Abstract
Objective
To investigate the impact of metformin on survival of diabetic patients following surgery for colorectal cancer (CRC).
Methods
This was a retrospective cohort study. From Taiwan’s population-based National Health Insurance Research Database (NHIRD) we identified 12,512 patients with CRC and type II diabetes who underwent curative surgery between 2000 and 2012. Of these, 6222 patients were included in a matched cohort. Using Cox regression models with time-dependent covariates we examined the impact of metformin on survival.
Results
Average duration of follow-up was 49 and 54 months for metformin users and non-users, respectively. Cox proportional hazard model showed that metformin was associated with 5-year overall survival benefit (Hazard ratio, 0.23 [95% CI, 0.20–0.26]) and inverse association with risk of liver metastasis (Hazard ratio, 0.79 [95% CI, 0.68–0.93]).
Conclusions
Metformin was associated with a survival benefit in diabetic patients with CRC following surgery, and an inverse association with risk of liver metastases suggesting a potential anti-tumorigenic effect.
Introduction
Despite advancements in healthcare, colorectal cancer (CRC) remains the third most common cancer in terms of incidence worldwide and ranks second in terms of mortality. 1 According to GLOBOCAN 2018 data, there were 1.8 million new CRC cases in 2018 and 881,000 deaths. 1 In Taiwan, CRC is the most commonly diagnosed cancer and the third leading cause of cancer-related death.1,2 The most common pathological type of CRC is adenocarcinoma (>90%) and the remaining rare types include neuroendocrine, squamous cell, adenosquamous, spindle cell and undifferentiated carcinomas. 3 Approximately 80% of CRCs are localized to the colorectal wall and/or regional lymph nodes, and so curative management of CRC relies primarily on surgical resection, including removal of the major vascular pedicles and lymphatic drainage of the involved colorectal segment. 4
Global prevalence of type 2 diabetes is projected to increase from 415 million in 2015 to 642 million by 2040.5,6 In Taiwan, epidemiological studies show an increasing prevalence of type 2 diabetes which is highly associated with obesity, especially in young age groups. 7 Importantly, studies have shown a high risk of death from CRC in diabetic patients. 2 Diabetes and CRC share several common risk factors, including obesity, inactivity and a Western diet. However, diabetes is regarded as an independent risk factor for developing CRC.8,9 Indeed, the risk of developing colon cancer among diabetic patients is approximately 38% higher than in non-diabetic patients and the risk of developing rectal cancer among diabetics is 20% greater than in non-diabetic patients. 10
Metformin is a biguanide antihyperglycemic agent and first-line treatment for type II diabetes. 11 Several studies have shown that metformin may help lower the risk of developing certain cancers, including CRC.11–14 In addition, studies have found that the antineoplastic effect of metformin can resolve the inhibition of the mammalian target of the rapamycin (mTOR) pathway and prevent cancer cells from growing.15,16 Therefore, metformin may potentially contribute to the primary prevention and treatment of CRC as an adjuvant therapy.16–18
We conducted a nationwide population-based retrospective cohort study using data from the National Health Insurance Research Database (NHIRD) in Taiwan to investigate the impact of metformin on survival of diabetic patients and the possibility of decreasing of liver metastases following surgery for CRC.
Methods
The National Health Insurance (NHI) program of Taiwan was established in 1995 and covers >99% of the Taiwan’s 23 million population; the system’s claims data are released as NHIRD. To identify participants, we used ICD-9-CM codes (153–154 for CRC and A181, 250 for type 2 diabetes). Using data from the registry for catastrophic illnesses, we analysed claims made between January 2000 and December 2012.
Eligible patients were at least 18 years of age, had a confirmed diagnosis of CRC and had undergone curative colorectal surgery. In addition, they had a confirmed diagnosis of type 2 diabetes before the date of surgery (i.e., index date) and had received oral diabetes medications ≥28 days within the year before the index date. Patients with history of other malignancies, unspecified malignant neoplasm of the anus (ICD-9-CM code: 154.3), liver metastases before date of surgery (i.e., index date), follow-up period <3 months and/or missing data were excluded from the study.
We extracted baseline data on age, sex, income, Charlson comorbidity Index (CCI), adapted diabetes complications severity index (aDCSI), diabetes medications, statin, and cancer treatment. The follow-up period began at the date of curative colorectal surgery and lasted for 12 months until the end of 2013. During this period, if the interval between two doses of metformin was within 14 days, the drug was defined as administrated. The primary endpoint was 5-year overall survival (OS) defined as the time from surgery to death or date of the last follow-up, and the secondary outcome was the presence of liver metastasis. Liver metastasis that occurred after surgery was defined as the date the patient had an ICD-9 code of 197.7.
The study followed the STROBE statement guidelines 19 and was approved by the Institutional Review Board of Kaohsiung Veterans General Hospital (VGHKS15-EM10-02). Written informed consent was note required because this was a retrospective study and data were anonymized.
Statistical analyses
Statistical analysis was performed using SAS for Windows v 9.4 (SAS, Inc., Cary, NC). A P-value <0.05 was considered to indicate statistical significance. Descriptive statistics were calculated for all variables, with categorical data shown as percentile values, and continuous variables as mean ±standard deviation.
The propensity-score method was used in an attempt to balance covariates between treatment groups. Standardized mean differences (SMD) were used to examine the balance of covariate distribution between the groups. A standardized difference ≥0.1 indicates that there may be differences between the two groups on certain variables, suggesting a potential imbalance. 20 Kaplan-Meier analysis was used to plot survival curves for metformin users and non-users, and the log-rank test was used to compare differences between groups.
Because diabetes patients may have changed medication during the observation period, we considered metformin use as a time-dependent covariate in the Cox proportional hazard model. The impact of metformin on survival was examined after adjusting for age, sex, income, CCI, and cancer treatment. In the time-dependent covariates of metformin use, the comparison represented each period for a single patient, and drug exposure variables were presented as binary variables. This approach differs from the traditional Cox proportional hazard model, which only considers a fixed time of metformin use. In addition, we conducted a subgroup analysis to investigate the dose and duration of metformin use. We separated patients into three groups based on their metformin use during the one-year observation period. Patients with an observation period of less than one year were excluded. We estimated the adjusted hazard ratio using the Cox proportional hazard model.
Results
The flow chart of study participants is shown in Figure 1. From the 91,649 patients we identified from Taiwan’s NHIRD as having CRC and curative surgery between 2000 and 2012, 12,512 patients had type II diabetes and met our eligibility criteria. Of these, 9,401 were metformin users and 3,111 non-users. Using propensity score matched pair sampling (1:1 ratio) to adjust for differences in baseline characteristics, we identified 6,222 patients in the matched cohort.

Flowchart showing the process of enrolment.
In the unmatched analysis, the average follow-up time was 40 ± 19 and 41 ± 19 months, and the duration from day of surgery (index day) to recurrence of CRC was 50 ± 37 and 54 ± 40 months for metformin users and non-users, respectively. Following propensity score matching, the average follow-up time was 42 ± 19 and 41 ± 19 months and duration from index day to recurrence of CRC was 56 ± 41 and 54 ± 40 months for metformin users and non-users, respectively.
Baseline demographic characteristics for unmatched patients are shown in Table 1 and as shown by SMDs there were significant differences between groups. Following propensity score matching, two balanced cohorts were identified 3,111 patients in each group. There were no significant differences in the demographic or treatment characteristics between the two groups, which was confirmed by SMDs (Table 2).
Demographic and clinical characteristics in the unmatched analysis.

Data are expressed as, mean ± standard deviation, n, or n (%).
aDCSI: adapted Diabetes Complications Severity Index; CCI: Charlson Comorbidity Index; NTD: New Taiwan Dollars; SMD, standardized mean difference.
*SMD ≥0.1 indicated that there may be differences between the groups on certain variables, suggesting a potential imbalance. 20
Demographic and clinical characteristics following propensity score matching.

Data are expressed as, mean ± standard deviation, n, or n (%).
aDCSI: adapted Diabetes Complications Severity Index; CCI: Charlson Comorbidity Index; NTD: New Taiwan Dollars; SMD, standardized mean difference.
*SMD ≥0.1 indicated that there may be differences between the groups on certain variables, suggesting a potential imbalance. 20
The log-rank test showed that metformin users had significantly superior 5-year OS rate compared with non-users (P = 0.0312; Figure 2) but there was no difference between groups in the incidence of 5-year liver metastasis (P = 0.3272; Figure 3).

Kaplan Meyer curves for 5-year overall survival (OS).
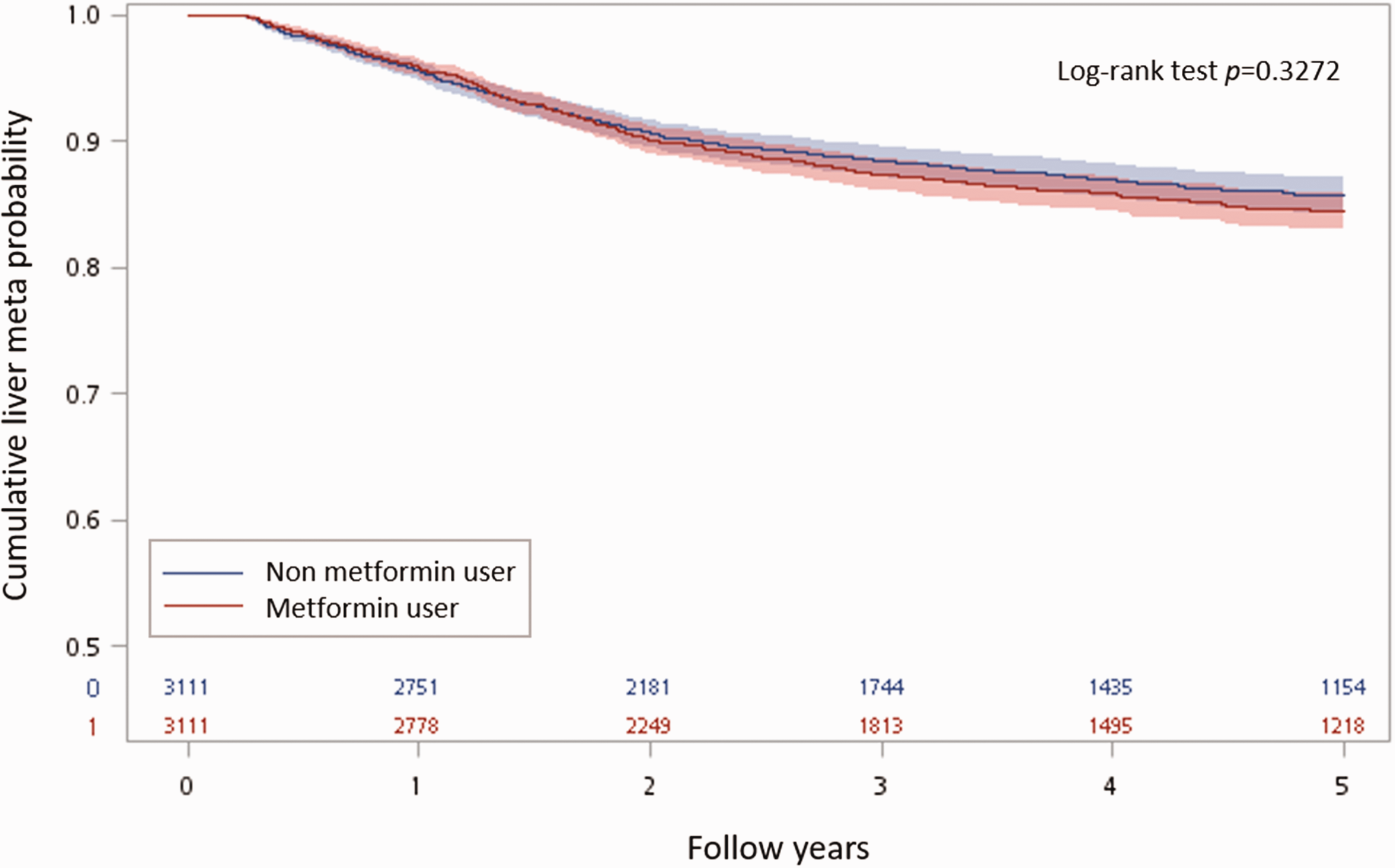
Kaplan Meyer curves showing 5-year incidence of liver metastasis.
The Cox proportional hazard model with time-dependent covariates showed that metformin use was associated with a 5-year OS benefit (HR: 0.23, 95% CI: 0.20–0.26, P < .0001; Figure 4). While the association was not as strong as OS, metformin use was associated with a decreased risk of liver metastasis (HR: 0.79, 95% CI: 0.68–0.93, P = 0.0037; Figure 5).
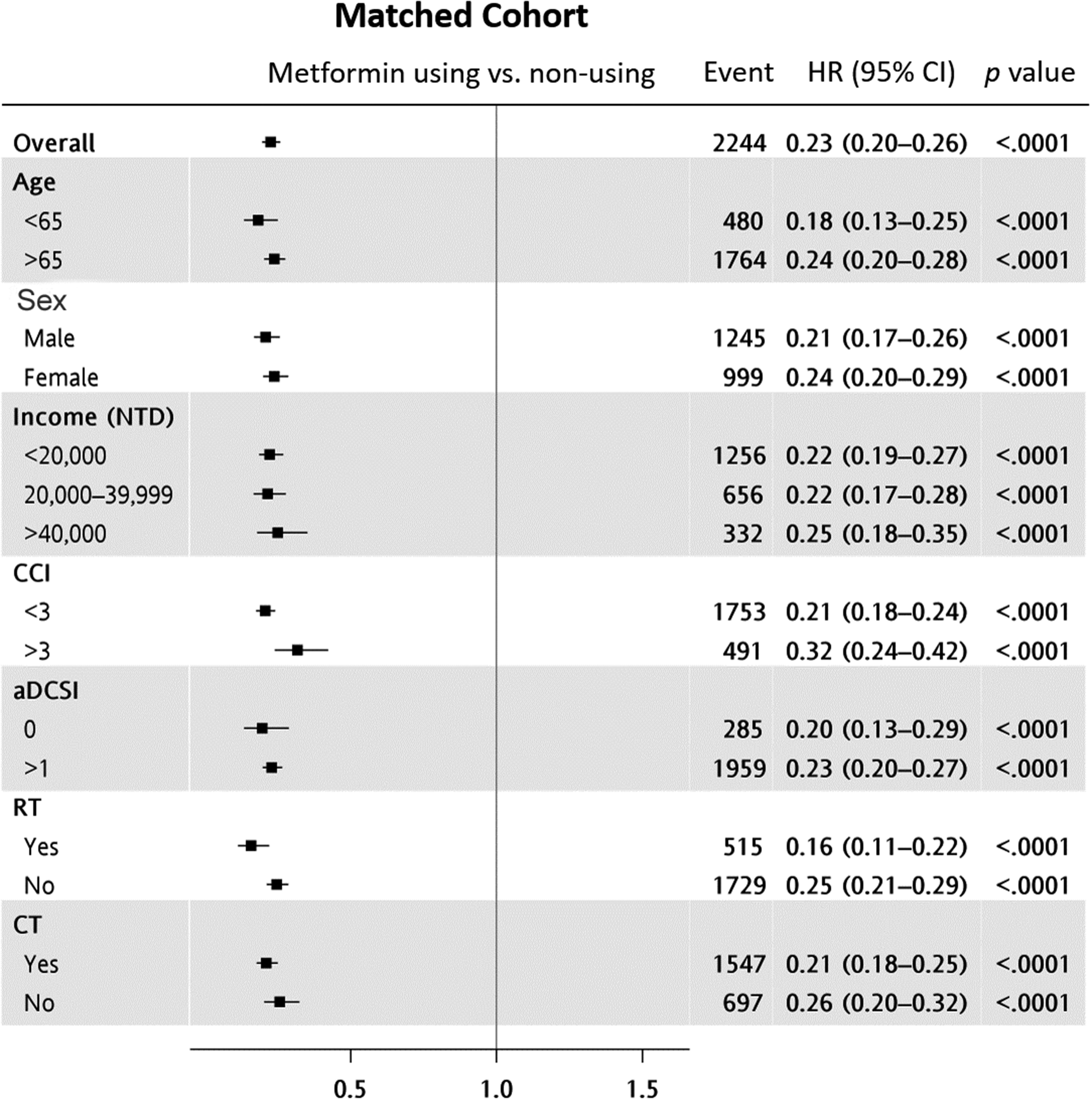
Cox proportional hazard analysis of 5-year overall survival in the matched cohorts.

Cox proportional hazard analysis of 5-year liver metastasis incidence in the matched cohorts.
In a sub-group analysis, we found that patients who used metformin for more than 270 days had a 5-year OS benefit (HR: 0.60, 95% CI: 0.52–0.70, P < .0001; Table 3). Although not statically significant we observed a trend for a protective effect against liver metastasis in patients who used metformin for more than 270 days (HR: 0.81,95% CI: 0.65–1.02, P = 0.0730; Table 3).
Cox proportional hazard analysis of 5-year overall survival and 5-year liver metastasis incidence in metformin users (n = 2,864*).

HR, hazard ratio; CI, confidence interval; ns, not statistically significant.
*All patients were separated into three groups based on their metformin use during the one-year observation period. Patients with an observation period of less than one year were excluded from the analysis.
aMetformin use (28–179 days) was used as the reference group for comparison with the other two groups.
Discussion
We found that in post-operative CRC patients with type 2 diabetes, administration of metformin was associated with a significant survival benefit and a reduction in the risk of liver metastasis suggesting a potential anti-tumorigenic effect of metformin. However, our results on liver metastasis using the Cox proportional hazard model differed from those using Kaplan–Meier analysis where the association was weaker. We believe it was because the Cox proportional hazard model considered metformin use as a time-dependent covariate, while the Kaplan–Meier analysis defined baseline metformin use as a fixed covariate. Using a fixed covariate design to analyse the influence of metformin may have underestimated the association.
Diabetes is associated with a high risk of developing CRC and inferior survival outcome following CRC.21–23 This public health issue is now a worldwide problem as the prevalence of type 2 diabetes in patients with CRC has been increasing in many countries. 24 Furthermore, the prognosis of patients with CRC is significantly affected by high blood sugar levels.25–28 Studies have suggested that diabetic patients with inadequate glycaemic control may be at high risk of CRC because they have high levels of circulating insulin and insulin-like growth factor 1 (IGF-1) which could enhance cell proliferation and inhibit apoptosis.28,29 Indeed, hyperinsulinemia, hyperglycaemia, and chronic inflammation are thought to be associated with risk of CRC.30–33
Metformin is one of the most extensively prescribed, first-line, orally administered drug for type II diabetes.16,34 In addition to its antidiabetic effect, metformin may lower the risk of developing certain cancers, including CRC.11–14,16 Although precise information on the molecular mechanisms by which metformin influences numerous cancers is lacking, it is known that metformin inhibits mTOR activity by activating ATM (ataxia telangiectasia mutated) and LKB1 (liver kinase B1) and then adenosine monophosphate-activated kinase (AMPK), and so prevents protein synthesis and cell growth.17,35 One study has shown that metformin and acetylsalicylic acid can act synergistically to supress cancer formation, but randomized controlled trials are required to verify this hypothesis. 36 Because of its tumour suppressor effects, it has been suggested that metformin may prevent certain types of cancer as well as being useful in radiotherapy, chemotherapy and androgen deprivation therapy (ADT). 35
Findings from previous studies investigating the association between metformin and reduced risk of CRC have been inconsistent. In agreement with our findings, some studies have shown high doses of metformin reduce mortality in patients with CRC.37,38 However, other studies have shown no association between diabetes or metformin and recurrence-free or disease-free survival following CRC surgery. 39 It has been speculated that differences in genetic polymorphisms may be a reason for the discrepancy in results. 35 More comprehensive research and investigation on metformin’s antitumor effects in nondiabetic patients is required, and further information is needed on biology and genetics of various cells and influence of radiotherapy. 35
The study had several limitations. For example, the NHIRD does not provide access to some potentially key clinical covariates, including diet type, smoking, and alcohol use. In addition, since the data are based on physician recorded diagnoses in medical claims, there may have been some potential misclassifications of disease and medication usage.
Nevertheless, data from this nationwide population-based retrospective cohort study showed that metformin was associated with a significant survival benefit and a reduction in the risk of liver metastasis in post-operative CRC patients with type 2 diabetes. Moreover, we found the continuous use of metformin was associated with a reduction in risk of liver metastases, which, to our knowledge, has not been reported elsewhere. Further prospective, randomized research is required to confirm our findings.
Research Data
Research Data for The impact of metformin on survival in diabetes patients with operable colorectal cancer: A nationwide retrospective cohort study
Research Data for The impact of metformin on survival in diabetes patients with operable colorectal cancer: A nationwide retrospective cohort study by Ping-Teng Chu, Tzu-Jung Chuang, Shu-Hung Huang, Tung-Ho Wu, Wei-Chun Huang and Jui-Ho Wang in Journal of International Medical Research
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
