Abstract
Background:
Pill crushing is a common practice in patient care settings. Crushing pills can disperse particulate matter (PM) into indoor air. The PM is a widespread air pollutant composed of microscopic particles and droplets of various sizes and may carry active and/or inactive ingredients nurses can inhale. This study aimed to quantify PM sizes and concentration in indoor air when pills are crushed and examine the role of a fume hood in reducing particulate pollution.
Methods:
Two scenarios (with and without a fume hood) representing nurses’ pill-crushing behaviors were set up in a positive-pressure cleanroom. Two acetaminophen tablets (325 mg/tablet) were crushed into powder and mixed with unsweetened applesauce. The PM sizes and concentrations were measured before and during crushing.
Results:
Different sizes of PM, including inhalable, respirable, and thoracic particles, were emitted during medication crushing. The total count of all particle sizes and mass concentrations of particles were significantly lower during crushing when a fume hood was used (p = .00).
Conclusion:
Pill crushing increases PM and should be considered a workplace safety health hazard for nurses. Healthcare professionals should work under a fume hood when crushing pills and wear proper protective equipment. The findings of significant particulate pollution related to pill crushing suggest that further research is warranted.
Background
Many nurses, especially those caring for pediatric or gerontological patients, are familiar with pill crushing and splitting. Although hospital pharmacies have the necessary engineering control system to control particulate pollution (Mulder, 2019), nurses crush pills where these engineering controls are lacking. Pill crushing leaves nurses at risk for chronic exposure to potentially harmful pharmaceutical particles, particularly active and inactive ingredients of pills.
As far back as 2004, the National Institute of Occupational Safety and Health (NIOSH) released an alert regarding handling hazardous drugs (antineoplastic agents) in which particulate exposure was recognized as a significant risk. The crushing of tablets was acknowledged as an exposure mechanism to hazardous drugs (NIOSH, 2004). There are no current recommendations concerning necessary personal protective equipment for drugs not listed as hazardous by NIOSH. There is no federal practice standard from the Occupational Safety and Health Administration (OSHA) about protecting nurses and others from residual medication that contaminates the air and the surrounding environment due to pill crushing.
The scientific name for airborne material or dust is particulate matter (PM). The PM is a widespread air pollutant consisting of solid and liquid particles, including inorganic ions, heavy metals, and biological debris. These microscopic particles are present in various sizes, mass concentrations (also called mass densities), and mixture contents in the air (EPA, 2022; Mills et al., 1996). All particles <10 μm are inhalable in humans (Brown, 2015). PM is classified into different groups based on size, and particle size is directly associated with health problems. In this investigation, we have used the following classifications: inhalable particles (<10 μm), the mass fraction of total airborne particles, which are inhaled through the nose and mouth; Thoracic (<2.5 μm), the mass fraction of inhaled particles penetrating beyond the larynx; and respirable (<1 μm), the mass fraction of inhaled particles penetrating the unciliated airways (Brown, 2015). Examples of particulate <1 μm are dust, combustion particles mostly from diesel engines, viruses, and bacteria. The known PM <1 μm can be classified as PM 0.1 to 0.3 μm (dust mites and allergens) and 0.3–1 μm (tobacco smoke, metallic fumes, and bacteria such as Staphylococcus). PM2.5, precisely PM <1 μm, can be inhaled and exhaled easily and directly enter the pulmonary and systemic circulations, trapped in the body and form deposits, and impact the human body in different ways (Ain & Qamar, 2021; BreathingSpace, 2023).
Chronic low-level exposures to crushed medications are associated with disease in healthcare workers. For example, a case study report relates the story of a pharmacist who crushed clozapine for several months in a closed room with inconsistent surgical mask usage. She was diagnosed with pneumonitis after complaining of shortness of breath. Her symptoms continued for several months but resolved when exposure to clozapine powder was stopped (Lewis et al., 2012). Multiple studies show the correlation between crushing benzodiazepine medications and nurse development of airborne occupational contact dermatitis (Gilissen et al., 2020; Hulst et al., 2010; Swinnen et al., 2014), but other drugs have also been correlated with contact dermatitis, such as beta-blockers, Angiotensin-converting enzyme (ACE) inhibitors, trazodone, ranitidine, and zolpidem (Gilissen et al., 2020; Swinnen et al., 2014). The allergic reactions might result from exposure to inactive ingredients of oral medications (Abrantes et al., 2016).
Nurses are exposed to PM of different sizes and concentrations due to various activities, such as cleaning, stocking medical supplies, and crushing pills. Crushing pills is common in hospitals and long-term care settings. Although chronic exposure to PM that carries drug particulates is considered a potential risk for nurses, the literature regarding the crushing medication and PM sizes and mass concentration released during pill crushing is scarce. This article aims to quantify PM size and mass concentration distribution dispersed in nurses’ breathing zones (within 10 inches radius of the crusher’s nose and mouth) when pills are crushed. In this study, the mixture content of particles released in the air during pill crushing was not studied.
Methods
An experimental study was conducted for crushing pills by simulating a medication room. To avoid the impact of other activities that may increase PM levels in a room, such as people walking around or packing or unpacking supplies, a positive-pressure cleanroom was used to replicate the experience of pill-crushing and quantify airborne PM in a controlled environment. The most common pill-crushing device, the screw type, was used. Since different pressures on pill crushers produce different sizes of particulates, a digital, adjustable torque wrench was applied for crushing pills to keep the amount of pressure on the pill crusher constant. For this purpose, a bolt was attached to the top of a generic screw-type pill crusher that fit a digital torque wrench (Figure 1). The wrench was set to 37 inches per pound for consistency among all trials. Only one person crushed tablets for all scenarios. We developed two pill-crushing scenarios, one with and one without a fume hood. Both scenarios were repeated 20 times. Two acetaminophen tablets (325 mg/tablet) were crushed in each scenario, and the resulting powder was mixed with the food vehicle (unsweetened apple sauce). The brands of acetaminophen and applesauce were kept the same for all trials.

Instruments, Medication, and Food Vehicles Used in the Project.
The PM size and mass concentration were measured every 6 seconds from the start and end of pill crushing using the Portable Aerosol Spectrometer Dust Decoder (PASDD) (Model 11-D, Grimm Inc.) (Figure 1), which is a real-time aerosol air measurement tool and does not require any chemicals, pumps, or radiation sources to operate (GRIMM: Aerosol Technik, n.d.). This unit enables data processing and communication via Bluetooth, USB, and Ethernet. This instrument measures the particle size distribution in 31 different sizes in the air ranging from 0.25 to 35.15 µm and classifies them as inhalable, thoracic, and respirable. It also presents the output as PM sizes in µm; in this manuscript, we reported sizes 1, 2.5, 4, and 10 µm. The reliability of this instrument has been compared with other devices and identified as a device with precise measurement of PM (Cheng, 2008). In addition, the rinsing air feature increases the reliability and long-term stability of the system by protecting the laser and detector in the optical cell (GRIMM: Aerosol Technik, n.d.). Other studies have also used this dust decoder (Laltrello et al., 2022; Sousan et al., 2016).
Before each trial, we measured the PM mass concentration. During each trial, the sampling head of the Grimm instrument was positioned within the breathing zone of the person crushing the pills. The crushing trial started with adding pills into the crusher, crushing, opening the crusher, and transferring the crushed medicine to apple sauce. During the trials, the person who crushed the medications wore a PPE (gown, gloves, and mask). The same type of PPE was used for each trial. In an additional trial and for photography purposes, we created a darkroom in the cleanroom to take photos of the dust released during pill crushing (Figure 2); note that those particles are large, approximately 25 µm in size; smaller particles are invisible with the naked eye. The data from this trial were not counted in the analysis.

Darkroom Photography During Crushing Medication Shows Suspended Particles in the Air With the Naked Eye.
The analysis was conducted with IBM SPSS Professional version 25 (Armonk, NY, USA) software. The fundamental dependent variable collected was the sum of particles counted at each crushing period and their concentrations. The multiple serial observations of different sizes of particle counts per trial and their concentrations facilitated each group’s calculation of Mean Ranks of Distribution. The non-parametric Mann–Whitney U (MWU) test was used to compare particle distributions and mass concentrations between two groups (with or without fume hood) because the particle counts were not normally or uniformly distributed.
Institutional review board (IRB) approval was obtained from the University of Alabama in Huntsville.
Results
Table 1 shows the particle counts (all categories) before and during crushing in two scenarios with or without a fume hood that repeated 20 times. The mean rank (MR) of the distribution of suspended particle counts (all 31 channels) before each trial (no crushing underway) and a fume hood in off mode was slightly lower but not significantly different than when sampled in the same situation with a fume hood turned on (N = 346, MWU= 15,142.500, p = .403). However, the MR of the suspended particle distribution that occurred during active crushing without a fume hood was significantly higher than when a fume hood was used (p = .000). Using the fume hood while crushing medication significantly lowered the mean of the overall 31-channel particle count from 1,747 to 7 (p = .000). In other words, comparing pre-crushing and during-crushing particle counts with the fume hood on and off shows that the particle counts increase during crushing and are much higher without a fume hood on (Figure 3).
Comparing Particle Counts (31 Channel Sizes) Pre-Crushing Versus During Crushing by Fume Hood Applications.

N: Number of observations of particle counts (the sum total of 31 particle size spectrograph channels in all 20 trials).
M (SD): Mean (standard deviation) of total 31 channel particle count.
MWU: Mann–Whitney U test of independent samples of mean ranks of count distributions.
Statistically significant difference at 95% confidence (p < .05).
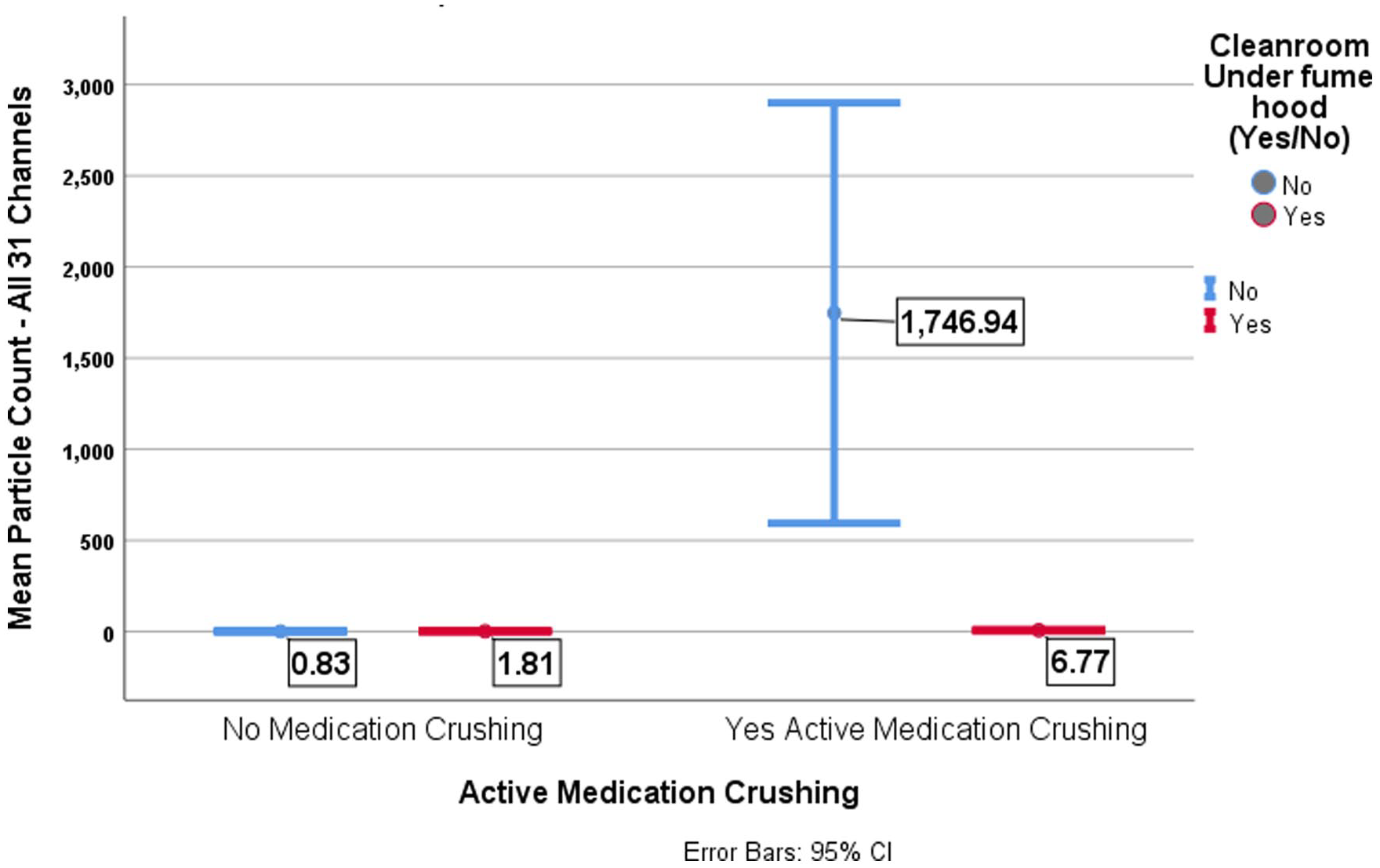
Means of All 31 Channels Particle Count by Active Medication Crushing Compared to Fume Hood Application.
Table 2 shows the mass concentrations of PM by the particle mass categories. The mean counts of particles for all particle mass size concentrations were significantly higher during medication crushing without the fume hood. Using the fume hood had a statistically significant impact on lowering suspended particle concentrations across all ranges of particle mass size concentrations. Similarly, using a fume hood significantly reduced the concentration of suspended particles across all categories (inhalable, respirable, and thoracic combined) (p = .00).
Comparison of Effects of Medication Crushing and Fume Hood Application Upon Means of Particle Counts by Particle Mass Size Concentration Categories (μg per cubic meter).
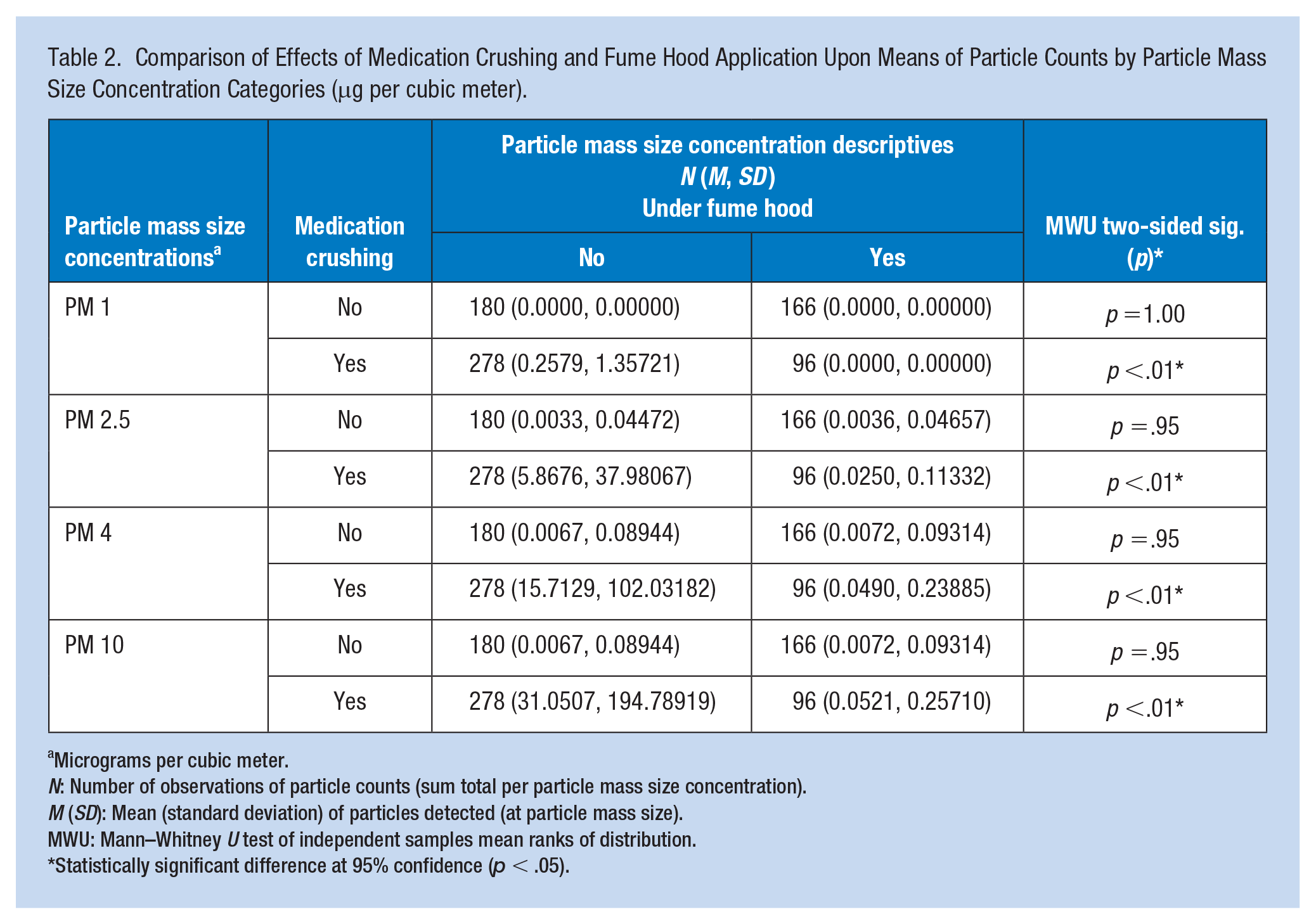
Micrograms per cubic meter.
N: Number of observations of particle counts (sum total per particle mass size concentration).
M (SD): Mean (standard deviation) of particles detected (at particle mass size).
MWU: Mann–Whitney U test of independent samples mean ranks of distribution.
Statistically significant difference at 95% confidence (p < .05).
Discussion
Studies on pill crushing are mainly focused on patients. They discuss whether patients receive the full dose of the medication after crushing or if administering the crushed pill in a food vehicle alters the pharmacokinetics and pharmacodynamics of the medicine (Fodil et al., 2017; Solberg et al., 2021). Patient safety and treatment are essential, but it seldom considers nurses exposure to particles released into the air during pill crushing. This study focused on measuring the size, count, and mass concentration of PM dispersed into the air during pill crushing and examined the benefit of crushing under a fume hood. This study showed that pill crushing contributes to the amount of PM suspended in the air within nurses’ breathing zone. Most of the particles released into the air were inhalable (<10 μm), thoracic (<2.5 μm), and respirable (<1 μm) (Table 2). When crushing was performed under the hood, fewer particles were found in the breathing zone, which means the food hood protects nurses’ exposure to PM released during pill crushing.
Crushing pills with tools such as a pill crusher, the back of a spoon, or a mortar and pestle releases PM into the surrounding air that contains measurable levels of active medication and also inactive ingredients, such as lactose, povidone, and carboxymethylcellulose (Reker et al., 2019). The potential for airborne exposure to pharmaceutical ingredients is more significant in powder form (Huyart et al., 1998). Champmartin and Clerc (2014) studied the crushing of medications in pharmaceutical settings compared with other activities associated with manufacturing medicines. They found that crushing pills creates the highest dust exposure levels. Maeda et al. (2016) simulated the crushing of Lanoxin and Neurovitan in a pharmacy draft chamber with the fan turned off and found that PM concentrations in the air samples were most significant after the pill crusher was opened and the drug powder was transferred from the pill crusher to another container holding the food vehicle (Maeda et al., 2016).
PM can act as a pro-allergic adjuvant, boosting the allergic immune response to inhaled allergens (de Haar et al., 2008; Inoue et al., 2010), and can, directly and indirectly, affect human health. The smaller particles not only penetrate deeper into the respiratory system but also have larger surface areas that enable particles to carry large quantities of hazardous materials, including organic chemicals and metals that can cause adverse health effects, such as exacerbation and promotion of asthma, chronic obstructive pulmonary disease (COPD), atherosclerosis, and cardiovascular diseases (Araujo & Nel, 2009; Li et al., 2008; Zhang et al., 2016).
In this study, we did not examine the health effects of chronic exposures to PM from pill crushing, but studies have shown correlations between crushing benzodiazepine, beta-blockers, ACE inhibitors, trazodone, ranitidine, and zolpidem and the development of airborne occupational contact dermatitis in nurses (Gilissen et al., 2020; Hulst et al., 2010; Swinnen et al., 2014). It is critical to note that allergic reactions may result from exposure to inactive ingredients of oral medications (Abrantes et al., 2016; Kelso, 2014). As discussed earlier, NIOSH has issued a warning about handling and crushing hazardous drugs, such as antineoplastics (NIOSH, 2004). NIOSH does not emphasize exposure to the chemical mixtures of dust dispersing from non-hazardous medicine during crushing or splitting, so it does not require precautions during pill crushing. In this study, we did not measure the chemicals carried by PM and released in the air while crushing but assumed that the active and non-active ingredients of the crushed medicine are released into the atmosphere through pollution particles. A critical mission of NIOSH is to promote worker health and safety. NIOSH’s (2019) strategic goals through the fiscal year 2023 include reducing occupational-related chronic disease and promoting safe workplace design.
Limitations of this study are that the focus was on PM size and not the chemical constituents, and there was no attempt to connect PM exposures, regardless of size, to any potential health outcomes. Further studies regarding both of these issues are warranted.
The evidence relevant to the chemical structure of particles released in the air after crushing medication is unclear. Nurses are asked to study the chemical mixtures of PM released in the air with specific attention to active and inactive ingredients of the commonly crushed medication. Also, analyzing biomarkers of these ingredients in urine or blood samples of nurses and their potential symptoms will help us understand how much of these particles might be transported to nurses’ bloodstreams.
Implications for Occupational Health Nursing Practice
Based on the results of this study crushing medication under a fume hood is the safest practice. A small fume hood in the medication room will significantly mitigate nurses’ exposure to potentially harmful particles. Basic ductless small fume hoods can cost as little as $2,000 and can fit in a medication room. Ductless fume hoods require changing the filter 2 to 3 times a year based on the number of crushing practices and manufacturer recommendations.
Moreover, we recommend nurses use an N95 mask and gloves while crushing medication, especially when not using a fume hood. N95 masks mitigate exposure to small particles, and surgical and cloth masks are partially effective (Rashid et al., 2022; Shakya et al., 2017). We recommend using a gown whenever possible to avoid carrying particles on clothing. For hazardous medication, nurses should use appropriate PPE, including N95 mask, gown, and gloves, and follow NIOSH and USP 800 guidelines on preventing occupational exposures to antineoplastic and other hazardous drugs (NIOSH, 2004, 2016; USP 800, 2017).
Applying Research to Occupational Health Practice
In hospitals and nursing homes, medications are frequently crushed in a small medication room or at the patient’s bedside by nurses with no masks or gloves. This practice leaves nurses at risk for chronic exposure to potentially harmful medication ingredients. This experimental study simulated a medication room in a positive-pressure cleanroom. We played a pill-crushing scenario, with or without a fume hood (20 times each), to measure the counts and mass concentrations of PM in different sizes that dispersed into the nurses’ breathing zone while pill crushing. We found inhalable, thoracic, and respirable PM emitting during pill crushing. The smallest measured particle was 0.253 μm, and the largest was 35.15 μm. The mass concentration of inhalable, thoracic, and respirable PM using a fume hood was significantly less than when a fume hood was not used (p < .001). We recommend adding a small ductless fume hood to the medication room and crushing medication under a fume hood. We also suggest using N95 masks and gloves to mitigate exposure to these particles.
Footnotes
Conflict of Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
