Abstract
Study Design:
Biomechanical investigation.
Objective:
To compare the biomechanical performance of nitinol memory metal rods and titanium rods when used as posterior spinal instrumentation in a synthetic model.
Methods:
Biomechanical testing was performed using ultra-high-molecular-weight polyethylene blocks. Nineteen spinal constructs were created to allow comparison of 5.5-mm nitinol rods with 5.5-mm titanium rods. Static compression and rotational testing were performed on an Instron 8874 and Instron 4202 at 37°C to simulate body temperature.
Results:
The average titanium construct stiffness under static compression or bending was 47.2 ± 9.1 N/mm while nitinol’s stiffness averaged 48.9 ± 12.4 N/mm (P = .83). During axial rotation testing, the nitinol rod system showed no torsional stiffness difference from the titanium system: 0.95 ± 0.03 Nm/deg versus 0.96 ± 0.17 Nm/deg, respectively (P = 0.91). There was a statistically significant difference between the average torsional yield point for the titanium constructs (14.4 ± 1.6 Nm/deg) and nitinol constructs (21.3 ± 0.8 Nm/deg) (P = .004). The torsional toughness of the nitinol constructs was also statistically greater than the titanium rods: 473 GN/m3 versus 784 GN/m3 (P = .0006). There was no statistically significant difference between the nitinol group sustaining a higher number of fatigue cycles until failure and the titanium group (181 660 cycles vs 64 104 cycles, respectively, P = .22).
Conclusions:
This study provides biomechanical evidence that nitinol rods used in a posterior construct are comparable to titanium rods with regard to compression and have increased torsional failure load and torsional toughness. While nitinol trended toward superior fatigue resistance, there was no significant difference in nitinol versus titanium construct fatigue resistance.
Introduction
Spinal instrumentation constructs have progressed from the use of stainless steel to titanium rods for stabilization. Now other materials are being considered for use as rods: polyetheretherketone (PEEK), cobalt chromium, and nitinol. 1,2 The ideal material for posterior instrumentation should provide enough stability for fusion to occur. It should not be too stiff, so as to accommodate bone graft loading and to minimize stress shielding. The ideal material has a Young’s modulus similar to that of cortical bone. 3 -5
Nitinol, a shape memory alloy (50% nickel/50% titanium), possesses unique properties among biomedical materials. Because nitinol memory metal can be chilled, contoured, and then heated to return to its precontoured shape, it has many potential applications for reducing fractures and correcting deformities. 6 The performance of nitinol for posterior spinal rod instrumentation has been studied in a plethora of animal studies and a handful of clinical studies. 3 -5,7 -9 Kok et al 1 studied the biomechanical properties of nitinol in a single rod construct and found improved torsional properties compared with titanium. To our knowledge, there has been no study that compared the biomechanical properties of nitinol in a double rod corpectomy model. The purpose of this study was to compare the biomechanical performance of nitinol memory metal rods with that of titanium rods when used as posterior spinal instrumentation in a synthetic model. It was hypothesized that nitinol memory metal rods would have a biomechanical performance comparable to that of titanium rods when used as posterior spinal instrumentation in a synthetic model.
Materials and Methods
Institutional review board approval was not needed as this was a biomechanical study with no human or animal tissue used. An a priori power analysis was performed to determine the minimum number needed in each group for a power of at least 0.80. Using a previous study comparing a single nitinol square rod construct versus a titanium construct, the effect size was determined. 1 Using the formula nA = (σA 2 + σβ 2/κ) ((z1-α + z1-β)/ μA – μβ))2, it was determined that the number needed in each group to compare static stiffness and torsional stiffness was 2 and 1, respectively. 2
The nitinol alloy composition selected was SE 508 manufactured by Nitinol Devices and Components (NDC; Freemont, CA). This alloy was chosen because of its transformational temperature range (–25°C to +30°C; information from NDC reference material data sheet) and potential in vivo applications. The rods obtained were 5.5 mm in diameter and cut to 100-mm straight lengths, creating 16 rods. The control group consisted of 10 rods composed of TiAl4V titanium alloy (Globus Medical Inc, Audubon, PA), 5.5 mm in diameter and 100 mm long. One nitinol rod was cut to 300 mm to test its modulus of elasticity.
No heat treatments were applied to the titanium control rods. The 16 nitinol rods were secured in a 0° curve and placed into a preheated oven to program the rods to maintain no radius of curvature. One of the 300-mm nitinol rods then underwent 3-point bending in an Instron 4202 single-axis mechanical testing frame (Instron Corp, Canton, MA) to verify that the material’s modulus of elasticity fell within the range specified by its manufacturer. Under displacement control, the rod had a total span of 200 mm and was bent 10 mm at a rate of 10 mm/min. Data was collected at a rate of 10 samples/s through a custom-written LabView program (National Instruments, Austin, TX). We used a heating lamp to achieve an in vivo testing temperature and attached a thermocouple to the rod to monitor maintenance of a temperature of 37°C (Figure 1). The stiffness value was then used to calculate the specific modulus of elasticity.

Nitinol construct in testing apparatus with Instron and heat lamp set to 37°C with thermocouple.
All testing involving both the nitinol and titanium control constructs was conducted in a temperature-controlled environment. In order to ensure that the nitinol constructs maintained their austenite state or more rigid state, testing was performed with the aid of a heat lamp and temperature probe, maintaining the construct temperature at 37°C (Figure 1).
Mechanical comparative testing of the rods was conducted according to the guidelines of the American Society for Testing and Materials (ASTM; F1717-04: Static and Dynamic Test Method for Spinal Implant Assemblies in a Corpectomy Model). A custom fixture was built and high-molecular-weight polyethylene (HMWPE) blocks were cut as described by ASTM. A single nitinol construct was composed of the 2 heat-treated nitinol SE 508 rods and four 6.5-mm Globus titanium polyaxial screws. A single titanium construct was composed of 2 TiAl4V titanium rods and four 6.5 × 45-mm Globus titanium polyaxial screws. In all constructs, rods were locked into the screws with a torque limiter set to 5.5 Nm. Each construct was mounted in 2 parallel HMWPE blocks with fixation points separated by 76 mm, in accordance with ASTM standards. 1,3 Groups were established consisting of 11 nitinol and 11 titanium constructs (Figure 2).

Nitinol construct (4 pedicle screws, 2 rods, and 2 polyethylene blocks) in testing apparatus with Instron.
Compressive bending tests were conducted on the Instron 4202 testing frame. Four nitinol and 4 titanium constructs were tested. Each construct was compressed at a rate of 10 mm/min; load and displacement data was collected in LabView at 10 samples/s until failure occurred. Testing was performed in a temperature-controlled environment. Failure was defined as either fracture of an implant or a noticeable reduction in stiffness. After displacement, the angle between the UHMWPE blocks was measured with a goniometer.
Torsional rotation testing was conducted on a biaxial Instron 8874 testing frame (Figure 2). Four nitinol and 4 titanium constructs were tested. Each construct was rotated at a rate of 60 deg/min to 30 deg or implant/fixture failure. The torque (Nm) and degrees of rotation were recorded throughout the test through Instron’s WaveMatrix software.
Fatigue testing was performed on an Instron E3000 in displacement control at 10 mm of compression at 5 Hz. A dry specimen temperature of 37°C during testing was maintained using the heat lamp. Three titanium and 3 nitinol constructs were tested until failure. The implants were cyclically loaded until failure or implant breakage occurred or runout of 1 000 000 cycles was achieved.
A Shapiro-Wilk test was performed to determine if the data was normally distributed. Both the titanium group and nitinol group were determined to have normally distributed data (P = .42 and P = .20, respectively). Statistical analysis compared the control group and the nitinol group with respect to torsional stiffness, bending stiffness, and fatigue cycles using a two-sample t test.
Stress, Strain, and Toughness Calculation
Angular strain or torque was converted to stress using the formula τ = TL / (Π/32 × d4 × θΠ/180). Angular deformation was converted to strain using the formula γ = τ/G. G = TL / (Π/32 × d4 × θΠ/180). The toughness was determined by calculating the area under the stress strain curve for each sample in Joules per cubic meter (J/m3). 6
Results
Young’s Modulus Calculation
A single 5.5-mm diameter rod (100 mm in length) was tested in order to obtain basic biomechanical data prior to spine construct testing. Using a custom 3-point bending apparatus, a peak stiffness value of 19.9 N/mm was measured with a 10-mm displacement at 12.5 mm/min. Inertia was calculated to be 44.92, using the formula I = πD4/16. 10 Using the formula for calculating Young’s modulus (E = [PL3] / [48Iv]), the Young’s modulus was calculated to be 74.21 GPa. 11
Static Compression Testing
The average titanium construct stiffness under static compression or bending was 47.2 ± 9.1 N/mm while nitinol’s stiffness averaged 48.9 ± 12.4 N/mm. There was no statistically significant difference between the stiffness of the constructs under static compression (P = .83). The average yield point for titanium versus nitinol constructs was 320 ± 87 versus 320 ± 36 N; the difference was not statistically significant (P = .98) (Table 1). All constructs failed at the screw-rod interface. Static compression load-displacement curves for all constructs can be seen in Figure 3.
Static Compression Structural Properties of Constructs With Titanium and Nitinol Rods.
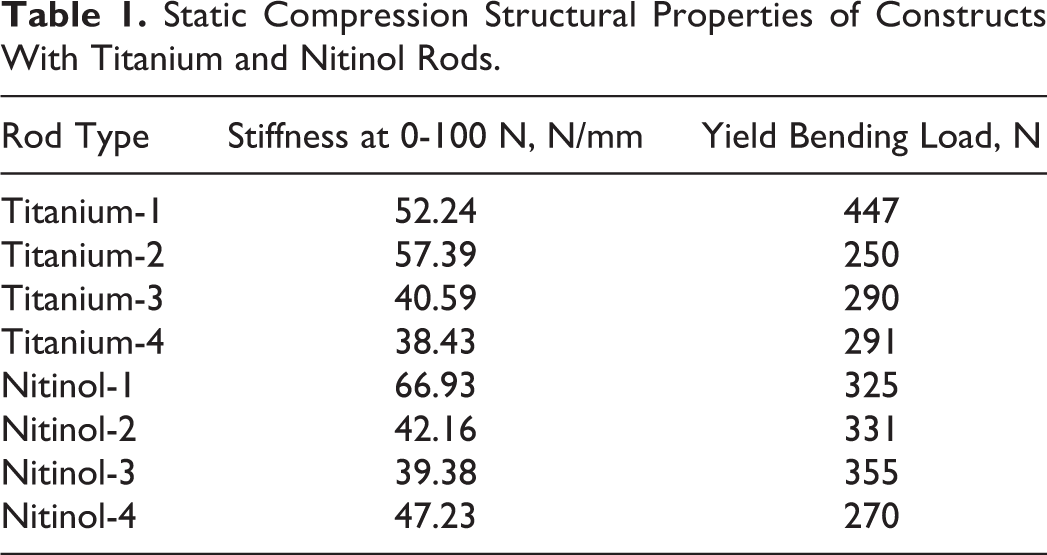
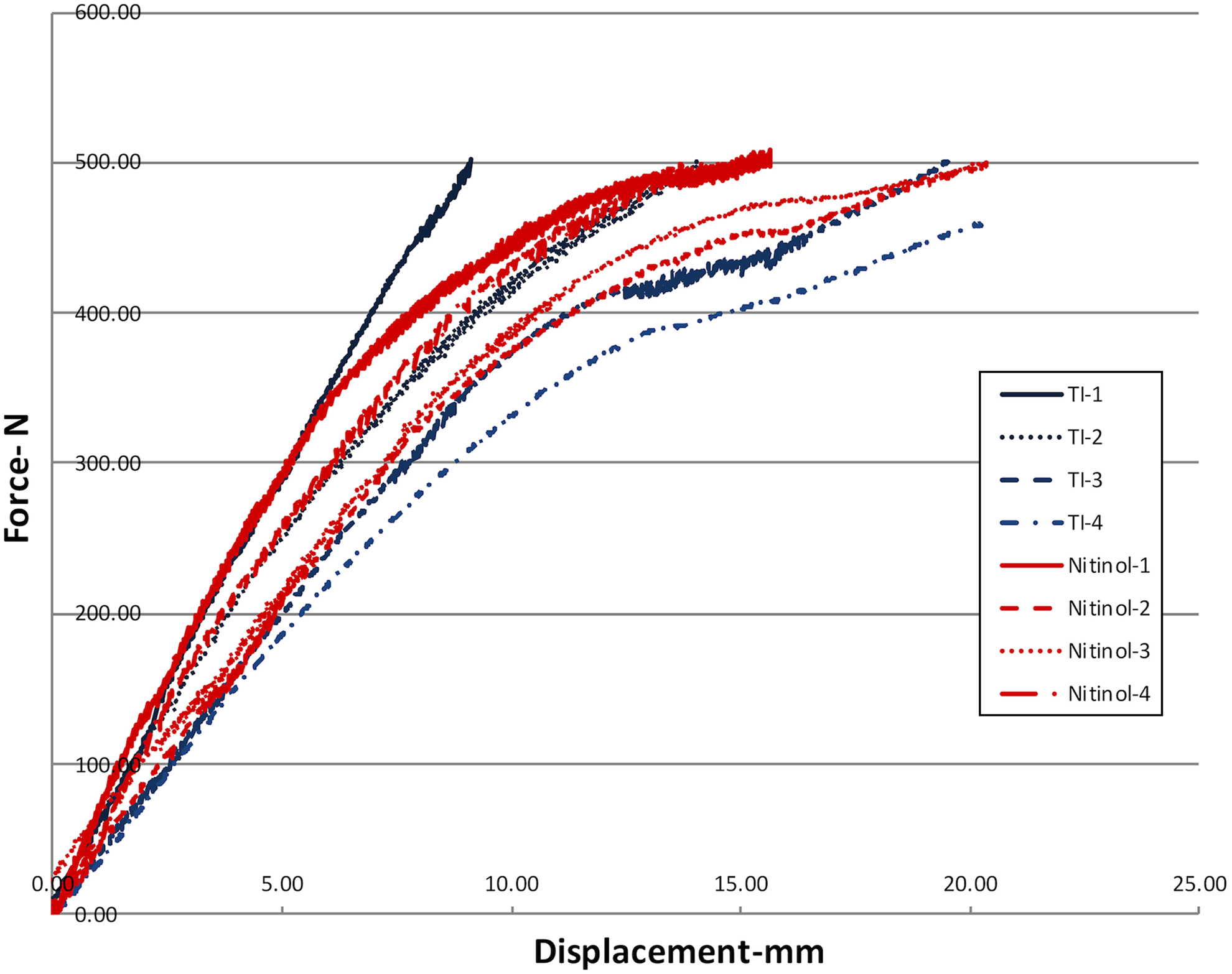
Load-displacement curves for titanium and nitinol constructs in static compression. There was no statistically significant difference between the stiffness (P = .83) or the yield point for titanium versus nitinol constructs (P = .98).
Axial Rotation Testing
During axial rotation testing, the nitinol rod system showed no torsional stiffness difference than the titanium system: 0.95 ± 0.03 versus 0.96 ± 0.17 Nm/deg, respectively, P = .91 (see Table 2). There was a statistically significant difference between the average torsional yield point for the titanium constructs (14.4 ± 1.6 Nm/deg) and nitinol constructs (21.3 ± 0.8 Nm/deg) (P = .004). Angular displacement curves for all constructs are presented in Figure 4.
Biomechanical Data of Titanium Versus Nitinol Posterior Spine Instrumentation System Tested in Torsional Mode.
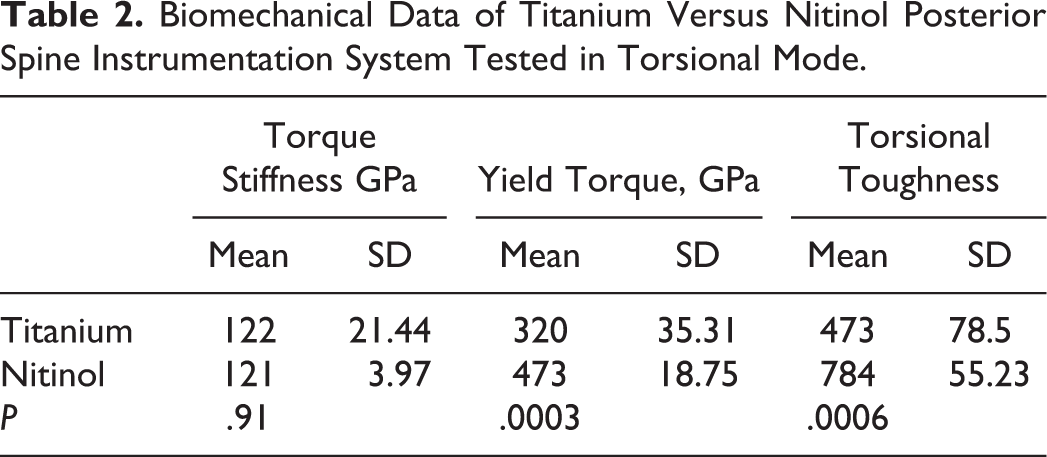
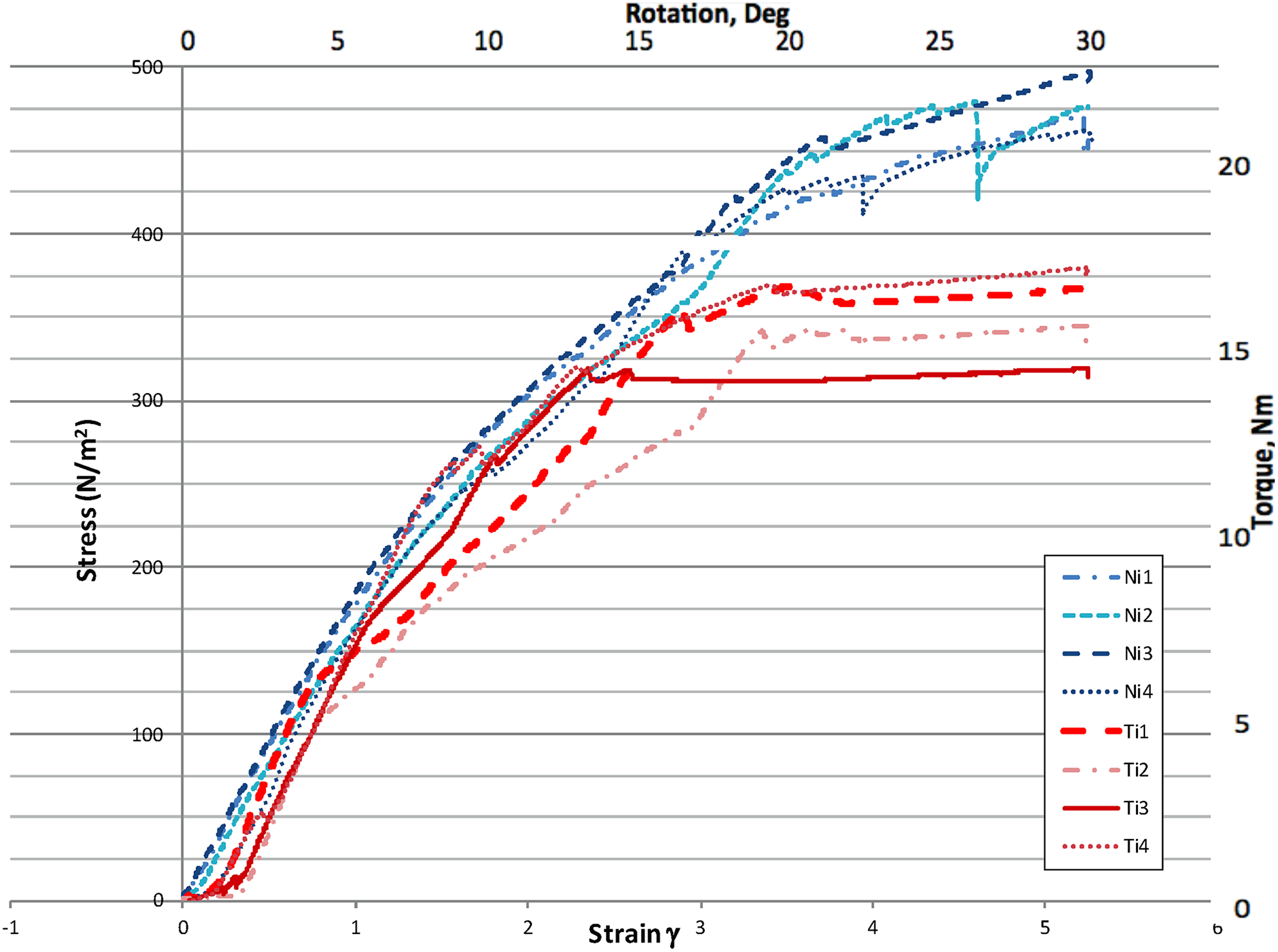
Torsional load displacement curves of titanium and nitinol samples. Values are expressed as both stress (N/m2) / strain and torque (Nm) / rotation (degrees). The nitinol rod system showed no difference with stiffness (P = .91) but a higher yield point than the titanium system (P = .004) with torsional testing.
Torsional force and angular displacement were converted to stress (N/m2) and strain in order to evaluate toughness. The torsional stiffness of the spine construct nitinol rods was not statistically different from the titanium rods: 121 versus 122 GPa (P = .91). The torsional toughness of the nitinol constructs was statistically greater than the titanium rods: 473 versus 784 GN/m3 (P = .0006) (see Table 2).
Fatigue Testing
There was no significant difference for the nitinol group sustaining a higher number of fatigue cycles until failure than the titanium group (181 660 cycles vs 64 104 cycles, respectively; P = .22) (see Table 3). All the titanium rods failed due to a single rod breaking. The nitinol constructs failed due to 1 rod breakage, 1 screw breakage, and 1 failure at the rod-screw interface.
Number of Fatigue Cycles Until Failure of Nitinol Group Versus Titanium Group.
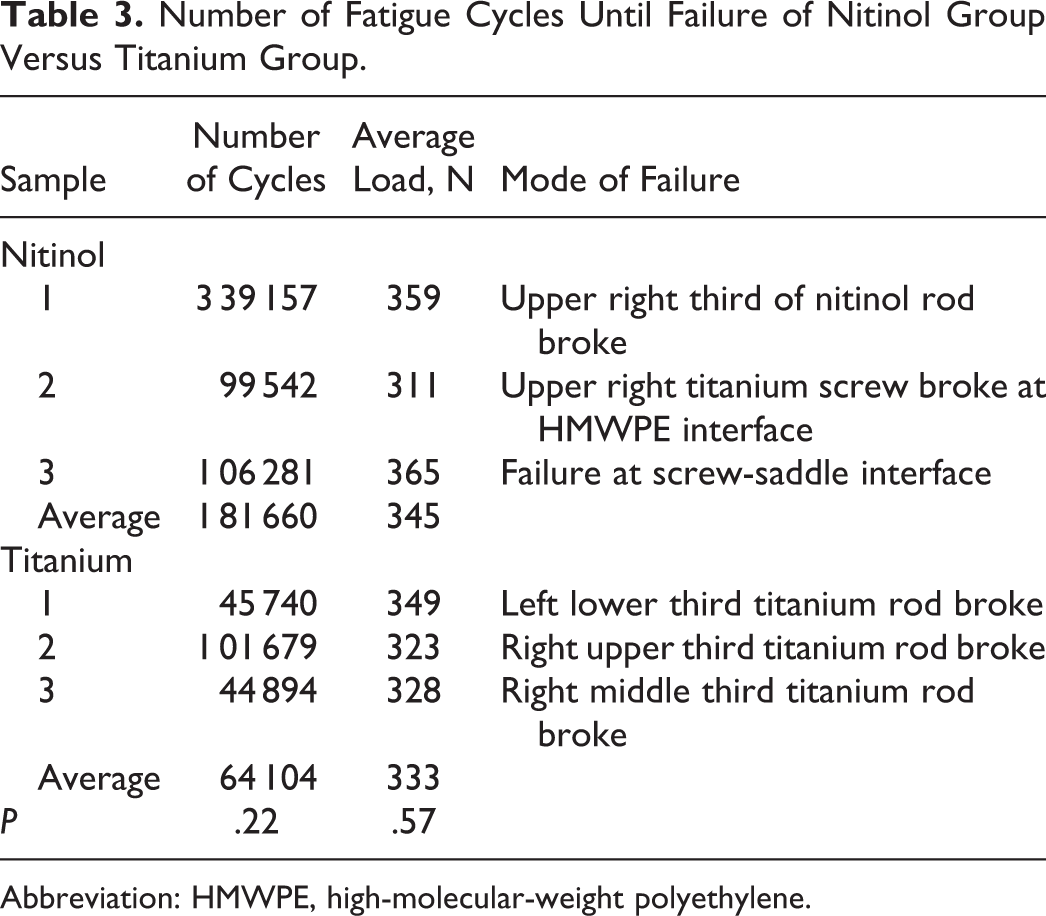
Abbreviation: HMWPE, high-molecular-weight polyethylene.
Discussion
In a corpectomy model, nitinol rods appear to have biomechanical properties comparable to those of titanium and may have more resistance to failure during torsion, which is similar to the results in the biomechanical testing demonstrated by Kok et al. 1 Nitinol exhibits variable rigidity and shape memory performance, depending on its temperature. These properties can be mapped over a temperature range over which 2 unique physical states exist. At lower temperatures, the metal exists in a “martensite” state, possessing less rigidity and thus being more flexible and more easily manipulated from its original shape. 12 When the metal is warmed into its transformational temperature range, it begins transitioning into an “austenite” state, which exhibits a significant increase in rigidity while simultaneously resuming its initial heat-programmed memory conformation. In other words, the metal can be programmed to return to a desired shape when it is heated.
A second novel characteristic of this material is that its warm austenite (maximally rigid) phase has an adjustable rigidity. This range is created by the specific temperature and time components of the metal’s heat programming or annealing process and can be pre-calculated and customized. 13
Nitinol has a well-established history of being safe for use in medical applications, the best known of which is for self-expanding vascular stents. Memory metals are also used safely for vena cava filters, atrial septal occlusion devices, and endoscopic tools. In a study by Rhalmi et al, 9 using an animal model, nitinol and titanium alloy interbody fusion devices were implanted into a rabbit spine. No nitinol ions were found in the animals’ lymph nodes, distant organs, or adjacent neural structures. 9 Nitinol has also been shown to have superior magnetic resonance imaging (MRI) compatibility over 316L stainless steel alloy. 8,14
The biocompatibility of nitinol is similar, if not superior, to that of titanium. As previously mentioned, implants should have a Young’s modulus similar to that of bone, which is between 4 and 30 GPa. Ti-6Al-4V, 316L stainless steel, and cobalt chromium have a Young’s modulus of 120 193.1, and 210 GPa, respectively. 15 -19 Previous reports described the Young’s modulus of nitinol in its austenite state to be 120 GPa. 20 In the current study, our nitinol alloy had a modulus of elasticity of 74.21 GPa, which is much closer to that of bone than other commonly used alloys. Recent evidence has shown that porous nitinol cages have more bony ingrowth than titanium intervertebral cages filled with autologous iliac crest bone graft, likely owing to its similar Young’s modulus. 4 Similar to titanium, nitinol is extremely biocompatible, undergoing self-passivation with a titanium oxide layer. 8
When examining corrosion properties of nitinol, it appears to have less chance of wear then other metals. Venugopalan et al 21 assessed the corrosion behavior of nitinol in vivo and in vitro and found that because of the self-passivation and formation of a titanium oxide layer, nitinol exhibits increased resistance to surface damage when compared with 316L stainless steel. Nickel released from nitinol was shown to be well below dietary levels and falls rapidly to nearly undetectable levels after a few days in a physiologic medium. 21 Similarly, Kazimierczak et al 22 evaluated the risk of galvanic corrosion in stent grafts and found that direct contact between stainless steel and nitinol alloys creates electric potential, but with minimal risk of galvanic corrosion.
Currently, no pedicle screws made of nitinol are available on the market. Studies investigating galvanic interactions between nitinol and other commonly implanted materials have shown high compatibility between nitinol and its base metals, including titanium. 23 Thus, it is possible to conclude that nitinol rods could be applied in conjunction with existing titanium pedicle screw products.
In addition to all these beneficial properties, for use in spinal instrumentation nitinol was recently tested for use in correction of spinal deformity. Lavelle et al 5 prevented scoliosis curve progression with vertebral body stapling. Nitinol staples were soaked in ice-cold water and then distracted. Next, they were inserted into pilot holes in the convex side of multiple vertebral bodies. After the patient’s body temperature heated the staples, they compressed the apex of the curve. 5
Nitinol has also been used for posterior instrumentation as a memory loop. A clinical case series resulted in successful fusion in 9 patients at a mean follow-up interval of 16.6 months. 24 Wever et al, 25 using a pig model, showed that nitinol rods could not only create a 40° curve but also maintain it until fusion. They placed cold straightened nitinol rods, programmed to a 40° curve, into multilevel constructs. After the rods were heated by the pigs’ body temperature, their spines deformed to 40°. 25 Another case control series showed that, compared with traditional correction techniques, patients with scoliosis curves corrected with nitinol shape memory alloy rod had less operative time and blood loss and improved correction in the coronal plane. 26
As previous studies have shown, nitinol rods can be applied as a unique “force of correction” intraoperative tool for scoliosis curves. 26 Traditional scoliosis curve correction methods can be cumbersome; nitinol memory metal scoliosis correction results in less blood loss, less operative time, and better correction of curve. For difficult curves, nitinol rods can be cooled, curved by hand, and then positioned with pedicle screws. As the body temperature heats the rods, they straighten the spine and maintain this correction until fusion. 25,27
One weakness of this study is the low sample size. While a power analysis showed a sufficient sample size, future studies should utilize larger numbers. Additionally, this study is solely a synthetic corpectomy model. Future studies should use cadaver specimens in addition to synthetic models. Finally, the fatigue testing could be performed at lower loads. One of the theoretic advantages of nitinol is that it has high resistance to fatigue. At lower loads, the nitinol constructs may have a statistically significant higher number of fatigue cycles until failure than the titanium constructs. Clearly, nitinol rods have diverse possible applications for spinal instrumentation. Yet, questions still need to be answered regarding their safety and efficacy. Further animal studies should be done to quantify the amount of nickel and titanium ions that enters the bloodstream. Several studies have demonstrated that this alloy is safe to use for spinal instrumentation, has low MRI artifact, and can be used to achieve successful clinical outcomes. 4,5,9,12 -14,24 -26
Conclusion
This study provides biomechanical evidence that nitinol rods used in a posterior construct are comparable to titanium rods with regard to compression and have increased torsional failure load and torsional toughness. While nitinol trended toward superior fatigue resistance, there was no significant difference in nitinol versus titanium construct fatigue resistance.
Footnotes
Acknowledgments
The manuscript was copyedited by Linda J. Kesselring, MS, ELS.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
