Abstract
Background:
Spinal anaesthesia is a common anaesthetic technique for caesarean sections. Neuraxial opioids such as intrathecal morphine may cause common adverse effects (pruritus, post-operative nausea and vomiting).
Objectives:
We investigated the incidence, severity and need for treatment of pruritus and post-operative nausea and vomiting following administration of intrathecal morphine in spinal anaesthesia for caesarean section at KK Women’s and Children’s Hospital, Singapore.
Methods:
We conducted a prospective study involving 124 parturients who received intrathecal morphine in spinal anaesthesia for caesarean section from October 2012 to October 2014.
Results:
Seventy patients (56.5%) had moderate or severe pruritus (score 4–10), while 54 patients (43.5%) had no or mild pruritus (score 0–3). Mean (SD) value of the worst pruritus score reported on a scale of 0–10 was 4 (2.59). Only seven out of the 124 patients (5.6%) required treatment for pruritus. With respect to distress and bother from itching in the past 24 hours on a score of 0–4, the mean score reported was 1.7 (1.23). Fourteen (11.2%) patients reported vomiting, dry-retching and nausea. Six (4.8%) patients had nausea that interfered with activities of daily living. Four (3.2%) patients had clinically significant post-operative nausea and vomiting. Seven (5.6%) patients received anti-emetics. The average Overall Benefit of Analgesia Score was 3.8 (SD 2.6, min–max: 0–15). The average (SD) maternal satisfaction with pain relief and side effects was 84.9% (9.9%).
Conclusion:
There is a high incidence of pruritus, with most women reporting moderate to severe pruritus. The incidence of post-operative nausea and vomiting is low, and women reported good maternal satisfaction.
Keywords
Introduction
Spinal anaesthesia is a common anaesthetic technique for caesarean section. In KK Women’s and Children’s Hospital, and many other centres, patients receiving spinal anaesthesia for caesarean sections are given intrathecal morphine 100 μg and intrathecal fentanyl 15 μg, in addition to hyperbaric bupivacaine, to provide effective and sustained postoperative analgesia. However, pruritus and post-operative nausea and vomiting (PONV) are common and troublesome side effects after intrathecal morphine. This can lead to maternal dissatisfaction and distress, adversely affecting the childbirth experience.
Previous studies on opioid adverse effects following neuraxial anaesthesia found the incidence of pruritus to be1–9 30–100% and incidence of PONV to be3,4,9– 13 22–67% after intrathecal morphine. Patients with severe pruritus and PONV may require treatment, and these adverse effects may result in poorer maternal satisfaction with their pain therapy. We investigated the incidence, severity and need for treatment of pruritus and PONV following administration of intrathecal morphine in spinal anaesthesia for caesarean sections at KK Women’s and Children’s Hospital.
Methods
We conducted a prospective study involving 124 parturients who received intrathecal morphine in spinal anaesthesia for caesarean section, over 2 years from October 2012 to October 2014 in KK Women’s and Children’s Hospital, Singapore. Singhealth Centralised Institutional Review Board (CIRB approval number: 2012/820/D) approval was obtained. Inclusion criteria included elective caesarean section under spinal anaesthesia, with administration of intrathecal morphine, aged 21–50 years old and American Society of Anesthesiologists (ASA) 1 and 2 patients. Exclusion criteria included patients with contraindication to spinal anaesthesia, drug allergy to opioid or contraindication to the use of intrathecal morphine, e.g. severe obstructive sleep apnoea.
Informed consent was obtained from all subjects for post-operative review 24 hours after intrathecal morphine administration in spinal anaesthesia for caesarean section. A standardised survey form was used, and data was collected on the incidence and severity of pruritus and PONV. The presence and site of pruritus, as well as the patient’s assessment of her distress from the pruritus, was obtained at the review. The primary obstetric team decided on the need and type of treatment for PONV and pruritus as well as post-operative analgesia at the clinician’s own discretion.
A patient is considered to have PONV if she experiences nausea, vomiting or retching post-operatively. The incidence and severity of PONV was scored using the PONV Intensity Scale 14 at 24 hours after surgery. A PONV Intensity Scale of ≥ 50 indicates clinically significant PONV. Data for the need for treatment for PONV and the Overall Benefit of Analgesia Score (OBAS) were also collected. OBAS is a validated scoring system to assess pain intensity and opioid-related adverse effects. 15 A low OBAS (min–max: 0–28) indicates high benefit. Patients were also asked to give a satisfaction score (ranging from 0% meaning very unsatisfied to 100% meaning very satisfied) for pain relief and side effects. The primary outcome was incidence of PONV at 24 h post-operatively and was treated as dichotomous data.
Post-operative well-being was also assessed using the Quality of Recovery (QoR) score, which ranges from a total score of 0 to 18, with higher score indicating better quality of recovery.
Categorical variables were presented as frequencies with corresponding proportions. Continuous variables were presented as medians, i.e. interquartile range (IQR), or mean (± standard deviation (SD)). Data analysis was performed using SPSS for Windows 18.0. We investigated whether patients with pruritus also had higher incidence of PONV. The study required a sample size of 124 based upon a difference of at least 20% in PONV between no or mild and moderate or severe pruritus groups based on the following assumptions: incidence of PONV as 20% and 45% in no or mild pruritus group and moderate or severe pruritus group, respectively, from department database; we used a level of significance of α = 0.05, allocation ratio of 1:1.
Results
A total of 150 patients was screened in this study and 124 patients gave informed consent to participate in the study. There were no withdrawals or dropouts. All the patients received spinal anaesthesia for caesarean section and completed all the surveys. The baseline characteristics of the participants are shown in Table 1. Of note, the body mass index of the patients prior to surgery was 29.974 (5.2026) mean (SD).
Baseline characteristics of study participants.

ASA: American Society of Anesthesiologists; IQR: Interquartile range; LSCS: lower segment caesarean section; SD, standard deviation.
Seventy patients (56.5%) had moderate or severe pruritus (score 4–10), while 54 patients (43.5%) had no or mild pruritus (score 0–3). The mean value of the worst pruritus score reported on a scale of 0–10 was 4 (2.59). With respect to the site of pruritus (see Figure 1), 51 patients (41.1%) reported itch over the face, 36 patients (29.0%) reported itch over the neck, 41 patients (33.1%) reported itch over the upper limbs, 31 patients (25.0%) reported itch over the lower limbs, 57 patients (46.0%) reported itch over the chest, 52 patients (41.9%) reported itch over the abdomen and 59 patients (47.6%) reported itch over the back. Only 7 out of the 124 patients (5.6%) required treatment for pruritus. With respect to distress and bother from itching in the past 24 hours on a score of 0–4, the mean score reported was 1.7 (1.23). With respect to distress and bother from itching in the 24 hours post-administration of intrathecal morphine, patients with moderate or severe pruritus reported a mean score of 2.5 out of 4 (SD 0.88) compared with patients with no or mild pruritus who reported a mean score of 0.7 (SD 0.86). This difference in distress and bother from itching based on the severity of pruritus was significant, with a
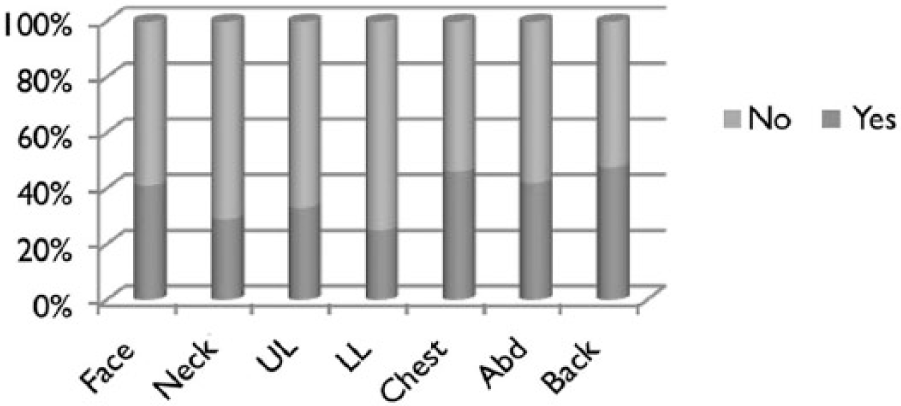
Reported sites of pruritus.
Incidences of vomiting, dry-retching or nausea
Fourteen (11.2%) patients reported vomiting, dry-retching or nausea (see Figure 2). Out of 11 patients who experienced nausea, 10 reported that nausea was intermittent in nature. Only 1 of the 11 reported that nausea was constant. Six (4.8%) patients had nausea that interfered with activities of daily living. The average duration of nausea was 2.6 h (minimum of 1 h, maximum of 6 h). Four (3.2%) patients had clinically significant PONV. Seven (5.6%) patients received anti-emetics. The average OBAS score was 3.8 (SD 2.6, min–max: 0–15). The average (SD) maternal satisfaction with pain relief and side effects was 84.9% (SD 9.9%). However, there is no significant difference in maternal satisfaction with pain relief and side effects regardless of the severity of pruritus. Patients with no or mild pruritus had a mean maternal satisfaction score (scored from 0 to 100%) of 85.6% (SD 9.81) and a median score of 90% (IQR 10.0). Those with moderate or severe pruritus had a mean maternal satisfaction score of 84.3% (SD 10.05) and a median score 85.0 (IQR 10.0). There was no statistically significant difference when the mean and median scores of both groups were compared, with

Incidence of vomiting, retching or nausea.
As post-operative analgesia given by the primary obstetric team could affect the incidence of pruritus and PONV, we collected data on the type of analgesia received by the parturients and their incidence of pruritus and PONV. Only one patient received morphine post-operatively. She had a pruritus score of 5 (average pruritus score in study group is 4.0) but did not require treatment for it. She did not have PONV. Four patients received tramadol, but did not experience any PONV, 117 patients received paracetamol and 114 patients received either mefenamic acid or naproxen.
In terms of OBAS score (from 0 to 28, lower score indicates higher benefit and lower side effects from opioid analgesia), the no- or mild-pruritus group has a mean score of 2.8 (SD 2.02) and a median score of 2.0 (IQR 3.0), which is significantly lower (
There was no difference in the incidence of PONV between the two pruritus groups (no or mild pruritus and moderate or severe pruritus groups). There was no statistically significant difference in the percentage of patients having vomiting, dry-retching or nausea regardless of the severity of pruritus. Two (3.7%) patients in the no- or mild-pruritus group vomited or had dry-retching, compared with eight (11.4%) patients in the moderate or severe pruritus group (
Discussion
In this study, most of the 70 patients (56.5%) had moderate or severe pruritus (score 4–10). There is a high incidence of pruritus, with most women having moderate-to-severe pruritus. The high incidence of pruritus following intrathecal morphine administration is similar to incidences reported previously.1–8
More patients complained of itch over the face, chest, back and abdomen, compared with the neck, upper and lower limbs (see Figure 1). This is in contrast to the findings of a previous study by Warwick et al. that found that all patients who experienced pruritus itched on the face, with the arms, legs and trunk less affected. 6 The need for treatment of pruritus is low, with only 7 out of 24 patients (5.6%) requiring treatment. This is in contrast to the relatively high number of patients treated for itch: 64% in the study by Warwick et al. 6 It should be noted that the dose of intrathecal morphine used in the latter study was 200 μg during spinal anaesthesia for caesarean section, while our patients received intrathecal morphine 100 μg. In a double-blind, randomised study by Girgin et al. 16 on 100 ASA physical status I and II term parturients undergoing caesarean delivery with spinal anaesthesia, where patients were randomised to one of five groups to receive 0.0, 0.1, 0.2, 0.3 or 0.4 mg intrathecal morphine in addition to low-dose (7.5 mg) heavy bupivacaine, they found that the dose of 0.1 mg intrathecal morphine produces analgesia comparable with doses as high as 0.4 mg, with significantly less pruritus when combined with low-dose bupivacaine. A meta-analysis by Gehling and Tryba. 17 found similarly that higher doses of 0.3 mg or more intrathecal morphine were found to increase the incidence of pruritus (risk ratio (RR) of pruritus with intrathecal morphine doses of 0.3mg or more; RR≥ 0.3 mg 5.0, 95% confidence interval (CI) 2.9–8.6) compared with doses lower than 0.3 mg (RR< 0.3 mg 1.8, 95% CI 1.4–2.2).
In another prospective, randomised and double-blind study involving 58 parturients receiving elective caesarean section under spinal anaesthesia, Yang et al.
18
compared intrathecal morphine 0.1 or 0.25 mg, and also found that 0.1 mg intrathecal morphine provided satisfactory postoperative analgesia following caesarean section when compared with 0.25 mg. This was associated with lower mean visual analogue scale pruritus scores and lower need for treatment of pruritus with nalbuphine (4/30) than in the 0.25 mg group (12/28), with
A recent meta-analysis by Sultan et al.
19
involving 480 patients from 11 randomised-controlled trials compared the effect of low dose (0.05–0.1 mg) with high dose (>0.1–0.25 mg) intrathecal morphine and found that the incidence of nausea or vomiting (odds ratio (OR) 0.44 [95% CI 0.27–0.73];
However, there have also been studies that found no statistically significant difference in treatment rate of pruritus between doses of 100 or 200 μg of intrathecal morphine for caesarean delivery. A retrospective chart review by Wong et al.
20
of 241 patients who had an elective caesarean delivery and received either 100 or 200 μg of intrathecal morphine found that 17% of patients required treatment with nalbuphine in the former group, whereas the treatment rate was 22% in the latter group. The difference was not statistically significant with
The effect of ondansetron on our rates of pruritus is debatable as there have been conflicting results from previous studies. Some studies had showed no effect of ondansetron when administered for either prevention or treatment of pruritus caused by intrathecal morphine after caesarean delivery.21–23 However, a systematic review and meta-analysis by George et al. 24 on the use of serotonin receptor antagonists for the prevention and treatment of pruritus, nausea, and vomiting in women undergoing caesarean delivery with intrathecal morphine found that the incidence of pruritus was not reduced with 5-HT3 receptor antagonists prophylaxis compared with placebo (80.7% versus 85.8%, relative risk (RR) [95% CI] = 0.94 [0.81–1.09]). However, their use reduced the incidence of severe pruritus and the need for treatment of pruritus (number needed to treat = 12 and 15, respectively).
The audit also shows that the incidence of PONV in our hospital appears to be lower than published average rates3,4,9–13 of 22–67% with good maternal satisfaction. Fourteen (11.2%) patients reported vomiting, dry-retching or nausea, with most (10/11) reporting nausea varying in nature; only one patient reported constant nausea. Only four (3.2%) patients had clinically significant PONV based on the PONV Intensity Scale, 14 and the rate of rescue treatment required was low, with only 7 (5.6%) patients requiring anti-emetics. We can conclude that most patients in our institution did not have clinically significant PONV requiring treatment. Owing to the nature of the audit, one limitation could be that several risk factors for PONV, such as smoking and previous history of PONV, were not collected.
In conclusion, there is a high incidence of pruritus after administration of intrathecal morphine in spinal anaesthesia for caesarean section. The severity of pruritus is associated with the degree of distress and bother from itching, the OBAS and QoR scores. However, there is no significant difference in reported maternal satisfaction and incidence of vomiting, dry-retching or nausea with respect to the degree of pruritus. Overall, the need for treatment of pruritus is low. This study allowed us to better understand and advise patients undergoing caesarean sections under spinal anaesthesia and improve their childbirth experience. The findings will also be useful in further studies to evaluate effectiveness of medications for the prevention and treatment of pruritus and PONV in our local obstetric population. It can serve as the groundwork for further studies to identify risk factors for pruritus and PONV in our local obstetric population, so that preventive and therapeutic guidelines can be instituted.
Footnotes
Declaration of Conflicting Interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Availability of Data and Materials
Participant’s data and statistical analysis are available upon request to the corresponding author.
Informed Consent
Written informed consents were obtained from all participants involved in the study.
Ethical Approval
Singhealth Centralised Institutional Review Board (CIRB approval number: 2012/820/D) approval was obtained.
