Abstract
Study Design:
Meta-analysis of randomized controlled trials (RCTs).
Objectives:
The aim was to analyze the efficacy of zoledronic acid (ZA) versus denosumab in the prevention of spinal cord compression in patients with spine metastases from advanced cancers, by evaluating all available RCTs on this subject.
Methods:
A systematic search of electronic databases (PubMed and MEDLINE) was performed to identify all published RCTs comparing ZA with denosumab in prevention of spinal cord compressions in spine metastases. Risk of bias of the studies was assessed. The primary outcomes evaluated were spinal cord compression.
Results:
Three RCTs (5274 patients) were included. Denosumab was not significantly superior to ZA in reducing the likelihood of spinal cord compression, when all tumor types were combined (odds ratio [OR] 0.92, 95% confidence interval [CI; 0.66, 1.28], P = .66). Denosumab was not significantly favored over ZA in endodermal origin (breast and prostate; OR 0.72, 95% CI [0.43, 1.19], P = .20) and mesodermal origin tumors (solid tumors and multiple myeloma; OR 1.10, 95% CI [0.72, 1.69], P = .66).
Conclusion:
Denosumab does not significantly reduce the likelihood of spinal cord compressions in comparison to ZA in patients with spine metastases. When spinal cord compressions were grouped by tumor origin (endodermal or mesodermal), there remained no significant difference between denosumab and ZA. Further long-term studies are needed to determine the effectiveness of these treatment regimens.
Introduction
Metastases to the bone is one of the most common complications associated with advanced cancer. Approximately 350 000 individuals in the United States die each year with bone metastases. 1 Bone metastases typically stem from malignant breast (73%), prostate (68%), and lung (36%) cancers. Patients with bone metastases are at risk of devastating skeletal-related events (SREs), including spinal cord compressions, requiring prompt referral to a spine surgeon for appropriate management. 2 -4
Bisphosphonates are the main treatment used to reduce the number of SREs in patients with multiple myeloma, or bone metastases from advanced cancers (breast, prostate, or solid tumors). 2,5,6 Zoledronic acid (ZA) is a bisphosphonate and regarded as one of the current gold standard treatments to reduce, but not completely eliminate, SREs. Systemic ZA has recognized side effects including an increased risk of osteonecrosis of the jaw and renal insufficiency. 5,7 -9 Therefore, alternate therapies are needed to further reduce the frequency of SREs with fewer adverse effects. Another systemic therapy, denosumab, a human monoclonal antibody that binds to the RANK ligand, has been shown as a noninferior alternative to ZA. 10
The aim of our study was to investigate the efficacy of ZA versus denosumab in the prevention of spinal cord compression in cancer patients by evaluating the randomized controlled trials (RCTs).
Source of Funding
There were no external funding sources for this study.
Material and Methods
We selected RCTs comparing bisphosphonates versus denosumab in patients with bone metastases from advanced cancer and reported outcomes of spinal cord compression prevention. Studies were excluded if they considered children (<16 years) or they had a follow-up of <12 months.
We searched the English literature using PubMed and MEDLINE on April 20, 2019, with different terms and synonyms for “bone metastases,” “Bisphosphonates,” and “Denosumab.” In addition, the reference lists of previously published randomized trials and systematic reviews were manually searched for additional eligible studies. The titles and abstracts of the search results were screened, and in case of presumed eligibility, full-text articles were reviewed by 2 independent reviewers (HF and AF).
The data extraction from each study includes year of publication, randomization method, and patient and treatment characteristics. Seven aspects of the studies related to the risk of bias were assessed, following the instructions in the Cochrane Handbook for Systematic Reviews of Interventions. 11 Also, the studies were evaluated specifically for publication bias using a funnel plot.
RevMan software (Version 5.3; The Cochrane Collaboration) 12 was used for the analysis. Treatment effects were estimated by calculating the odds ratio (OR) with 95% confidence interval (CI) for dichotomous variables, and the mean difference with 95% CI for continuous variables. Studies were weighted by the inverse of the variance (IV) of the outcome, and a fixed-effects model was used for all analyses.
Results
The search terms, as described above, identified 119 references (Figure 1). Of the 8 articles eligible for analysis, 5 studies needed to be excluded because the number of SREs were not listed by type, specifically the number of spinal cord compressions. Three RCTs were included in the meta-analysis with a total of 5274 patients (denosumab group = 2636 and ZA group = 2638).
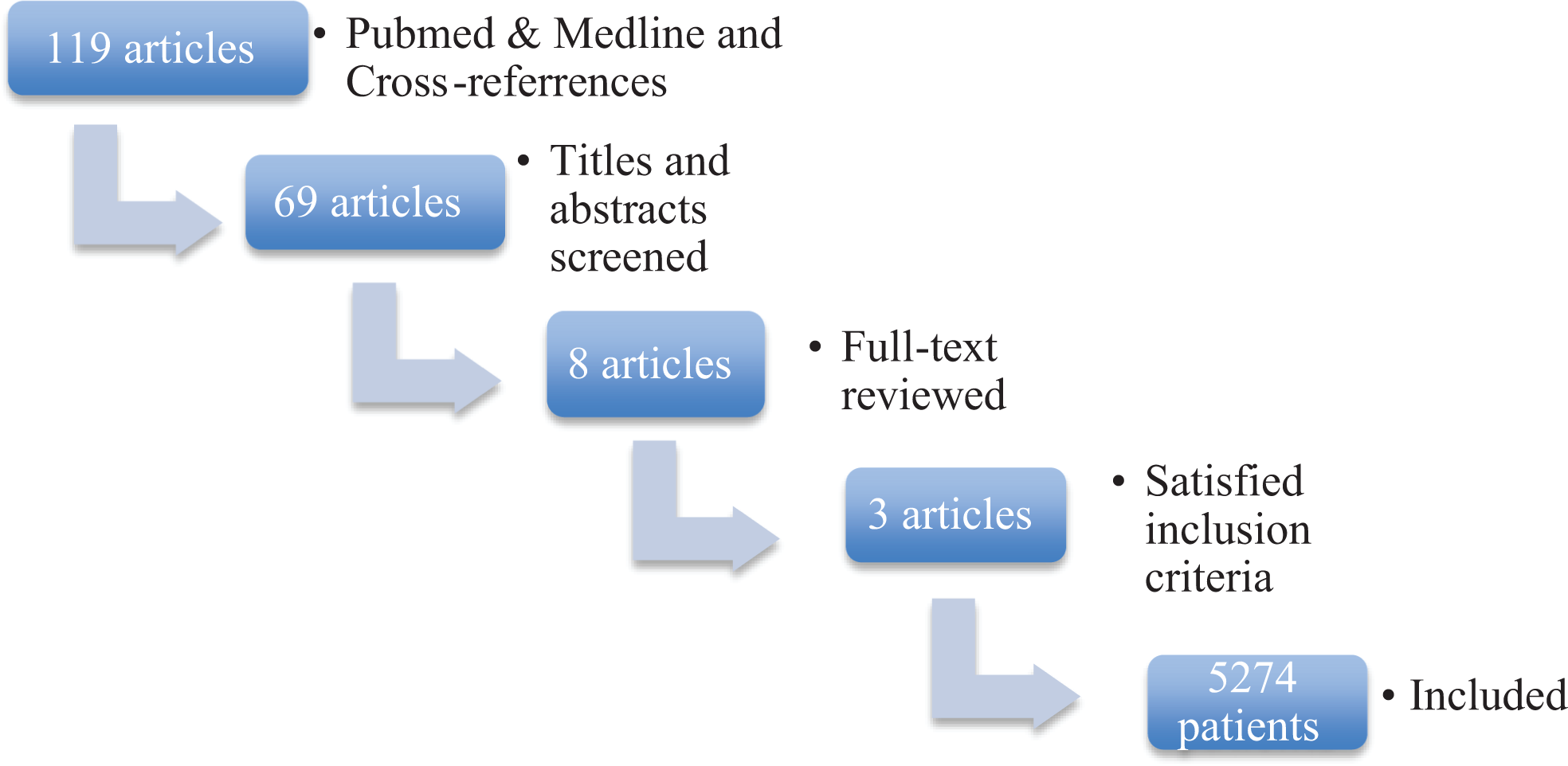
Flow chart illustrating the article screening process.
Study Characteristics and Quality
The sample size of the included trials ranged from 797 to 951 patients (Table 1). Two trials included patients with mesodermal tumors (or solid tumors and multiple myeloma), 13,14 and the other trial included patients with endodermal tumors (breast or prostate cancer). 15 In addition, one study did not specifically mention the number of spinal cord compressions, only denoting the total number of SREs. 14 Amgen was consulted to determine the number of spinal cord compression in both the denosumab and ZA groups.
Characteristics of Studies Included in Meta-Analysis.
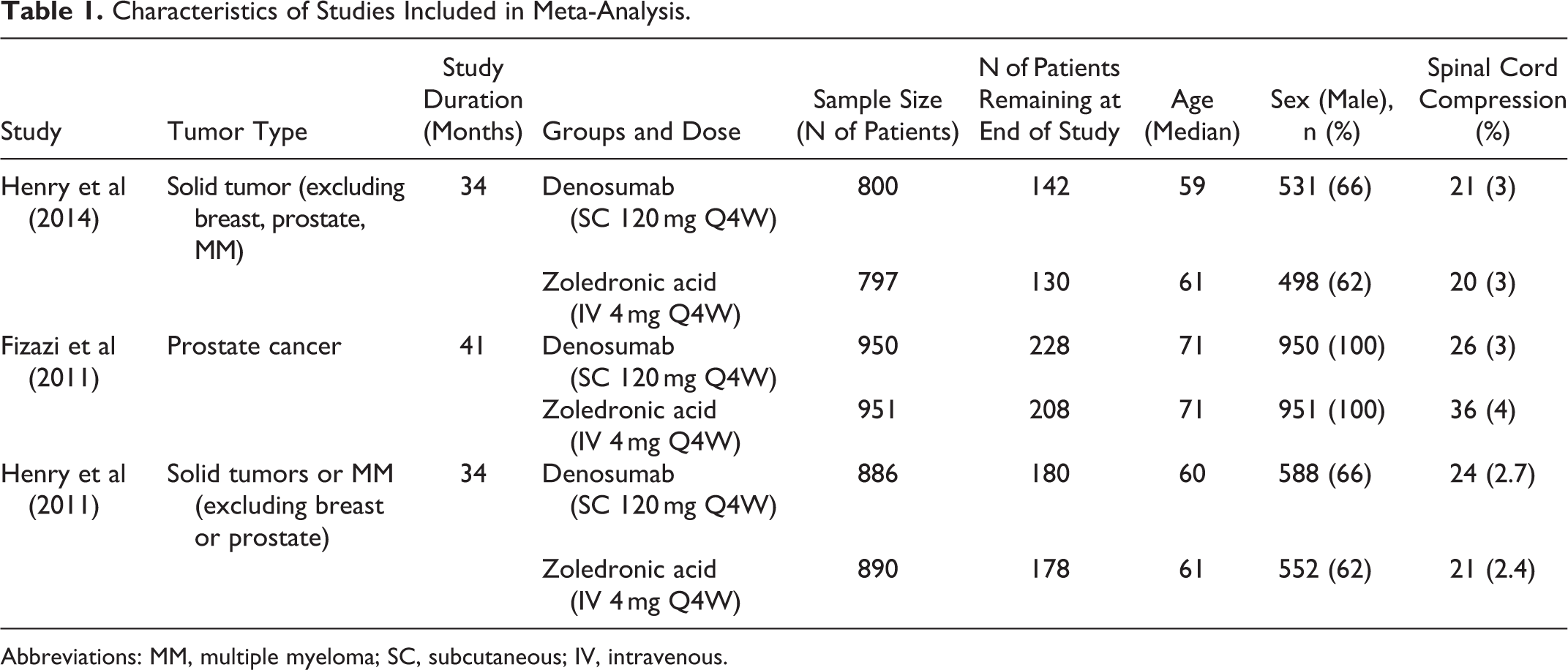
Abbreviations: MM, multiple myeloma; SC, subcutaneous; IV, intravenous.
In each of the 3 trials, denosumab was administrated subcutaneously at 120 mg every 4 weeks, and ZA was administered intravenously at 4 mg every 4 weeks. Furthermore, all of the 3 trials concluded that denosumab was significantly more effective in preventing SREs in patients with bone metastases in comparison to ZA.
A summary of the risk of bias in the included studies is presented in Figure 2 along with a funnel plot in Figure 3. The included RCT studies were all high quality, based on the Cochrane bias risk assessment. From the funnel plot, it is shown that the 3 studies included are well distributed on both sides, in terms of publication bias (Figure 3).

Risk of bias summary according to Cochrane risk of bias assessment tool. “+” = low risk of bias; “−” = high risk of bias; “?” = minimal information and cannot judge risk of bias.
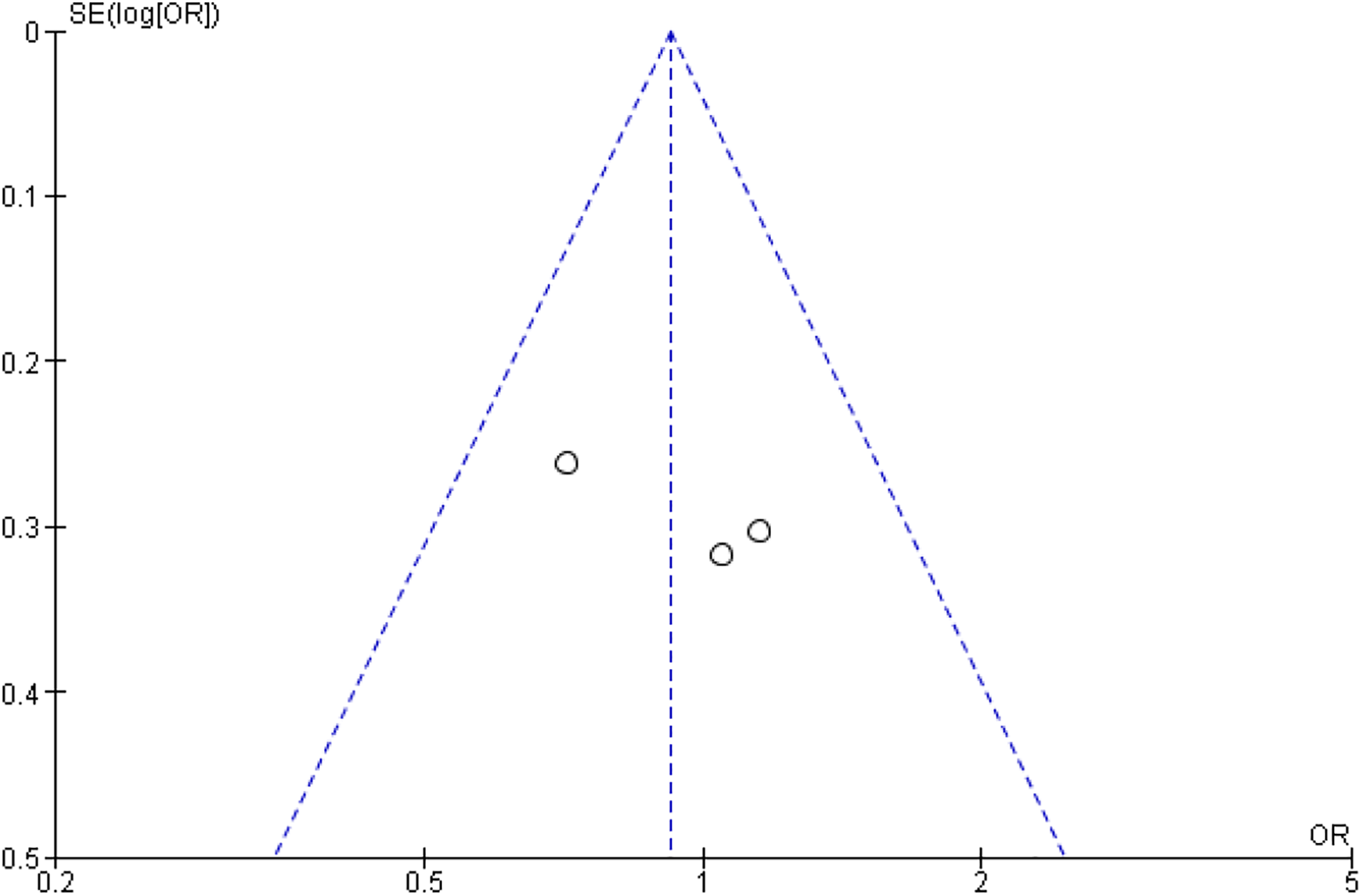
Funnel plot illustrating level of publication bias.
Effect of Denosumab in Comparison to Zoledronic Acid
From all studies combined, independent of tumor origin, the effect size estimate favored the denosumab group over ZA in spinal cord compressions, although not significantly (OR 0.92, 95% CI [0.66, 1.28], P = .66; Figure 4). Similarly, when malignancies were divided by tumor origin, denosumab was not significantly favored over ZA in endodermal origin (breast and prostate; OR 0.72, 95% CI [0.43, 1.19], P = .20; Figure 5A) and mesodermal origin tumors (solid tumors and multiple myeloma; OR 1.10, 95% CI [0.72, 1.69], P = .66; Figure 5B).
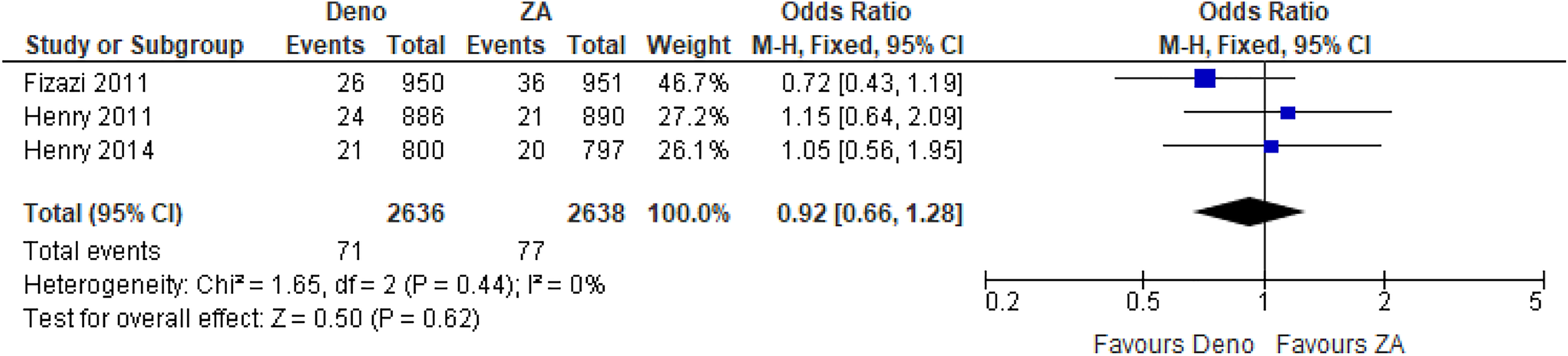
Spinal cord compression events presented as odds ratio with 95% confidence interval for 2 treatment groups in total, after denosumab versus zoledronic acid (ZA) treatment.
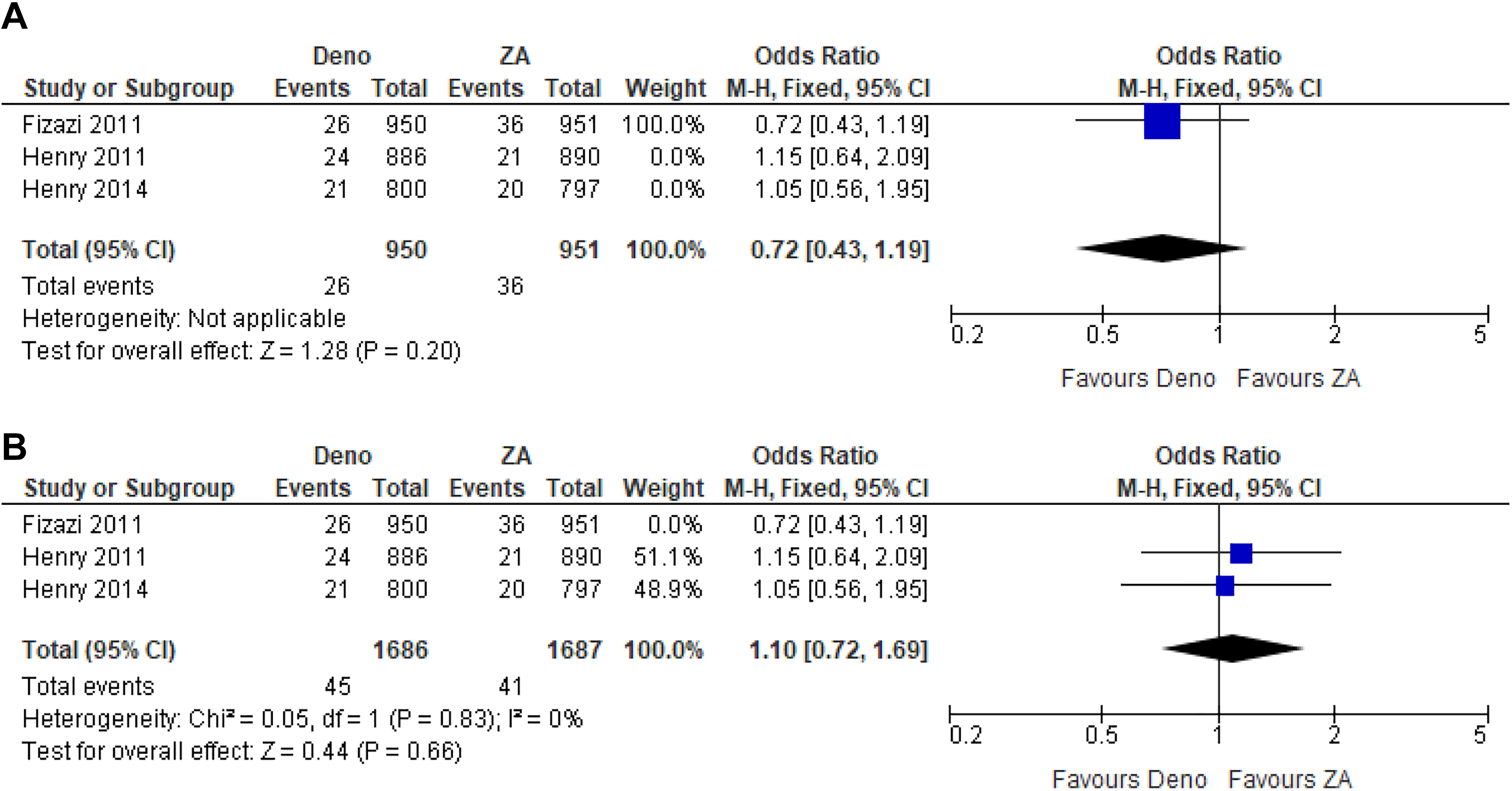
Spinal cord compression events presented as odds ratio with 95% confidence interval for 2 treatment groups: (A) in endodermal cancers (breast and prostate) and (B) in mesodermal cancers (solid and multiple myeloma), after denosumab versus zoledronic acid (ZA) treatment.
Discussion
Current modalities for management of metastatic spine disease include radiotherapy, surgery, and systemic chemo-/antiresorptive therapy. 16 Surgery has proved to be the most effective intervention in patients with neurological deficits and bony instability. 17,18 However, this is not without risk, as more than 10% of these patients must be re-operated on often due to hardware failure or other complications, 19,20 which can become a costly burden on the health care system. 21 Additionally, tumor recurrence and continuous local bone loss lead to the importance requirement for systemic chemo- and antiresorptive therapy.
This meta-analysis of 3 RCTs that evaluated a total of 5274 patients shows that denosumab reduced, but not significantly, the likelihood of spinal cord compression by 8% in comparison to ZA in the treatment of spine metastases. There was also no significant difference between the 2 groups when patients were categorized by tumor origin, as either endodermal or mesodermal origin. However, previous meta-analyses concluded denosumab as significantly superior to bisphosphonates, reporting an effect estimate favoring denosumab in terms of SREs, and time to first SRE. 22,23
While there was reduction in the number of spinal cord compression, denosumab is not a superior alternative to ZA for advanced tumors in the prevention of spinal cord compression, a major cause of morbidity associated with significant bone pain and paralysis. 2 Adverse effects of denosumab and ZA were not evaluated in this study, and thus, we cannot comment on the safety profile of these respective treatment regimens. The treatments may not be suitable depending on the patients’ health conditions.
ZA and denosumab are 2 antiresorptive treatments with differing mechanisms of action, both helping reduce the likelihood of SREs, which includes spinal cord compressions. ZA is a bisphosphonate that directly inhibits osteoclastic-mediated activity, through accumulation in the mineralized bone matrix and subsequent release during bone resorption. Studies have also suggested that ZA may exhibit antitumor effects, including inhibition of tumor cell migration, invasion, proliferation, and viability, further reducing skeletal tumor burden and bone metastasis. 24 -27 In comparison, denosumab is a monoclonal antibody that binds with high affinity to RANKL, a key mediator in osteoclastic formation and activity, thereby disrupting bone resorption. 28,29 The disruption of the RANKL signaling pathway by denosumab may be a possible explanation for the small reduction of spinal cord compression with denosumab, in comparison to ZA, despite this reduction being not significant.
The strength of this meta-analysis was the comprehensive and robust search of the literature. This search ultimately yielded 3 high-quality RCTs, based on the Cochrane risk of bias summary. The funnel plot did not have a skew, although this was difficult to judge with the small number of studies. Since funnel plots are often skewed and asymmetrical in the presence of publication bias, 30 the presence of publication bias is unlikely.
Despite the comprehensive search of the literature from electronic databases, the main limitation with this meta-analysis is the small number of included RCTs. Effect estimates of drug treatments overall, and in subgroups categorized by tumor origin, yielded no significant results, since few RCTs were included in this study. These effect estimates of drug treatments may vary with more studies and, thus, a greater number of patients included. Furthermore, many others studies have published data comparing denosumab and ZA in terms of SREs; however, the number of SREs by type, such as spinal cord compression, were not described. 14,31 -34 Amgen was consulted for data concerning SREs by type in one study, allowing for inclusion of this data within the analysis. 14
In conclusion, this meta-analysis shows that denosumab is not significantly superior in reducing the likelihood of spinal cord compression compared to ZA in patients with spine metastases. Furthermore, denosumab was not superior to ZA in the reduction of spinal cord compressions when advanced tumors were grouped by origin. Therefore, we cannot recommend generalized use of denosumab over ZA in the reduction and prevention of spinal cord compression in patients with spine metastases. Further large-scale studies are required to determine the effectiveness of the medications to reduce spinal cord compressions in advanced cancers.
Footnotes
Authors’ Note
We had full access to all the data in the study and take responsibility for the integrity and accuracy of the data, as well as the decision to submit for publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
