Abstract
Objectives
To investigate paraoxonase-1 (PON1) lactonase activity, myeloperoxidase (MPO) activity (as a marker of inflammation) and antioxidant status in plasma of patients with type 1 diabetes mellitus.
Methods
Whole blood and plasma samples were collected from patients with diabetes and healthy control subjects. PON1 lactonase and MPO activities and total antioxidant capacity (TEAC) were determined in plasma. Glycosylated haemoglobin (HbA1c) was quantified in whole blood.
Results
Plasma PON1 lactonase and MPO activities were significantly higher and TEAC was significantly lower in patients with diabetes (
Conclusions
Increased lactonase activity may inefficiently compensate for the high level of chronic inflammation and low antioxidant capacity in the plasma of patients with type 1 diabetes mellitus.
Introduction
The paraoxonase (PON) family comprises three enzymes (PON1, PON2 and PON3) with structural homology and common antioxidant properties. 1 PON1 has been widely studied due to its anti-atherogenic function and role in cardiovascular disease. 1 The enzymatic activities of PONs include paraoxonase, arylesterase and lactonase, which catalyse the hydrolysis of organophosphates, aromatic esters and lactones, respectively. 2 Alterations in circulating PONs (for example, the concentration and activity of PON1) have been reported in a variety of diseases involving oxidative stress, including diabetes mellitus. 3
Chronic inflammation is common in patients with type 1 diabetes, 4 manifesting as elevated inflammatory markers, 5 immune activation 6 and oxidative stress.7,8 These factors are strongly associated with the initiation and progression of atherosclerosis and cardiovascular disease. 9 Plasma myeloperoxidase (MPO) is a valuable marker of chronic inflammation, endothelial dysfunction and cardiovascular disease. 10
Diabetes is associated with high levels of oxidative stress and damage. 11 Antioxidant systems are major mechanisms for protecting cells from oxidative stress-induced damage, and imbalances in the redox mechanism may contribute to the pathogenesis or complications of diabetes mellitus.12,13 There is conflicting information regarding the antioxidant status of patients with diabetes, however.14,15
In spite of a lack of information regarding PON1 lactonase activity in patients with type 1 diabetes, its aryl esterase and paraoxonase activities are known to be decreased.16,17 Factors including increased oxidative stress and protein glycation have been found to be at least partially responsible for these changes. 3
The aim of the present study was to investigate PON1 lactonase activity, plasma MPO (as a marker of inflammation) and antioxidant status of patients with type 1 diabetes.
Patients and methods
Study population
This case–control study recruited sequential patients with type 1 diabetes mellitus (defined according to criteria of the American Diabetes Association 18 ) undergoing treatment at the NC Paulescu National Institute of Diabetes, Nutrition and Metabolic Diseases, Bucharest, Romania, between January 2011 and January 2012. Patients were examined for specific chronic complications of diabetes, including microangiopathy, macroangiopathy and hypertension. Exclusion criteria for all study participants were: acute hyperglycemia crisis; inflammatory or infectious disease; active liver disease; acute vascular disease; current immunosuppressive therapy; and vitamin C or E supplementation. All patients were receiving subcutaneous insulin for glycaemic control, with a target glycosylated haemoglobin (HbA1c) level of <7%. Healthy staff members were recruited as volunteers and included as control subjects. No attempt was made to age- or sex-match the control population.
The study was approved by the ethics committee of the NC Paulescu National Institute of Diabetes, Nutrition and Metabolic Diseases, Bucharest, Romania. Written informed consent was obtained from all study participants.
Plasma preparation
All participants provided peripheral blood samples after an overnight fast. Blood (5 ml) was collected into sterile tubes containing 34 IU lithium heparin and centrifuged at 548 ×
Sample analysis
All assays were performed on a Perkin-Elmer Lambda EZ 210 spectrophotometer (Perkin-Elmer, Boston, MA, USA), and were carried out on duplicate samples.
Total protein was assayed via the Bradford method (Bio-Rad Laboratories, Hercules, CA, USA). Optical density was measured at 595 nm against a bovine serum albumin standard curve.
Plasma PON1 lactonase activity was determined using dihydrocoumarin (DEPCyMC; Sigma-Aldrich Chemie, Steinheim, Germany), as described previously, 19 with absorbance measured at 270 nm. Enzymatic activity was expressed as U/ml.
MPO activity was determined with the
Total antioxidant activity was determined using the 6-hydroxy-2,5,7,8-tetramethylchroman-2 carboxylic acid (Trolox) equivalent antioxidant capacity (TEAC) assay developed by Miller et al, 21 with modifications. 22 Optical density (absorbance) was read at 734 nm against 5 mmol/l phosphate-buffered saline (pH 7.4). The percentage inhibition of absorbance (directly proportional to antioxidant activity) was calculated. The assay was calibrated against a Trolox standard curve. Plasma TEAC was expressed as mmol of Trolox equivalent per litre of plasma.
Whole blood HbA1c was quantified by standardized immunoturbidimetry (Cobas Integra®; Roche Diagnostics, Mannheim, Germany) in accordance with the manufacturer’s instructions. 23
Statistical analyses
Data were presented as adjusted predictive values based on a linear regression model, using age as the independent variable. No data imputations were performed. The data were, therefore, corrected for age as a potential confounder. Between-group differences were determined using parametric (two-tailed Student’s
Results
Clinical and demographic characteristics of patients with type 1 diabetes mellitus and healthy control subjects included in a study to investigate plasma paraoxonase-1 lactonase activity in diabetes.
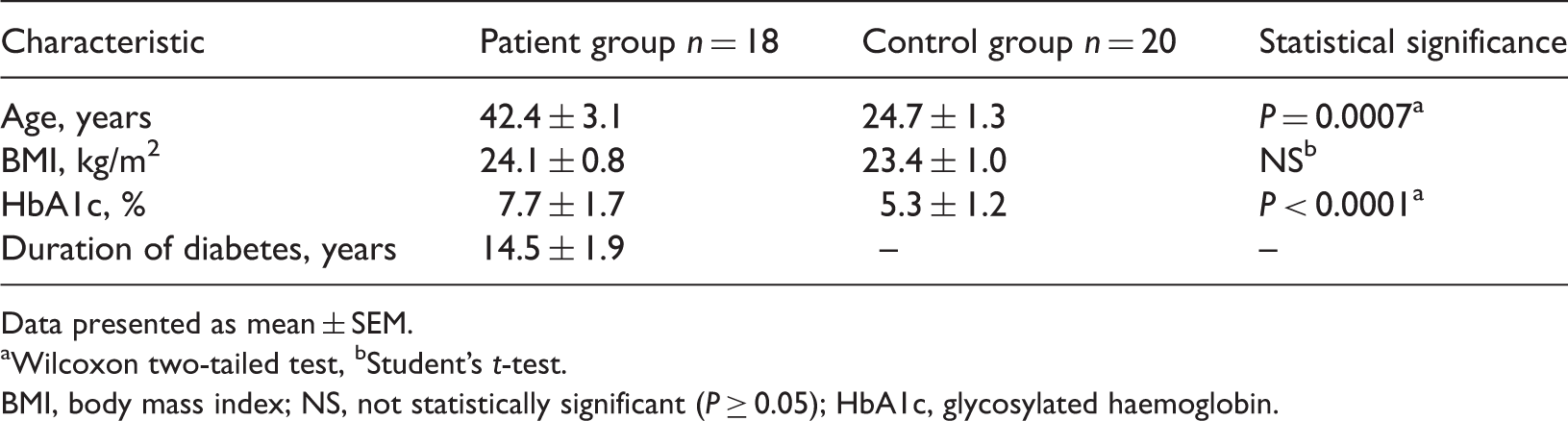
Data presented as mean ± SEM.
Wilcoxon two-tailed test, bStudent’s
BMI, body mass index; NS, not statistically significant (
Paraoxonase-1 (PON1) lactonase and myeloperoxidase (MPO) activities and total antioxidant capacity in plasma from patients with type I diabetes mellitus and healthy control subjects.

Data presented as mean ± SEM.
Wilcoxon two-tailed test.
TEAC, Trolox equivalent antioxidant capacity.
In the patient group, there were significant positive correlations between PON1 lactonase activity and MPO activity (
Discussion
The present study identified increased levels of PON1 lactonase activity and decreased total antioxidant capacity in the plasma of patients with type 1 diabetes mellitus compared with healthy control subjects. The TEAC assay is a validated, standardized method for determining total antioxidant capacity of a biological sample. 24 Our data, therefore, suggest defective antioxidant function in type 1 diabetes despite apparent upregulation of PON1 lactonase. It is possible that this may be due to the known decrease in activity of PON1 when dissociated from high-density lipoprotein (HDL) to the lipoprotein-free serum fraction under diabetic conditions. 25 It has been shown that the higher the concentration of glucose in plasma from patients with diabetes, the higher the rate of PON1 dissociation from HDL. 26
In vitro studies have found that that various oxidative stress systems differentially impair PON1 activities, 27 and that HDL-bound PON1 lactonase is less sensitive than HDL-bound PON1 arylesterase in the same specific oxidative environment. 27 Similar data have been found in patients with type 2 diabetes. 28 PON1 lactonase activity has also been shown to decrease with higher carotid intima–media thickness, suggesting that this enzyme is an independent risk factor for atherosclerosis in patients with diabetes. 28 Substrate availability may also influence PON1 lactonase activity in diabetes. 29 Homocysteine and homocysteine–thiolactone are natural substrates of PON1 lactonase, 30 and high homocysteine concentrations are an independent risk factor for cardiovascular disease. 31 Homocysteine–thiolactone is a naturally occurring metabolite of homocysteine in all human cells, and may be responsible for its deleterious effects. 32 Clinical studies have indicated that PON1 lactonase activity and homocysteine levels vary in diabetes and some metabolic diseases. 33
Plasma MPO activity was also increased in plasma from patients with diabetes compared with control subjects in the present study, and MPO activity was positively correlated with HbA1c. This suggests that diabetes is characterized by chronic inflammation, in accordance with data showing the importance of plasma MPO concentration and chronic inflammation as predictors for atherosclerosis progression in coronary disease.34,35 Studies have found that MPO activity is a source of reactive oxygen species (ROS), 36 amplifying the ROS-induced impairment of endothelium-dependent relaxation via reduction of nitric oxide bioavailability. 37 Furthermore, MPO is a well established inflammatory marker related to endothelial dysfunction and coronary disease, 10 and ROS are considered to be mediators of chronic inflammation in diabetes. 38
PON1 lactonase reduces lipid peroxides and exhibits peroxidase-like activity. 39 The positive correlation between PON1 lactonase activity, MPO activity and HbA1c in our patients with type 1 diabetes therefore suggests a protective role of PON1 in the development of atherosclerosis, in accordance with data indicating a role for PON1 in the prevention of atherosclerosis and cardiovascular disease. 40 It could be speculated that the increased peroxidase-like activity of PON1 lactonase may reflect a compensatory response against ROS overproduction generated by MPO activity.
There are several limitations of our study. The incomplete evaluation of all activities of PON1 results in limited characterization of PON1 function in type 1 diabetes. The quantification of homocysteine or homocysteine–thiolactone would allow better characterization of PON1 lactonase activity or other activities, such as arylesterase. Isolation of serum lipoproteins would permit analysis of serum PON1 distribution between HDL and lipoprotein-deficient serum, and the comparison of PON1 biological functions in these fractions. This study was also limited by its small sample size, although this is more likely to bias the results toward a negative finding (due to insufficient statistical power) than a false-positive result.
In conclusion, our study suggests that increased lactonase activity may inefficiently compensate for the high level of chronic inflammation and low antioxidant capacity in the plasma of patients with type 1 diabetes mellitus. This observation underlies the antioxidant properties described previously in relation to the lactonase activity of PON1.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
