Abstract
Globally, hepatitis C virus (HCV), the cause of one of the most common infectious diseases, infects approximately 4 million to 5 million Americans with approximately half of infected individuals undiagnosed. Some workplaces screen employees for HCV exposure and other bloodborne pathogens (BBP) after needlestick injuries, but it is not well known whether employers screen employees for HCV without an occupational exposure. New guidelines from the Centers for Disease Control and Prevention (CDC) recommend that all individuals born between 1945 and 1965 should be screened for HCV regardless of risk; this provides an opportunity at the worksite for HCV outreach to employees, dependents, and retirees. To understand this recommendation, the management of HCV in the age of direct-acting antivirals (DAAs) should be reviewed. Now that new DAA treatment can cure HCV, occupational health nurses should identify potential HCV-positive individuals at the worksite via HCV education, screening, testing, and, if positive, linking to specialists for treatment.
Keywords
Globally, the hepatitis C virus (HCV) is one of the most common infectious agents with a worldwide impact; approximately 185 million Americans have tested positive for the hepatitis C antibody (Mohd, Groeger, Flaxman, & Wiersma, 2013). In the United States, approximately 4 million to 5 million individuals are infected with HCV annually; approximately half of these adults are undiagnosed (Yehia, Schranz, Umscheid, & Lo Re, 2014). In a recent Global Burden of Disease Survey, deaths from complications of cirrhosis and hepatocellular carcinoma (HCC) due to chronic HCV are rising despite a decreasing incidence of HCV disease (GBD 2013 Mortality and Causes of Death Collaborators, 2014).
The median age of individuals with chronic HCV is increasing, reflecting the aging baby boomer generation. As this generation, who have been infected with HCV for the past 30 years to 60 years, grows older, the complications arising from cirrhosis due to HCV also increases as HCV complications develop over time. According to some estimates, the number of individuals who will develop end-stage liver disease will quadruple without effective treatment (Younossi, Singer, Mir, Henry, & Hunt, 2014). The use of second-generation direct-acting antivirals (DAAs) has achieved successful outcomes. More than 90% of individuals with chronic hepatitis C (CHC) are cured of HCV infections within 12 weeks (www.hcvguidelines.org) with interferon-free regimens.
Hepatitis C virus is transmitted from individual to individual primarily through infected blood contacting exposed mucosa. Hepatitis C virus is not found in water, feces, urine, saliva, vaginal secretions, or semen, unless blood is present. The prevalence of HCV is highest among individuals who have injected drugs, had blood transfusions before 1992, engaged in high-risk sexual activity, or had HCV-positive mothers (Ghany, Strader, Thomas, & Seeff, 2009). However, individuals born between 1945 and 1965 may have been exposed to HCV without being in one of those risk groups. In addition, health care workers are at increased risk of HCV infection compared with other workers due to the nature of their duties (Askarian et al. 2011).
Occupational health nurses screen employees for HCV and other bloodborne pathogen (BBP) exposures after needlestick injuries; however, it is not well known whether occupational health nurses educate and screen employees, dependents, and retirees for HCV in circumstances other than post-injury. With new guidelines from the Centers for Disease Control and Prevention (CDC) recommending that all individuals born between 1945 and 1965 be screened for HCV regardless of risk, occupational health nurses must be up-to-date on the management of hepatitis C in the age of DAAs. To follow is a brief review of occupational HCV transmission and a clinical update on CHC screening, management, and treatment as well as barriers to screening and treatment, applicable to all workplaces beyond health care settings.
Occupational Transmission of the Hepatitis C Virus
Occupational exposure to HCV and other BBP is a recognized risk for all health care workers. It is reported that approximately 380,000 to 400,000 occupational exposures to human immunodeficiency virus (HIV), hepatitis B virus (HBV), or HCV occur annually in the United States (Naggie, Holland, Sulkowski, & Thomas, 2017; Porta, Handelman, & McGovern, 1999).
Reported rates of transmission from work-related patient care exposures to HCV vary from 0% to 10% (Naggie et al., 2017).
Exposure to HCV results in chronic infection in up to 75% to 85% of those exposed (Ghany et al., 2009). Persistence of HCV ribonucleic acid (RNA) is found in individuals’ serum for 6 months or more after HCV exposure that results in CHC infection (Ghany et al., 2009). For occupational needlestick injuries, the highest transmission of HCV occurs with hollow-bore needles that can transfer more blood and better penetrate muscle tissue (Tomkins et al., 2012). The Massachusetts Department of Public Health (2017) reported similar sharps injuries in the hospital between physicians and nurses; however, sharp injuries due to hollow-bore needles were 3 times higher in nurses than physicians (Massachusetts Health and Human Services, 2017). In a systematic review of needlestick injuries in the United States, the United Kingdom, Germany, France, Italy, and Spain, the number of percutaneous injuries due to needlesticks was 10 times higher in registered nurses than physicians (Motaarefi, Mahmoudi, Mohammad, & Hasanpour-Dehkordi, 2016).
The CDC recently reviewed and updated the 2001 guidance on occupational HCV transmission and provided an algorithm for testing and decision making (CDC, 2017a). No post-exposure treatment for HCV is needed if the source patient is unknown or if HCV antibody/RNA testing is positive because, if infected, the health care worker could spontaneously clear the virus in the subsequent 6 months; if not, treatment can then be initiated with high cure rates as outlined below.
Natural History of the Hepatitis C Virus
Chronic hepatitis C is often asymptomatic until individuals develop signs and symptoms of cirrhosis. Unfortunately, lack of symptoms explains why half of all individuals who have CHC infection are unaware of their status (Yehia et al., 2014). Individuals with CHC infection have an all-cause mortality more than double that of non-HCV infected individuals. Hepatitis C virus-related causes remain the most frequent reason for liver transplantation in the United States (Roche & Samuel, 2012). Furthermore, chronic HCV accounts for 50% of all hepatocellular carcinoma (HCC) cases in the United States (Mehta et al., 2013).
The HCV is a single-stranded, enveloped RNA virus from the Flaviviridae family. Without effective HCV treatment, CHC infection causes liver inflammation that can lead to progressive liver scarring and cirrhosis. On average, cirrhosis develops 20 years to 30 years in immunocompetent adults. Alcohol, HIV, co-infection with other hepatitis viruses, and other insults to the liver will increase the likelihood of developing cirrhosis and hasten its development (Ghany et al., 2009).
Hepatitis C has 6 major genotypes (1-6) with subtypes within each genotype. Genotype 1 is the most common genotype in the United States, accounting for approximately 70% of the infected population. Genotypes 2 and 3 account for 25% of the infected population, and Genotypes 4, 5, and 6 are rare in the United States and account for the remaining 5% of the infected population. Genotypes do not influence the progression of liver disease, but do predict successful HCV treatment and determine which treatments are preferred.
Screening
Screening for CHC uses the serum hepatitis C antibody (HCV Ab) test. If this test is positive, then a confirmatory test, HCV RNA also known as HCV viral load test, is used. If the HCV RNA is positive for more than 6 months, then CHC infection is confirmed. If the HCV RNA test is negative, then individuals have either cleared HCV infection without treatment (approximately 15%-25% of exposures clear within 6 months of exposure) or individuals have already achieved sustained virological response (SVR) with successful HCV treatment and are cured.
In 1998, the CDC released recommendations on HCV screening based on risk factors (e.g., intravenous drug use [IVDU] and blood transfusion history; Table 1). However, risk-based screening was limited as 50% of individuals who had HCV infection did not know of their HCV infection status. In 2012, the CDC released a new recommendation based on birth cohort HCV testing. Current recommendations are to screen all individuals born between 1945 and 1965, the baby boomer generation, with a one-time HCV Ab test regardless of the presence or absence of risk factors. The recommendations target this age group because 75% of all HCV infections in the United States affect members of this generation (www.cdc.gov/hepatitis/hcv/guidelinesc.htm). In following years, the U.S. Preventive Services Task Force (USPSTF) and the American Association for the Study of Liver Diseases (AASLD)/Infectious Diseases Society of America (IDSA; 2017) endorsed birth cohort HCV testing.
CDC Risk-Based HCV Screening Recommendations

Source. CDC (2017b).
Note. CDC = Centers for Disease Control and Prevention; HCV = hepatitis C virus.
Evaluation
After initial diagnosis of chronic HCV with a positive HCV Ab test and a positive HCV RNA test, evaluation of CHC status is determined by baseline laboratory tests, HCV-specific laboratory tests, and additional assessment of relevant co-infections and other liver disease etiologies. General laboratory testing includes a complete blood count (CBC), a basic metabolic panel (BMP), liver function tests (LFT), and a coagulation panel (prothrombin time/international normalized ratio [PT/INR]). Baseline laboratory testing and imaging determine liver inflammation and dysfunction, and establish baseline values with which to monitor liver function during HCV treatment (Sidebars 1-3).
History and Physical Examination
Occupational health nurses should assess employees, dependents, or retirees for the following:
When and how workers were infected
History of or current IVDU, blood transfusions before 1992, tattoos or piercings, sexual transmission, vertical transmission (mother to baby at birth), occupational needlestick injury, born between 1945 and 1965, lived in an endemic country, and/or had surgeries or blood transfusions in endemic country
Co-morbidities that may affect progression of liver disease and HCV treatment Viral co-infections: HIV, HBV Chronic diseases: diabetes, cardiac disease, pulmonary disease, neurologic disease Alcohol use/abuse Working with solvents and/or other hepatotoxic chemicals
Prescription, herbal, and over-the-counter (e.g., acetaminophen) medications
History of HCV treatment failures
Family history of liver disease
Review of systems
Any decompensation signs of liver disease: jaundice, ascites, pedal edema, confusion, or gastrointestinal bleeding Fatigue, abdominal or right upper quadrant (RUQ) pain, malaise, nausea Extrahepatic manifestations of HCV including purpura, fever, renal insufficiency, arthralgias, peripheral neuropathy
Immunizations
Hepatitis A and hepatitis B immunizations if not already immune Pneumococcal vaccination in workers with cirrhosis
Physical Examination
A complete physical examination is indicated to evaluate individuals with HCV. However, more specific signs of advanced liver disease, which may suggest cirrhosis but are not specific to liver disease alone, are listed.
Spider angioma: dilated arterial blood vessels just below the surface. Angiomas will blanch with pressure because the cirrhotic liver cannot excrete excess estrogen.
Palmar erythema is typically seen in the thenar area of the palm with sparing of the central area of the palm, due to excess estrogen.
Jaundice, also called icterus, is yellow discoloration seen best in the sclera of the eye and the skin. Jaundice is typically present when the total bilirubin is greater than 2 mg/dL to 3 mg/dL.
Ascites and caput medusa: the presence of portal hypertension can lead to dependent edema in the flank areas when a worker is supine. Caput medusa refers to the swelling of abdominal veins with increased pressure of distended abdomen from tense ascites. The dilated and engorged veins will radiate from the umbilicus, looking like the hair on the head of Medusa.
Pedal edema is dependent edema due to portal hypertension.
Hepatomegaly/splenomegaly is enlargement of the liver and spleen that can be felt with palpation.
Encephalopathy produces changes in mental status, varying from mild to severe confusion and coma due to increased ammonia levels in the brain.
Gynecomastia is more commonly seen in alcoholic cirrhosis, due to the liver’s inability to breakdown estrogens.
Treatment
The goal of HCV therapy is a sustained virologic response (SVR), meaning no virus (HCV RNA) detectable in the blood 12 weeks after completion of HCV treatment. Once SVR is achieved, the individual is cured of HCV because SVR is durable in 99% of individuals followed 5 years or more (Manns et al., 2013). Achieving SVR yields many health benefits, including improving all-cause mortality (van der Meer et al., 2012). Sustained virologic response has been associated with less liver inflammation and less risk of progression to fibrosis and cirrhosis. Achieving SVR is also associated with a 90% reduction in liver-related death and liver transplantation (Morgan et al., 2013). Perhaps most importantly, SVR has been associated with better quality of life as measured by social, physical, and emotional health indicators (Boscarino et al., 2015).
The AASLD/IDSA (2017) recommend that all individuals with CHC benefit from HCV treatment except those individuals with limited life expectancy (less than 12 months) from non-liver-related conditions. Sustained virologic response success rates are greater than 90% with new DAA medications. The success rates are lower in individuals with histories of previous HCV treatment failures, presence of cirrhosis, and non-structural (NS) 5A mutations in individuals with Genotype 1a HCV.
The new DAAs fall into three main classes of target-specific drugs that disrupt the viral replication cycle of HCV: NS5A inhibitors, NS5B inhibitors (i.e., nucleoside and non-nucleoside), and NS3 protease inhibitors. Treatment of HCV usually combines at least two of the three drug classes with or without ribavirin for a total of 12 weeks to 24 weeks of therapy. Typical treatment for a treatment-naive CHC worker who does not have cirrhosis is 12 weeks, with new treatments demonstrating cure in 8 weeks. Longer treatment (24 weeks) with the addition of ribavirin is reserved for those workers who are more difficult to cure (e.g., workers with cirrhosis and workers who have failed HCV treatment in the past). Treatment options and SVR rates grouped by genotype are described in Table 2. Due to the rapidly changing landscape of HCV therapy, the following website is updated frequently with the newest guidelines: www.hcvguidelines.org/full-report/hcv-testing-and-linkage-care.
Treatment of HCV and SVR Rates
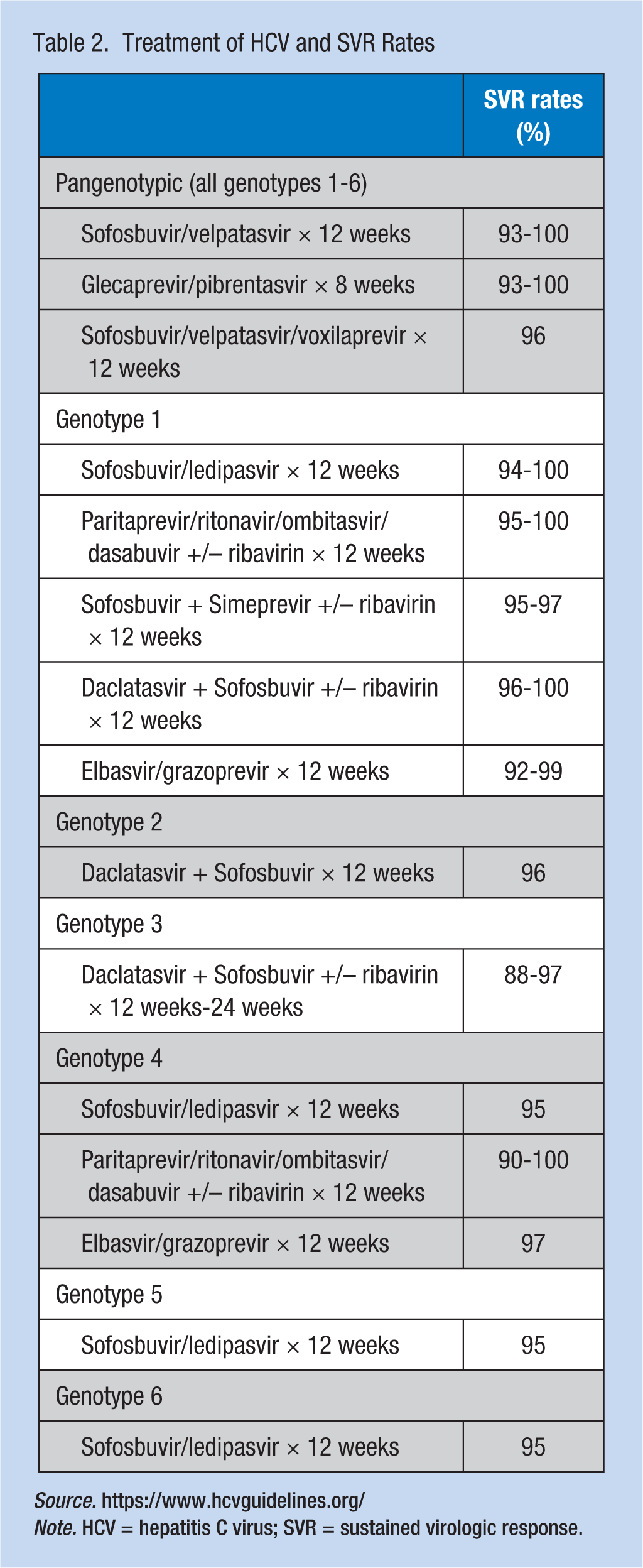
Source. https://www.hcvguidelines.org/
Note. HCV = hepatitis C virus; SVR = sustained virologic response.
Achieving SVR means achieving a virologic cure but does not bestow immunity. If an individual is cured of HCV but exposed to HCV in the future, they are at risk for re-infection. To determine if an individual has been re-infected, an HCV RNA test can detect re-infection. The HCV Ab test will not reliably identify re-infection as the test remains positive even after SVR is achieved because HCV Ab marks exposure to the HCV and will not return to negative, once positive.
If SVR is not achieved (i.e., a positive HCV RNA persists after HCV therapy), then workers have relapsed and will need to be reassessed for additional treatment. Currently, it is not recommended to retreat using the same drug classes that the relapsed individual has already received. Retreatment with the same classes of drugs will not be as effective because resistance to these drug classes is common. Fortunately, with the new DAAs, relapse rates are low and a virologic cure or SVR is achieved in 94% to 99% of cases. In the case of relapse, multiple triple-drug treatment regimen, Vosevi (sofosbuvir/velpatasvir/voxilaprevir), has recently been approved (2017) for rescue therapy.
Treatment also includes ensuring that hepatitis A and B vaccinations are current. Health education includes individuals not sharing razors or toothbrushes while RNA HCV positive, and avoiding alcohol during treatment.
Referral
Any individual with CHC who is a candidate for HCV therapy should be evaluated and referred to an appropriate health care provider for treatment. Prior to the development of DAAs, HCV treatment required pegylated interferon and year-long therapy with very low SVR rates (<40%). The pegylated interferon and ribavirin era was also complicated by a multitude of medication side effects (e.g., flu-like symptoms, neutropenia, anemia, depression, anxiety, alopecia, and anorexia). Individuals with comorbid conditions (i.e., heart disease, lung disease, thyroid disease, and depression) were often refused HCV therapy due to side effects (Ghany et al., 2009). Direct-acting antivirals are much easier for individuals to tolerate due in part to shorter treatment schedules (8-12 weeks), minimal side effects from HCV treatment with DAA (e.g., mild headache and fatigue), and SVR rates above 90% (Liang & Ghany, 2013). Now, during HCV treatment with DAAs, individuals are usually able to work without restrictions.
Historically, almost all individuals with CHC were treated by gastroenterologists and hepatologists during the pegylated interferon and ribavirin era. However, with new DAA therapies, HCV treatment can be managed by primary care providers (PCP). Primary care providers managing and treating HCV infection increase the number of providers available to treat CHC. One of the barriers to successful HCV treatment has been the lack of specialty providers available for referral in certain areas of the United States. According to the Association of American Medical Colleges (2016), about 14,000 gastroenterologists practiced in the United States in 2015 compared with over 200,000 family medicine and internal medicine physicians. Educating PCPs, both physicians and nurse practitioners, to not only screen for HCV, but to effectively evaluate, manage, and treat HCV infection, will greatly increase access to HCV treatment for all individuals with CHC.
An example of medicine and technology working together to amplify the capacity to treat CHC is Project ECHO (Extension for Community Healthcare Outcomes). Project ECHO, initiated by a hepatologist in New Mexico, is a movement to expand treatment for HCV. By pairing local clinicians with specialist teams at academic medical centers via weekly virtual clinics, Project ECHO has expanded HCV treatment capacity. In 2011, a New England Journal of Medicine article documented the success of Project ECHO, stating that the quality of care received through Project ECHO was similar to the CHC care received through academic institutions (Arora et al., 2011).
Beyond access to qualified providers, an additional barrier to HCV treatment is the cost of HCV therapy. The cost savings, after SVR is achieved, of preventing fibrosis and improving quality of life should be factored into the cost of therapy. In various mainstream news reports, the high cost of DAA medication combinations used to treat HCV has been highlighted (Table 3). These figures usually include the wholesale acquisition cost (WAC) of the drug and not the negotiated cost of the drug to insurance companies via pharmacy benefit managers (PBMs) or through state and federal programs (e.g., Medicaid and the Veterans Health Administration). The negotiated costs of the drugs are usually much lower than costs reported in the news (Saag, 2015). Unfortunately, little transparency in the final negotiated cost of the drugs exists because contracts between drug companies and various PBMs are not publicized. Occupational health nurses should know the scope of HCV treatment coverage provided by group health plans for employees, dependents, and retirees. In addition, patient assistance drug medication programs should be accessed to assist with worker out-of-pocket costs.
Estimated Cost of HCV Therapy Based on Wholesale Acquisition Cost
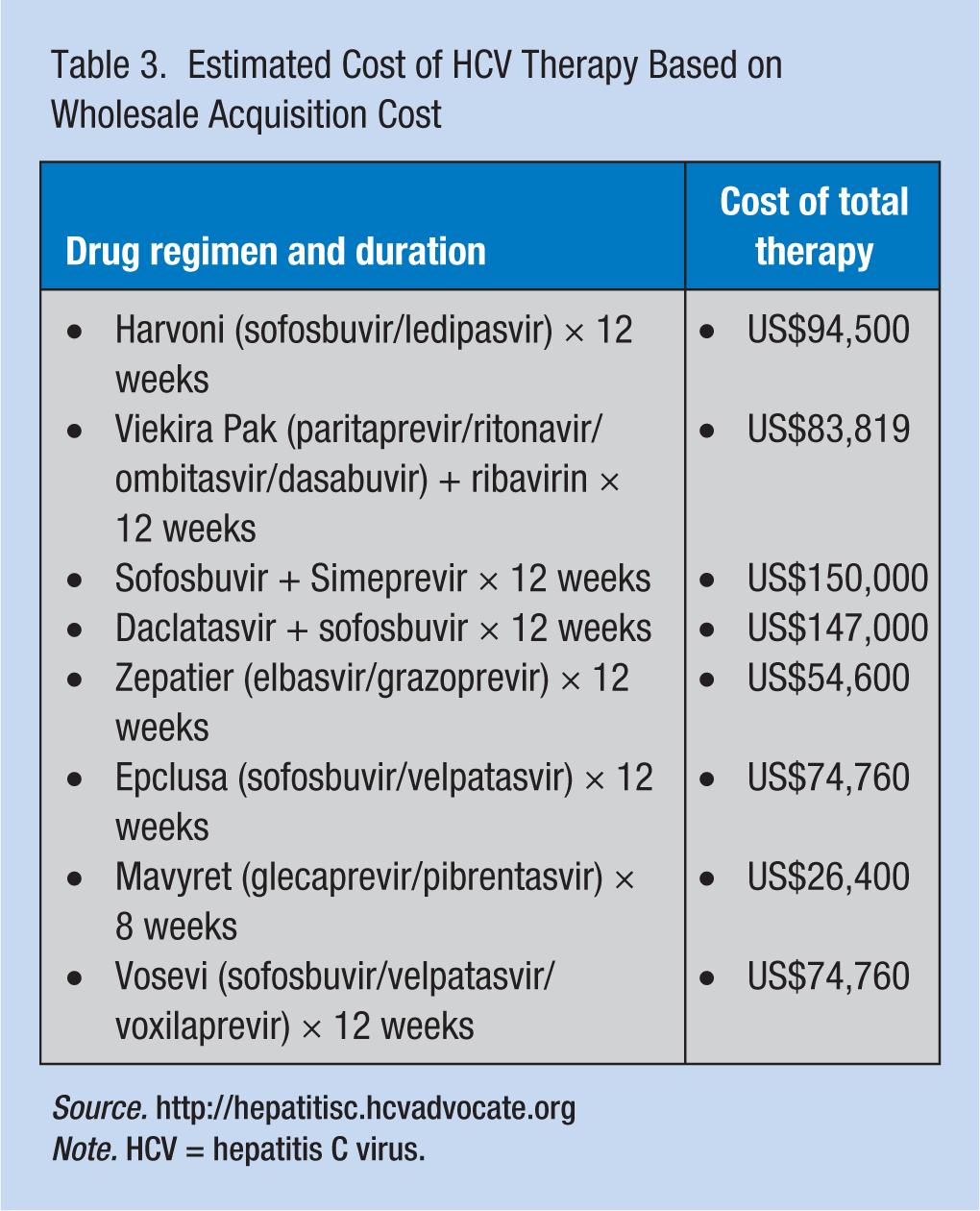
Note. HCV = hepatitis C virus.
Multiple reports have described the cost effectiveness of DAA treatment on outcomes such as disease progression. The results vary from cost savings to non-cost savings (Chhatwal, Roberts, & Dunn, 2015; Chidi et al., 2016). In general, using DAA to treat individuals with advanced fibrosis or cirrhosis provides more value than treating individuals with mild liver disease, likely due to direct cost savings from eliminating cirrhosis and decompensated liver disease progression as well as lessening complications from liver failure.
Implications for Health Care Providers
Traditionally, PCPs have been screening clients for HCV. However, occupational health services offer a unique opportunity to educate and potentially screen employees who meet CDC guidelines but may not know they are at risk for HCV. Healthy working individuals, who do not have any serious health concerns, may not see their PCPs routinely or know of new developments in HCV treatment. Because CHC often do not present with symptoms, it is imperative that all health care providers, whether in traditional health care facilities or occupational health centers, offer HCV screening to all individuals who meets CDC guidelines. Implications for occupational health nurses can be found in Table 4.
Implications for Occupational Health Nursing Practice

Note. HCV = hepatitis C virus.
Summary
With the success of new HCV treatments, more attention has been focused on HCV screening and linkage to chronic HCV infection care. Occupational health nurses and nurse practitioners should be on the frontlines of screening and diagnosing HCV infections in addition to educating employees, dependents, and retirees regarding HCV risk factors and HCV treatment and cure rates. In 2012, the CDC released new guidelines that recommended a one-time HCV screening for all adults born between 1945 and 1965 regardless of risk factors. Seventy-five percent of all individuals with HCV infections in the United States were born in these two decades.
Being diagnosed with HCV can still be stigmatized in many US communities. However, with education regarding HCV treatment, CHC can be considered less of a chronic illness and more a cured disease after SVR is achieved. With the development of new DAA therapies, HCV treatment is shorter (8 weeks-12 weeks total) and successful (SVR rates of 93%-100%). Achieving a cure or SVR has been associated with improved outcomes for all-cause mortality.
With new DAA treatment resulting in cure and easier to tolerate, the majority of workers with HCV can be successfully treated by a PCP. Education regarding HCV, through conferences, webinars, or linking with Project ECHO, can increase provider confidence in advocating for HCV treatment for their clients. With treatment to cure HCV and exceptional cure rates, it is possible that all HCV in the United States will be eradicated.
Hepatitis C Virus Laboratory Tests
HCV genotype: Six distinct genotypes (1-6) with subtypes vary with respect to the geographic location of where workers are infected. In the United States, Genotype 1 accounts for approximately 70% of HCV cases.
HCV RNA: The HCV viral load not only confirms chronic infection but also establishes baseline viral load at least 6 months prior to starting HCV treatment. Outside of HCV treatment monitoring, it is not necessary to monitor HCV RNA as it is not a prognostic indicator of liver disease progression.
Non-structural (NS) 5A resistance: In workers with Genotype 1a, baseline NS5A resistance test can determine if any baseline mutations may influence HCV treatment options.
Serologic markers for fibrosis: Currently, several mathematical models use serologic markers to estimate the amount of scarring or fibrosis in the liver. The main benefit of these markers is that the test is non-invasive. FibroSure or FibroTest uses six serum tests to generate a score that indicates the degree of liver inflammation and fibrosis. In addition to FibroSure, ordered through the laboratory, two additional free online tools can assess for fibrosis (i.e., the Fibrosis-4 [FIB-4] calculator and the aspartate aminotransferase (AST) Platelet Ratio Index (APRI) calculator):
Co-Infection and Liver Disease Etiology Laboratory Tests
HIV antibody
Hepatitis A antibody
Hepatitis B surface antigen, hepatitis B core antibody, hepatitis B surface antibody
Non-alcoholic fatty liver disease (NAFLD): lipid panel, hemoglobin A1c (HgA1c)
Hemochromatosis, a disorder of iron metabolism: serum iron, ferritin, hemochromatosis gene testing if indicated
Autoimmune hepatitis: anti-nuclear antibody (ANA), serum immunoglobulin (IgG), anti-smooth muscle antibody (ASMA)
Other Testing
Imaging/ultrasound: May identify presence of nodularity on the liver surface indicating cirrhosis; spleen size; size of the portal vein; and liver lesions (e.g., hepatocellular carcinoma [HCC]). For workers with cirrhosis, it is recommended to screen for HCC with ultrasound every 6 months.
Transient ultrasound elastography (FibroScan): Determine degree of liver fibrosis. The non-invasive FibroScan, by examining a large mass of liver tissue, can provide a more accurate assessment of liver fibrosis than serologic markers.
Liver Biopsy: Long considered the gold standard for liver fibrosis assessment, the liver biopsy is not needed to diagnose and treat CHC. The biopsy, if done, provides information on degree of inflammation and fibrosis, but is invasive and has associated risks. Liver biopsy is diagnostic for liver disease, especially in cases where two co-existing liver diseases (e.g., HCV and NAFLD) exists.
Footnotes
Conflict of Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Miranda Surjadi is a member of the Gilead Sciences HCV Speakers Bureau.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author Biography
Miranda Surjadi, RN, MS, ANP, assistant clinical professor at the University of California, San Francisco (UCSF), School of Nursing, is a faculty in the Adult-Gerontology Primary Care Nurse Practitioner (AGNP-PC) Graduate Program and is a nurse practitioner at the San Francisco Department of Public Health (SFDPH) Liver Clinic, with expertise in hepatitis C virus diagnosis and management. She has been in the field of hepatology for over 10 years and primarily manages viral hepatitis patients.
