Abstract
An employee undergoing cancer treatment was prescribed metformin, a medication used in the treatment of type 2 diabetes and currently used as adjunct therapy for cancer. Metformin’s anti-inflammatory properties inhibit tumor growth. It is speculated that cancer cells develop a metabolism that enhances aerobic glycolysis, which promotes cell growth and increases the need for glucose. Metformin decreases insulin in the blood, which decreases the amount of glucose available to cancer cells, thus starving the cells of nutrition, resulting in cell death. This case study explores the role of metformin in cancer treatment, with implications for occupational health nurses.
JC is a 55-year-old female who works as a nurse and is returning to work after 6 months. JC was diagnosed with cancer of the right breast 7 months ago. She underwent a total mastectomy of the right breast as well as several cycles of chemotherapy and radiation. JC denied any history of smoking or alcohol use. She has taken a statin for lipid control for the past 5 years. JC has a body mass index (BMI) of 26. She has two children in their late 20s and, with both pregnancies, she had gestational diabetes; since her pregnancies, she has never had an issue with glucose. JC was cleared to return to work and reported to the occupational health clinic. During the interview, the occupational health nurse noticed that JC was prescribed metformin. No mention of diabetes was included in JC’s past health history. JC explained that she did not have diabetes but the oncologist prescribed metformin as adjunct therapy to her cancer treatment. JC stated that the metformin was prescribed to “starve the cancer cells of glucose and enhance the chemotherapy’s ability to kill the cancer cells.”
Obesity and type 2 diabetes are considered risk factors for a variety of cancers (Gong et al., 2016). Adults with type 2 diabetics have poor cancer survival rates compared with nondiabetic adults (Gong et al., 2016). However, diabetic individuals prescribed metformin had a lower rate of cancer and improved cancer survival rates (Kasznicki, Sliwinska, & Drzewoski, 2014). Metformin is usually well tolerated and is associated with a lower incidence of hypoglycemia, which led oncologists to consider prescribing metformin as adjunct therapy to cancer treatment.
Metformin
Metformin decreases glucose in the blood, decreases insulin levels, and increases insulin sensitivity and oxygen delivery to tissues (Figure 1) that inhibit tumor growth. Specifically, metformin reduces the production of glucose by inhibiting gluconeogenesis in the liver. It is a medication from the biguanide class and is used to treat type 2 diabetes. Metformin activates adenosine monophosphate–activated protein kinase (AMPK) in the mitochondrial respiratory chain, which inhibits gluconeogenesis (Viollet et al., 2012). Aenosine monophosphate–activated protein kinase is an enzyme that decreases glucagon, which reduces the production of cyclic adenosine monophosphate, responsible for the regulation of glucose. Aenosine monophosphate–activated protein kinase also has an inhibitory effect on the production of glucose by the hepatic cells. Metformin increases the activity of AMPK in the skeletal muscle via glucose transporter (GLUT) 4, which improves glucose utilization by skeletal muscles. Glucose transporter (GLUT) 4 regulates insulin in adipose, skeletal, and cardiac tissue. Metformin increases the peripheral utilization of glucose by improving insulin binding to insulin receptors which results in less blood glucose released by the liver (Viollet et al., 2012)
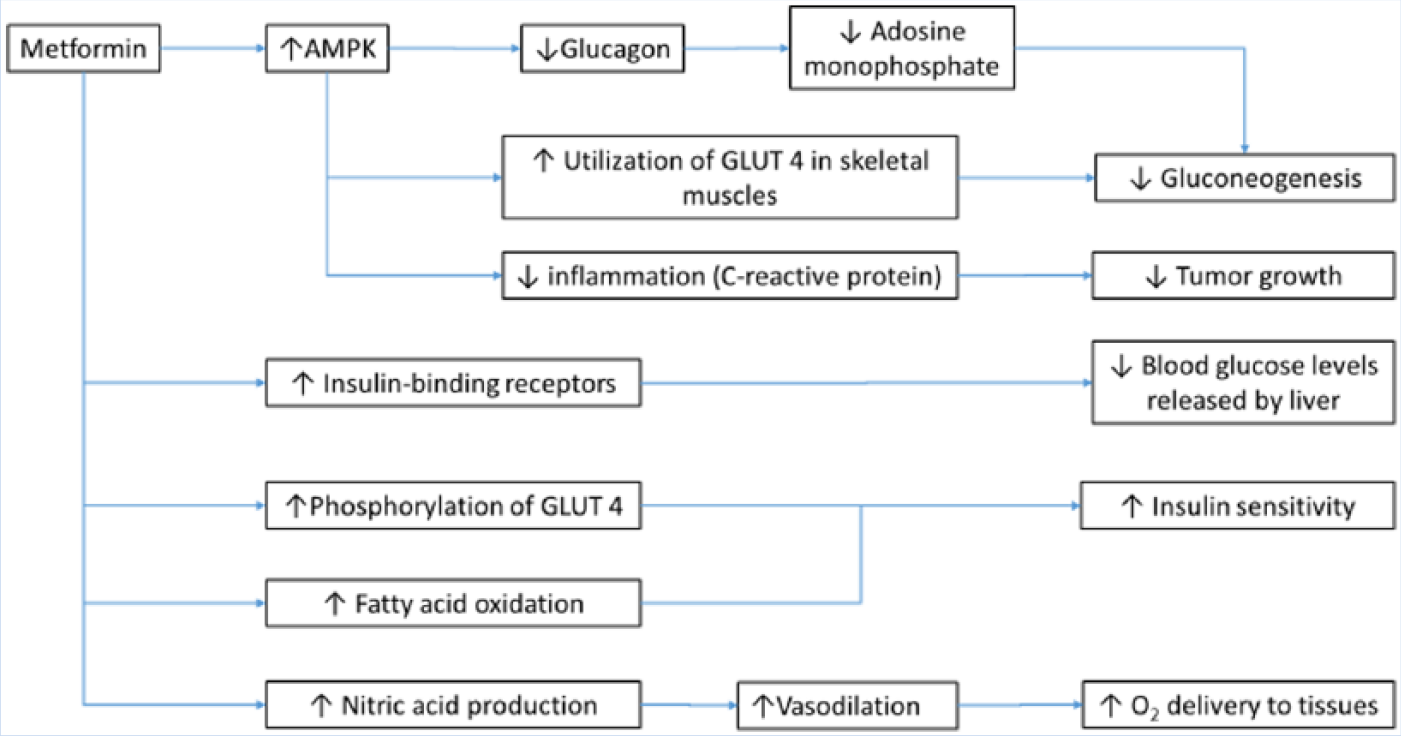
Effects of metformin on metabolism.
Metformin also increases insulin sensitivity by inducing the phosphorylation of GLUT4 and decreasing insulin-induced suppression of fatty acid oxidation (Viollet et al., 2012). Metformin enhances nitric oxide production, which supports vasodilation and the delivery of oxygen to the tissues (Liu et al., 2014) as well as lessening C-reactive protein, an inflammatory marker. Metformin may also decrease the risk for some cancers (Viollet et al., 2012). Insulin resistance and associated elevated insulin levels are related to elevated levels of insulin-like growth factors, steroid sex hormone, inflammatory responses, and adipokines, secretious cytokines associated with obesity and cancer risks (Viollet et al., 2012).
Cancer Cell Metabolism
Cancer cells are more metabolically active than noncancer cells and therefore must consume more glucose for survival. To do this, cancer cells rely on a high rate of aerobic glycolysis that results in lactic acid fermentation in cytosol (Choi, Collins, Gout, & Wang, 2013). Lactic acid fermentation requires glucose processing that results in elevated blood lactate levels. It is speculated that tumor lactate levels may be a predictor of metastasis and play a role in the inflammatory state that promotes tumor growth (Hirschhaeuser, Sattler, & Muellar-Klieser, 2011).
Aerobic glycolysis in cancer cells is a substitute for cellular respiration involving oxygen in the mitochondria. It is hypothesized that cancer cells adapt this metabolic pathway because not enough oxygen is available for cancer cell growth. In addition, cancer cells damage the mitochrondria, affecting the cell’s ability for apoptosis and the use of glucose (Boland, Chourasia, & Macleod, 2013).
The role of nitric oxide and its effects on cancer cell metabolism is controversial (Choudhari, Chaudhary, Bagde, Gadbail, & Joshi, 2013). It is speculated that high levels of nitric oxide derived from macrophages, Kupffer cells, natural killer cells, and endothelial cells inhibit tumor cell growth by suppressing DNA (Choudhari et al., 2013). In addition, high levels of nitric oxide may repair DNA damage and restore the normal function of tumor suppressor cells (Xu, Liu, Loizidou, Ahmed, & Charles, 2002).
The Effects of Metformin on Cancer Cells
Metformin contributes to cancer cell apoptosis by supporting nutrient deprivation and hypoxia. Metformin improves insulin sensitivity and lowers blood glucose and circulating insulin levels, which may lead to less growth factors that stimulate tumor formation (Kasznicki et al., 2014). Metformin acts in the mitochrondria by decreasing cellular respiration, thus lowering energy requirements; cancer cells cannot tolerate lower metabolism. Metformin also inhibits aerobic glycolysis, the main source of energy production in cancer cells.
Metformin reduces chronic inflammation and stimulates fatty oxidation, a major component of cancer development and progression (Kasznicki et al., 2014). The production of AMPK inhibits the inflammatory response and thus leads to tumor reduction. Metformin also inhibits angiogenesis by decreasing levels of vascular endothelial growth factor, responsible for the delivery of oxygen to tissues.
Metformin has been associated with less expression of oncoproteins by activating the AMPK pathway, and promotes apoptosis in cancer cells. Metformin has been associated with the loss of p53 (Li, Zhao, Parris, Feng, & Yang, 2015), a tumor suppressor protein that mutates in human cancers and thus does not promote apoptosis. With the loss of p53, cancer cells cannot adapt to lower glucose levels, which results in massive ATP depletion and apoptosis. Metformin reduces the inflammatory response that may interfere with the proper function of p53 (Li et al., 2015).
Implication for Occupational Health Nurses
Occupational health nurses must be prepared when employees return to work while undergoing or having completed cancer treatment. Nurses must be knowledgeable about the medications that are prescribed for cancer treatment, the reason they are prescribed, and their side effects. Occupational health nurses must understand the pathology of cancer and how cancer diagnoses affect employees and their ability to do their jobs safely.
To ensure a healthy workforce, occupational health nurses should educate employees about the health effects of obesity, elevated glucose levels, and insulin resistance that may contribute to the development of cancer. Emphasis should be placed on the importance of glucose management and compliance for those employees diagnosed with type 2 diabetes to reduce the risks for cancer and heart disease.
Insulin resistance may be more common in post-menopausal women (Davis et al., 2012). Due to the lack of potent estrogens, postmenopausal women may lose their sensitivity to insulin contributing to more abdominal girth, a risk factor for insulin resistance. Abdominal fat secretes adipokines, which are inflammatory cytokines that contribute to the development of insulin resistance and cancers (Viollet et al., 2012). Postmenopausal women also secrete lower levels of nitric oxide (Mercuro et al., 2006). Finally, menopause affects mitochrondria function, impairing glucose metabolism (Velarde, 2013). Exercise can improve insulin resistance, increase levels of nitric oxide, and optimize mitochrondria function (Jornayvaz & Shulman, 2010; Moreau, Stauffer, Kohrt, & Seals, 2013; Overmyer et al., 2015; Spangenburg, Wohlers, & Valencia, 2012).
Conclusion
Metformin has been shown to improve survival in adults with cancer by lowering insulin levels and improving glucose levels, which may be protective for cancer (National Institutes of Health [NIH], 2013). Metformin does not have the risk of hypoglycemia as do other diabetic medications; however, any individual prescribed metformin must eat an appropriate number of calories as prescribed by a dietitian, and monitor blood glucose levels. Contraindications to metformin are severe liver and kidney disease (Nasri & Rafieian-Kopaei, 2014); alcohol consumption should be limited when taking metformin.
Calories should be based on activity and include fruits and vegetables, whole grains, and lean meats (Centers for Disease Control and Prevention [CDC], 2016). Exercise and weight loss are encouraged as they reduce inflammation and improve immunity via T-cell mediated tumor suppressor cells. Metformin may be considered a weight loss medication due to its ability to enhance glucose metabolism; however, more research is needed to support this claim (Tan, Alquraini, Mizokami-Stout, & MacEachern, 2016)
JC reported that she will continue to use metformin and engage in regular exercise because these interventions may contribute to her survival after breast cancer treatment. She was instructed to monitor blood glucose once a day and follow the dietitian-prescribed diet. The occupational health nurse asked JC to report to the health clinic if she identified any issues with glucose readings or other measures.
Footnotes
Conflict of Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author Biography
Ann R. Lurati is a nursing lecturer at California State University, Monterey Bay.
