Abstract
Study Design:
In vitro biomechanical study.
Objectives:
The objective of this in vitro biomechanical range-of-motion (ROM) study was to evaluate spinal segmental stability following fixation with a novel anterior cervical discectomy and fusion (ACDF) device (“novel device”) that possesses integrated and modular no-profile, half-plate, and full-plate fixation capabilities.
Methods:
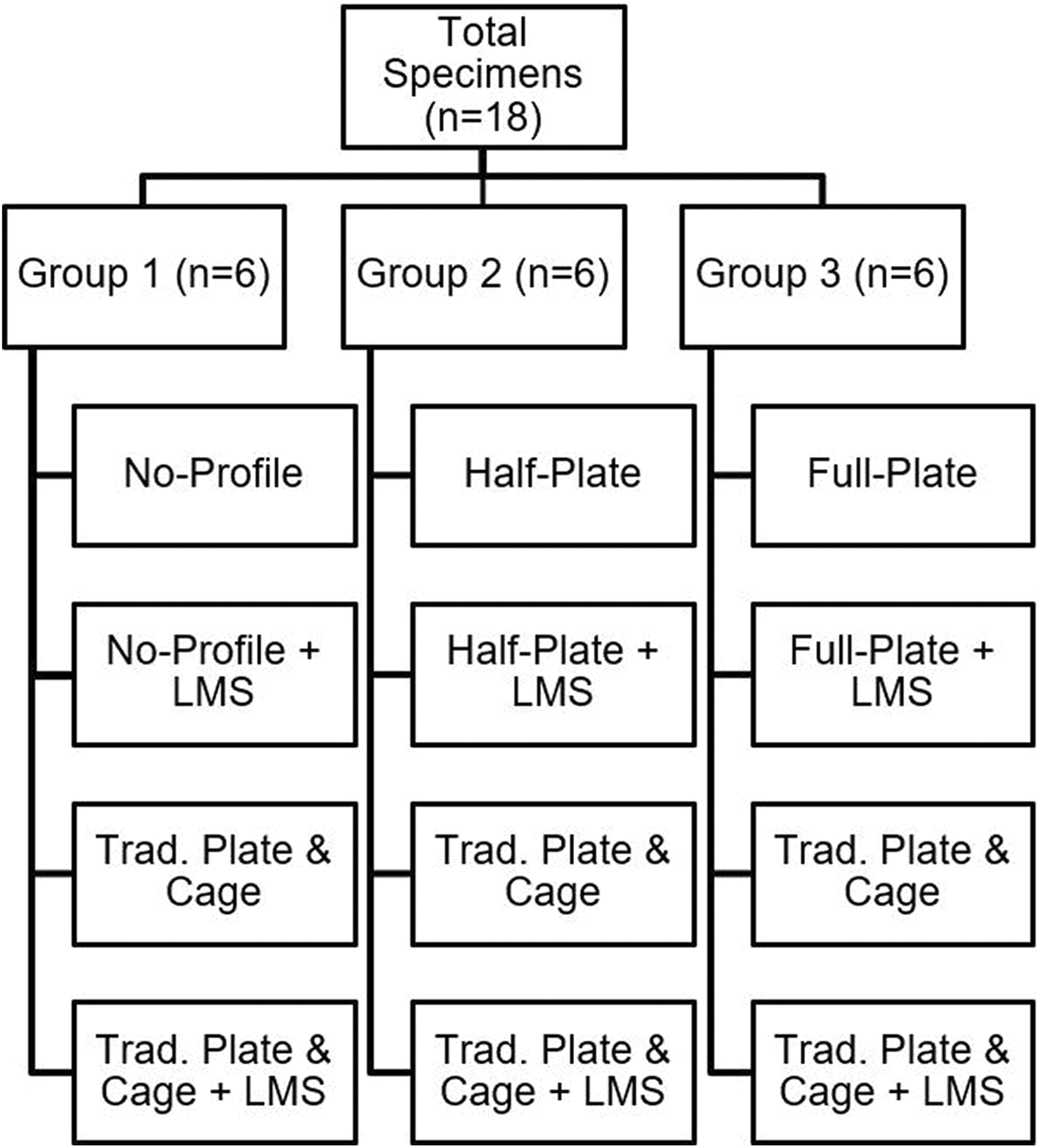
Human cadaveric (n = 18, C3-T1) specimens were divided into 3 groups (n = 6/group). Each group would receive one novel device iteration. Specimen terminal ends were potted. Each specimen was first tested in an intact state, followed by anterior discectomy (C5/C6) and iterative instrumentation. Testing order: (1) novel device (group 1, no-profile; group 2, half-plate; group 3, full-plate); (2) novel device (all groups) with lateral mass screws (LMS); (3) traditional ACDF plate + cage; (4) traditional ACDF plate + cage + LMS. A 2 N·m moment was applied in flexion/extension (FE), lateral bending (LB), and axial rotation (AR) via a kinematic testing machine. Segmental ROM was tracked and normalized to intact conditions. Comparative statistical analyses were performed.
Results:
Key findings: (1) the novel half- and full-plate constructs provided comparable reduction in FE and LB ROM to that of traditional plated ACDF (
Conclusions:
The novel ACDF device may be a versatile alternative to traditional no-profile and independent plating techniques, as it provides comparable ROM reduction in all principle motion directions, across all device iterations.
Keywords
Introduction
Anterior cervical discectomy and fusion (ACDF) is a common surgical technique in treating symptomatic disc degeneration, segmental instability, and trauma of the cervical spine. Characterized by acute segmental motion reduction, ACDF has demonstrated robust fusion capabilities and subsequent clinical improvement. 1,2 While the placement of an intervertebral graft alone aides in anterior fusion by stretching the annulus, breach of the anterior and posterior longitudinal ligaments diminishes stability in flexion and extension. Accordingly, an anterior cervical plate is readily used in ACDF and has been documented to further improve sagittal stability and rate of fusion. 3,4 However, both surgical exposure and plate profile/footprint have been shown to play a role in subsequent adjacent soft tissue injury (dysphagia and dysphasia) and contiguous level pathology (heterotopic ossification). 5 -7
Consequently, the ACDF paradigm has seen a considerable shift toward techniques possessing a decreased access window and a minimal anterior device footprint/profile. Next-generation short stature plates and integrated “low-profile” or “no-profile” anchored devices have emerged. 8 -11 While a number of biomechanical studies have documented good mechanical stability with such devices, the majority have suggested that the integrated design provides less stability in flexion and extension than traditional cage and plate constructs. 10 -17 Moreover, when considering the limitations of instrument angulation, presence of adjacent and/or previous hardware, and the degree of sagittal correction desired, it remains apparent that a “one size fits all” approach to ACDF is not possible.
More recently, a novel ACDF construct capable of both integrated screw and modular plate fixation (MPF) was developed to provide greater device versatility beyond the current spectrum of ACDF constructs (Figures 1 and 2). The device supports in situ transitioning between no-profile, half-plate, and full-plate fixation. This provides a more comprehensive ACDF strategy that may better accommodate pathological and anatomical variables on a patient-specific level. Furthermore, the connected MPF design, which creates a singular and continuous body about the index level, facilitates optimal plate orientation/alignment, significantly diminishes potential for cage migration and/or subsidence, and promotes physiological compression of the cage/graft.

Modular anterior cervical discectomy and fusion (ACDF) construct with integrated fixation: no-profile (left), half-plate (middle), full-plate (right).
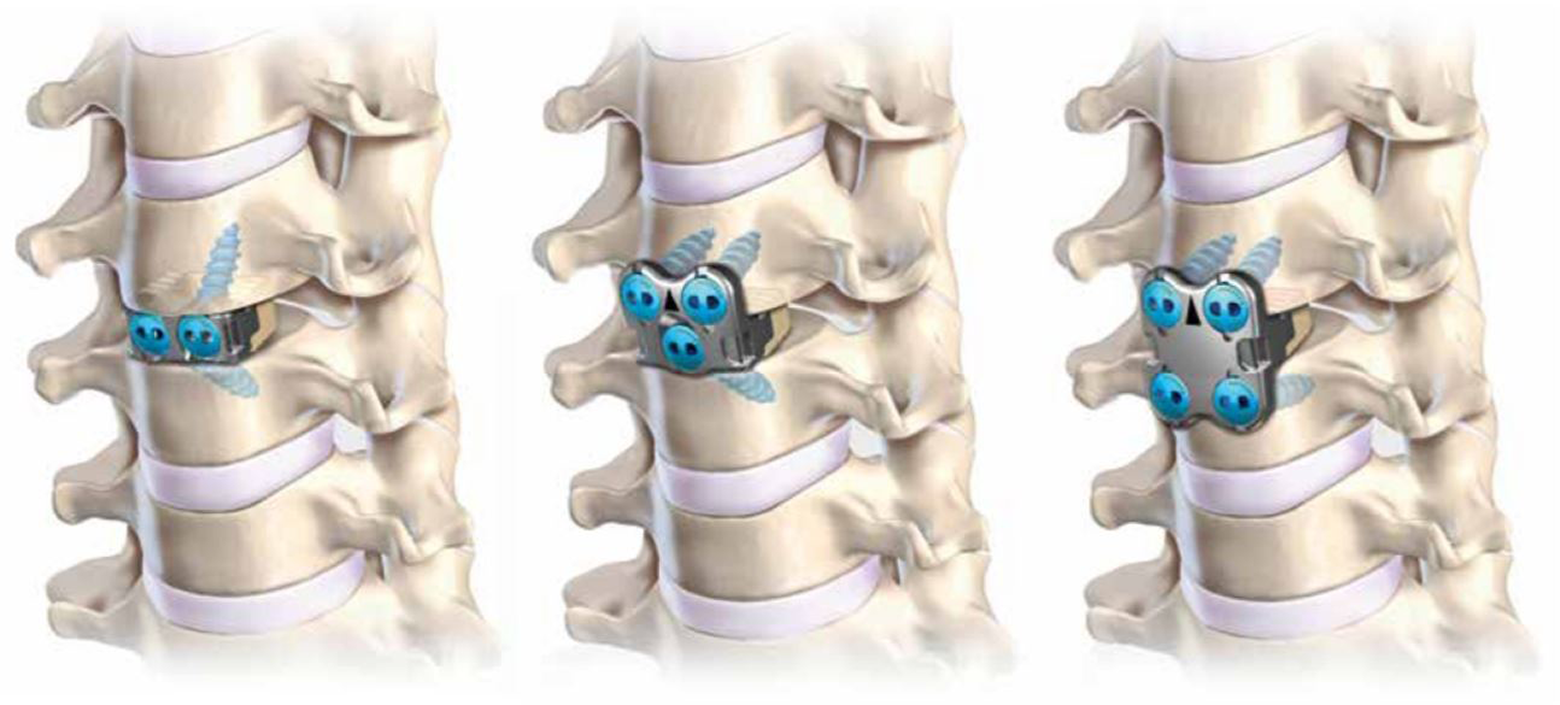
Anatomical renderings of modular anterior cervical discectomy and fusion (ACDF) construct with integrated fixation: no-profile (left), half-plate (middle), full-plate (right).
While these features present with an array of perceived biomechanical, ergonomic, and clinical benefits, no data exists yet in the literature. The objective of this study was to assess the segmental stability achieved with the novel ACDF (“novel”) construct, across all iterations, utilizing traditional plated ACDF and anteroposterior cervical fusion constructs as controls.
Materials and Methods
Specimen Preparation and Instrumentation Technique
Eighteen (n = 18) fresh-frozen human cadaveric spine specimens (C3-T1) were tested (7 females, 11 males; mean age 59.1 ± 11.1 years). Each spine was thawed at room temperature, the cervicothoracic specimens were resected, the ligamentous structures were maintained, and the residual musculature and adipose tissue was removed. Structural integrity was confirmed via standard anteroposterior and lateral radiographs. Any specimens exhibiting previous surgery or anatomical discrepancy were excluded. Bone mineral density (BMD) evaluations were performed by dual-energy X-ray absorptiometry (DEXA) scans. The terminal ends of each specimen were potted for test apparatus attachment using standard wood screws placed in the vertebral bodies and anchored within high-strength resin (Bondo Body Filler; 3M, St Paul, MN, USA). The potted specimens were sealed in plastic bags and maintained frozen at –20°C until approximately 10 hours before testing, at which time they were thawed at room temperature (∼25°C).
Specimens were divided into 3 groups such that the mean BMD values for each group were comparable. The 3-group protocol was used such that each specimen group received a single iteration of the novel ACDF device (full-plate, n = 6; half-plate, n = 6; or no-profile, n = 6) (Alta ACDF System; Zimmer Biomet Spine, Westminster, CO, USA) in addition to the traditional (control) plate (MaxAn Anterior Cervical Plate System; Zimmer Biomet Spine, Westminster, CO, USA) and cage (C-THRU Anterior Spinal System; Zimmer Biomet Spine, Westminster, CO, USA) construct. It should be noted that the novel ACDF device is a commercially available (nonprototype) ACDF device, approved by the US Food and Drug Administration for indication as a stand-alone cervical fusion device in skeletally mature patients with degenerative disc disease of the cervical spine (C3-T1) at one level. Additionally, in all 3 modular iterations, the device is composed of a PEEK-OPTIMA LT1 (Invibio Inc, West Conshohocken, PA, USA) intervertebral spacer, with titanium plate and screws (Figures 1 and 2).
Separate grouping allowed for greater bone preservation during iterative testing, as single specimen instrumentation with all 3 modular device conditions would greatly compromise bone quality for each subsequent construct. Mean BMD values for each group were as follows: group 1 (novel no-profile), 0.79 ± 0.14 g/cm2; group 2 (novel half-plate), 0.73 ± 0.11 g/cm2; and group 3 (novel full-plate), 0.75±0.15 g/cm2; respectively. Mean
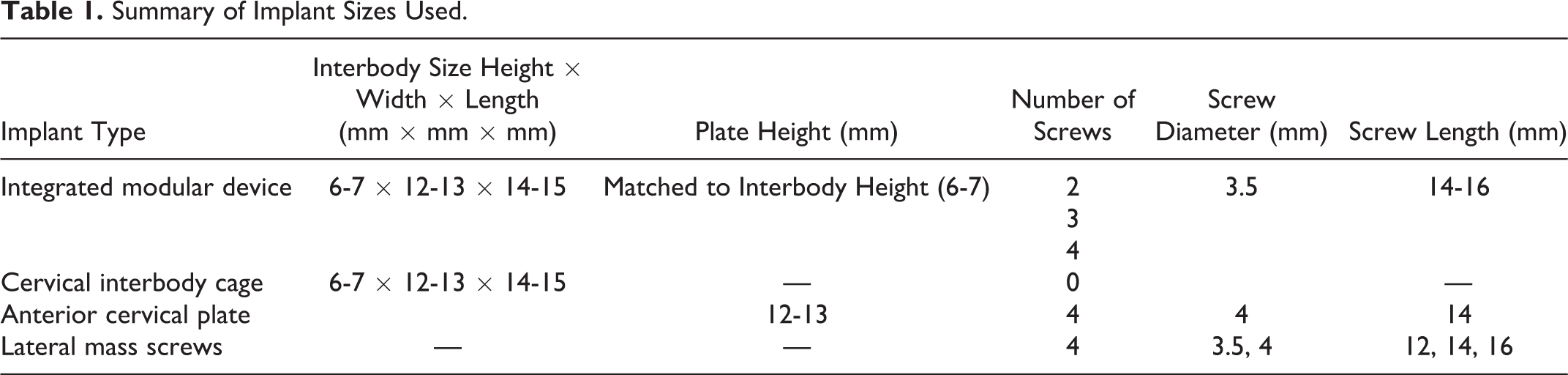
All procedures and instrumentation were performed under fluoroscopic guidance by a board certified, fellowship-trained, spine surgeon in a clinically representative manor (Figure 3A-C; Table 1). Lateral mass screws (LMS) were placed bilaterally (Lineum OCT Spine System; Zimmer Biomet Spine, Westminster, CO, USA) (screw diameters, 3.5, 4 mm; screw lengths, 12, 14, 16 mm). Range of interbody cage (all iterations) height, width, and length were 6-7 mm, 12-13 mm, and 14-15 mm, respectively, and the size of the corresponding modular plate was matched to that of the interbody cage. Traditional anterior plates were either 12 or 13 mm in height and received 4.0 mm (diameter) by 14 mm (length) screws. All novel device iterations received 3.5 mm (diameter) by 14 or 16 mm (length) screws. All novel device half-plate constructs were placed with the plate oriented caudally. It is emphasized that the half-plate construct can be oriented cranially or caudally, however, for sake of consistency, only the caudal orientation was chosen in the present study. This rationale was based on evidence showing a greater risk for adjacent level ossification at the cranial level when using a traditional plate. 5,18 Furthermore, given the inherent lordosis of the cervical spine, less bony work is necessary to position a plate over the caudal body.
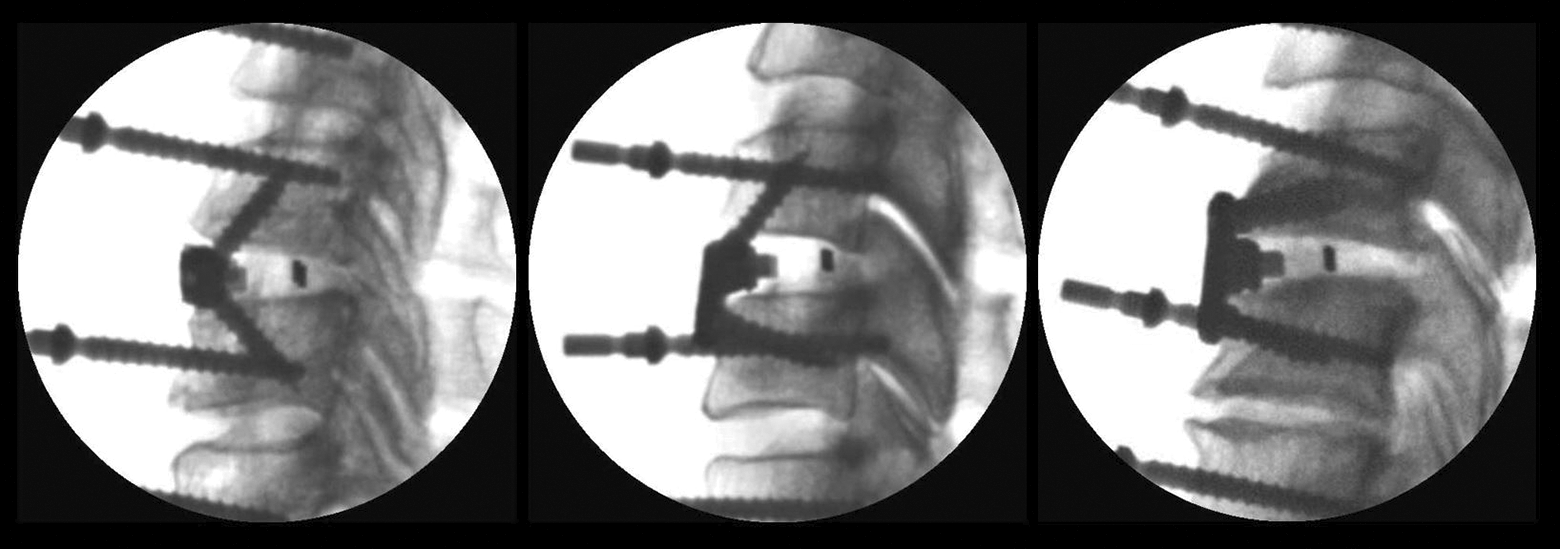
Lateral fluoroscopic images of modular anterior cervical discectomy and fusion (ACDF) construct with integrated fixation, taken prior to range-of-motion (ROM) testing: no-profile (left), half-plate (middle), full-plate (right).
Summary of Implant Sizes Used.
Testing and Motion Analysis Protocol
A 6 degree-of-freedom (6-DOF) kinematic testing machine (Bionix Spine Kinematics System, MTS Corporation, Eden Prairie, MN, USA) was used to apply nonconstraining, nondestructive, pure-moment loading in the 3 principal motion directions. Specimens were affixed with optoelectronic markers and mounted within the test apparatus at the C3 and T1 pots (Figure 4). The caudal pot attachment afforded translation in the

Potted C3-T1 specimen affixed with optical markers within the kinematic testing system (Bionix Spine Kinematics System, MTS Corporation, Eden Prairie, MN, USA).
Specimen distribution and testing order. LMS = lateral mass screws
Three-dimensional motion of each vertebral body was recorded, in all cycles, relative to their adjacent caudal level (C3-C4, C4-C5, C5-C6, C6-C7, C7-T1), as well as the 3-dimensional motion of the cumulative specimen (C3-T1) using an optoelectronic motion measurement system (Optotrak Certus Motion Capture System; Northern Digital Inc, Waterloo, Ontario, Canada). Each optoelectronic triad maker was coupled to its respective level to establish a local coordinate system. Additionally, two optoelectronic markers were rigidly attached to the static test frame to define the +
Statistical Analysis
Intact data outcomes for each ROM within each group were compared using 3 one-way analyses of variance. The natural log of the ROM outcome data was taken to force a normal distribution within the data sets. Next, a linear mixed model framework was used to model the natural log of each ROM outcome (flexion-extension, lateral bending, and axial rotation) as a function of the 9 interventions, using intact ROM as a reference category. A random intercept was included in the model to account for correlation due to multiple measurements taken on each sample. All pairwise comparisons of the change from the intact state were made between the interventions. To maintain a .05 type 1 error rate, a Bonferroni correction was made to the
Ethics Approval
No medical ethics committee approval or institutional review board was required for this cadaveric study. All specimens were obtained from an accredited tissue bank service (Science Care, Inc, Phoenix, AZ, USA). All donors and the next of kin were fully legally competent and consented to use for research. Written informed consent from the donor or the next of kin was obtained.
Results
Mean ROM outcomes are summarized in Table 2, and in Figures 6, 7, and 8, respectively. No significant differences existed within a ROM outcome and between groups for the intact data sets (
Summary of Mean Range-of-Motion (ROM) Outcomes.a
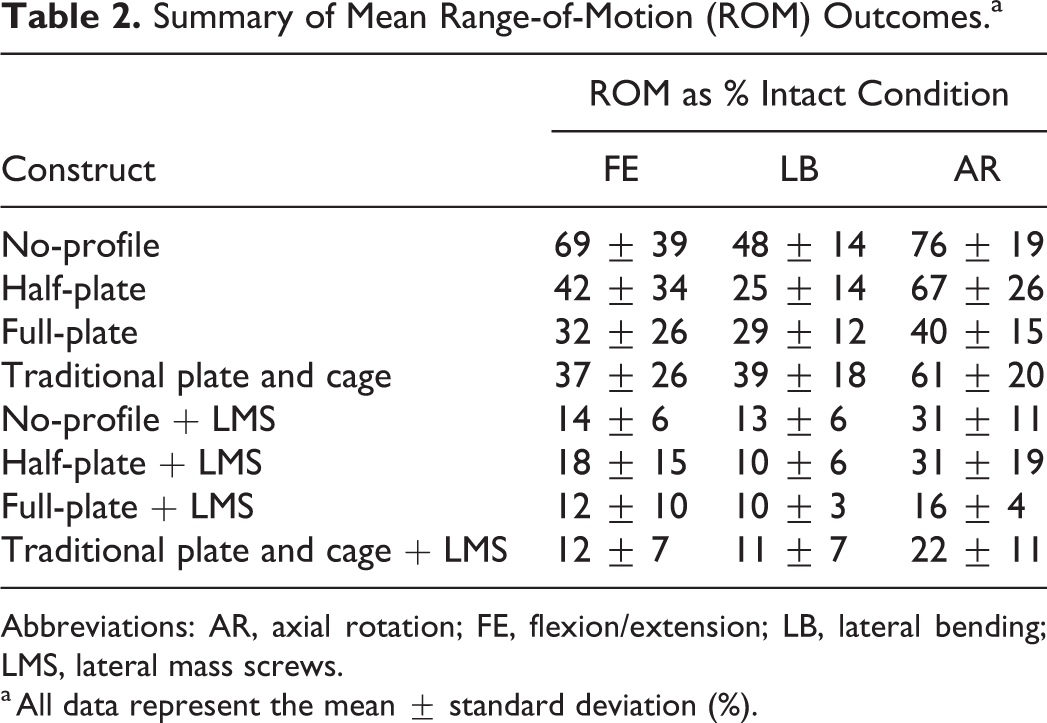
Abbreviations: AR, axial rotation; FE, flexion/extension; LB, lateral bending; LMS, lateral mass screws.
a All data represent the mean ± standard deviation (%).
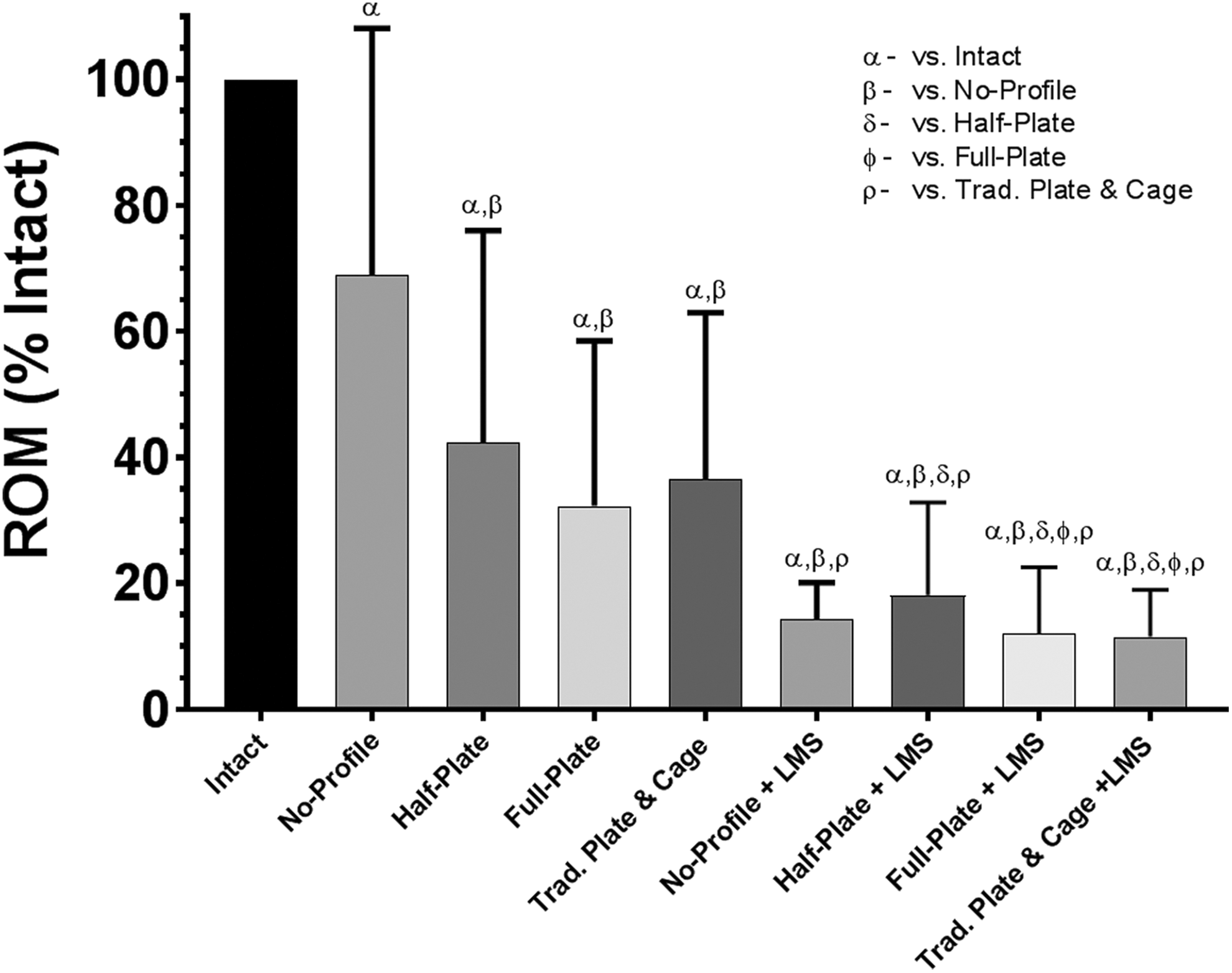
Range of motion (ROM), relative to intact conditions, when loaded in flexion-extension under a pure moment of ±2.0 Nċm. Bars represent the mean and error bars are standard deviation. Symbols denote significant differences between groups according to a repeated-measures analysis of variance with Bonferonni’s correction for multiple comparisons.
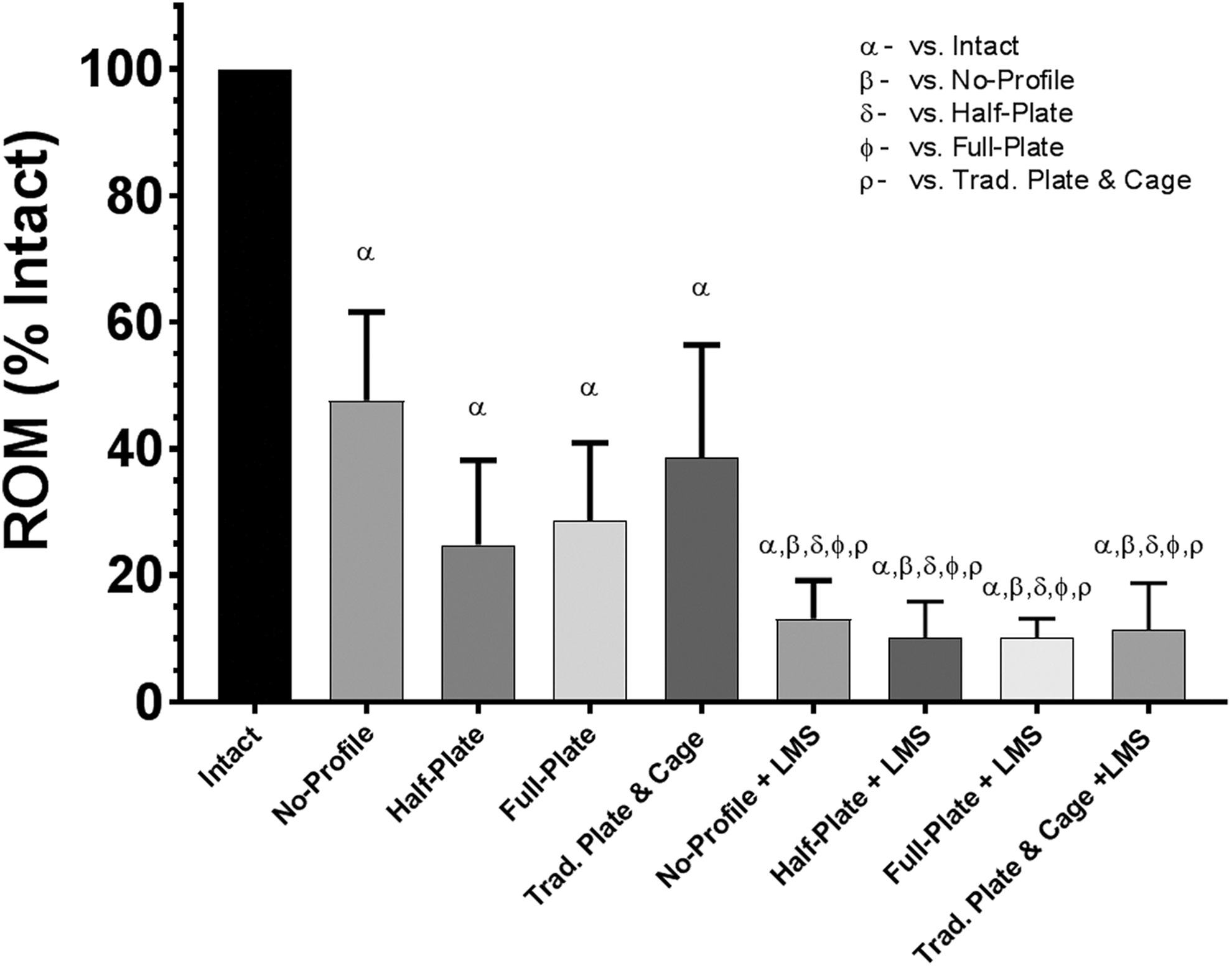
Range of motion (ROM), relative to intact conditions, when loaded in lateral bending under a pure moment of ±2.0 Nċm. Bars represent the mean and error bars are standard deviation. Symbols denote significant differences between groups according to a repeated-measures analysis of variance with Bonferonni’s correction for multiple comparisons.
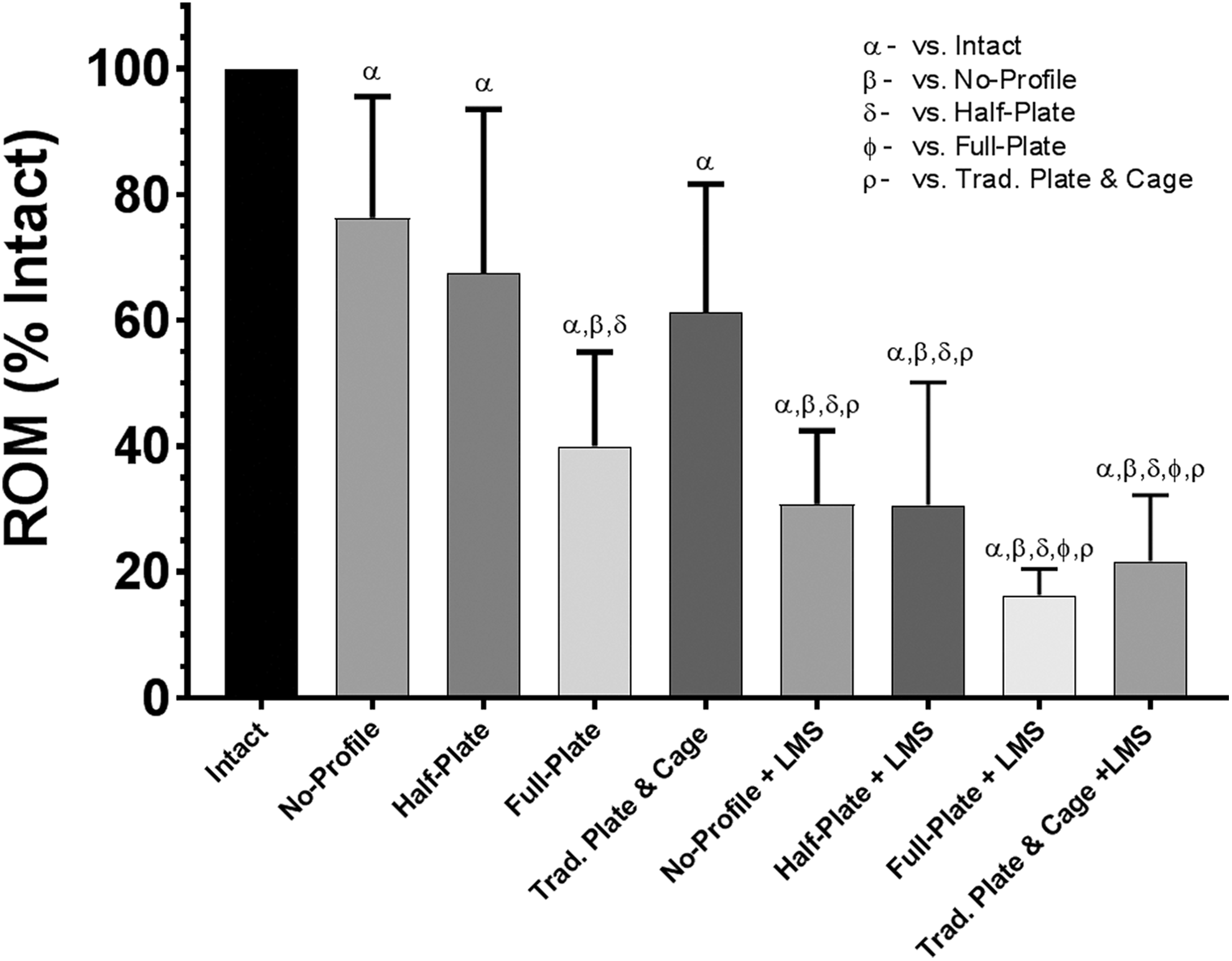
Range of motion (ROM), relative to intact conditions, when loaded in axial rotation under a pure moment of ±2.0 Nċm. Bars represent the mean and error bars are standard deviation. Symbols denote significant differences between groups according to a repeated measures analysis of variance with Bonferonni’s correction for multiple comparisons.
Flexion/Extension
All plated constructs (+/− LMS) achieved significance reduction in ROM over the novel no-profile device (
Lateral Bending
Anterior-only constructs significantly reduced ROM from that of intact (
Axial Rotation
No significant differences in ROM reduction were observed between the novel no-profile, half-plate, or traditional plate constructs (
Discussion
The present biomechanical study assessed a novel integrated and modular (“novel”) ACDF device, possessing multiple face-plate fixation options (no-profile, half-plate, and full-plate), using a traditional ACDF (plate + cage) as a control. While integrated fixation features have become prevalent in ACDF, no device has been studied in which multiple face-plate fixation mechanisms can be leveraged and further adapted in situ. 9 -13,15 -17,22 -24 It was postulated that such a design may improve the ease and safety of implantation, while also improving mechanical stability.
Pertaining to the latter, the current study found that the novel full-plate construct provided comparable stability to traditional plated ACDF in all motion planes, reaching significance in AR (
However, the comparable stability of the novel full-plate construct, considered in the context of a consolidated insertion/implantation technique and streamlined cage/plate orientation, may still be of unique clinically utility. Given that emerging evidence suggests that both retraction time and end-stage sagittal alignment play a role in subsequent pathology following ACDF, future clinical assessment of potential ergonomic and biomechanical benefits resultant from the integrated half-plate design is warranted. 27 -29
The novel half-plate construct may also help in avoiding the limitations introduced by preexisting adjacent level instrumentation. Means to achieving secondary adjacent level instrumentation have traditionally involved the removal, replacement and/or extension of the existing plate, use of axillary posterior instrumentation, and/or selection of a no-profile construct. These techniques often require additional and/or lengthier surgical interventions, leading to longer operative time and possibly compromising segmental stability. In the current study, the novel half-plate construct supported lower mean ROM (greater reduction) in comparison to the no-profile construct in all principle directions (FE ROM, 42% ± 34% vs 69% ± 39%; LB, 25% ± 14% vs 48% ± 14%; AR 67% ± 26% vs 76% ± 19%), achieving significance in FE. However, it should be noted that the no-profile construct in the current study possessed 2 screws, while many commercially available no-profile/zero-profile devices often possess 3 or 4 screws. Scholz et al,
10
assessing a 4-screw no-profile ACDF device, found that device ROM reduction was comparable to that of traditional ACDF plate and cage constructs (
Moreover, when used in a primary index procedure, the comparable stability of the novel half-plate construct (vs no-profile) may be of value in the high and low cervical segments when the mandible and sternum/clavicle can obstruct high-angled screw instrumentation. Additionally, given its comparable stability to traditional/full-plate constructs, the novel half-plate construct may be an effective alternative when preservation of cranial vertebral face is desired, avoiding a potential etiology for adjacent level ossification. 5,19,28
Finally, while the outcomes demonstrated by the novel no-profile construct appear of less novelty than those of the half- and full-plate constructs, the no-profile technology remains a powerful option within ACDF. As previously noted, the continued development of no- or low-profile anchored cages has been fueled by the desire to simplify surgical technique and reduce implant prominence. 8 -10 While biomechanical studies have demonstrated that the stability of no-profile constructs may be comparable to traditional plated ACDF, diminished stability is still typically observed in flexion and extension. 10 -14,22,24 Outcomes of the current study are consistent with this trend, as the novel no-profile construct was markedly less stable in flexion/extension. However, despite these biomechanical discrepancies with no-profile devices, several clinical studies have demonstrated excellent efficacy with the technology. 12,15 -17,22,23 These findings further support the assertion that ACDF is far from a “one size fits all” technique and that more versatile technologies, and subsequent characterization, is needed to support this paradigm and delineate standards of care.
The authors acknowledge several limitations in this study that are consistent with those previously reported in the literature. 10 -14,22,24 Limitations include use of cadaveric specimens, use of a non–diseased state model, no accounting for fatigue behavior, and no accounting for the presence of boney fusion. Given the already intrinsic variability across specimens, further introduction of anatomical and/or biomechanical variables can result in significant irregularities. Accordingly, the aforementioned design parameters were deemed confounding for the purpose of the present evaluation. An additional study limitation is that of the no-profile device design, which leveraged 2-point screw fixation. No-profile ACDF devices possessing 3- or 4-point screw fixation are commercially available, which can have implications on the amount of segmental stability afforded by this technique. The authors emphasize that the novelty of the data presented here pertains primarily to the performance of the integrated half- and full-plate iterations in comparison to traditional plate ACDF, irrespective of the no-profile iteration.
Conclusion
Use of an integrated and modular ACDF device, particularly in half- and full-plate fixation, can provide comparable ROM reduction to that of a traditional ACDF plate and cage. Given this finding, future clinical evaluation is warranted to better understand device design implications on perioperative and patient outcomes.
Footnotes
Acknowledgments
The authors would like thank Amy Claeson, PhD, and Vijay Permeswaran, PhD for their help in editing the article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Anup Gandhi, Sam Farmer, and Jeremy Hansmann are employees (salary) of the funding source. Ripul Panchal and John Wanebo have received consulting fees (hourly) from the funding source. John Wanebo is a design surgeon (royalty bearing) of the device examined in this study (Alta ACDF System).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Zimmer Biomet Spine.
