Abstract
Background
We describe the study design and protocol of a pragmatic randomized controlled trial (RCT) Acupressure for Children in Treatment for a Childhood Cancer (ACT-CC).
Objective
To describe the feasibility and effectiveness of an acupressure intervention to decrease treatment-related symptoms in children in treatment for cancer or recipients of a chemotherapy-based hematopoietic stem cell transplant (HSCT).
Design
Two-armed RCTs with enrollment of 5 to 30 study days.
Setting
Two pediatric teaching hospitals.
Patients
Eighty-five children receiving cancer treatment or a chemotherapy-based HSCT each with 1 parent or caregiver.
Intervention
Patients are randomized 1:1 to receive either usual care plus daily professional acupressure and caregiver delivered acupressure versus usual care alone for symptom management. Participants receive up to 20 professional treatments.
Main Outcome
A composite nausea/vomiting measure for the child.
Secondary Outcomes
Child’s nausea, vomiting, pain, fatigue, depression, anxiety, and positive affect.
Parent Outcomes
Depression, anxiety, posttraumatic stress symptoms, caregiver self-efficacy, and positive affect. Feasibility of delivering the semistandardized intervention will be described. Linear mixed models will be used to compare outcomes between arms in children and parents, allowing for variability in diagnosis, treatment, and age.
Discussion
Trial results could help childhood cancer and HSCT treatment centers decide about the regular inclusion of trained acupressure providers to support symptom management.
Introduction
Background and Rationale
Despite advances in pharmacologic symptom management, most children with cancer1,2 or receiving a hematopoietic stem cell transplant (HSCT)3–6 suffer from multiple treatment-related symptoms including nausea, vomiting, 7 pain, 8 and fatigue. 3 Pharmacologic symptom management can be effective, but can have side effects that decrease quality of life.9,10 Symptom distress is an important predictor of poor health-related quality of life among children treated for cancer 11 or receiving HSCT.6,12 Severe symptoms can result in interruptions in planned treatment, dose reductions, or other therapy changes with the potential to compromise survival.13,14 A systematic review documented frequent undertreated symptoms during oncology and HSCT treatment. 1
A growing body of high-quality evidence from large meta-analyses and systematic reviews of adult acupuncture point stimulation supports the use of acustimulation for the management of adult pain,15–19 headache,20–22 postoperative nausea/vomiting (PONV), 23 presurgery anxiety, chemotherapy-related nausea/vomiting (CINV),24–35 cancer-related pain,24,36 cancer-related anxiety,32,37,38 chemotherapy-related fatigue,38,39 and cancer treatment-related symptoms. 24 (The term acustimulation is used throughout the protocol and refers to the use of needles, laser, or pressure stimulation.) A National Institutes of Health (NIH) Consensus Conference assessed acupuncture as effective in reducing CINV and PONV. 40 Compared to pharmacologic symptom management alone, in adults, acustimulation may provide equivalent 41 or superior 30 symptom relief and decreased the use of rescue antiemetics and opioids. 29
Fewer studies support the use of acustimulation in children. Systematic reviews reported that acupuncture decreased pain, 42 and a Cochrane review reported decreased PONV. 41 Three randomized controlled trials (RCTs) reported no statistically significant effect of acupressure on CINV43–45 or PONV.46,47 Two well-designed, but small RCTs of children receiving highly emetogenic chemotherapy for treatment of a childhood cancer reported decreased nausea and vomiting48,49 as well as reduced the use of antiemetic medications following acupuncture. 50 An RCT of children with leukemia showed decreased postchemotherapy fatigue with the application of acupressure to a single point (St36), but results lasted less than 24 hours. 51 A review article of acustimulation for nausea, vomiting, or rescue antiemetics describes the quality of evidence as low but having nonsignificant moderate effect sizes (ESs) in acupuncture versus sham in children. 52 Studies consistently report few side effects and a strong safety profile.41,42,48,53 Most acustimulation studies described side effects from acupuncture (not acupressure), and these included pain on needle insertion, redness, irritation, bleeding, swelling, itching, or fatigue.42,48,53
In its supportive care guidelines The Children’s Oncology Group (COG) states “that acupuncture, acupressure … may be effective in children receiving antineoplastic agents” to reduce CINV. 54 However, the strength of the COG recommendation is rated as “weak” and the quality of evidence is categorized as “very low.” 54 Acustimulation has the potential strengths of being safe, having few side effects, and addressing multiple symptoms with one treatment. However, the majority of systematic reviews have concluded that additional high-quality trials are needed among children to assess the effectiveness of acustimulation to reduce pain, nausea, and vomiting in general populations,42,55–59 as well as in childhood cancer.29,30,60,61
Trial Objectives/Aims
The primary objective of Acupressure for Children in Treatment for a Childhood Cancer (ACT-CC) is to describe the feasibility, benefits, and risks of an acupressure intervention plus usual care versus usual care alone for symptom management in children in treatment for a childhood cancer and/or receiving a chemotherapy-based HSCT. Aim 1 assesses whether patients in the acupressure arm, compared to usual care alone arm, will report decreased nausea/vomiting (primary outcome) and improved management of treatment-related symptoms such as nausea, vomiting (separately), pain interference and intensity, fatigue, anxiety, depression, and greater positive affect (secondary outcomes). It is hypothesized that rescue antiemetic and analgesic use will be lower in the acupressure arm and that greater acupressure dose will lead to greater improvement of primary and secondary outcomes. Aim 2 investigates whether parent involvement in the delivery of acupressure reduces posttraumatic stress symptoms, anxiety and depression, and increases positive affect and caregiving self-efficacy.
This protocol manuscript is based on the SPIRIT (Standard Protocol Items: Recommendations for Interventional Trials) guidelines to ensure comprehensiveness of reporting 62 with recommended additions from the SPIRIT protocol to Traditional Chinese Medicine (TCM). 63 The study protocol will be made available on the ACT-CC study website once the trial is complete.
Trial Design
This is a 2.5-year pragmatic RCT among children receiving treatment for a childhood cancer or a HSCT. We will enroll 85 dyads consisting of a child with a parent/caregiver for a total of 170 participants. Study staff and health-care providers are blinded to study arm assignment. Participants will be randomized 1:1 into the 2 arms and will be followed for up to 30 days of hospital-based oncology or HSCT treatment (continuous or intermittent days) within 2 calendar months from enrollment. If a patient finishes treatment before 30 study days (ie, end of stem cell transplant hospitalization), the study enrollment period ends. Children in Arm A will receive professional acupressure 5 days/week along with usual care, and their parent/caregiver will be trained in how to provide additional acupressure to manage symptoms. Arm B participants will receive usual care alone. (The caregiver will be trained in how to provide acupressure at the end of study participation.)
Stakeholder Involvement
Stakeholders were engaged with the original design for the pilot study during study development, and have ongoing involvement in the study implementation (recruitment, outreach, intervention, and reviewing patient materials). Stakeholders will continue their involvement in the interpretation of the results and in discussions on framing or interpretation of findings.
Methods: Participants, Intervention, and Outcomes
Study Setting
Inpatients and outpatients are enrolled from a United States, Northern California Pediatric Teaching Hospital with 2 sites. Patients are enrolled from hematology-oncology, neuro-oncology, or HSCT units.
Participants’ Eligibility Criteria
The ACT-CC study aims to enroll 85 patients, most with 1 caregiver. Recruitment started in October 2017. Inclusion criteria for ACT-CC require that the child is either receiving treatment for childhood cancer (curative or supportive care) or a chemotherapy-based HSCT (nonmalignant central nervous system tumors are considered a childhood cancer); be age 5 through 24 years old; be receiving hospital-based treatment (inpatient or outpatient); English or Spanish speaking; and availability and willingness of a parent or caregiver to deliver acupressure for patients aged 5 to 17 years. (For young adults aged 18–24 years, participation of a parent, close friend, or family member/caregiver is preferred but not required.) Children are excluded if the treating oncologist, health-care provider, or a study investigator advises against study participation for physical health, mental health (parent or child), or logistical reasons. Examples of exclusion reasons include when the health-care provider thinks participation might be burdensome, there are serious mental health issues for child or caregiver, family stress, a child is receiving a radioactive treatment and is in isolation, or hospital visits are not long enough or frequent enough to provide the intervention.
Intervention
The protocol is based on adherence to the STRICTA reporting guidelines (Standards for Reporting Interventions in Clinical Trials of Acupuncture) 64 which are an extension of the CONSORT Statement 65 and the CONSORT guidelines for nonpharmacologic interventions. 66 The STRICTA guidelines were developed to improve the completeness and transparency of reporting acupuncture interventions (see Table 1). 64
Summary of the Semistandardized Intervention Details.

Abbreviations: ACT-CC, Acupressure for Children in Treatment for a Childhood Cancer; TCM, Traditional Chinese Medical; TENS, Transcutaneous Clectrical Nerve Stimulation.
aBarriers to use of bilateral points include limb amputation and medical equipment obstructing access.
Acupoint treatment order
Most points used are located distal to the elbows or knees. Treatment is applied based on primary symptom/complaint using standard points (termed “platform points”) prescribed for each symptom. Additional points (+) are included in the protocol and can be chosen based on secondary symptoms, experience, and TCM diagnosis (see Table 2).76,77
Points for Each Symptom/TCM Diagnosis.

Abbreviations: TCM, Traditional Chinese Medical; +, additional points that can be used to supplement the platform of points.
aParents only use bolded points.
Acupressure points are applied in the following order:
Upper limbs—yin (medial) side (distal to proximal) then yang (outer) side (distal to proximal) Lower limbs—yin side (distal to proximal) then yang side (distal to proximal) Prone (face down)—lower torso working up to head Supine (face up)—abdomen working up to chest and then face
Delivery of Intervention for Each Group
Intervention precautions
Before proceeding with the acupressure session, the provider verifies platelet counts, assesses for skin breakdown associated with graft versus host disease (GVHD), or infection precautions. If platelet counts are <10 000/mL, usual supportive care guidelines recommend a transfusion. If the patient has not received a transfusion, the acupressure provider instructs parents to defer acupressure until platelets recover. Professional acupressure will continue to be delivered by the practitioner using gentle touch as per recommendations from the oncology team. In the case of infection precautions, the acupressure provider follows designated institutional infection control protocols including wearing gloves, mask, and gown as indicated. Acupressure will not be performed when there is skin breakdown or performed only on unaffected areas.
Patients are treated in their clothing or hospital gowns either lying in bed or seated in a chair according to patient preference. Lotion is not used. To assess intervention fidelity, randomly selected sessions are observed by study investigators.
Parent acupressure intervention
Caregivers are trained by the acupressure providers in the performance of acupressure for the patient. The training occurs at the same time as the professional acupressure intervention is initiated. Parents are encouraged to provide acupressure on a daily basis or as requested by the child. Teaching materials include a brochure with instructions including pictures of each point, a description of recommended points for each symptom, and point names (in English and Chinese). Video teaching instructions are also available on the study website for parents in Arm A (using a website log-in). During the first week of regular child acupressure, the provider begins by teaching parents the safety precautions and acupressure points for specific symptoms (eg, nausea, vomiting, pain, and fatigue). Teaching is updated each time the acupressure provider gives a session until parents are confident. Parents in Arm B (controls) are given acupressure instructions and materials when they complete the study.
Usual care for symptom management applied to both groups
Usual care is defined according to Children’s Oncology Group endorsed supportive care guidelines for nausea or vomiting 54 with updated guidelines7,96–100 and guidelines for management of pain 101 and fatigue. 102 Usual care for symptom management to reduce nausea and vomiting involves the classification of the antineoplastic agents into varying levels of emetogenicity (highly emetogenic chemotherapy [HEC], moderately emetogenic chemotherapy [MEC], low and minimal emetic risk) and the provision of appropriate antiemetic agents on a regular schedule. The goal is to achieve optimal control of acute CINV, defined as “no vomiting, no retching, no nausea, no use of antiemetic agents other than those given for CINV prevention, no nausea-related change in the child’s usual appetite and diet.” 103 Different antiemetic agents are recommended based on the age of the child, treatment factors (ie, avoidance of drug interactions), and the emetic risk. Guidelines to treat anticipatory and breakthrough nausea and for the prevention of refractory nausea are also followed.7,99
Discontinuation of Intervention
The intervention will be discontinued if continued participation is no longer in the patient’s best interest (ie, in response to an adverse event as needed; emergence of skin breakdown due to GVHD), upon patient or caregiver request, when exclusion criteria occurs (even mid-enrollment) or in the case of concurrent illness that prevents acupressure from being delivered (ie, when a patient is moved to the Pediatric Intensive Care Unit).
Intervention Fidelity
The principal investigator (PI) does periodic observations of the acupressure providers for fidelity to the written intervention protocol.
Outcomes
Patient-Reported Outcomes Measurement Information System (PROMIS) measures are used when available consistent with the study interest in patient-reported outcomes (PROs). They are valid, easily understood and interpreted, 104 comparable across studies, often used in clinical trials, 105 demonstrate strong reliability (≥.85) for each domain measured,106–108 and when a repeated measure, are responsive to changes in status in pediatric cancer patients 109 and in adults. 110 Both adult and pediatric measures are designed for those with chronic health conditions (as well as general populations), aged 8 to 17 years, 111 and child proxy measures can be filled out by parents to assess symptoms in children aged 5 to 7 years or when children are unable to fill out forms.103,112 Studies have verified the feasibility and efficiency 113 of administering PROMIS instruments via tablet computers (see Table 3). 114
Study Child and Parent Outcomes.
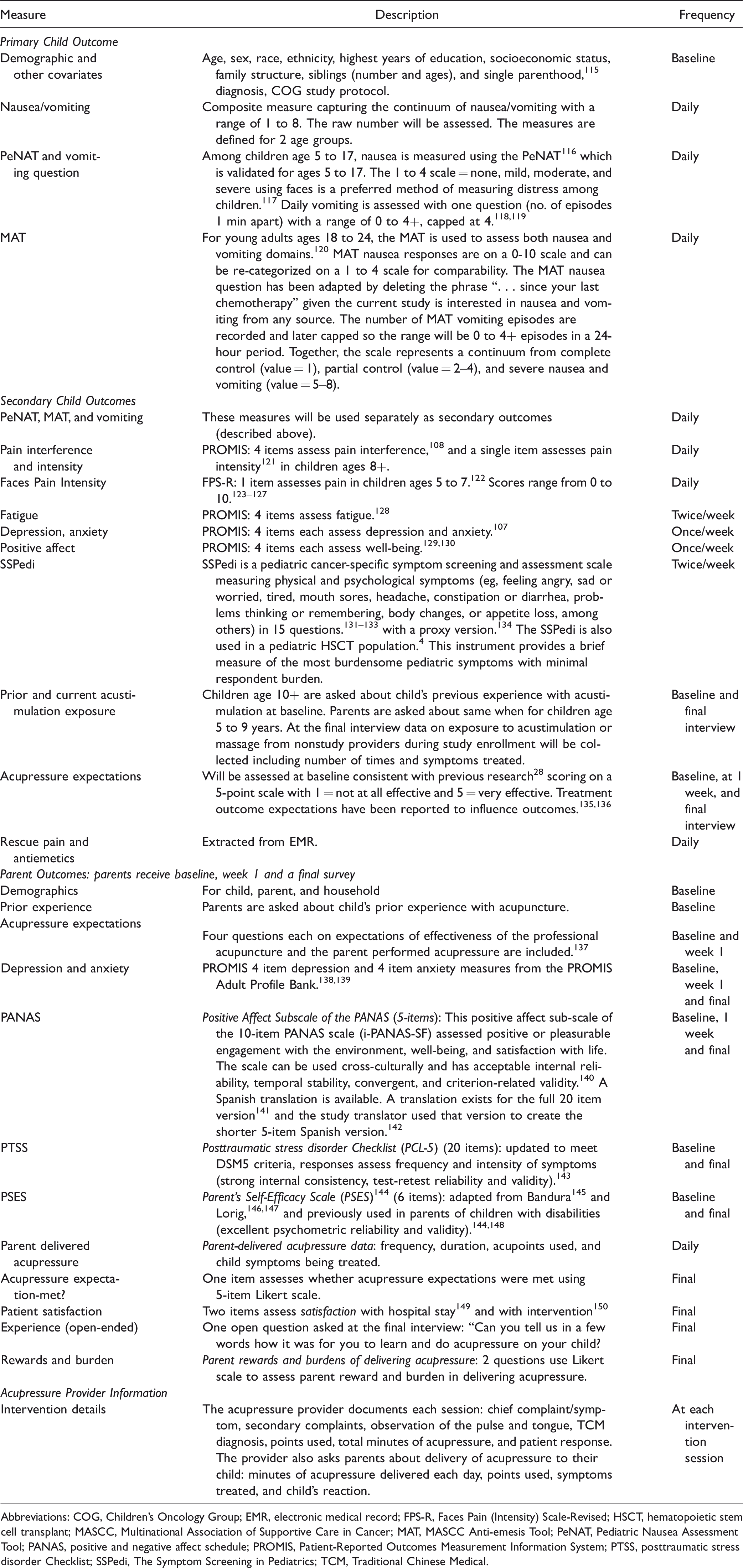
Abbreviations: COG, Children’s Oncology Group; EMR, electronic medical record; FPS-R, Faces Pain (Intensity) Scale-Revised; HSCT, hematopoietic stem cell transplant; MASCC, Multinational Association of Supportive Care in Cancer; MAT, MASCC Anti-emesis Tool; PeNAT, Pediatric Nausea Assessment Tool; PANAS, positive and negative affect schedule; PROMIS, Patient-Reported Outcomes Measurement Information System; PTSS, posttraumatic stress disorder Checklist; SSPedi, The Symptom Screening in Pediatrics; TCM, Traditional Chinese Medical.
Additional data related to child outcomes are collected from the electronic medical records (EMRs) including supportive care pain and antiemetic medications; medications for sleep; vomiting episodes; pain; ability to drink; parenteral nutrition; fever; chemotherapy complications (infection/sepsis, bleeding, GVHD, or hepatic veno-occlusive disease). Laboratory data being collected include the absolute neutrophil count (ANC) and platelet count. Additional information will be collected on likely precipitants for nausea/vomiting including chemotherapy (agent, dose, timing), surgery (length of surgery, length of time since surgery, and body location), and investigational drugs. Related to the timing of treatment, variables will be created based on the following conditions: CINV will be categorized for children reporting nausea during administration and up to 24 hours following chemotherapy.151,152 For delayed CINV, we will analyze CINV scores in groups (HEC, MEC, vs lower intensity) up to 4 days after the end of chemotherapy administration based on recommendations from a National Institute for Health Research report. 151 For PONV, we will categorize a participant at risk for nausea/vomiting following surgery for up to 24 hours. 153
Participant Timeline
Table 4 details the participant timeline.
Participant Timeline: Schedule of Enrollment, Interventions, Assessments, and Visits to Participants.

Abbreviations: CRC, clinical research coordinators; CTSI, Clinical & Translational Science Institute; EMR, electronic medical record; HIPPA, Health Insurance Portability and Accountability Act; PI, principal investigator.
aClinical and Translational Science Institute services doing digital downloads of specified data.
Sample size
The target sample size is 85 participants along with ∼85 parents/caregivers. Participants are randomized on a 1:1 basis into each of the 2 study arms. Under some simplifying assumptions, detailed below, we estimate that this sample of 85 patients, randomized 1:1 to acupressure versus usual symptom management, will provide 80% power in 2-sided tests with α of .05 to detect average reductions of 0.52 to 0.58 standard deviations (SDs) in pairwise comparisons between the acupressure arm and the usual care only arm, depending on the intraclass correlation (ICC) of the repeated outcome measures (assumed to be 0.6–0.8). (With a more conservative assumption where ICC = 0.9, the minimal detectable effects [MDE] would increase to 0.61.)
MDEs in context
In a systematic review of acupressure studies of CINV, the standardized ES for vomiting episodes was 0.53 SDs, for retching episodes ES = 0.44 SDs, for severity of nausea, ES = 0.73 SDs, for the use of rescue antiemetics ES = 0.46 SD’s, 35 child-reported pain ES = 0.42 SDs, and fatigue ES = 0.78 SDs. 90 A composite score for nausea/vomiting should fall within the ES range for CINV in previous studies. Additional ESs for secondary symptoms fall within a similar range where pain ES = 0.37–0.55. 93 Thus, our calculations suggest that, in the case where the ICC = 0.6, with a sample size of 85 we expect to have 80% power to detect plausible and clinically significant effects for nausea, vomiting, and fatigue outcomes and may be able to detect differences in pain. If the ICC = 0.8, we expect to be able to detect differences for nausea and fatigue.
Recruitment
Patients are identified and recruited at varying stages of their disease course: at the time of diagnosis; during a new or repeat hospitalization, mid-treatment, or at relapse.
The study nurse monitors all planned and unplanned admissions and outpatient appointments, and prescreens for eligibility including age, diagnosis, language, and availability of a parent/caregiver. Approval of a treating health-care provider is sought before eligible patients are approached during inpatient or outpatient visits to the hospital. Consent is obtained from parents/caregivers and patients who are 18 years and older, and assent is obtained from patients aged 8 to 17 years following UCSF institutional review board (IRB) guidelines (see Figure 1).
Methods: Assignment of Interventions for Controlled Trials
Randomization, Allocation Concealment Mechanism, and Blinding
After collection of baseline data, the study nurse (who does not see study patients) performs 1:1 simple randomization into 2 groups: acupressure + usual care versus usual care alone using a Qualtrics computer-based assignment program in which the study nurse enters the participant ID number before randomization is revealed. Assignment is immediately locked into the database. Study staff who carry out the symptom assessments and health-care providers are blinded to study arm assignment. The use of simple randomization is considered the strongest design to prevent selection bias. 154 The acupressure provider, floor nurses, patients, and caregivers are not blinded due to the type of intervention. Floor nurses are informed before the acupressure provider enters the room to ensure that there are no medical issues that need to be addressed. Families are asked not to disclose their study arm to the staff and periodic checks assure us that the physicians remain unaware of allocation. Allocation is communicated to the family by the acupressure provider who naturally knows the allocation.
Methods: Data Collection, Management, and Analysis
Data Collection
Study staff were trained in the collection of survey data. Data are monitored post interview for accuracy and completeness. Survey data collection can occur in 4 ways: paper survey administered by staff, paper survey filled out by patient or parent, staff administered assessment using a portable device (ipad) with a link to a programmed questionnaire, or patient/parent filling out survey using link to the online questionnaire. See Table 3 for survey outcomes and timing. Study assessments are allowed a window of ±3 weekdays, ±4 days between consent and baseline, and ±10 days for final surveys. Electronic Medical data for each patient participant for each study day are collected using digital methods.
Patient retention is promoted by efforts of study staff to form a trusting relationship with participants, the use of a financial incentive of $50 each (caregiver and child) upon study completion, and, for the control arm, by providing instruction and training for caregivers to deliver acupressure at the end of participation.
Data Management
Survey data are entered into the Qualtrics database and recruitment and enrollment tracking data are recorded and stored using secure HIPAA compliant software. The study project manager monitors data entry for accuracy and completeness for both sites. Survey data include study ID with no protected health information (PHI) with the exception of the baseline and follow-up survey. Baseline and follow-up surveys are deidentified before the analysis phase. Information linking the study ID and patient PHI are kept in a HIPAA secured environment separate from survey data. Physical records are kept in an area accessible only to research staff and in a locked file cabinet. No names or individual identities will be used in publications resulting from the study. Research data are stored on a secure, HIPAA-compliant server and drive with monitored and controlled access for study staff and investigators.
Statistical Analysis
Aim 1: Child Outcomes
Summary of proposed modeling approach
This study will use linear mixed models (LMMs), an extension of least squares regression to longitudinal data. Among other advantages, this approach will accommodate differences in treatment timing, intensity and emetogenicity between the treatment groups, and differences related to treatment uptake and the semistandardized intervention, while also accounting for within-patient correlation of repeated measures, optimally weighting data for patients with different numbers of responses, and providing valid estimates in the presence of missing data under relatively mild assumptions about how the missing data arise. 155 In brief, we will flexibly model the time course of the composite nausea/vomiting severity scale for each treatment group and by emetogenicity, which will allow us to estimate average differences between these trajectories by randomized study intervention assignment (acupressure vs usual care). The model will intrinsically weight patients in proportion to the length of the course of treatment.
Technical description of analysis approach
We will flexibly model the average time course of composite nausea/vomiting scores (and severity of symptoms for other outcomes) for each combination of treatment modality and intervention assignment, using group-specific restricted cubic splines (RCS) in time since the start of study enrollment. The model will also include random intercepts and RCS components, to flexibly model patient-specific departures from the average group-specific trajectory. In addition, to capture spikes in symptom severity in the first few days after surgery or chemotherapy, the model will include time-dependent covariates flexibly modeling the effect of time since the most recent chemotherapy, surgery, and/or radiation, depending on the outcome (ie, for CINV chemotherapy will be dominantly factored in). The function of these adjustments is to explain variability in the outcome, increasing model efficiency. Normalizing transformation of the composite nausea/vomiting symptom severity scale will be used if necessary. We will assess the adequacy of both the fixed and random parts of the model and check for modification of the effects of the acupressure by both time and treatment modality. We will perform intent-to-treat analyses (by original assigned groups), which is the recommended method for describing clinical trial results, 65 and additional analyses will report on groups as treated.
Assumptions and sensitivity analyses
This approach relies on the plausible assumption that the timing and duration of treatment will not be affected by assignment to or intensity of the use of acupressure. Sensitivity analyses will be conducted assessing evidence for violations of this assumption, using between-arm comparisons of treatment patterns, as well as models for the association of lagged acupressure intensity with the timing of subsequent treatments.
Dose–response analysis using marginal structural models (Aim 1)
Our primary analyses for each symptom will be by intent-to-treat, according to allocation assignment, without regard to the intensity or duration of acupressure actually received. To assess dose effects as an exploratory analysis, the effects of total minutes of professional acupressure (as well as total minutes of professional plus caregiver provided acupressure) received per week, treated as an ordinal category with 3 to 4 levels, will be estimated in a secondary analysis. Because acupressure duration may depend on earlier levels of the outcome, marginal structural models will be used.
Aim 2: Parent Outcomes
We will determine whether parents differ in posttraumatic stress symptoms (total symptom scores), depression, and anxiety (T scores) and caregiver self-efficacy (score) between groups at 2 points: at week 1 for both arms regardless of pattern of hospital-based treatment; and at 1 month post-study enrollment, which will vary depending on whether patients have 1 month of continuous hospitalization followed by 1 month for follow-up or intermittent hospitalization or outpatient treatment over 2 months followed by a 1 month follow-up. In both cases, analysis will control for baseline values as appropriate. We will use similar analytic approaches as in Aim 1, with LMM as the primary analysis approach.
Exploratory analysis of heterogeneity of treatment effects
Using LMMs we will explore heterogeneity of treatment effects by demographics (age, sex, race/ethnicity), diagnosis, treatment agent/modality, or initiation of most recent treatment dose, with the goal of assessing variation in benefit from acupressure by subgroup. Our overall hypothesis in regard to heterogeneity of treatment effect is that groups with more severe symptoms of nausea and vomiting will experience greater benefit. 156 If certain groups benefit more from acupressure, recommendations may target provision of the intervention to these groups. Given our anticipated sample size of n = 85, within-subgroup effect estimates will likely have wide confidence intervals, and power to detect between-subgroup differences may be low. Since previous research has not documented point estimates for differences between subsamples within pediatric populations, these findings will provide preliminary estimates of ESs within subgroups which will be the basis for future studies of subsamples.
Missing Data Approaches
Some outcome data may be missing not at random (MNAR)—in particular, when missing symptom assessments occur due to early hospital discharge, moving into hospice care, severe symptoms or illness, refusal to answer, or conflicts with other medical procedures. To address this difficulty, we will perform sensitivity analyses using multiple imputation of missing outcomes under plausible MNAR scenarios as well as under the standard MAR assumptions for key outcomes. Where feasible, we will utilize appropriate information on nausea, vomiting, and pain available in the EMR in multiple imputation models to take advantage of data that may be available even when patient-reported symptom data cannot be collected directly.
Definition of populations
Intent to treat is defined as the entire randomized sample.
Methods: Monitoring
Data Safety Monitoring and Audits
The study is monitored by the Helen Diller Family Comprehensive Cancer Center (HDFCCC) Regulatory Unit for quality and regulatory compliance with yearly audits of 20% of participants (guideline for minimal risk trial) and biannual review by the Data Safety Monitoring Board (DSMB) reporting on data quality, subject safety, serious adverse reactions, and yearly accrual. The DSMB is independent from PCORI and the Investigators have no competing interests. The DSMB is made up of members who are knowledgeable in the conduct of research, with backgrounds in cancer, biostatistics, experimental design, or bioethics. The HDFCCC DSMB charter can be found at http://cancer.ucsf.edu/itr/itr-dsm.
Interim Analyses and Stopping Rules
The primary clinical outcome variable, nausea/vomiting, is a symptom associated with exposure to pediatric cancer treatment and HSCT conditioning. Continued nausea/vomiting is not an outcome that would justify early termination rules for a study of this nature, and it is highly unlikely that there would be differences in mortality or any other major health difference between arms. No interim analyses related to outcome will be performed and we do not anticipate stopping the trial early.
Adverse Events
Any adverse events (AEs) related to the administration of acupressure, specifically including bruising or skin irritation, are recorded. AEs can be reported by the family or the medical provider. All AEs are entered into the study records. Study AEs are graded. Serious (grade 3 and above) AEs are reported to IRB and the DSMB. AEs are monitored and discussed at every study staff and Co-Investigator meeting.
Ethics and Dissemination
The ACT-CC study has been approved by UCSF Helen Diller Family Comprehensive Cancer Center Protocol Review Committee and the IRB at UCSF Benioff Children’s Hospital Mission Bay site (9-26-17) and Benioff Children’s Hospital-Oakland site (11/3/17). HIPAA permissions, Patient Bill of Rights and consent/assent is obtained by trained study staff who receive 6 weeks of Cancer Center training in addition to training from the PI. No biological samples are collected for this trial.
This study includes a vulnerable population of children with life-threatening conditions. Thus efforts are made to reduce patient burden in the following way: after baseline regular assessments are short (<5 min) and enrolled patients can decline acupressure sessions or assessments; parents in Arm A are taught to provide acupressure and training is available throughout enrollment; surveys can be performed using the most convenient survey method. A pediatric cancer or HSCT population can be challenging to access given severe symptoms, fatigue, the need to have both the child and parent present for consent and intervention delivery for the youngest group, and the need to work around curative treatment procedures, medical equipment, and health-care personnel visits to the room. Thus, study staff receive training and ongoing monitoring as recruitment and enrollment occurs to ensure that patient needs are honored and study procedures are transparent, clear, and optional. Acupressure training is provided to caregivers in the control arm after the family finishes the trial to ensure that everyone enrolled has the opportunity to benefit from the intervention if desired. We ask participants to refrain from receiving acupuncture, acupressure or massage from outside providers during the trial unless medically indicated, in which case, patients can remain in the trial and receive integrative medicine services. The use of integrative medicine services is documented.
Protocol changes related to eligibility or any significant change in study procedures are reported to the study funder, the HDFCCC Site Committee, the IRB, and updated on clinicaltrials.gov.
Study results will first be presented to our stakeholders for evaluation and discussion. After incorporating stakeholder feedback, final study results related to primary and secondary outcomes will be published in peer-reviewed journals and presented at one or more scientific conferences.
Confidentiality
At recruitment, personal information is collected through secure e-mails (to get treating health-care provider permission to approach). Enrollment tracking is recorded using HIPAA compliant software or stored in HIPAA compliant secure folders. No names or personal identifiers are available during the analysis phase.
Access to Data
Access to the data involves a formal request including an abstract, proposed aims, background, outcomes, analysis plan, and investigator qualifications. Upon approval a complete, cleaned, and de-identified copy of the final dataset will be provided. Data sharing will be coordinated to avoid overlapping analyses.
Discussion
The results of the ACT-CC trial will provide both feasibility and effectiveness data so that Hematology-Oncology and HSCT treatment centers can assess the logistics, risks, and benefits of integrating acupressure into symptom management along with usual care in inpatient and outpatient settings treating childhood cancer or providing a chemotherapy-based HSCT. To our knowledge, this study represents the largest study employing a semistandardized daily acupressure intervention for pediatric oncology and HSCT-patients.
In 2002, the NIH convened a 14-member multidisciplinary expert panel to examine symptom management dilemmas for patients with cancer.157,158 The panel stated that all patients should have optimal symptom control throughout the course of their illness and suggested that pain, depression, and fatigue are inadequately treated in most cancer patients. Additional data show that effective management of nausea and vomiting in children remains challenging.3,96 This trial will provide data on an integrative medicine approach to improve symptom management.
Although the trail has employed rigorous methodology, nonetheless, it has limitations. A sample size of 85 may make it harder to detect differences between groups for pain. It is likely to be difficult to determine the effectiveness of the intervention between subgroups such as those receiving inpatient versus outpatient care, hematology-oncology versus HSCT, by age, or by diagnosis. A future larger multisite trial would be important to increase the sample size so that the effectiveness in subgroups could be explored in more depth.
Trail Status
The trial is currently active enrolling and was opened in October 2017. No posttrial care is required.
Footnotes
Appendix 1
Author Contributions
EAL drafted the manuscript. All remaining authors reviewed and edited versions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by: Patient Centered Outcomes Research Institute (PCORI) R-1602-34557 (Lown); Pierre’s Birthday Fund for acupressure delivery; National Cancer Institute, P30CA082103 (UCSF Helen Diller Family Comprehensive Cancer Center); and UCSF Clinical & Translational Science Institute (CTSI) UL1 TR991872. The scientific direction and publication is determined by the investigator team.
