Abstract
Study Design:
Retrospective cohort study among Medicare beneficiaries who underwent posterior lumbar interbody fusion (PLIF) surgery.
Objective:
To identify the complication rates associated with the use of bone morphogenetic protein 2 (BMP2) in PLIF. Human BMP2 is commonly used in the “off-label” manner for various types of spine fusion procedures, including PLIF. However, recent studies have reported potential complications associated with the recombinant human BMP2 (rhBMP2) use in the posterior approach.
Methods:
Medicare records within the PearlDiver database were queried for patients undergoing PLIF procedure with and without rhBMP2 between 2005 and 2010. We evaluated complications within 1 year postoperatively. Chi-square was used to compare the complication rates between the 2 groups.
Results:
A total of 8609 patients underwent PLIF procedure with or without rhBMP2. Individual complication rates in the rhBMP2 group ranged from 0.45% to 7.68% compared with 0.65% to 10.99 in the non-rhBMP2 group. Complication rates for cardiac, pulmonary, lumbosacral neuritis, infection, wound, and urinary tract (include acute kidney failure and post-operative complications) were significantly lower in the rhBMP2 group (
Conclusion:
Our data showed that the patients who received rhBMP2 had lower complication rates compared to the non-rhBMP2 group. However, use of rhBMP2 was associated with a higher rate of pseudarthrosis. We did not observe any difference in radiculitis and central nervous system complications between the groups.
Introduction
For many years, the use of iliac crest bone graft (ICBG) was a staple for most of the spine fusion procedures as an effective way to achieve solid fusion. However, studies have shown that ICBG harvest is associated with the increased patient morbidity, blood loss, and length of stay. 1 Since then, various classes of bone substitutes were developed to eliminate these complications.
Bone morphogenetic proteins (BMPs) were discovered by Marshall Urist in 1965, and are involved in tissue morphogenesis, cell differentiation, normal skeletogenesis, cancer and several other processes. 2,3 Large number of studies have explored BMP2 capabilities in promoting osteogenesis and bone formation in vitro and in vivo. In an animal study, Sandhu et al 4 demonstrated that group with rhBMP2 had earlier fusion, better bone formation observed via histology and biomechanical properties when compared with the autograft group. In 2002, rhBMP2 saw an increase in the popularity after Burkus et al 5 reported similar fusion rates between the ICBG and rhBMP2 in the anterior lumbar fusion. Initially, rhBMP2 was approved for single-level anterior fusion approach, 6 and subsequently extended for revision surgeries including the posterolateral fusion. High fusion rates led to its “off-label” use with 30% of the surgeries being posterior lumbar interbody fusion (PLIF) and transforaminal lumbar interbody fusions (TLIF). 7,8 Although the use of rhBMP2 led to high fusion rates, 9 there is a large body of literature on potential adverse events associated with its application. 10 –14 Two recent meta-analyses challenged the benefits and adverse events associated with rhBMP2. 15,16 They reported that the previous data on complications was misrepresented, inconclusive, and often biased by industry-sponsored clinical trials. 15,16
The aim of our study was to compare the complication rates in patients receiving rhBMP2 for PLIF with those without rhBMP2, using a large insurance database.
Materials and Methods
Data Extraction
Our study was deemed exempt from institutional review board review as all patient information was deidentified and we did not receive individual patient information. Data was derived from the Orthopaedic subset of Medicare database (January 2005 to December 2010) within the PearlDiver Patient Records Database (http://www.pearldiverinc.com; Pearl-Diver Inc, Fort Wayne, IN). The database was queried using ICD-9 (International Statistical Classification of Diseases, 9th Revision) diagnosis and Current Procedural Terminology (CPT) codes. We used CPT-22630 (arthrodesis performed using a posterior interbody technique that includes laminectomy and discectomy to prepare the interspace at a single level) to isolate patients undergoing PLIF procedure. ICD-9 P-8452 (insertion of bone morphogenetic protein) was then added as an inclusion and exclusion criteria to create 2 cohorts: patients undergoing PLIF with rhBMP2 and PLIF without rhBMP2. The specific complications were isolated within 1 year postoperatively. Complication groups included cardiac, pulmonary, thromboembolism, neuralgia and radiculitis, lumbosacral neuritis, pseudarthrosis, postoperative infection, hemorrhage and hematoma, wound, central nervous system, and urinary tract (include acute kidney failure and postoperative complications, Table 1). Complication rate of each given complication were calculated as following: the number of PLIF patients (with or without rhBMP2) with a specific complication/number of PLIF patients (with or without rhBMP2) × 100 (to create percentage).
ICD-9-D Complication Codes.

Abbreviation: ICD-9, International Classification of Diseases, 9th Revision.
Statistical Analysis
Chi-square was used to compare the complication rates between the rhBMP2 and non-rhBMP2 groups. SPSS statistical software (IBM Corporation, Armonk, NY) was used for the data analysis. Statistical significance was set at
Results
A total of 8609 patients within the Medicare database underwent PLIF procedure between 2005 and 2010. Of this subset of patients, 5497(63.9%) had a PLIF surgery without rhBMP2 and 3112 (36.1%) had it with rhBMP2. Demographics were not part of this analysis; however, all the patients were aged ≥65 years.
Complication rates were significantly higher in the non-rhBMP2 group (0.95%-10.99%) compared with the rhBMP2 group (0.45%-7.68%,
The number of complications significantly higher in non-rhBMP2 group included postoperative infections (2.86% non-rhBMP2 vs 2.09% rhBMP2), wound complication (2.31% non-rhBMP2 vs 1.35% rhBMP2), urinary tract (4.73% non-rhBMP2 vs 3.54% rhBMP2), and lumbosacral neuritis (10.99% non-rhBMP2 vs 7.68% rhBMP2). Similarly, significant differences were observed for cardiac complications in the non-rhBMP2 group (3.75%) when compared with the rhBMP2 group (2.22%,
Complication Rates in PLIF With and Without rhBMP2, With Associated
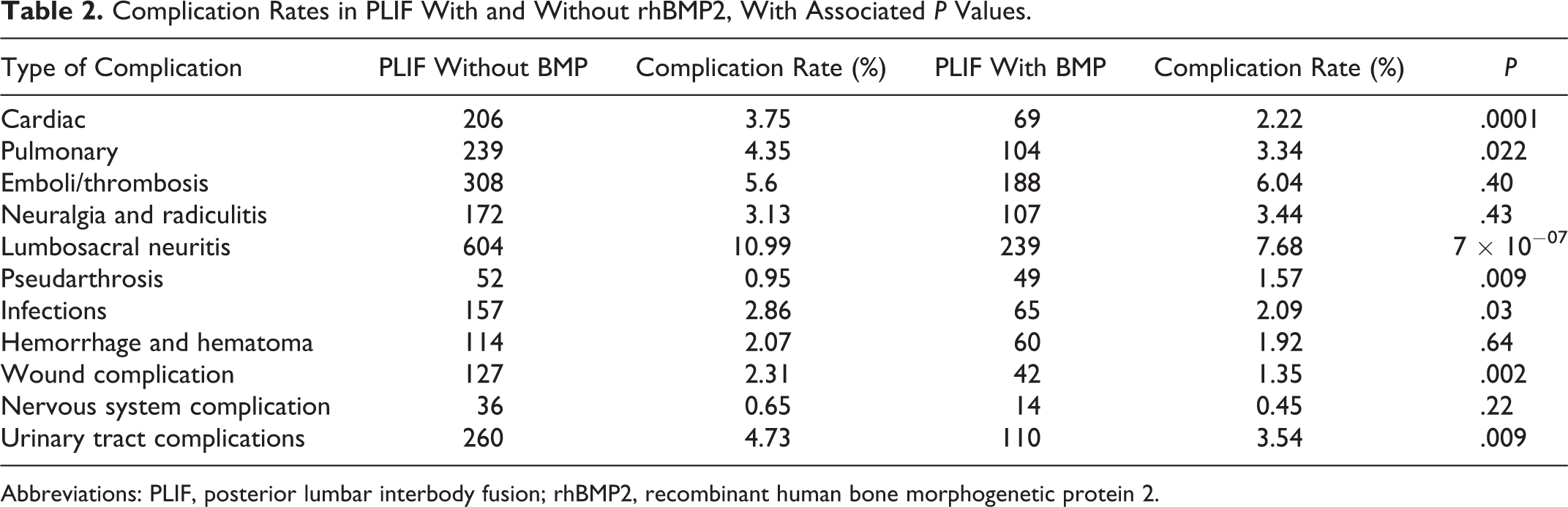
Abbreviations: PLIF, posterior lumbar interbody fusion; rhBMP2, recombinant human bone morphogenetic protein 2.
Discussion
In our study, the use of rhBMP2 in PLIF procedures was not associated with an increased rate of complications compared to PLIF without rhBMP2. In fact, we found the rate of complication to be significantly higher in those individuals who underwent PLIF procedure without rhBMP2. On the other hand, Deyo et al 14 reported no significant difference in the complication rates between patients undergoing spinal fusion for stenosis with and without rhBMP2 and non-rhBMP2 within the Medicare database. A potential explanation for this discrepancy might be due to the follow-up period, in the study done by Deyo et al, 14 complication and reoperation rates were recorded within first 30 days.
The incidence of infection in our study was significantly lower in the rhBMP2 group. Similarly, Singh et al
17
reported a 2.1% infection rate in rhBMP2 group compared with 9.1% in the control group (
In our study, pseudarthrosis rates were higher in rhBMP2 group, which is different from the previous reports that suggest decreases the rate of pseudarthrosis with rhBMP2. 17 An explanation for this may be the nature of the database and if this complication is coded, as well as our population profile, older individuals with lower bone density and osteoinductive activity. Furthermore, rhBMP2 tends to be used in more complex cases, which potentially carry a higher risk of pseudarthrosis. 16,18 Neurological and wound complications were lower in the rhBMP2 group.
Similar rates of hemorrhagic complications in our study are in accordance with previous literature. 19 In our study, no significant difference was found for thromboembolism, neuralgia and radiculitis, and other nervous system complications. Chrastil et al 20 conducted a systematic review on several complications with rhBMP2 use and reported the rates of postoperative radiculitis between 14% and 20% in patients undergoing PLIF/TLIF with rhBMP2 compared with 3% to 5% without rhBMP2.
There were several limitations to our study. This was a retrospective database study searched by ICD-9 and CPT codes and it is subject to failure in recording specific complications. Furthermore, previous medical history, clinical and radiographic outcomes, and rhBMP2 dose were not available limiting us to account for any confounding variables between the groups.
Despite these limitations, we believe that our study provides additional support to the notion that the rate of complications associated with rhBMP2 use in PLIF is lower than in PLIF procedures without rhBMP2. Further studies on biologics focusing on the mechanism of complications, optimal dosing and long-term outcomes are warranted.
Footnotes
Acknowledgments
Our study was supported by the AOSpine. It is a clinical division of the AO Foundation—an independent medically guided nonprofit organization. The AOSpine Knowledge Forums are pathology focused working groups acting on behalf of AOSpine in their domain of scientific expertise. Each forum consists of a steering committee of up to 10 international spine experts who meet on a regular basis to discuss research, assess the best evidence for current practices, and formulate clinical trials to advance spine care worldwide. Study support is provided directly through AOSpine’s Research Department.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ZB—Xenco Medical (consultancy), AO Spine (consultancy, past). HJM—Dr Meisel is consultant (money paid to institution) - Regenerate Life Sciences GmbH for Zyga, DiFusion (ongoing), Co.don (paid to Dr. Meisel past); royalties from: Medtronic, Fehling Aesculap (past); owns stocks (money paid to institution) - Regenerate Life Sciences GmbH in DiFusion. STY—Dr Yoon owns stock in Phygen, Alphatec; Meditech, royalties Meditech Advisors, Stryker Spine (Paid directly to institution/employer), grant from AOSpine (Paid directly to institution/employer), research support from Biomet (Research support given to AREF), non financial research support from Nuvasive and Medtronic. JAY—Royalties: NuVasive, Osprey Medical, Amedica, Integra; Stock Ownership: Benvenue Medical, Paradigm Spine, Promethean Surgical Devices, Spinal Ventures, VertiFlex, Spinicity, ISD, Providence Medical; Private Investments: Amedica, VertiFlex, Benvenue, NuVasive; Consulting: Integra, NuVasive, Amedica, HealthTrust; Board of Directors: Durango Orthopedic Associates (None); Research Support (Staff and/or Materials): Globus Medical (Paid directly to institution/employer), NuVasive (Paid directly to institution/employer), VertiFlex (Paid directly to institution/employer), Integra (Paid directly to institution/employer). DB—Consultant – Vallum, Royalties – America, DePuy Synthes, Medtronic, Fellowship Support – AOSpine (paid directly to institution). JCW—Royalties: Aesculap, Biomet, Amedica, Seaspine, Synthes; Stock Ownership: Fziomed; Private Investments: Promethean Spine, Paradigm spine, Benevenue, NexGen, Vertiflex, electrocore, surgitech, expanding orthopaedics, osprey, bone biologics, curative biosciences, pearldiver; Board of Directors: North American Spine Society (nonfinancial, reimbursement for travel for board meetings, courses, etc.), North American Spine Foundation (nonfinancial), Cervical Spine Research Society (nonfinancial, reimbursement for travel for board meetings), AO Spine/AO Foundation (honorariums for board position); Fellowship Support: AO Foundation (spine fellowship funding paid to institution).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
