Abstract
Background: Most studies that have evaluated the impact of infection-control measures (ICM) reported a decrease in latent tuberculosis (TB) and not in TB. The objective of this study was to evaluate the impact of ICM on TB incidence among Health Care Workers (HCW’s). Methods: We conducted a retrospective record review study in a general, tertiary care, university-affiliated hospital. All TB case reports among HCWs in the hospital from 2005 to 2018 were reviewed. The TB incidence was measured before and after 2012 to evaluate the impact of ICM implemented. Findings: In total, there were 53 TB cases. The number of TB cases before and after the implementation of ICM was 42 (incidence: 100.0 cases/100,000 HCWs/year) and 11 (incidence: 26.2 cases/100,000 HCWs/year), respectively (p < .0001). Conclusions/Application to Practice: TB incidence among HCWs reduced significantly after the implementation of ICM. The establishment of ICM, such as written TB infection control plan, monitoring, screening, training, and education, can reduce TB incidence.
Background
Tuberculosis (TB) is a major public health problem worldwide, especially in low- and middle-income countries. Brazil is among the 30 high TB burden countries which are responsible for 87% of TB cases globally, with a incidence of 44 cases/100,000 population in 2018 (World Health Organization [WHO], 2019a). In 2015, the WHO introduced the “End TB” Strategy, aiming to reduce TB incidence by 95% by 2030. Infection control in health care settings was included as a key component of this strategy. In 2017, 9,299 TB cases were reported among health care workers (HCWs), and Brazil accounted for 11% of these cases (WHO, 2019b).
Several studies indicate that HCWs have a higher frequency of TB (Baussano et al., 2011; Menzies et al., 2007; Xue He et al., 2010). Some factors, such as increased exposure at work and suboptimal infection-control measures (ICM) in health care settings in countries with high TB burden, increase the HCWs’ occupational risk (Menzies et al., 2007; Xue He et al., 2010). A systematic review showed that the TB incidence in Latin American’s HCWs is 91 cases/100,000 inhabitants, higher than in the general population (82 cases/100,000 inhabitants) (Baussano et al., 2011).
The objectives of this study were to examine the TB incidence among HCWs in a tertiary care hospital in a region with high TB incidence and to evaluate the impact of the implementation of ICM in TB incidence.
Methods
Study Design and Location
We conducted a retrospective record review to evaluate the impact of the implementation of ICM in TB incidence. We evaluated the health records of HCWs who had been diagnosed with TB before and after 2012 (2005–2011 and 2012–2018)—the date when new ICM were implemented. The study was conducted in the city of Porto Alegre, in Southern Brazil, which is the fourth Brazilian capital of a state with the highest number of TB cases, with an incidence of 80.4 cases/100,000 inhabitants in 2019 (Ministério da Saúde, 2019). The location of the study was a general, tertiary care, university-affiliated hospital with 750 beds. This hospital was considered a medium risk for TB transmission as defined by the CDC criteria for size (has more than 200 beds and the number of TB cases in the past year is more than six) (Gerberding et al., 2005). The study was approved by the Ethics Committee at the Hospital de Clínicas de Porto Alegre.
Participants and Data Collection
All HCWs were evaluated at least once a year (and as needed if they had any symptoms) at the Occupational Health Department. Upon hire and annually they are asked about respiratory symptoms, and they are encouraged to come to the Occupational Health Department whenever they experience respiratory symptoms. All consultations were registered in an electronic health record system (EHS) dedicated to the hospital’s HCWs. If they had symptoms suggestive of TB, a sputum smear and a chest X-ray were conducted and additional specific examinations in cases of extrapulmonary TB. TB was diagnosed according to Brazilian guidelines (Conde et al., 2009). All bacteriologically confirmed (smear or culture) cases, pulmonary or extrapulmonary, were considered as TB cases. All TB case reports among HCWs registered in the EHS from 2005 to 2018 were retrospectively reviewed by a trained team; all employee TB cases were also logged in a database. All staff working at the hospital were considered as HCWs: nurses, physicians, laboratory technicians, and administrative personnel. HCWs who had contact with a TB patient were considered to have contracted TB at work (Inchai et al., 2018). Outside exposure was considered when the HCW had an intradomicile contact with TB.
A standardized form was completed for each employee, with the following information: demographic data; work place including department served with the following options: administrative, internal medicine, surgery, emergency, laboratory, radiology, pulmonology, pediatrics, and ICU; and staff category with the following options: nurse technician, administrative, cleaners, nurse, medical doctor, student/resident, duration of work (years of work as an HCW), previous TB (yes or no) or TB contact (yes or no), comorbidities (yes or no, and which one), TB symptoms cough (yes or no), fever (yes or no), weight loss (yes or no), night sweats (yes or no), hemoptysis (yes or no), other symptoms (yes or no, and describe), radiographic findings (noncavitary nonbilateral pulmonary involvement, bilateral pulmonary involvement with cavitary lesions, cavitary lesions, bilateral pulmonary involvement, pleural effusion), methods of diagnosis (sputum, bronchoalveolar lavage, biopsy), clinical form of TB (pulmonary or extrapulmonary TB), and treatment outcome (cure, loss to follow-up, death). Cure was defined by at least two negative smears, one in the maintenance phase and one at the end of treatment. If sputum smear examinations were not possible, a patient who completed the treatment without evidence of treatment failure, and who was discharged based on clinical and radiological criteria, was also considered as cure. Loss to follow-up was considered when the patient was more than 30 days without using the treatment.
Before 2012, there were some ICM including negative pressure TB isolation rooms; HCWs were evaluated only when they had contact with a TB patient, but they were not screened regularly; respirators (e.g., N95 disposable respirators) were available, but HCWs were not trained on how to use it. After 2012, additional ICM were implemented, based on WHO recommendations (WHO, 2009). These included the following: (a) written TB infection-control plan, specific for HCWs, to ensure prompt detection, airborne precautions, and treatment; (b) training and educating HCWs regarding TB, with specific focus on prevention, transmission inside and outside the hospital, and symptoms (4 hours of online training and 1 hour of face to face training, both once a year); and (c) screening and evaluating HCWs who are at risk for TB disease or who might be exposed to Mycobacterium tuberculosis (the screening was upon hire and then once a year, unless they experienced respiratory symptoms or had unprotected contact with a TB patient—then they were evaluated at any time). We also trained HCWs on how to use respirators (e.g., N95 disposable respirators). The hospital’s Occupational Health Department was in charge of implementation and oversight of these ICM.
Data Analysis
Data analysis was performed using IBM SPSS Statistics for Windows, version 22.0 (IBM Corp., Armonk, NY). Data were presented as number of cases, mean ± standard deviation (SD), or median with interquartile range (IQR). Categorical comparisons were performed by chi-square test using Yates’s correction if indicated or by Fisher’s exact test. Continuous variables were compared using the t-test or Wilcoxon test. A two-sided p-value < 0.05 was considered significant for all analyses.
We calculated the overall incidence (2005–2018) and the incidences before (2005–2011) and after (2012–2018) the implementation of ICM. We used the mean number of HCW at the hospital (6,000 HCWs) as the denominator. Thus, to calculate the incidence, in the numerator we enter the number of cases in the period of interest (2005–2018 or 2005–2011 or 2012–2018) and in the denominator the average number of HCWs in the hospital (6,000) multiplied by the number of years considered in the period (7 or 14 years). The result was multiplied by 100,000.
Sample size calculation for the comparison between TB incidence before and after 2012 was based on a previous study (Baussano et al., 2011). We determined that in order to detect a modest effect with 80% of power and a 95% confidence level, we estimated a sample size of at least 10 patients per group.
Results
In total, there were 53 TB cases in HCWs from 2005 to 2018 (Figure 1). There was a decrease in TB cases even before 2012 when the program was implemented most likely because in 2010–2011, the preventive treatment of TB (latent TB infection [LTBI] treatment) was intensified in the community, with more people receiving treatment indication.
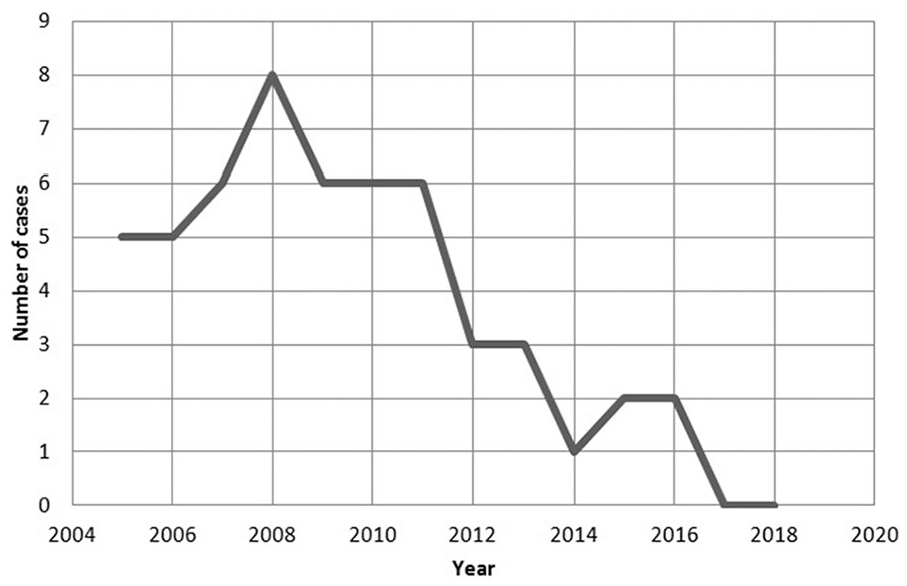
Number of cases of confirmed TB among HCWs between 2005 and 2018 in Hospital de Clínicas de Porto Alegre hospital.
The characteristics of the study population are shown in Table 1. The mean age of the HCWs was 48.2 years. There were 20 males (37.7%) and they had a mean experience of 19.9 years. The most common staff categories were as follows: nurse technician (n = 21; 39.6%), administrative (n = 12; 22.6%), and cleaners (n = 9; 17.0%). The departments most commonly served were as follows: administrative (n = 19; 35.8%) and internal medicine (n = 18; 34.0%).
Characteristics of Health Care Worker Population.
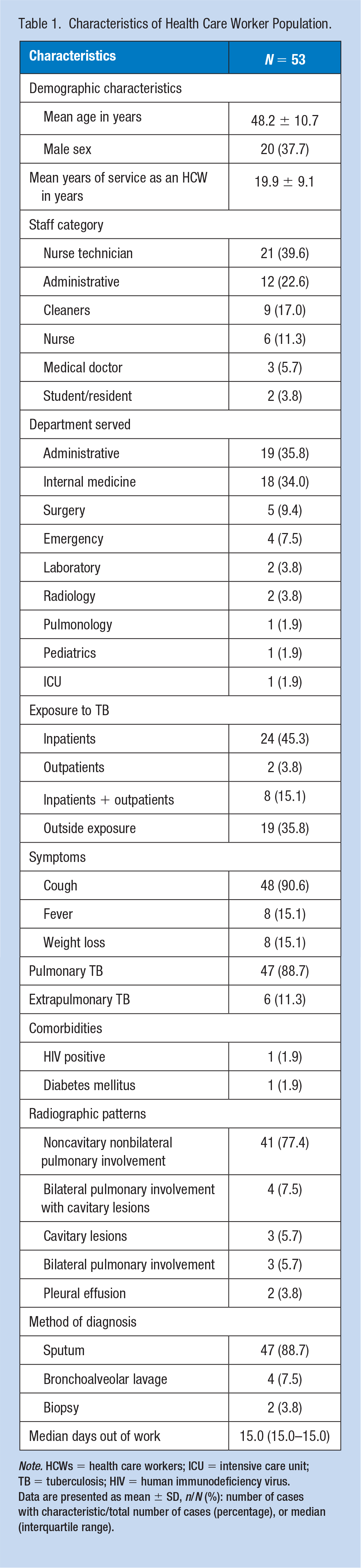
Note. HCWs = health care workers; ICU = intensive care unit; TB = tuberculosis; HIV = human immunodeficiency virus.
Data are presented as mean ± SD, n/N (%): number of cases with characteristic/total number of cases (percentage), or median (interquartile range).
The most common exposures to TB were as follows: only inpatients (n = 24; 45.3%), outside exposure (n = 19; 35.8%), both inpatients and outpatients (n = 8; 15.1%), and only outpatients (n = 2; 3.8%). In total, 47 (of 53; 88.7%) HCWs had pulmonary TB. The median time out of work was 15 days. All HCWs were cured after treatment.
The TB incidence for total study period (2005–2018) was 63.1 cases/100,000 HCWs/year. The TB incidence before (2005–2011) and after (2012–2018) the implementation of ICM was 100.0 and 26.2 cases/100,000 HCWs/year, respectively (p < .0001) (Table 2).
TB Incidences Over Study Periods.

Note. TB = tuberculosis; HCWs = health care workers.
Discussion
In this retrospective study, we found a high TB incidence of 63.1 cases/100,000 HCWs/year from 2005 to 2018 among HCWs in a tertiary care hospital. Nurse technicians and administrative personnel were the most common categories affected. Also, the most common exposures to TB were inpatients only, and outside exposure. In addition, the TB incidence after the implementation of ICM reduced significantly.
Health care facilities are common settings for TB transmission and HCWs have been shown to have a high TB risk higher than that of the general population (Baussano et al., 2011; Joshi et al., 2006; Menzies et al., 2007; WHO, 2010). In the present study, we found a TB incidence of 63.1 cases/100,000 HCW/year. The TB incidence among HCWs is highly variable among studies. In a meta-analysis (Uden et al., 2017) including 21 studies with data on 30,961 HCWs across 16 countries, the pooled estimate for TB incidence was 97 cases/100,000 HCWs/year, ranging from 42 to 4,393 cases/100,000 HCWs/year. In a study conducted in Malaysia, a high TB burden country, the incidence rates ranged from 135.18 to 156.50 cases/100,000 HCWs/year (Liew et al., 2019).
Our study hospital has been evaluated every 4 years since 2013 by the Joint Commission International (JCI) which requires a TB program for accreditation. The implementation of comprehensive ICM requiring administrative, engineering, and personal protective measures is effective in reducing TB transmission (Bock et al., 2007; Dharmadhikari et al., 2012). We found a significant reduction in TB incidence—from 100.0 to 26.2 cases/100,000 HCWs/year—after those interventions were implemented in the study hospital. Most studies that evaluated the impact of those measures in HCWs reported a decrease in LTBI and not in TB disease (Harries et al., n.d.; Wenger et al., 1995; Yanai et al., 2003). Yet, in our view, TB incidence among HCWs as well as among patients could be a proxy for the effectiveness of ICM (Claassens et al., 2013). In a study (Harries et al., 2002) that evaluated the impact of multiple ICM in hospitals in Malawi, the authors found a non-significant decrease in TB incidence after 1 year of implementation. In another study (Yanai et al., 2003), conducted in Thailand, ICM resulted in a significant drop in the incidence of LTBI; however, they also showed a non-significant increase in the incidence of TB disease 1–2 years after initiation of these measures, which could be related to the concomitant increase in HIV-TB patients treated in this hospital. Tiemersma et al. (2016) demonstrated a decrease over time in TB notification rates, associated with an increase over the same period in the availability of personal protective equipment.
Nurse technicians and administrative personnel were the most common categories affected in our study, and the departments most commonly served by the HCWs were administrative and internal medicine. It is well known that the risk of TB among HCWs varies according to their occupations and work settings. Those HCWs working in TB clinics, laboratories, and emergency departments have a higher TB risk (Grobler et al., 2016; Joshi et al., 2006; Sharma et al., 2016). Nurses, laboratory personnel, and physicians are the categories most commonly affected (Joshi et al., 2006). In a study conducted in a tertiary hospital in South Africa, exposure was more frequent in medical wards where known TB patients are present, and also in emergency department (Malotle et al., 2017). In our hospital, TB patients are mostly concentrated in internal medicine department, one of the departments most commonly served by the HCWs with TB in this study. In a retrospective cohort study conducted in Thailand, the occupation categories with the highest TB incidence were nurses, physicians, and paramedics (Inchai et al., 2018). Health professionals and technicians represented 82.5% of the TB cases among Peruvian HCWs (Soto-Cabezas et al., 2016). In 203 hospitals in China, the TB prevalence ratio among medical professionals was significantly higher than other hospital staff (Du et al., 2017). In our study, nurses and physicians were not the most commonly affected; nurse technicians, administrative personnel, and cleaners were the most frequent occupations. This is in contrast with previous studies that demonstrated that TB incidence was lowest among administrative staff (Inchai et al., 2018; Malotle et al., 2017; Menzies et al., 2007). Nevertheless, in high burden countries, many of these TB cases could be attributed to community exposure and not occupational exposure (Inchai et al., 2018; Jones, 2017). In fact, almost 40% of TB cases in our study had history of outside exposure only. In accordance with our findings, a study conducted in Belarus founded that the most common places of work for HCWs with TB were non-medical departments (Klimuk et al., 2014).
Our study has some limitations. First, it was conducted at a single center; however, we think the results may apply to other settings with similar TB incidence. Second, we could not evaluate which of the ICM contributed more to reduce the TB incidence, once they were all established at the same time, but this was not a study objective. Despite these concerns, the results of the present study increase awareness of TB incidence in HCWs. Large, multicenter studies evaluating ICM individually are recommended.
In conclusion, we found high TB incidence among HCWs in a tertiary care hospital. Nurse technicians and administrative personnel were the most common categories affected, and almost 40% had history of outside exposure. Also, after the implementation of ICM, the TB incidence reduced significantly. These findings highlight the need for regular monitoring of TB cases, not only among nurses, physicians, and laboratory personnel working in TB wards and emergency departments but also among administrative personnel/settings.
Conclusion
The study findings highlight the need for regular monitoring of TB cases in HCWs not only among nurses, physicians, and laboratory personnel working in TB wards and emergency departments but also among administrative personnel. As demonstrated, the establishment of a set of ICM, such as written TB infection-control plan, monitoring, screening, training, and education, can contribute significantly to reducing the TB incidence.
Application to Professional Practice
We found a high TB incidence of 63.1 cases/100,000 HCWs/year in a tertiary care hospital during the study period (2005–2018). Nurse technicians (39.6%) and administrative personnel (22.6%) were the most common categories affected. In addition, the TB incidence after the implementation of ICM reduced significantly. The results of the present study increase awareness of TB incidence in HCWs and highlight the need to establish a comprehensive set of ICM, such as written TB infection-control plan, monitoring, screening, training, and education, to reduce TB cases.
Footnotes
Acknowledgements
The authors would like to acknowledge the intellectual support from the International Clinical Operational Health Services Research Training Award (ICOHRTA/Fogarty International Center/National Institutes for Health [NIH]) and Johns Hopkins University (Johns Hopkins Bloomberg School of Public Health).
Author Contributions
A.C.V.A. designed the study, collected data, and helped to draft the manuscript. S.R.H., E.G.C.A., O.A.G.D.C., M.M.L., A.A.F., C.A., F.D.M., and F.F.D.F. collected and analyzed data, and helped to draft the manuscript. D.R.S. designed the study, analyzed data, and wrote the paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author Biographies
Ana Cláudia Vasconcellos Azeredo is an occupational doctor at Hospital Nossa Senhora da Conceição. She earned a MSc degree at Programa de Pós-Graduação em Ciências Pneumológicas, Universidade Federal do Rio Grande do Sul (UFRGS).
Sizuane Rieger Holler is a medical student at Medical School, Universidade Federal do Rio Grande do Sul (UFRGS).
Ellis Gabriela Correa de Almeida is a medical student at Medical School, Universidade Federal do Rio Grande do Sul (UFRGS).
Otávio Augusto Gonçalves Dias Cionek is a medical student at Medical School, Universidade Federal do Rio Grande do Sul (UFRGS).
Marcelle Martinez Loureiro is a medical student at Medical School, Universidade Federal do Rio Grande do Sul (UFRGS).
Alana Ambos Freitas is a medical student at Medical School, Universidade Federal do Rio Grande do Sul (UFRGS).
Camila Anton is a pulmonologist at Hospital de Clínicas de Porto Alegre. She does a MSc at Programa de Pós-Graduação em Ciências Pneumológicas, Universidade Federal do Rio Grande do Sul (UFRGS).
Felipe Dominguez Machado is a pulmonologist at Hospital de Clínicas de Porto Alegre. He does a MSc at Programa de Pós-Graduação em Ciências Pneumológicas, Universidade Federal do Rio Grande do Sul (UFRGS).
Fábio Fernandes Dantas Filho is an occupational doctor at Hospital de Clínicas de Porto Alegre. He earned a MSc degree at Universidade Federal do Rio Grande do Sul (UFRGS).
Denise Rossato Silva is a professor of Medicine at Medical School, Universidade Federal do Rio Grande do Sul (UFRGS) and at Programa de Pós-Graduação em Ciências Pneumológicas, Universidade Federal do Rio Grande do Sul (UFRGS). She is also a Pulmonologist at Hospital de Clínicas de Porto Alegre. She earned a PhD degree at Programa de Pós-Graduação em Ciências Pneumológicas, Universidade Federal do Rio Grande do Sul (UFRGS).
