tomography, these studies do not specifically mention the means of diagnosis. Furthermore, the natural history of incidental pulmonary findings on imaging is not well described in the literature. For example, patients with clinically insignificant pleural effusions or atelectasis likely suffer no long-term sequelae. Whether or not these findings constitute postoperative pulmonary “complications” is unclear.
Conclusions
Pulmonary complications have traditionally been thought to be more common in anterior approaches compared with posterior approaches to the anterior thoracic spine, but this observation may not be the case. Based on our analysis, there is insufficient data to determine the risk of pulmonary complications following anterior, posterior, or lateral approaches to the thoracic spine. Methods for assessing pulmonary complications were not well reported, and data was sparse. As such, the risk factors for the development of pulmonary complications following any approach to the anterior thoracic spine remains unclear.
Disclosures
Brandon C. Gabel: none
Eric C. Schnell: none
Joseph R. Dettori: none
Shiveindra Jeyamohan: none
Rod Oskouian: none
Gabel et al provided an important contribution to our current understanding of access-related morbidity resulting from anterior spinal thoracic spinal column surgery relative to posterolateral, traditional anterior, and “less invasive” laterally based approaches. Our
Straightforward questions are still in need of answers. How long can we expect a chest tube to be left in place? When is a chest effusion or a hematothorax expected and when is it a complication? How do we diagnose pulmonary complications? Do we rely on chest radiographs, chest computed tomography? What role do clinical parameters such as decreased oxygenation or need of supportive respiratory therapy play? What kind of effects do comorbidities such as restrictive lung diseases, chronic obstructive pulmonary disease, pulmonary fibrosis, and others have on pulmonary complications?
Without such definitions, we cannot (yet) answer questions such as the ones asked by the authors: are the more recently introduced laterally based exposures as safe or safer than conventional transthoracic, endoscopically based, or posterolaterally based exposures? What are the outcomes of the various surgical approaches from a patient's perspective?
In our era of increased focus on establishing normative data sets for complications related to procedures, the absence of core definitions in any entity creates a fluid environment unsuitable for meaningful analytics. Larger prospective data sets that use clear consensus-based operational definitions could provide important comparison opportunities that the current published spine knowledge base currently does not provide.
Footnotes
Acknowledgments
Analytic support for this work was provided by Spectrum Research, Inc. with funding from the Swedish Neuroscience Institute.
