Abstract
Objective
Parents of young children with type 1 diabetes (T1D) are at risk of experiencing elevated stress due to their responsibilities as caregivers. Despite this, there are limited interventions designed to enhance resilience in this population of parents. This pilot randomised controlled trial aimed to examine the acceptability, appropriateness, and feasibility of the Promoting Resilience in Stress Management for Parents (PRISM-P) intervention in parents of young children with T1D.
Method
Parents of children (aged 11 years and younger) with T1D participated in this study (N = 30). Participants were randomised (1:1) to a waitlist control or intervention group, with the intervention group participating in three one-on-one sessions via telemedicine with a trained coach. Intervention acceptability and appropriateness were assessed through a survey and semistructured interview. Feasibility was defined by a minimum study enrolment rate of 50% and a minimum retention rate of 70%. Validated instruments to assess psychosocial wellbeing were completed as secondary outcomes.
Results
Results provided strong support for the acceptability and appropriateness of PRISM-P. The enrolment rate for the study was 27% and the retention rate was 87%. The direction of change across psychosocial outcomes also provided preliminary support for the efficacy of the intervention.
Conclusions
PRISM-P was deemed an acceptable and appropriate intervention, as it was met with high satisfaction among the participants who completed it. There was mixed support for the feasibility of PRISM-P as completion was high, but enrolment rates were modest. Future large-scale trials should employ additional recruitment strategies to increase study enrolment rates.
Implications for Impact
Parents and caregivers play an essential role in the medical management and health outcomes of young children living with type 1 diabetes. This pilot trial highlights the potential value of the Promoting Resilience in Stress Management for Parents (PRISM-P) intervention in supporting the psychological wellbeing of parents. With further large-scale evaluation, interventions such as PRISM-P could serve as an accessible adjunct to paediatric diabetes care by providing targeted psychosocial support for parents and caregivers.
Type 1 diabetes (T1D) is one of the most common chronic health conditions affecting children (Australian Institute of Health Welfare, 2020). Management of this condition is highly complex and involves maintaining stable and in-range blood glucose levels, which is fundamental in mitigating the risk of adverse health outcomes. As T1D is commonly diagnosed during childhood, parents (including primary caregivers) play a central role in the management and responsibility for diabetes care and thus bear the primary treatment responsibility. This is particularly relevant to parents of young children who are generally not capable of independently managing their diabetes and may struggle to communicate symptoms of high and low blood glucose (Rankin et al., 2014). Therefore, parents of young children with T1D are faced with unique stressors related to the daily management of their child’s diabetes and the constant vigilance that is required (Kimbell et al., 2021). For example, fluctuations in dietary intake, activity levels, and emotional states can all impact insulin requirements, making diabetes management particularly stressful (Rifshana et al., 2017). Even when parents are away from their children, having to rely on secondary caregivers (e.g. teachers, family, and babysitters) who have minimal experience with T1D can be challenging (Commissariat et al., 2020). Due to these caregiving responsibilities, parents of children with T1D are vulnerable to experiencing elevated stress compared to parents of children without a chronic condition (Thomson et al., 2018).
In the general population, there is strong evidence to suggest that parenting stress is associated with increased internalising and externalising problems in children (Fang et al., 2024). This finding is consistent in parents of children with chronic conditions, whereby parental stress is associated with poorer psychological adjustment in children (Cousino & Hazen, 2013), and negative health outcomes in children with T1D (Helgeson et al., 2012). Given that living with T1D involves inevitable exposure to stressors, parental resilience has the potential to buffer the adverse effects of these diabetes-related stressors.
Resilience is a construct that describes the process of maintaining stable and healthy functioning in the face of adversity (Southwick et al., 2014). Individuals do not respond uniformly to stressful life events, and positive adaptation is often the result of several interacting protective factors (Southwick et al., 2014). Rosenberg and Yi-Frazier (2016) propose that resilience is facilitated through a set of ‘resilience resources’, which may be internal (e.g. dispositional qualities and learned skills), external (e.g. social support and socioeconomic status), and existential (e.g. faith and capacity to make meaning). This perspective suggests that targeting modifiable resilience resources has the potential to improve one’s ability to adaptively respond to stressors. Further, parental resilience has been associated with improved psychological and physical health outcomes in both parents and children with T1D (Luo et al., 2022), making it an important target for intervention. Despite this, interventions aimed at improving psychosocial outcomes for parents of children with T1D have historically focused on improving diabetes management or parenting practices (e.g. conflict management with children; Zhao et al., 2019). While these remain important targets, there are fewer interventions that specifically target resilience resources at the level of the parent.
PRISM-P Intervention Content
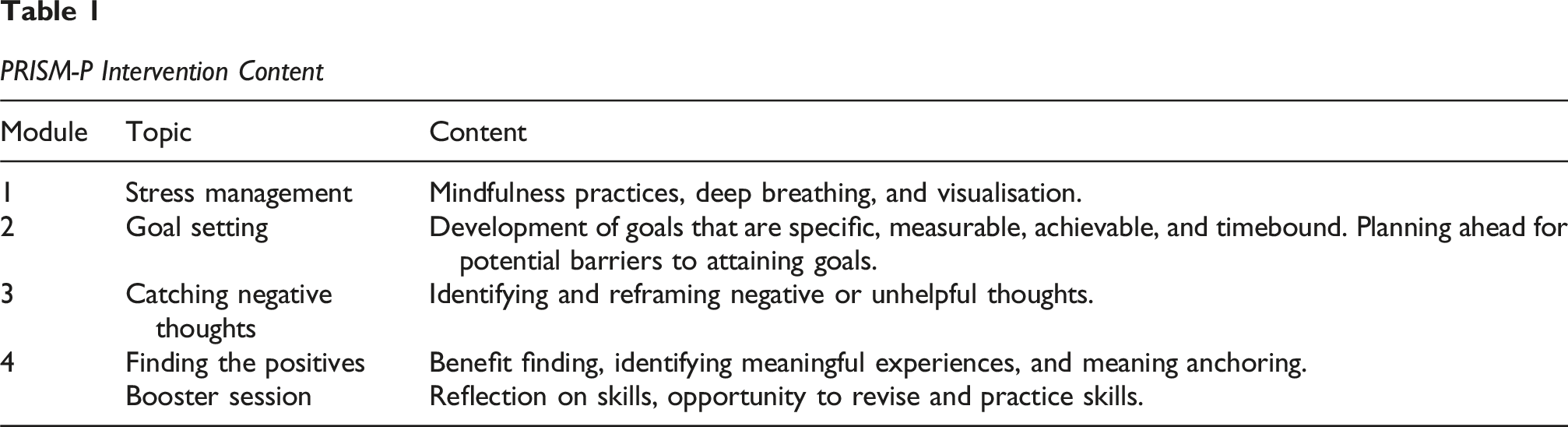
The PRISM-P modules are broadly applicable; however, examples drawn on within the intervention are tailored to a specific parent population. Select skills within PRISM-P are commonly employed in third wave cognitive behavioural therapy, including mindfulness practices (Module 1) and cognitive restructuring (Module 3), while other skills provide more practical guidance through support with goal setting (Module 2). Additionally, gratitude practices within Module 4 encourage parents to reflect on positive experiences that have come from living with T1D (i.e. benefit finding), alongside identifying meaning in challenging experiences associated with the T1D journey. The format and mode of delivery of PRISM-P is designed to be flexible, with previous trials involving both one-on-one and group formats, as well as in-person, telemedicine (i.e. video-calling), and phone delivery (Rosenberg et al., 2019; Yi-Frazier et al., 2017).
A previous feasibility study in the United States established that qualitative satisfaction for the PRISM-P intervention was high, and it was well-accepted by parents of youth with T1D (aged 12–25 years; Yi-Frazier et al., 2017). Moreover, in full-scale trials involving parents of children with cancer (aged 2–24 years), PRISM-P has led to improvements in psychosocial outcomes, specifically resilience and benefit finding (Rosenberg et al., 2019). However, PRISM-P has not been trialled in parents of young children (i.e. aged 11 years and younger) with T1D, who face unique stressors relating to their child’s developmental stage and caregiving responsibilities.
It is also important to evaluate PRISM-P in a telemedicine format. This form of health care delivery is becoming increasingly common in diabetes management (de Kreutzenberg, 2022), and when applied to mental health interventions has demonstrated comparable efficacy in improving symptomatology (Bulkes et al., 2022). Further, remote intervention delivery has been preferred by parents in prior feasibility trials (Yi-Frazier et al., 2017), and is likely to improve intervention accessibility in the present study.
The primary aim of this pilot randomised controlled trial (RCT) was to examine the acceptability, appropriateness, and feasibility of the PRISM-P intervention, delivered via telemedicine, in parents of young children with T1D. A secondary aim was to examine changes in both general and diabetes-specific psychosocial outcomes.
Method
Trial Design
This was an RCT that utilised a parallel design in which participants were randomly allocated (1:1) to an intervention or waitlist control group.
Eligibility
Parents of children aged 11 years and under, who had been living with T1D for longer than 6 months, were eligible to participate in this study. Inclusion criteria were being a primary caregiver (defined as undertaking the majority of diabetes-related tasks) and residing in Western Australia. Exclusion criteria for the study were not being able to speak and understand English (e.g. requiring a family member to translate information) and not having access to a reliable internet connection to participate in intervention sessions.
Recruitment
Potentially eligible participants were identified through the Western Australian Children’s Diabetes Database based on the T1D status of their child, the child’s age, and the child’s duration of T1D. They were sent a personalised email explaining the study and providing them with a link to enrol. If no response was received, and the recruitment target had not been met, parents were followed up by phone at random and provided with a verbal study explanation. Recruitment continued until there were 30 parents had enrolled in the study.
Procedure
Interested participants provided informed consent to participate in the study by digitally signing a consent form. Following this, they completed a baseline questionnaire, involving demographic questions and psychosocial measures, and were randomised to the intervention or control arm. Participants in the intervention group received three PRISM-P sessions conducted approximately 2 weeks apart. At 6 weeks postbaseline, participants completed outcome measures again. The intervention group also completed additional measures related to acceptability and appropriateness and took part in a one-on-one interview via video call. Participants then completed outcome measures for the final time at 12 weeks postbaseline. Participants were sent a text message at 6- and 12-weeks postbaseline with a link to complete the survey and received no more than two follow-up reminders. All consent forms and surveys were completed online via REDCap software (Version 13.10.0). Upon completion of the 12-week survey, the waitlist control group were offered the PRISM-P intervention. Recruitment and data collection took place between April 2022 and December 2022. This trial was undertaken in accordance with the Consolidated Standards of Reporting Trials (CONSORT) guidelines for pilot and feasibility trials (Eldridge et al., 2016).
Intervention
There are four modules within PRISM-P. The first two modules were covered in session 1 and the third and fourth modules were covered in session 2. The third session was a brief booster session. All sessions were delivered one-on-one via video call with one of three PRISM-P coaches, consisting of researcher JI and coaches OK and PB (see acknowledgments). The coaches completed 7 hours of training with the creators of PRISM-P. This training occurred over video call across three separate days and involved learning about the background of PRISM-P, education around specific coach skills (e.g. active listening), practice and feedback from facilitators, and learning from trainers’ experience delivering PRISM-P. Coaches were also oriented to the intervention manual, which outlined the session components and included scripted content. Coaches had access to ad hoc supervision from an endorsed Clinical Psychologist. All coaches had completed an undergraduate degree in psychology, although there was no indication that this psychology-specific knowledge was essential for delivering PRISM-P.
Primary Outcomes
Acceptability and Appropriateness
Acceptability was defined as the extent to which PRISM-P was agreeable or satisfactory for parents (Proctor et al., 2011). It was measured through the Acceptability of Intervention Measure which consisted of 4 items (e.g. ‘The Promoting Resilience in Stress Management intervention is appealing to me’; Weiner et al., 2017). Appropriateness was defined as the extent to which PRISM-P was relevant and compatible with parents’ needs (Proctor et al., 2011). It was measured through the Intervention Appropriateness Measure which also consisted of 4 items (e.g. ‘The Promoting Resilience in Stress Management intervention seems fitting’; Weiner et al., 2017). Responses for both scales were given on a 5-point Likert scale ranging from 1 (completely disagree) to 5 (completely agree). Total scores were obtained through the mean of all items, with higher scores indicating greater intervention acceptability and appropriateness, respectively (range: 1–5). Both scales demonstrated sound psychometric properties including high internal reliability (Cronbach’s α = 0.85–0.91; Weiner et al., 2017). A median score of >3 on both scales, which represents the midpoint of the scale, was considered evidence that the intervention was acceptable and appropriate. There are no established cut-offs for the Acceptability of Intervention Measure and Intervention Appropriateness Measure, however; this midpoint has previously been used to evaluate a psychological intervention (Leyenaar et al., 2023).
Information about the acceptability and appropriateness of PRISM-P was also obtained through semistructured postintervention interviews (conducted by researcher JI). Participants were encouraged to provide honest feedback on their experience of the intervention and were asked questions about the helpfulness of specific modules and the booster session (e.g. Did you find the stress management strategies in session 1 helpful? Why/why not?). Participants were also asked about general improvements (e.g. What aspects of the program could be improved?) and the timing of the intervention (i.e. Would you have preferred it closer or further from when your child was diagnosed? Why?). In addition to questions relating to acceptability and appropriateness, participants were asked about their motivations to participate in the study (i.e. What made you want to take part in this program?) and barriers to engagement (i.e. What made it difficult to take part in this program?). The full interview guide is presented in Table 1 of the Supplementary Material.
Feasibility
Feasibility was defined as the extent to which a full-scale RCT of PRISM-P could be successfully carried out (Proctor et al., 2011) and was assessed through enrolment and retention rates. It was decided a priori that an RCT of the intervention would be considered feasible if ≥ 50% of eligible participants who were approached enrolled in the study and if ≥ 70% of consenting participants completed all PRISM-P intervention sessions. These cut-offs were based on what was considered feasible in the clinical service at Perth Children’s Hospital, which was influenced by the known number of potentially eligible participants.
Secondary Outcomes
Psychosocial outcomes, including resilience, parental diabetes burden, diabetes benefit finding, and negative emotional symptoms, were measured at baseline, 6 weeks postbaseline, and 12 weeks postbaseline. Resilience was measured through the brief version of the Connor-Davidson Resilience Scale (CD-RISC; Campbell-Sills & Stein, 2007). The brief CD-RISC is a 10-item questionnaire designed to measure personal resilience resources. Higher scores on this scale indicate greater resilience (range: 0–40). Diabetes-related distress was measured through the Problem Areas in Diabetes Survey – Parent Revised version (PAID-PR; Markowitz et al., 2012). The PAID-PR is an 18-item questionnaire that measures challenges and worries associated with caring for a child with T1D. Higher scores on this scale indicate greater distress in relation to caring for a child with T1D (score range: 18–90). Diabetes benefit finding was measured through the Diabetes Benefit Finding Scale, Parent Version (DBFS-P; Pierce et al., 2019). This is a 16-item questionnaire that measures the extent to which parents utilise benefit finding (i.e. the process of finding positive meaning during times of adversity) as a means of coping with diabetes-related stressors. Higher scores on this scale indicate greater use of benefit finding coping strategies (score range: 16–80). Negative emotional symptoms were measured through the Depression, Anxiety and Stress Scale (DASS-21; Antony et al., 1998). Higher scores indicate greater symptomatology within each respective scale (score ranges: 0–21). All scales measuring psychosocial outcomes have demonstrated good construct validity through high correlations with related scales and good internal reliability (Cronbach’s α = .87–.97; Antony et al., 1998; Campbell‐Sills & Stein, 2007; Markowitz et al., 2012; Pierce et al., 2019). Changes in HbA1c, an indicator of glycaemic control, between baseline and 12 weeks postbaseline were planned to be a secondary outcome measure. However, this was excluded due to missing data and poor alignment between blood tests and study endpoints.
Sample Size
A sample size of 10–15 participants per treatment arm is recommended for pilot studies when a small-to-medium effect size is expected for the full-scale RCT (assuming 80% power and two-sided 5% significance; Whitehead et al., 2016). Therefore, 30 participants were recruited for the present study.
Randomisation
Participants enrolling in the study were sequentially randomised using a computer algorithm. During recruitment, participants were not immediately enrolled in the study and were sent a link to enrol in their own time, such that researchers were masked to the eventual group allocation. Adaptive covariate randomisation was used to ensure that important prognostic factors (i.e. baseline stress and gender) were balanced across the intervention and control group (Pocock & Simon, 1975).
Analyses
Demographic and clinical characteristics were summarised descriptively. Scores on measures assessing acceptability, appropriateness, and feasibility were compared to cut-offs defined a priori. Exploratory analyses of covariance were used to examine pre-post psychosocial change for each time point. Correlation analyses were also completed for the purpose of informing power calculations in future large-scale trials. All analyses were completed in R (Version 4.2.3).
Interview recordings were transcribed verbatim and analysed using qualitative content analysis (Hsieh & Shannon, 2005), in NVivo software (Version 14.23.2). Analysis of the interviews primarily involved a deductive approach, whereby pragmatic information relevant to future trials of PRISM-P informed the development of categories, including feedback on specific modules, intervention timing, and motivators for participation. Analysis of the transcribed text focused on manifest content by directly interpreting participants’ responses, meaning there was minimal abstraction and interpretation of underlying meanings. Researcher JI completed the category allocation, which involved identifying meaningful units of text, describing the text to develop a condensed meaning unit, and assigning them to a category or subcategory (see Table 2 in Supplementary Material for an example). Category and subcategory allocations were checked by researcher JO, and amendments were made by mutual agreement where necessary.
Ethical Approval
Ethical approval to conduct this study was granted by the Child and Adolescent Health Service Human Research Ethics Committee based at Perth Children’s Hospital (RGS4818). Reciprocal ethical approval was granted by the Human Ethics Office at the University of Western Australia (2021/ET001099).
Results
Participants
Demographic and Clinical Characteristics of Participants and Their Children, N = 30
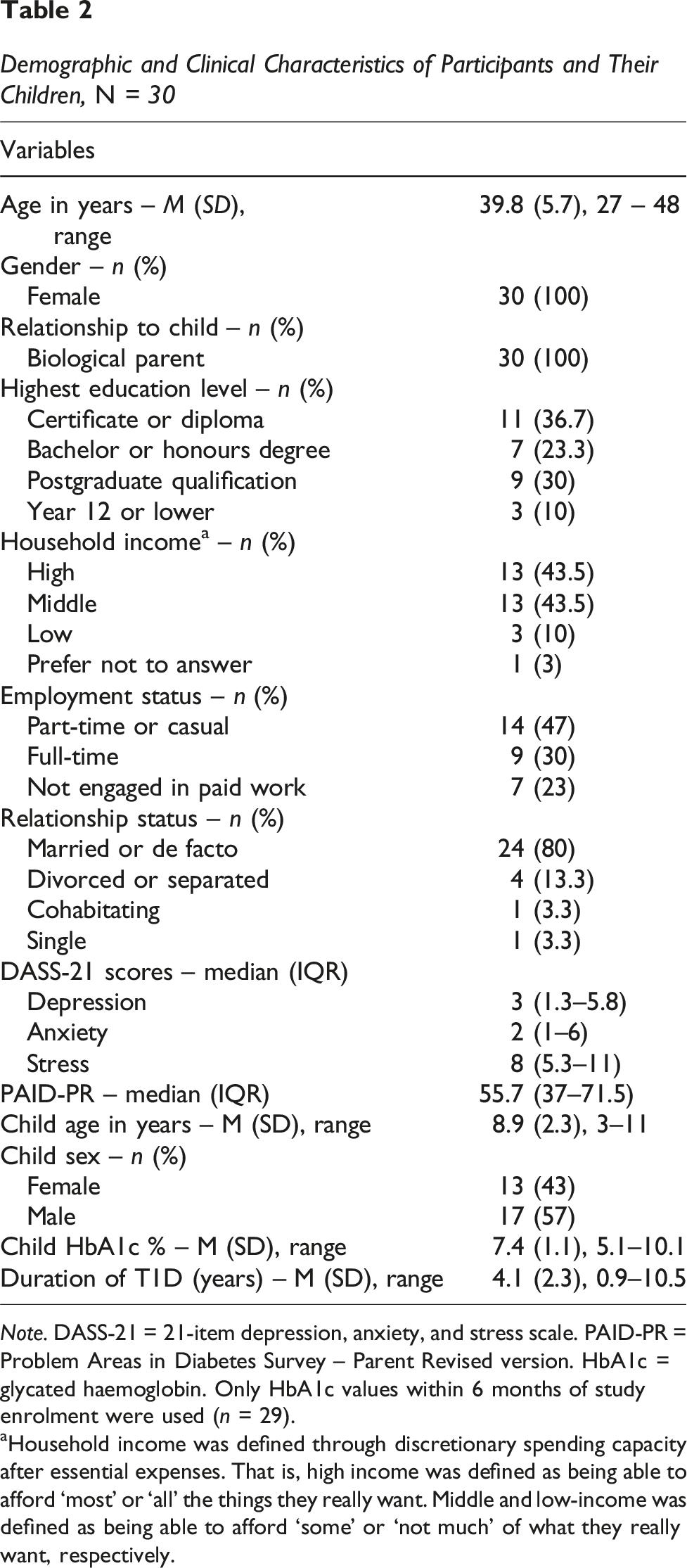
Note. DASS-21 = 21-item depression, anxiety, and stress scale. PAID-PR = Problem Areas in Diabetes Survey – Parent Revised version. HbA1c = glycated haemoglobin. Only HbA1c values within 6 months of study enrolment were used (n = 29).
aHousehold income was defined through discretionary spending capacity after essential expenses. That is, high income was defined as being able to afford ‘most’ or ‘all’ the things they really want. Middle and low-income was defined as being able to afford ‘some’ or ‘not much’ of what they really want, respectively.
Intervention Fidelity
Fidelity checks were completed by researcher KB who did not deliver any intervention sessions. Two participants for each coach were selected at random and fidelity checks were completed on all three PRISM-P sessions. The fidelity criteria involved assessments of rapport being established, as well the appropriate content being delivered and practiced in session. Fidelity for all three coaches was 100% based on the participants reviewed. Additionally, there were no adverse incidents reported that required follow-up.
Primary Outcomes
Study Enrolment Rate
A total of 368 parents were initially invited to participate in the study via email, and 11 participants enrolled through this contact. Due to the low uptake, parents were followed up by phone (n = 73). During recruitment, it was apparent that many parents did not recall receiving an email and were not aware of the study. Therefore, when determining the study enrolment rate, only eligible participants who had the study verbally explained to them (n = 71) and subsequently enrolled (n = 19) were included. This resulted in an enrolment rate of 27%. Reasons for declining study participation included not having enough time and feeling as though they did not require help with stress management (see Figure 1). CONSORT flow diagram showing study stage and participant numbers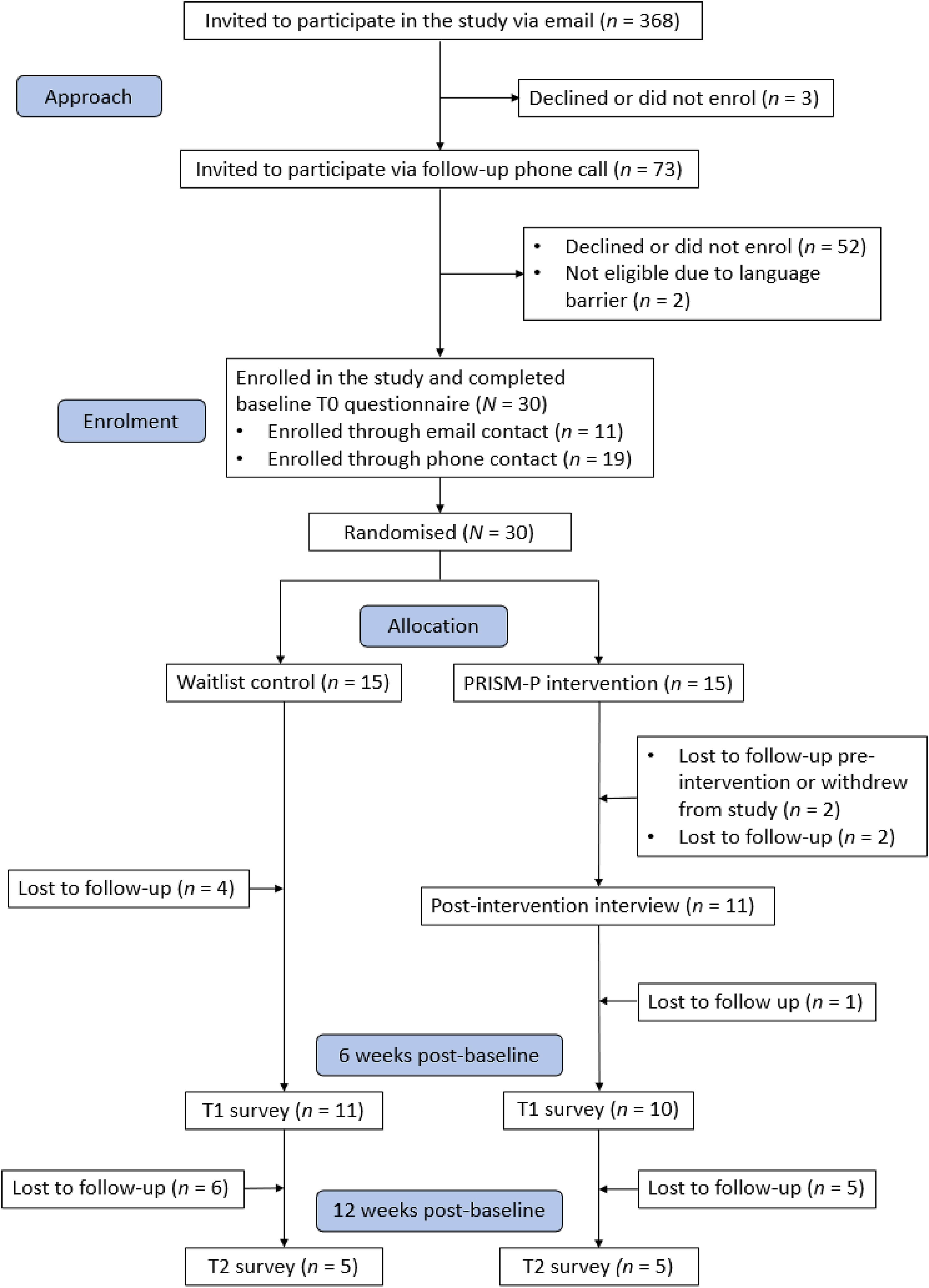
Retention Rate
Of the 15 participants randomised to the intervention group, 13 completed all three sessions, resulting in a retention rate of 87%. One participant was lost to follow-up prior to starting the intervention, and another withdrew after one PRISM-P session, citing time constraints. Figure 1 depicts the flow of participants from the recruitment phase to the final data collection point. Upon completion of the T2 measures, the five remaining participants in the waitlist control group were invited to contact the research team should they want to engage with PRISM-P; however, no expressions of interest were received.
Acceptability and Appropriateness
Questionnaire Measures
Median scores for the Acceptability of Intervention Measure and the Intervention Appropriateness Measure were 4.9 (range: 4–5) and 4.1 (range: 4–5), respectively. These scores were compared to an a priori cut-off of 3.
Postintervention Interviews
Of the 13 participants who completed all three sessions of PRISM-P, 11 were interviewed as soon as possible after the booster session. All interviews were conducted by researcher JI and the average length of time of the interviews was 12 minutes. From the postintervention interviews, four main categories were identified – module experiences, earlier intervention delivery, motivators for participation, and barriers to participation. Within the module experiences category, participants reported either positive or neutral feedback and also provided general feedback applicable to all modules. Within the motivators for participation category, subcategories included wanting to contribute to research and wanting to improve their stress management. Lastly, within the barriers to participation category, subcategories included limited time and changing availability.
Module Experiences
Subcategories are presented for individual modules to assist with interpreting separate areas of the intervention.
Stress Management Module
All participants reported finding one or more strategies in the stress management module in session one to be helpful. Deep breathing was reported to be particularly helpful as it helped them achieve relaxation, was easy to implement, and provided them with a way to physically reset their body and counter stress reactions. Related to deep breathing, one parent noted ‘It’s just easy to implement on a day-to-day basis without having to think too much about it’ (participant 16), and another expressed that ‘I feel like it just helps physically with your, you know, your reaction to stress like it just comes and is that reset’ (participant 12).
Although feedback for the stress management module was notably positive, some improvements were suggested. Two participants commented that the mindfulness and visualisation strategies were potentially uncomfortable for people to complete over a video-call and that these practices may be more suited to an in-person setting or could be completed through a smartphone app in their own time. One participant noted that there was a lot of content to cover in session one and suggested splitting these components into separate sessions.
Goal Setting Module
Eight participants reported finding the goal setting module in session one helpful. Participants reported finding value in making a conscious effort to identify goals that were important to them but would not typically prioritise. Further, participants found it helpful to write their goals down and approach them in a structured way, with one parent noting that ‘…writing them down and formalising it and making almost a list really, of doing those things, rather than it just being an idea. Actually formalising it really helped’ (participant 1). Three participants reported that the goal setting module included familiar strategies that they often used in the workplace and, thus, found them less helpful than other strategies.
Cognitive Restructuring Module
Nine participants reported that the cognitive restructuring module in session two was helpful. Participants noted that it increased their awareness of negative thoughts and was helpful in preventing them ‘spiralling’. Related to the practice of catching negative thoughts, one participant noted that ‘Obviously our brains are unfortunately a bit hard-wired to go to the negative so it’s really about re-training it. But I think, yeah, it really helped when I did catch them’ (participant 18). Conversely, one participant reported difficulty with increasing their awareness of negative thoughts and instead expressed a preference for positive thinking.
Finding the Positives Module
Eight participants reported that the finding the positives module was helpful. For some participants, it aligned with how they generally approach stressful situations, while others noted that it helped them generally achieve a more positive outlook. Related to the gratitude practice, one participant noted that ‘…rather than looking at the dark side of things, it’s just nice to be able to do that and refresh yourself with it and then feel like you’ve just got more of a positive outlook rather than a negative one’ (participant 17). Two participants reported neutral feedback for this module. One participant noted that it did not resonate with them as they do not have difficulty finding the positives, whereas another participant found it difficult to find positives from the experience of caring for a child living with T1D.
Booster Session
Feedback for the booster session was mixed. Five participants found it to be a useful recap, and it also served as a useful reminder to engage in the skills; for example, one parent expressed ‘Yeah, it was good. It reminded me that I had forgotten a lot of the breathing techniques. So I had to have a recap. If you’re not repeating them, you do forget’ (participant 25).
Conversely, four participants reported neutral feedback for the booster sessions and did not feel it was necessary to refresh the skills, in part due to the resources provided throughout the intervention (e.g. the worksheets and the app).
General Module Experiences
Participants often reported some pre-existing familiarity with the strategies introduced across the modules. In these instances, participants mostly reported the strategies to be helpful, as the sessions served as a reminder or gave them a different way to approach the strategies (e.g. a different style of deep breathing). For example, one participant expressed that ‘I’ve sort of learnt different things and forgotten them over the years and I think it’s that getting yourself in a habit of doing things or having some kind of contact with someone who can remind you how to do it’ (participant 1). However, participants occasionally reported neutral feedback, as they felt a strategy was not helpful for them. These responses may reflect a differential preference for novel stress management strategies versus familiar strategies that require some accountability to engage with or would benefit from a different approach.
Earlier Intervention Delivery
All interviewed participants expressed a preference to take part in PRISM-P earlier in the diabetes journey, as that is when they felt stress management strategies were needed most. Diabetes duration in the present study ranged from 0.9 to 10.5 years, and when asked about an optimal timeframe to engage with PRISM-P, suggestions from participants ranged between 3 and 18 months postdiagnosis. ‘I think having it closer, yeah, so we’re 18 months in and I feel like getting to the first anniversary became a bit of a milestone and I felt a bit more confident, but I think more support earlier is helpful’ (participant 12).
However, participants also expressed a preference for allowing families time to adjust to their child’s T1D diagnosis. This was to allow for time to process the grief of being newly diagnosed alongside the significant learning curve associated with T1D management, with one parent expressing a preference for PRISM-P being delivered ‘probably not too early on because you are so bombarded with information and trying, trying to, to work out your own path’ (participant 6).
Motivators for Participation
Nine interviewed participants expressed motivation to participate in the study to contribute to research and improve outcomes for other families. Some participants also indicated that not needing to involve children was a motivating factor for them, as their children have expressed reluctance to participate in studies that involve additional procedures beyond their routine clinical care (e.g. additional blood tests). ‘I want to be able to do things to help with regards to trials and things that might help other families and people with type 1 diabetes. But things that have been offered before have involved my daughter and in some ways I don’t want to burden her any further’ (participant 17).
Additionally, six participants also expressed a desire to improve their stress management as a reason for participating, with one parent noting ‘you’re always learning so I just thought it would be interesting to know and to pick up and things to deal with stress, ‘cause it’s in everyone’s life, so I just thought it would be a great reference point’ (participant 6).
Barriers to Participation
Not all participants could identify personal barriers to participation. Of the barriers that were noted, four participants cited limited time to take part in the three PRISM-P sessions, with one parent noting ‘even if there’s only three sessions over a short period of time, between work, kids, and life, it’s just difficult’ (participant 16). Similarly, two participants also noted that changing availability due to other responsibilities made participation difficult at times (e.g. illness in the family).
Secondary Outcomes
Exploratory Analyses for Changes in Psychosocial Outcomes at Baseline, 6 Weeks, and 12 Weeks
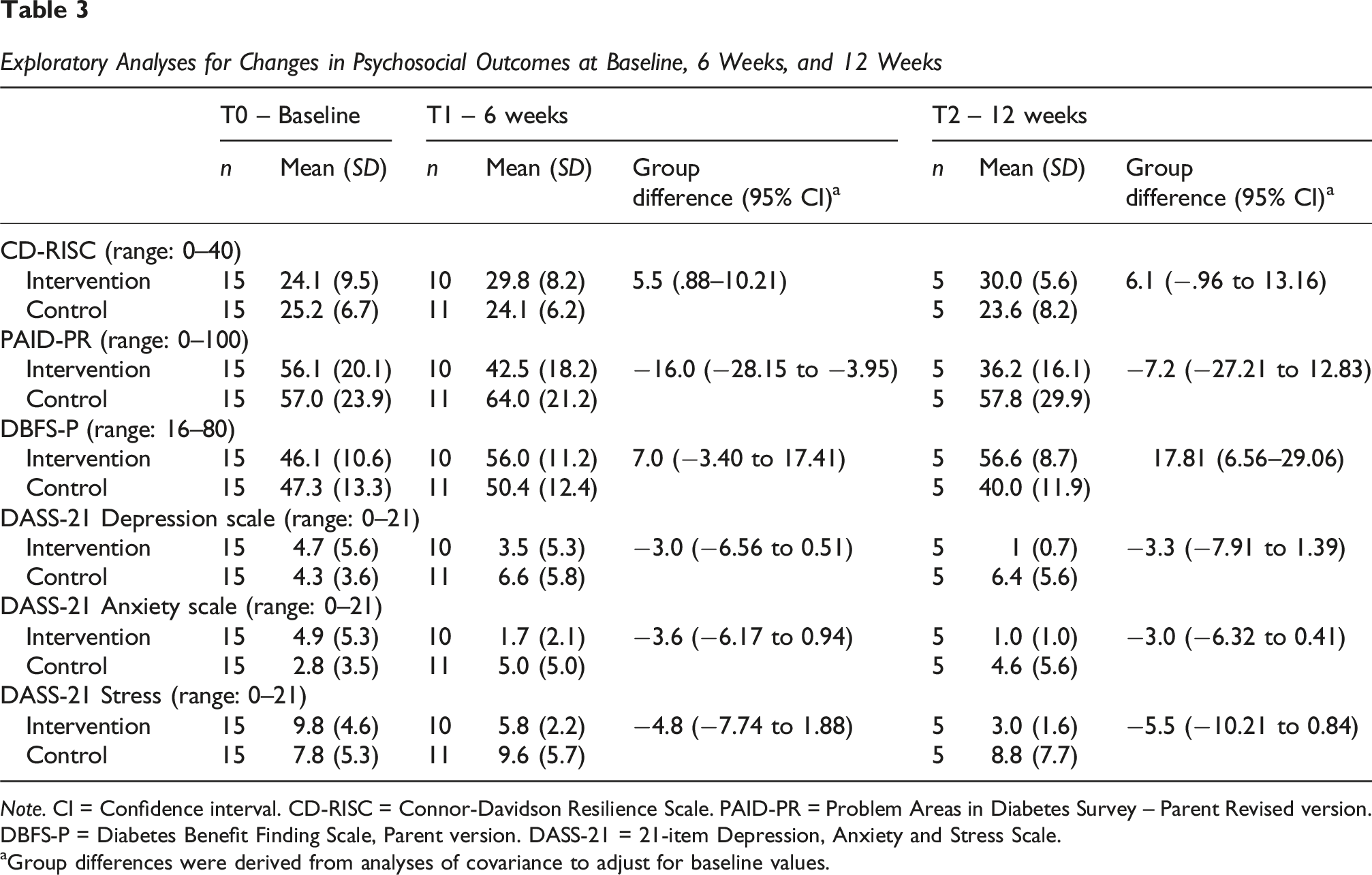
Note. CI = Confidence interval. CD-RISC = Connor-Davidson Resilience Scale. PAID-PR = Problem Areas in Diabetes Survey – Parent Revised version. DBFS-P = Diabetes Benefit Finding Scale, Parent version. DASS-21 = 21-item Depression, Anxiety and Stress Scale.
aGroup differences were derived from analyses of covariance to adjust for baseline values.
Discussion
This pilot RCT aimed to examine the acceptability, appropriateness, and feasibility of the PRISM-P intervention for parents of children with T1D, finding positive results.
Primary Outcomes
Acceptability and Appropriateness
Quantitative and qualitative outcomes supported both the acceptability and appropriateness of PRISM-P. Acceptability, defined as the extent to which PRISM-P was agreeable or satisfactory, was supported as the lower-bound range of the Acceptability of Intervention Measure score exceeded the scale midpoint of 3. Further, there were no negative or adverse experiences reported in postintervention interviews. Regarding appropriateness, defined as the extent to which PRISM-P was relevant and compatible with parents’ needs, results were also favourable. The lower-bound range of the Intervention Appropriateness Measure score exceeded the scale midpoint of 3, indicating that all participants reported PRISM-P to be appropriate. Positive feedback was also provided in the postintervention interviews. Of note, the strategies in PRISM-P allowed parents to achieve a state of relaxation, identify and prioritise important goals, and minimise repetitive negative thinking. These acceptability and appropriateness findings align with prior pilot trials of PRISM-P involving other parent populations (Fladeboe et al., 2023; Yi-Frazier et al., 2017), whereby feedback about the intervention was likewise highly positive.
While parents’ views about PRISM-P were mainly positive, constructive feedback was provided that can guide future amendments to the delivery and content of PRISM-P. In particular, parents unanimously indicated that they would have benefited more from the intervention earlier in the diabetes journey. This feedback suggests that it may be worthwhile to offer PRISM-P to parents after they have completed the necessary education for newly diagnosed families and had time to adjust to the practicalities of managing T1D, for example, 3 months postdiagnosis. This suggestion also aligns with internationally recognised guidelines that recommend psychosocial support for families living with T1D during critical transition points, such as postdiagnosis (de Wit et al., 2022). Additionally, some practices in PRISM-P (i.e. mindfulness) may be more suited to in-person delivery; however, this inevitably raises practical barriers to arranging childcare, particularly for parents of young children, and restricts access for rural and regional parents.
Regarding the content of PRISM-P, areas that could benefit from amendments include the goal setting module and the booster session. For the goal setting module, feedback indicated that some participants did not find value in the practice as a stress management tool. Amendments to this module could focus on facilitating goal setting towards a specific concern that is a source of stress, rather than identifying any goal that interests them. This amendment may improve the relevance of the skill as a stress management tool and orient them towards stressors in their life that can be addressed through actionable steps. Additionally, some parents indicated that they did not find the booster session necessary, as the intervention provided adequate resources to revise the skills. Amendments to this part of the intervention could involve making the sessions optional rather than being embedded in the standard delivery.
Feasibility
Quantitative outcomes provided mixed support for the feasibility of carrying out PRISM-P as a full-scale RCT. The enrolment rate in this study was 27%, indicating that slightly over one-quarter of eligible parents who had the study verbally explained to them consented to participate. It is difficult to know how much this represents actual intervention enrolment, as this study involved randomisation and completion of research-related measures, which might have influenced the decision to enrol. Nonetheless, this enrolment rate was lower than the cut-off of 50% and was also lower than that of prior PRISM-P pilot studies involving different parent populations, which ranged between 50 and 60% (Fladeboe et al., 2023; Yi-Frazier et al., 2017). This enrolment rate difference may be attributed to different recruitment approaches, the absence of participant remuneration in the present study, and the single-arm nature of the trials mentioned earlier; that is, participants were not consenting to randomisation and potentially being on a waitlist.
While the study enrolment rate might not adequately represent intervention enrolment rates, future randomised trials should consider strategies to enhance enrolment. For example, having PRISM-P embedded in the standard model of care offered to families, such that clinicians can increase parents’ awareness of the importance of stress management, could enhance enrolment rates (Caldwell et al., 2010). Including PRISM-P in standard care has also been supported by parents of children with other chronic conditions (Rosenberg et al., 2021). Additionally, making potential participants aware of study remuneration could also increase study interest (Caldwell et al., 2010). Finally, having multi-centre trials, expanding the eligibility criteria, and a longer time frame for recruitment could mitigate the problem of low enrolment.
Conversely, the retention rate in this study was 87%, which well exceeded the expectation of 70%, indicating that nearly all parents allocated to the intervention group completed all three sessions of the intervention. An important factor that likely facilitated this high retention rate was the scheduling flexibility of PRISM-P coaches and the intervention being via telemedicine. These two factors allowed changes in the parents’ availability to be accommodated and limited the commitment required to attend the sessions. The retention rate was higher than that of prior PRISM-P pilot studies, which ranged between 67% and 75% (Fladeboe et al., 2023; Yi-Frazier et al., 2017).
Additional considerations relevant to the feasibility of PRISM-P as a full-scale RCT include survey completion rates at 6- and 12-weeks postbaseline. At 6 weeks postbaseline, survey completion rates were 67% (n = 10) for the intervention group and 73% (n = 11) for the control group. Further, at 12 weeks postbaseline, completion rates within both groups dropped to 33% (n = 5 per group). These lower survey completion rates may be due to limited incentives and the increased time that had elapsed since their initial interest in the study. While the present study prioritised participant convenience by sending direct survey links via text message (including reminders), future studies should incorporate additional strategies to increase survey completion rates. For example, making direct contact with participants through phone calls and remunerating participants for their time completing the surveys (Bower et al., 2014).
Secondary Outcomes
The direction of change across psychosocial outcomes at each time point provides preliminary support for PRISM-P’s efficacy in improving parents’ psychological wellbeing. However, due to low participant numbers and potential dropout biases, these estimates are limited in precision and accuracy. Full-scale trials with higher participant numbers are required to examine intervention efficacy in this population of parents.
Implications
A major clinical implication of this study relates to improving the design and delivery of a full-scale randomised controlled trial of PRISM-P, or similar psychosocial intervention in this parent population. This will ensure that intervention efficacy can be evaluated by avoiding pitfalls common to full-scale RCTs, for example, suboptimal statistical power (de Vries et al., 2023). Additionally, findings from this study suggest that brief stress management interventions, that are delivered remotely and can accommodate scheduling flexibility, are a promising avenue for supporting parents of young children with T1D.
Limitations and Future Directions
A limitation of this study includes the generalisability of the findings due to the sample of participants that were recruited. Of note, all participants were female and 87% identified as middle or high-income. There are several reasons this may have occurred, including high female representation in caregiving roles, which extends to parents of children with T1D (Kobos et al., 2023). Additionally, parents from low-income households may face more barriers to volunteering their time to engage in research compared to parents with more financial resources. Nonetheless, future large-scale trials should seek to increase participant diversity to ensure that findings related to PRISM-P are relevant to the underlying population. Another limitation of this study relates to the absence of interview data for participants who withdrew from the study after partially completing the intervention (n = 1) or were not reachable after completing all three sessions (n = 2). Information about these participants’ experiences may have provided important insights into future study improvements. Additional research may also seek to isolate specific components of PRISM-P that received notably positive feedback from parents, for example, mindfulness practices.
Conclusion
This pilot RCT supports the acceptability and appropriateness of the PRISM-P, delivered via telemedicine, in parents of young children with T1D. Further, the direction of change in psychosocial outcomes provides preliminary support for the efficacy of PRISM-P. While there was mixed support for the feasibility of the intervention due to low enrolment rates, changes in recruitment approaches could overcome this logistical challenge to ensure adequate participant numbers. Overall, PRISM-P appears to be a promising intervention for this parent population and a valuable adjunct to supportive paediatric clinical care.
Supplemental Material
Supplemental Material - The Promoting Resilience in Stress Management for Parents (PRISM-P) Intervention: A Pilot Randomised Controlled Trial in Parents of Young Children With Type 1 Diabetes
Supplemental Material for The Promoting Resilience in Stress Management for Parents (PRISM-P) Intervention: A Pilot Randomised Controlled Trial in Parents of Young Children With Type 1 Diabetes by Jesse A. Ingram, Jeneva L. Ohan, Joyce P. Yi-Frazier, Craig Taplin, Amelia J. Harray, Amy Finlay-Jones, Timothy W. Jones, Elizabeth A. Davis, Keely Bebbington in Clinical Practice in Pediatric Psychology
Footnotes
Acknowledgements
The authors would like to extend their sincerest appreciation to the parents who took part in this research study. The authors would also like to acknowledge Olivia Kingsley and Phoebe Blackley for their time as intervention coaches, Dr. Courtney Junkins for her role in training the coaches, Alison Roberts for her assistance with recruitment emails, Heather Roby for her assistance with project management, Grant Smith for providing statistical advice, and Rigel Paciente for transcribing interviews. This study was conducted at the Rio Tinto Children’s Diabetes Centre, A JDRF Global Centre of Excellence. This Centre is situated within Perth Children’s Hospital and the Kids Research Institute Australia on Whadjuk Noongar country.
Author Contributions
Jesse Ingram: Methodology, Investigation, Project administration, Formal analysis, Writing – original draft, Writing – Review & Editing. Jeneva Ohan: Supervision, Writing – Review & Editing. Joyce Yi-Frazier: Funding acquisition, Conceptualisation, Writing – Review & Editing. Craig Taplin: Funding acquisition, Conceptualisation, Writing – Review & Editing. Amelia Harray: Funding acquisition, Writing – Review & Editing. Amy Finlay-Jones: Funding acquisition, Writing – Review & Editing. Timothy Jones: Funding acquisition, Writing – Review & Editing. Elizabeth Davis: Funding acquisition, Writing – Review & Editing. Keely Bebbington: Funding acquisition, Conceptualisation, Methodology, Supervision, Writing – Review & Editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by an Australian Government Research Training Program and a JDRF Australia Top-up PhD Scholarship (grant number 5-SRA-2021-1088-M-X). This study was funded by a Diabetes Australia Research Program grant (Y21G-TAPC).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Trial Registration
This study was prospectively registered with the Australian New Zealand Clinical Trials Registry (ACTRN12622000096752).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
