Abstract
Study Design:
Medicare database analysis.
Objective:
The purpose of this study was to investigate whether neurologic disorders represent a risk factor for revision after lumbar spine surgery.
Methods:
Patients who underwent lumbar spine surgery were identified from 5% Medicare Part B claims between 2005 and 2008. Cox regression analysis was used to evaluate risk factors for revision within the 7 years after the index lumbar surgery. Covariates included age, gender, race, census region, Medicare buy-in status, Charlson score, year, prior lumbar fusion within 2 years of index surgery, prior diagnosis of cervical spondylotic myelopathy treated with or without cervical spine surgery, and diagnoses of other neuromuscular conditions.
Results:
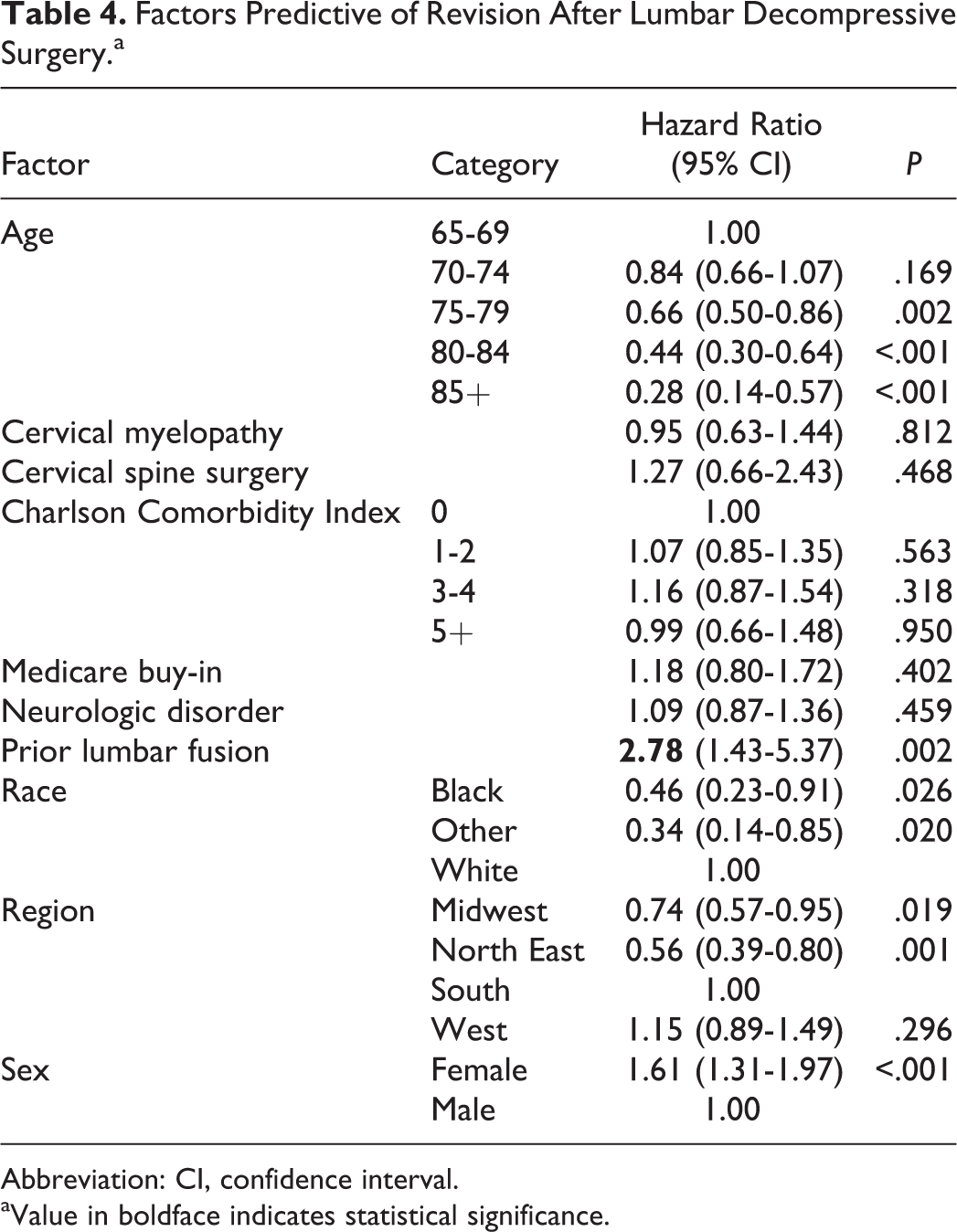
Of 8665 cases who had decompression only, 401 (5%) had a revision within 7 years after the index surgery. Factors predictive of revision were prior lumbar fusion (hazard ratio [HR] = 2.78, confidence interval [CI] = 1.43-5.37, P = .002) and being female (HR = 1.61, CI = 1.31-1.97, P < .001). Of 5501 cases who had a decompression and fusion, 752 (14%) had a revision surgery within 7 years after the index surgery. Factors predictive of revision were the presence of a neurologic disorder (HR = 1.24, CI = 1.05-1.46, P = .010), prior lumbar fusion (HR = 3.09, CI = 2.05-4.63, P < .001), and being female (HR = 1.35, CI = 1.15-1.57, P < .001).
Conclusions:
An increase in revision rate (P = 0.01, HR = 1.24) was seen in patients with neurologic disorders undergoing lumbar decompression and fusion, although not for patients undergoing decompression alone. This suggests an opportunity to improve clinical outcome and reduce revision rate through improved surgical decision making or treatment of the neurologic disorder.
Introduction
Risk stratification has been advocated as a strategy to optimize outcomes, limit complications, and increase the likelihood of a cost-effective result. 1 –3 Analysis of large datasets including the National Surgical Quality Improvement Program, the National Inpatient Sample, the Washington State Surgical Quality Outcomes Assessment Program, and the Neurosurgery Quality Outcomes Database have led to the development of risk calculators and other tools designed to aid the clinician in patient selection for surgical treatment, prediction of treatment outcomes, and the likelihood of complications. 1,2,4 –8
This strategy has been most successful in identifying risk for complications. However, the promise of risk stratification has yielded less tangible benefit in terms of either improved clinical outcome or cost optimization. Age, gender, and race have been identified as predictors of clinical outcome, 4,8 but since they cannot be modified, the operational benefit of this information is limited. Other predictive risks such as obesity, diabetes, or osteoporosis may theoretically be altered, but the time course and treatment requirements necessary to reduce the comorbid effect may conflict with timely management of the spinal disorder. 9,10 As an example, obese patients with severe spinal stenosis have difficulty exercising, and thus struggle to lose weight.
In order to leverage the value of predictive models, it is optimal to identify risk factors that can be altered, either preoperatively, postoperatively, or both. These factors can be described as modifiable risk. Relevant change in modifiable risk might influence health-related quality of life, complication profile, or revision rate. A known example is cigarette smoking in patients undergoing lumbar fusion. Smoking increases nonunion rate and adversely affects clinical outcome, whereas smoking abatement blunts those adverse effects. 11,12
This study examines the potential impact of neurologic comorbidity on revision rate in lumbar spine surgery. The presence of neurologic disease that affects standing balance is an identified, but poorly understood, predictive factor in the spine deformity population. Previous studies have shown that patients with Parkinson’s disease have a high failure rate after spinal fusion. 13,14 Furthermore, patients with a range of other neurologic diseases have a higher incidence of revision secondary to proximal junctional kyphosis. 15,16 The purpose of this study was to investigate whether neurologic disorders that impair standing balance might represent a relevant, and in some instances modifiable risk, for lumbar spine surgery.
Methods
Patients who underwent lumbar spine surgery were identified from 5% Medicare Part B (physician/carrier) claims between January 1, 2005, and December 31, 2008 (Table 1). Medicare is a publicly funded health insurance program in the United States for people age 65 or older, people under age 65 with certain disabilities, people of all ages with end-stage renal disease, and people with amyotrophic lateral sclerosis. The 5% dataset contains individual claims records for a random sample of Medicare beneficiaries, totaling about 2.4 million enrollees.
Current Procedural Terminology Code and International Classification of Diseases Code Used to Identify Cases.
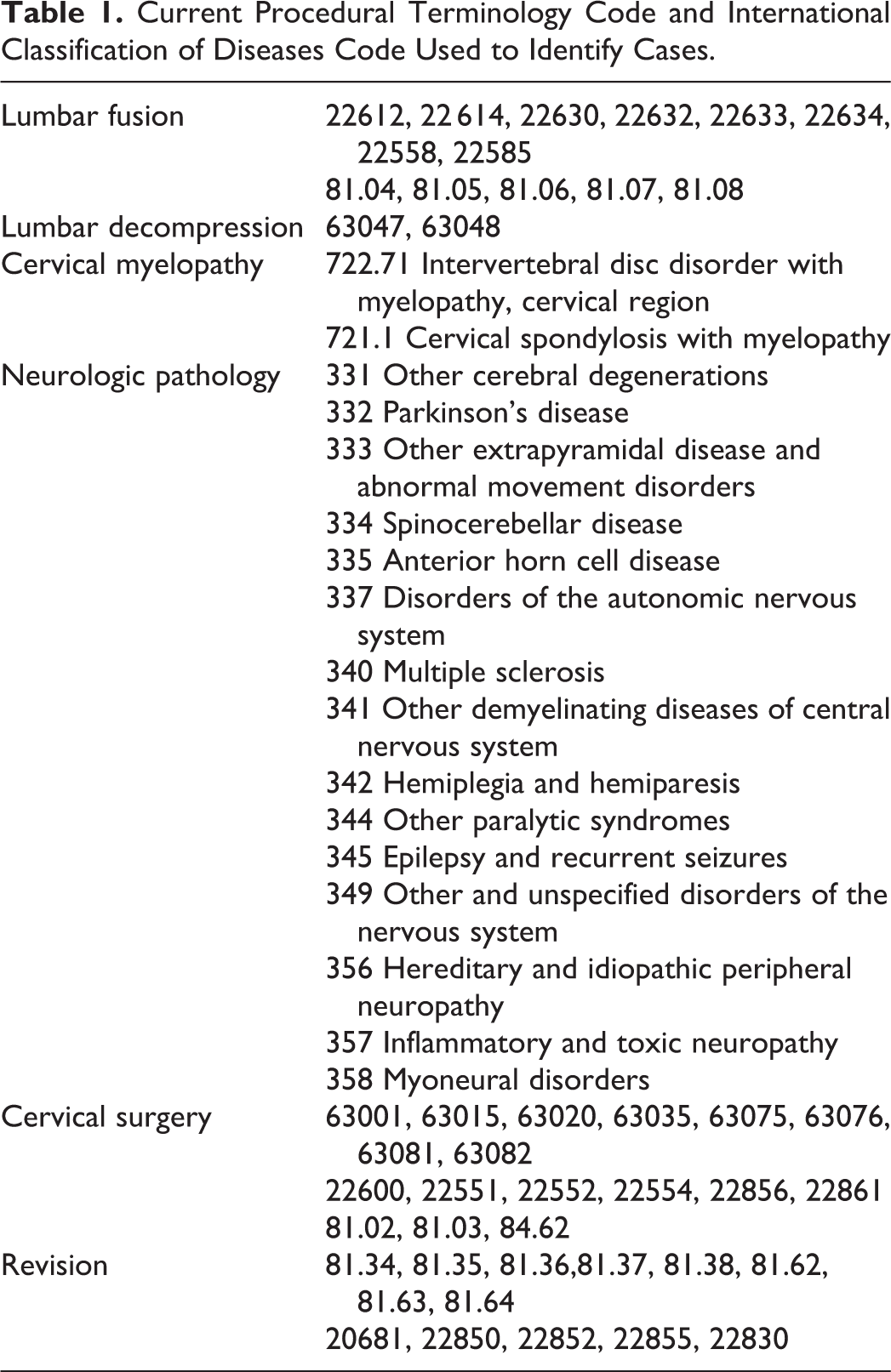
Two cohorts were identified using Current Procedural Terminology (CPT) codes: patients who had a lumbar decompression only as an index surgery (CPT codes 63 047 and 63 048) and those who had a decompression and fusion (CPT codes 22 612, 22 614, 22 630, 22 632, 22 633, 22 634, 22 558, 22 585, 81.04, 81.05, 81.06, 81.07, and 81.08). Those aged under 65 years receive Medicare insurance coverage due to their disabilities and were excluded from the study. Patients who received their Medicare health benefits through health maintenance organizations (HMOs) were also excluded because their health care expenses were not submitted to the Centers for Medicare and Medicaid Services for payment, and therefore, claims from these beneficiaries were not complete or available from the database.
Patients from both the decompression and decompression and fusion index cohorts were tracked for an additional 7 years. Multivariate Cox regression analysis was used to evaluate the risk factors for a revision lumbar surgery, either a fusion or a repeat decompression (CPT codes 81.34, 81.35, 81.36, 81.37, 81.38, 81.62, 81.63, 81.64, 20 681, 22 850, 22 852, 22 855, 2283) within the 7 years after the index lumbar surgery. The covariates in this model included age, gender, race, census region, Medicare buy-in status, Charlson score, year, prior lumbar fusion within 2 years of index surgery, prior diagnosis of cervical spondylotic myelopathy (CPT codes 722.71 or 721.1) treated with or without cervical spine surgery (CPT codes 63 001, 63 015, 63 020, 63 035, 63 075, 63 076, 63 081, 63 082, 22 600, 22 551, 22 552, 22 554, 22 856, 22 861, 81.02, 81.03, and 84.62), and diagnoses of other neuromuscular conditions (CPT codes 331 332 333 334 335 337 340 341 342344 345 349 356 357, and 358) (Table 2).
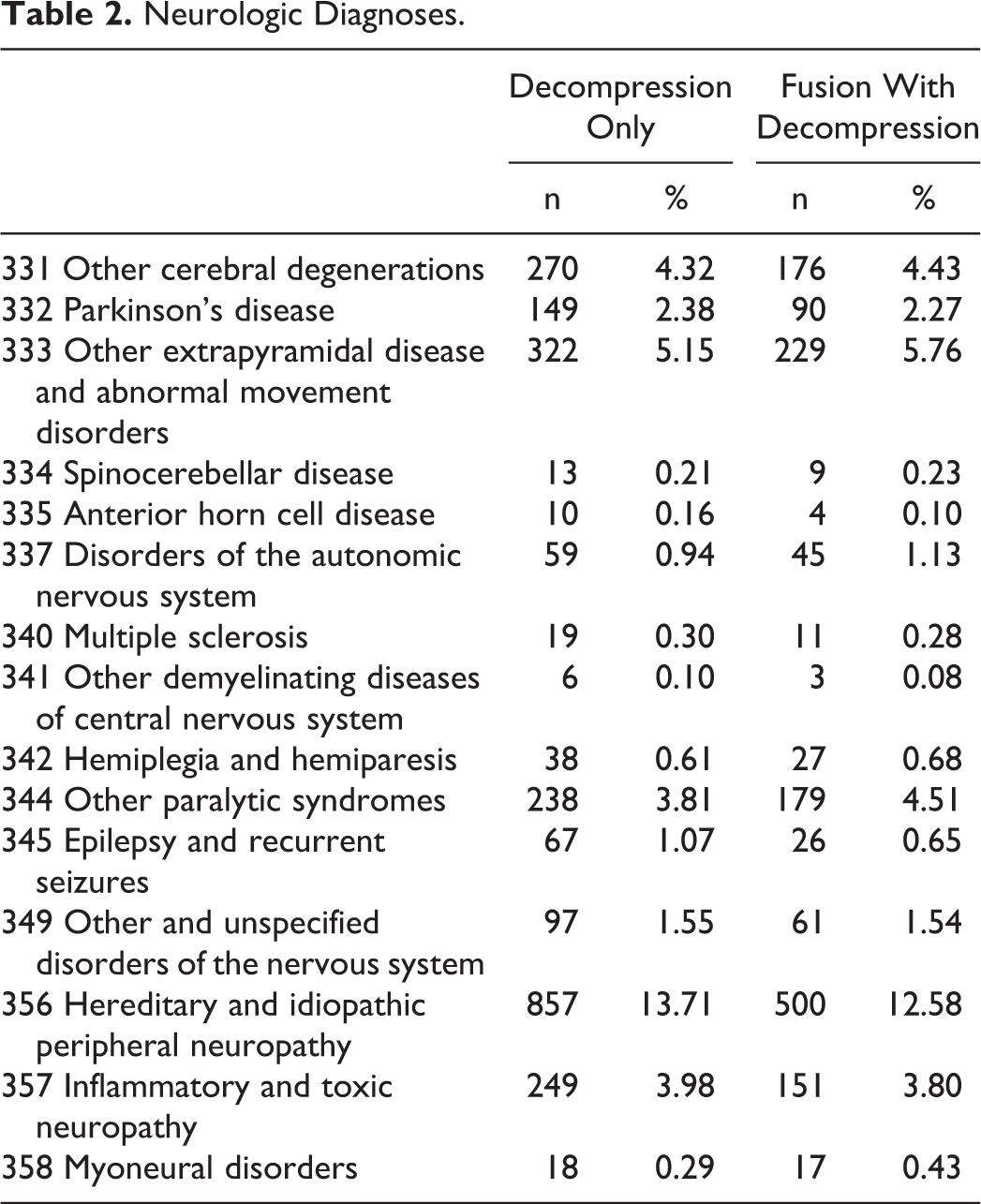
Neurologic Diagnoses.
Results
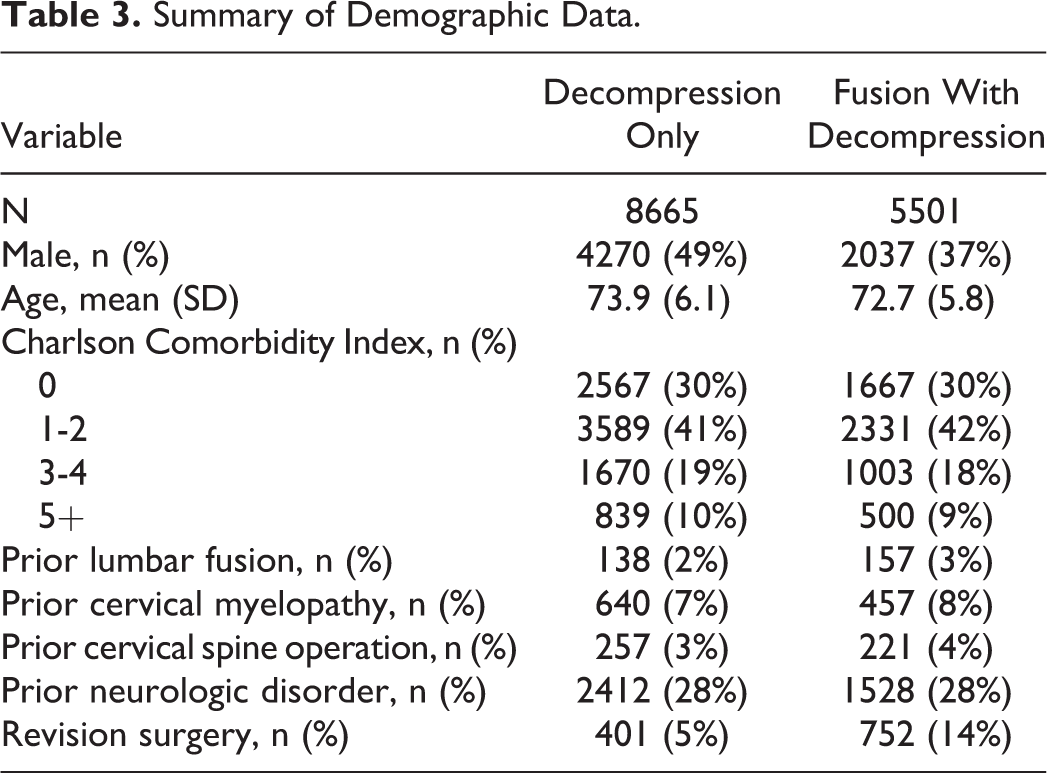
From 5% Medicare Part B (physician/carrier) claims, 14 166 cases that met inclusion were identified. Summary of demographic data for cases that had decompression only, or decompression and fusion, are presented in Table 3. As to be expected in this cohort, patients were relatively old, with a mean age of 74 years in the decompression only cohort and 73 years in the decompression and fusion groups. In general, the patients were fairly healthy with the majority of patients having a Charlson score of 2 or less. Only a small proportion of patients had prior lumbar fusion, cervical myelopathy, or cervical surgery. Almost 30% of patients in both cohorts had a neurologic disease (Table 2).
Summary of Demographic Data.
Of the 8665 cases who had decompression only surgery, 401 (5%) had a revision surgery within 7 years after the index surgery. Factors that were predictive of a higher risk of revision surgery were a prior lumbar fusion (hazard ratio [HR] = 2.78, confidence interval [CI] = 1.43-5.37, P = .002) and being female (HR = 1.61, CI = 1.31-1.97, P < .001). Older age, non-whites, and being in the North East lowered the risk of a revision surgery (Table 4). Of 5501 cases who had a decompression and fusion, 752 (14%) had a revision surgery within 7 years after the index surgery. Factors that were predictive of a higher risk of revision surgery were the presence of a neurologic disorder (HR = 1.24, CI = 1.05-1.46, P = .010), prior lumbar fusion (HR = 3.09, CI = 2.05-4.63, P < .001), and being female (HR = 1.35, CI = 1.15-1.57, P < .001), while older age lowered the risk of a revision surgery (Table 5). No single neurologic disease was associated with increased risk for revision (Table 2).
Factors Predictive of Revision After Lumbar Decompressive Surgery.a
Abbreviation: CI, confidence interval.
aValue in boldface indicates statistical significance.
Factors Predictive of Revision After Lumbar Decompression and Fusion Surgery.a
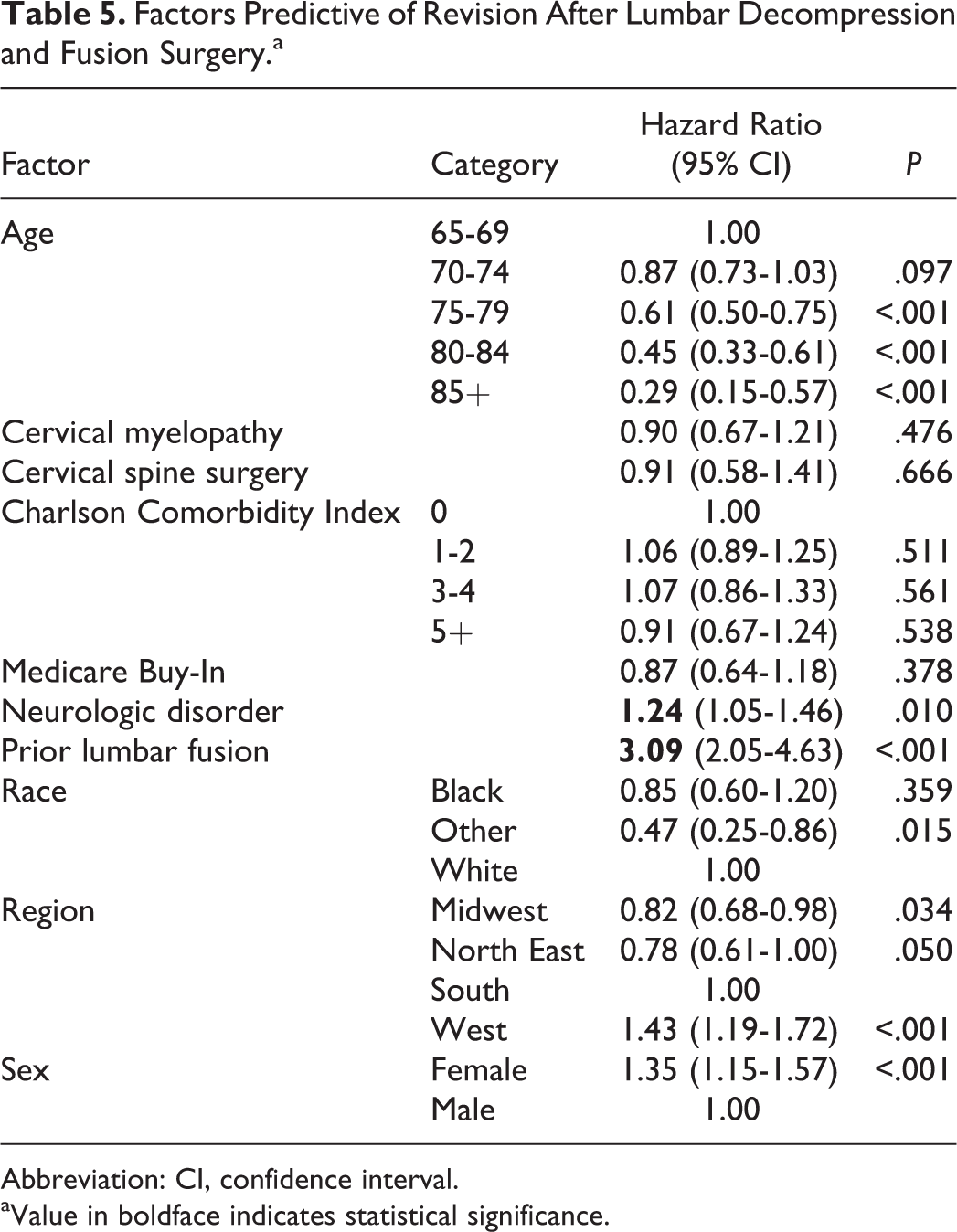
Abbreviation: CI, confidence interval.
aValue in boldface indicates statistical significance.
Discussion
While the advent of large multicenter studies and national datasets has led to a focus on risk stratification, the clinical impact of these efforts has been relatively limited. 17 Choosing a single metric to assess the impact of risk stratification is also difficult. For this study, we selected revision surgery, a readily identifiable serious adverse event that negatively affects both cost and outcome. In practice, avoiding revision surgery depends on either limiting care in high-risk patients or identifying risk factors that can be effectively modified preoperatively or postoperatively. These factors can be described as modifiable risks.
Previously identified modifiable risks for lumbar spine surgery include HgbA1C > 7.0, 9 cigarette smoking, and nonsteroidal anti-inflammatory drug use. 11,12 In this study, we confirmed previously identified associations between several nonmodifiable risk factors such as age, gender, race, and history of prior lumbar fusion (all P < .001) and revision surgery.
In an effort to identify additional contributing risk factors, we examined whether neurologic disorders associated with standing balance dysfunction might be a modifiable risk for revision surgery following either lumbar decompression or lumbar fusion. The rationale is that standing imbalance has been identified as a risk for proximal junctional kyphosis and revision surgery in the spinal deformity population, most notably in patients with Parkinson’s disease, 13,14 and more recently with lesser neurologic impairment. 15,16 Review of the Medicare database revealed an increase in revision rate for females, patients with a prior lumbar fusion, and patients with associated neurologic disorders undergoing lumbar decompression and fusion (P = .01, HR = 1.24). No difference was seen for patients undergoing decompression alone. Increased risk of revision surgery was not correlated with any single neurologic disease, although the number of patients in each of these subsets was small.
While this association has not been previously documented in the degenerative population, observation of this trend in the Medicare database suggests that hypothetical mechanisms and potential remedies should be considered. In theory, standing imbalance or gait instability might predispose to accelerated adjacent-level degeneration secondary to increased micromotion at the facet joint level. Alternatively, patients with otherwise asymptomatic radiographic findings of adjacent-level degeneration might be more likely to seek surgical consultation and undergo revision surgery because of symptomatic muscle fatigue associated with gait instability. This may represent an opportunity to effectively improve clinical outcome and reduce revision rate through improved surgical decision making or with preoperative or postoperative balance training.
As is the case with many large database analyses, the observations in this study are relatively nonspecific. This study identifies opportunities for more focused investigation rather than providing clearly defined clinical guidance. While the Medicare database offers the ability to track revision rate for a large number of patients over a significant period of time, specific patient-level data is limited. In particular, clinical and radiographic details for the index procedure are unknown. Did the patient start with significant adjacent-level pathology? Was preoperative sagittal alignment acceptable? All of these factors might substantially influence risk for revision surgery.
Despite these limitations, this study suggests that neurologic disorders associated with standing or gait imbalance, previously identified as a predictive risk for treatment failure in the spinal deformity population, may also represent an important risk factor for revision surgery following spinal fusion for lumbar degenerative disease. At least in some instances standing or gait imbalance may be altered preoperatively, whether by a complex intervention such as decompression for cervical myelopathy or more simply with physical therapy for balance and gait training. Future study should determine whether these or other interventions might reduce the attendant risk for revision spine surgery.
Lumbar spine surgery has the potential to be cost-effective, but only in well-selected patients. 18 –22 Obtaining this favorable outcome requires that the intervention provide durable clinical benefit and avoid costly complications or frequent revisions. Unfortunately, this paradigm has been difficult to achieve in a reproducible manner. Achieving cost-effective outcomes with lumbar spine surgery in a consistent manner will require optimal patient selection including appropriate risk stratification. Identification of modifiable risk will be an important part of that process. This is likely to comprise risks that we presently understand, and risks that we have yet to fully define.
Footnotes
Authors’ Note
This study was reviewed and approved by the University of Louisville Institutional Review Board.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SDG is an employee of Norton Healthcare; received consulting and royalties from Medtronic. LYC is an employee of Norton Healthcare; Member, Editorial Advisory Board, Spine Deformity, Spine, and Spine Journal; Institutional Review Board Member, University of Louisville; Research Committee Member, Scoliosis Research Society; receives research funds from the Orthopedic Research and Educational Fund, 2013 to present; received funds for travel for Study Planning Meetings from the Center for Spine Surgery and Research of the University of Southern Denmark; University of Louisville. JRD is an employee of Norton Healthcare; receives royalties, consulting and speaker fees from Medtronic; Member of Board of Directors of the Scoliosis Research Society. JLG receives royalties from Acuity; receives consulting fees from Medtronic, DePuy, Alphatec, Stryker, Acuity, K2M, Nuvasuve, PacMed; receives honoraria from Pacira. MD is an employee of Norton Healthcare; receives consulting fees from Medtronic and Nuvasive.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
