Abstract
To evaluate the benefits of cognitive training via CogniFit® in adults over 60 years and its effects on quality of life, insomnia, depression, and daily memory failure. Quasi-experimental pretest–posttest study with a control group. Seventy-four participants were recruited, and 45 completed the study (23 intervention, 22 control). Data were collected at baseline and 8 weeks post-intervention. The intervention involved 12 weeks of CogniFit® cognitive training (three 20–30-min sessions per week). Instruments included the EuroQol EQ-5D-3L, Oviedo Sleep Questionnaire, GDS-15, Memory Failures of Everyday Questionnaire, MoCA, and CogniFit® battery. Analyses were conducted using chi-square, t, Mann–Whitney U, Wilcoxon, and Spearman tests (p < .05). The intervention group showed significant improvements in global cognition (CogniFit® and MoCA). While memory failures did not improve, deterioration was prevented compared to the control group. No significant changes were found in depression, sleep, or quality of life. CogniFit® training may enhance cognitive function in adults over 60 and help prevent cognitive decline. The program mitigated memory deterioration observed in controls. Participants engaged in 12 weeks of cognitive training to promote cognitive health in older adults. The nonrandomized design, small sample, and self-reported measures limit internal validity. Nonetheless, findings support the utility of digital cognitive training as an accessible, nonpharmacological strategy to foster cognitive health and active aging.
Background
According to the World Health Organization (WHO), the population worldwide has an average life expectancy equal to or greater than 60 years (WHO, n.d.). The proportion of elderly people is increasing in most countries, generating an aging population, and it is estimated that in 2050, the number of people older than 60 years will more than double that in 2020, from 1 billion to 2.1 billion worldwide. In Spain, the country in which the study was conducted, in 2021, 24.78% of the population was over 60 years of age, according to data from the National Institute of Statistics (INEbase, n.d.).
The normal aging process entails age-related neurocognitive changes, such as changes in the speed of information processing, working memory, and executive cognitive function. Some diseases associated with age also affect the rate of neural dysfunction, neural loss, and cognitive impairment, all of which increase the loss of these abilities (Harada et al., 2013; Murman, 2015). Cognitive abilities progressively decrease beginning in adulthood and not only when individuals reach old age (Sabbagh et al., 2022; Tapia et al., 2022).
According to the WHO (2025), approximately 55 million people live with dementia globally, with nearly 10 million new cases each year. Cognitive decline, a precursor to dementia, compromises autonomy, increases dependency, and negatively affects emotional well-being and quality of life. These outcomes highlight the urgent need for early, nonpharmacological interventions. A recent meta-analysis revealed that mild cognitive impairment, a preclinical stage between normal aging and dementia, affects approximately 15% to 23% of adults aged ≥50 years living in the community (Bai et al., 2022). In Spain, representative epidemiological studies report a prevalence of 9.6% among individuals aged ≥50, with higher rates observed in women and older age groups (Lara et al., 2016). Mild cognitive impairment has a tangible impact on daily life: it impairs memory and executive functioning without compromising independence, but it increases the effort required for routine tasks and predicts progression to dementia in up to 46% of cases within 3 years (Pais et al., 2020). Consequently, the high prevalence and functional consequences of early cognitive decline justify the implementation of preventive strategies, such as cognitive training, to delay deterioration and preserve autonomy.
The worsening of cognitive skills caused by aging affects daily life memory failure and is an even stronger predictor of mood and perceived health than actual memory performance (Montejo, Montenegro, Fernández-Blázquez, et al., 2014). These memory failures can be improved through cognitive training, promoting the health and well-being of elderly individuals (Montenegro-Peña et al., 2025).
Following this, cognitive impairment can be prevented through lifestyle changes. At least 40% of dementia cases are associated with modifiable causes (Coley et al., 2022; Sabbagh et al., 2022). Cognitive deterioration can be prevented through the management of cardiovascular risk, physical activity, sleep, nutrition, social activity, and cognitive stimulation. In the aging process, memory failure, including the cognitive skill known as working memory, affects cognition, which is why working memory is used in tests of attention and problem solving (Ma & Chan, 2020; Wilson et al., 2013). Additionally, cognitive impairment is related to physical frailty and increased disability in old individuals, which leads to a loss of independence in performing activities of daily living and significantly lowers the quality of life of elderly people and their sense of well-being (Ma & Chan, 2020; Wilson et al., 2013).
Through one of the most widely used questionnaires to measure quality of life, the EuroQol (EQ-5D), cognitive impairment has been correlated with quality of life and depressive states (S. Lee & Ho Chung, 2022). Given the need to prevent and address cognitive decline, we find that, according to the systematic review carried out by Nguyen et al. on commercially available cognitive training programs for the general population for the prevention of cognitive decline (Nguyen et al., 2022), the only tools that yielded significant results from the seven included studies were BrainHQ and CogniFit®. In fact, the CogniFit® tool has been shown in several studies to improve cognitive abilities such as working memory, processing speed, and decision-making (Gigle et al., 2013; Nguyen et al., 2022; Peretz et al., 2011). Training with the CogniFit® program has shown improvements that are maintained over time and are reflected in the quality of life of elderly people (Shatil, 2013). Sleep is a mediator between depression and cognitive decline (Liu et al., 2022). The quality of sleep can be improved through continuous learning programs such as CogniFit® (Haimov & Shatil, 2013), thus minimizing the risk of dementia (Bahar-Fuchs et al., 2020; Sanchez-Castañeda et al., 2021).
Cognitive training has emerged as a promising strategy to maintain or improve cognitive performance in older adults, and increasing evidence suggests that such interventions may delay or reduce the risk of dementia by targeting modifiable risk factors in its early stages. Therefore, nursing professionals must be aware of the advantages of cognitive training for elderly people since they are responsible for promoting actions aimed at improving the cognitive activity of the people they care for, especially those working in primary-care positions and centers where more actions can be carried out and, by the continuity of care, can achieve early detection and prevention of cognitive deterioration and its associated factors (Arora et al., 2023; Hayajneh et al., 2020; J. Lee et al., 2020).
As mentioned above, previous studies have reported the benefits of cognitive training programs through CogniFit, which is one of the tools that yields the best results in the cognitive sphere. These cognitive benefits are related to different health parameters, such as depression and quality of life. No similar studies have been identified in the literature, no studies have investigated whether this improvement applies to memory failure in everyday life, and no known studies have been carried out in Spain to determine the benefits of this training program on the cognitive sphere in older adults.
The main objective of this study was to evaluate the benefits of cognitive training via CogniFit® in improving nine cognitive abilities frequently affected in old age, namely, phonological short-term memory, visual short-term memory, naming, working memory, visual perception, spatial perception, processing speed, hand–eye coordination, and response time, in adults over 60 years of age. The secondary objectives were to determine how cognitive training influences quality of life, insomnia, depression, and especially memory failure in everyday life.
Therefore, the following hypotheses are proposed:
(
(
Methods
Design
A quasiexperimental pretest with a control group was carried out from October 2022 to May 2023. The present study follows the TREND Statement recommendations for nonrandomized studies (TREND Statement | CDC, n.d.).
The quasiexperimental design was chosen because of the practical and ethical impossibility of randomly assigning participants to the experimental and control groups. In studies with an older adult population, random assignments may be limited by the availability, willingness or clinical conditions of the participants, which could compromise the external validity of the study if strict randomization is attempted.
The quasiexperimental design allows the effect of an intervention (in this case, cognitive training with CogniFit®) to be evaluated under more natural conditions representative of the real context while maintaining some control over the variables through the comparison between preestablished groups and the use of pre- and postintervention measures.
Sample/Participants
Participants were selected through nonprobabilistic convenience sampling from neighborhood associations, senior organizations and advertisements for elderly individuals through informative talks, information brochures, and referrals. Eligible individuals were those aged 60 years or older, with no diagnosed neurocognitive disorders and who agreed to participate voluntarily.
Owing to ethical and logistical considerations, random assignments to groups were not feasible, and participants were nonrandomly allocated to the intervention or control group. Therefore, participants were allocated to the experimental or control group on the basis of their availability and interest in participating in the cognitive training program. This approach is consistent with the quasiexperimental design of the study.
The inclusion criteria were as follows:
- Over 60 years of age.
- Total autonomy for the basic and instrumental activities of daily life.
The exclusion criteria were as follows:
- Cognitive impairment and/or dependence.
- Significant audiovisual or auditory disabilities prevent the use of electronic devices, which are necessary to carry out assessments and/or exercises.
- Major medical or neurological disease.
To calculate the size of the sample, the study by Gigle et al. (2013) was referenced, where general memory was improved by 0.3 points through cognitive training with CogniFit®.
The sample size was calculated with the GRANMO program (version 7.12, April 2012) for paired means. With an alpha risk of .05 and a beta risk of .2 taken in bilateral contrast, 12 subjects were needed in each group to detect a difference equal to or greater than 0.3 units. A standard deviation of 0.35 was assumed. A loss to follow-up rate of 10% was estimated.
The final sample consisted of 45 participants older than 60 years, 23 in the intervention group and 22 in the control group. All the participants could use the tablet to perform the cognitive assessments/exercises of CogniFit®. The participants were consecutively assigned to the intervention group or the control group.
Intervention
The participants in the intervention group participated in a personalized cognitive training program at home through 36 sessions on an electronic device for 12 weeks (three sessions per week), each session lasting 20 to 30 min, through the CogniFit® app. The CogniFit® cognitive training program (CogniFit Ltd., New York, USA) is a personalized cognitive training program carried out through an app that has been validated in various populations (Haimov & Shatil, 2013; Nguyen et al., 2022; Shatil, 2013; Yaneva et al., 2022). The test begins with a general cognitive assessment, the CogniFit computerized neurocognitive assessment, whose results determine the individual level of training each participant will receive. The difficulty of the exercises adapts automatically to elderly people’s practices and training. The wide variety of activities that make up this multidimensional resource has been fine-tuned to continuously measure user performance and automatically regulate the complexity and typology of the tasks. The different types of neurological stimulation and rehabilitation exercises that make up this complete battery can be performed by any computer or mobile device. Because the program is tailored to each person’s strengths and weaknesses, it is unlikely that two participants will receive the same training regimen. The CogniFit® training program for this study involved the training of nine cognitive abilities commonly affected in old age, namely, phonological short-term memory, visual short-term memory, naming, working memory, visual perception, spatial perception, processing speed, hand–eye coordination, and response time. The challenge level is readjusted after each training session according to the progress of the participant in the tasks (CogniFit, n.d.). The participants were allowed to choose the timing of their training sessions to enhance adherence and accommodate personal routines, as the CogniFit® platform offers flexibility in scheduling. The program automatically records user activity, including the completion of each session, duration, and performance.
Variables
The following independent variables were collected:
- Sociodemographic variables: age, gender, marital status, educational level, and children.
- The health-related variables included blood pressure and heart rate (objective physiological indicator), manual grip strength (indicator of physical function), health-related quality of life (measured through the EuroQol-5D health questionnaire; Herdman et al., 2001), sleep satisfaction (measured through the Oviedo Sleep questionnaire (COS); García et al., 2000), and depression (measured through the Yesavage Geriatric Depression Scale; Martínez de la Iglesia et al., 2002).
The following dependent variables were also analyzed:
- Memory failure was measured through the Memory Failures of Everyday Questionnaire (Sunderland et al., 1984).
- Cognitive ability was assessed through the Montreal Cognitive Assessment (MoCA; Delgado et al., 2019).
- Cognitive evaluation was performed through CogniFit®.
Instruments
- Sociodemographic variables questionnaire: self-completed questionnaire for the collection of sociodemographic data.
- The 5-dimensional, 3-level EuroQol questionnaire (EQ-5D-3 L) was used to assess health-related quality of life (Herdman et al., 2001). The questionnaire consists of a series of questions for the evaluation of five dimensions: mobility, self-care, habitual activities, pain/discomfort, and anxiety/depression. It also includes a visual analog scale (VAS) known as the EQ-VAS. A lower score on the scale and questionnaire represents a worse state of participants’ perceived health status, whereas a higher score indicates a better state of participants’ perceived health status. The authors reported Cronbach’s α values between 0.72 and 0.83 in residents with cognitive impairment (Pérez-Ros & Martínez-Arnau, 2020), and in the present study, the Cronbach’s α value was 0.511.
- The Oviedo Sleep Questionnaire (COS; García et al., 2000) consists of 15 items in three subscales: subjective satisfaction with sleep (one item), insomnia (nine items), and hypersomnia (three items). The two remaining items provide information on the use of sleep aids or the presence of adverse phenomena during sleep. The insomnia subscale ranges from 9 to 45, where a higher score corresponds to a greater severity of insomnia. Therefore, it provides a subjective assessment of satisfaction with sleep and indicates the presence of insomnia and hypersomnia. Previous studies in clinical populations have shown that it has high reliability, with α = .90 overall, α = .91 for the insomnia dimension, and α = .88 for hypersomnia (Paz García-Portilla et al., 2009); in the present study, the alpha value was .690.
- The GDS-15 (Martínez de la Iglesia et al., 2002) consists of 15 yes/no questions, in which each item is scored 0 or 1, depending on the answer. A score of 0 to 4 is considered normal, a score of 5 to 12 points is considered moderate depression, and a score of more than 12 points is considered severe depression. In Spanish primary care, the GDS-15 has demonstrated acceptable internal consistency, with a Cronbach’s α = .82 in individuals aged ≥65 years (Fernández-San Martín et al., 2002), and in the current study, the alpha coefficient was .492. The Memory Failures of Everyday (MFE) questionnaire is a 28-item scale used to assess memory failure in everyday life (Sunderland et al., 1984). The categories of forgetfulness collected were “speaking, reading and writing,”“names and faces,”“actions,” and “learning new things.” The questionnaire is answered via a Likert-type scale with nine response options ranging from “not once in the last 3 months” to “more than once a day.” Given the difficulty of choosing between nine answer options, in Spain, we use the three-option version (Montejo, Montenegro, Sueiro, & Huertas, 2014). The lowest values of the questionnaire represented the least amount of forgetfulness, and the highest values represented the greatest amount of forgetfulness. It has good internal consistency, with a Cronbach’s alpha of 0.83 (Montejo Carrasco et al., 2012), and in the present study, the Cronbach’s alpha was .913 for the Montreal Cognitive Assessment (MoCA; Delgado et al., 2019). This instrument consists of 30 items that examine the following skills: attention, concentration, executive functions (including the capacity for abstraction), memory, language, visuoconstructive capacities, calculation, and orientation. The administration time required was approximately 10 min. The maximum score was 30; a score equal to or greater than 26 was considered normal. The Montreal Cognitive Assessment (MoCA) tool has been used to evaluate early cognitive impairment, and its initial use has shown high validity and diagnostic precision in older people with regard to assessing abnormal cognitive performance (Bernier et al., 2023). In Spanish clinical samples, the MoCA-S displayed acceptable to good internal consistency, with Cronbach’s alpha values ranging from .702 to .822 (Gomez-Moreno et al., 2022).
- CogniFit® computerized neurocognitive assessment battery. In addition to the global score, 22 cognitive indicators, such as working memory, divided attention, processing speed, visual scanning, contextual memory, and hand–eye coordination, were used. The CogniFit® application used in our study was indeed available in the participants’ native language (Spanish). The CogniFit® evaluation battery has been validated and has good internal consistency (Cronbach’s alpha = .85–.88) and test–retest reliability (r = 0.69–0.92; Researchers Assessment Batteries Reliability and Validity Description of CogniFit Assessment Batteries. CogniFit Assessment Battery CogniFit©, 2022); in the current study, the Cronbach’s alpha was .930.
Procedure
A questionnaire was designed that included different tests and was accompanied by an information sheet and an informed consent form. Once a participant had been recruited, the study design was explained. At the beginning of the study, all participants filled out the questionnaire, which included the abovementioned instruments, and participants completed the form on paper in the presence of the researchers with informed consent before beginning the intervention. In addition, before the start of the intervention and at 8 weeks after the end of the intervention, the cognitive performance of the participants was evaluated via the CogniFit® computerized neurocognitive assessment, which lasted 40 min, to evaluate the changes in preintervention and postintervention cognitive function. During the 12-week intervention period of the study, participants in the intervention group performed a cognitive training program, with at least three trainings per week, via the CogniFit® app. A member of the research group was available throughout the period to help install the CogniFit® app, solve computer problems, and address possible doubts. The participants in the control group did not receive any intervention. Eight weeks after the intervention, a new CogniFit® computerized neurological evaluation was carried out, and the questionnaire was filled out again. The intervention and participant recruitment started in October 2022 and finished in May 2023.
In addition, the possible carry-over effects of repetitive training were taken into account by trying to reduce their effect. Baseline assessments and posttraining assessments with a lag period (8 weeks after the intervention) were included to distinguish the immediate effects of practice from longer-term cognitive improvements. Comparison with the control group made it possible to compare changes in performance and to assess whether the improvements were due to training rather than repeated exposure to the tasks. Furthermore, the fact that the tool automatically readjusts the difficulty level according to the progress of the individualized training makes this difficult.
Statistical Analysis
For the statistical analysis, SPSS version 28 (IBM Corp. Armonk, NY, USA) was used. The qualitative variables are expressed as n (%). The quantitative variables are expressed as arithmetic means (m) and standard deviations (SD). An inferential analysis was performed to determine the relationships of the independent variables with the dependent variables. Pearson’s chi-square test of independence was applied to examine associations between categorical variables, and Student’s t test or the nonparametric Mann–Whitney U test was used if the data did not follow a normal distribution (tested through the Shapiro–Wilk test and the Levene test).
For the evaluation of the cognitive sphere, two measures were used:
- The CogniFit® Global Cognitive Score is an aggregate measure of the current cognitive strength of different cognitive abilities. It ranges from the minimum of 0 to the maximum of 800. The cognitive score provides a quick way to determine where the user is cognitively conscious and to help assess progress over time (CogniFit, n.d.).
- The specific score of each domain of the cognitive sphere of the CogniFit® questionnaire, with its mean and standard deviation at the beginning and after the intervention, was normalized (z scores) from the CogniFit® database. This measure has been used in most of the previous articles (Bahar-Fuchs et al., 2020; Embon-Magal et al., 2022; Gigle et al., 2013; Haimov & Shatil, 2013).
Differences in the cognitive scores were examined via repeated-measures tests between intervals via the Wilcoxon rank test because the data were not normally distributed. Spearman’s correlation coefficient (rho) was calculated to explore the relationships among the different variables studied (through the EQ-5D-3 L, COS, GDS-15, MFE, MoCA, and CogniFit questionnaires). To control for the influence of education level and age, a partial correlation was performed. All hypothesis tests were bilateral. In all the statistical tests, those with a confidence level of 95% (p < .05) were considered to be significantly different.
Results
The flow of participants throughout the study, including reasons for dropping out, is shown in Figure 1. Among the 60 participants who completed the initial evaluation, 45 (75%) returned to the final evaluation 8 weeks after completing the intervention. The completion rates were similar in both groups (76.6% in the intervention group vs. 73.33% in the control group). Therefore, the final sample consisted of 45 elderly people, the majority of whom were women (64.4%), married (53.3%), had completed university studies (44.4%), and had children (84.4%). Table 1 shows the sociodemographic characteristics of the 45 participants in the two groups; no statistically significant differences were found between the groups in the key variables before the intervention.

Flow chart of the participants.
Sociodemographic Characteristics of the Participants.
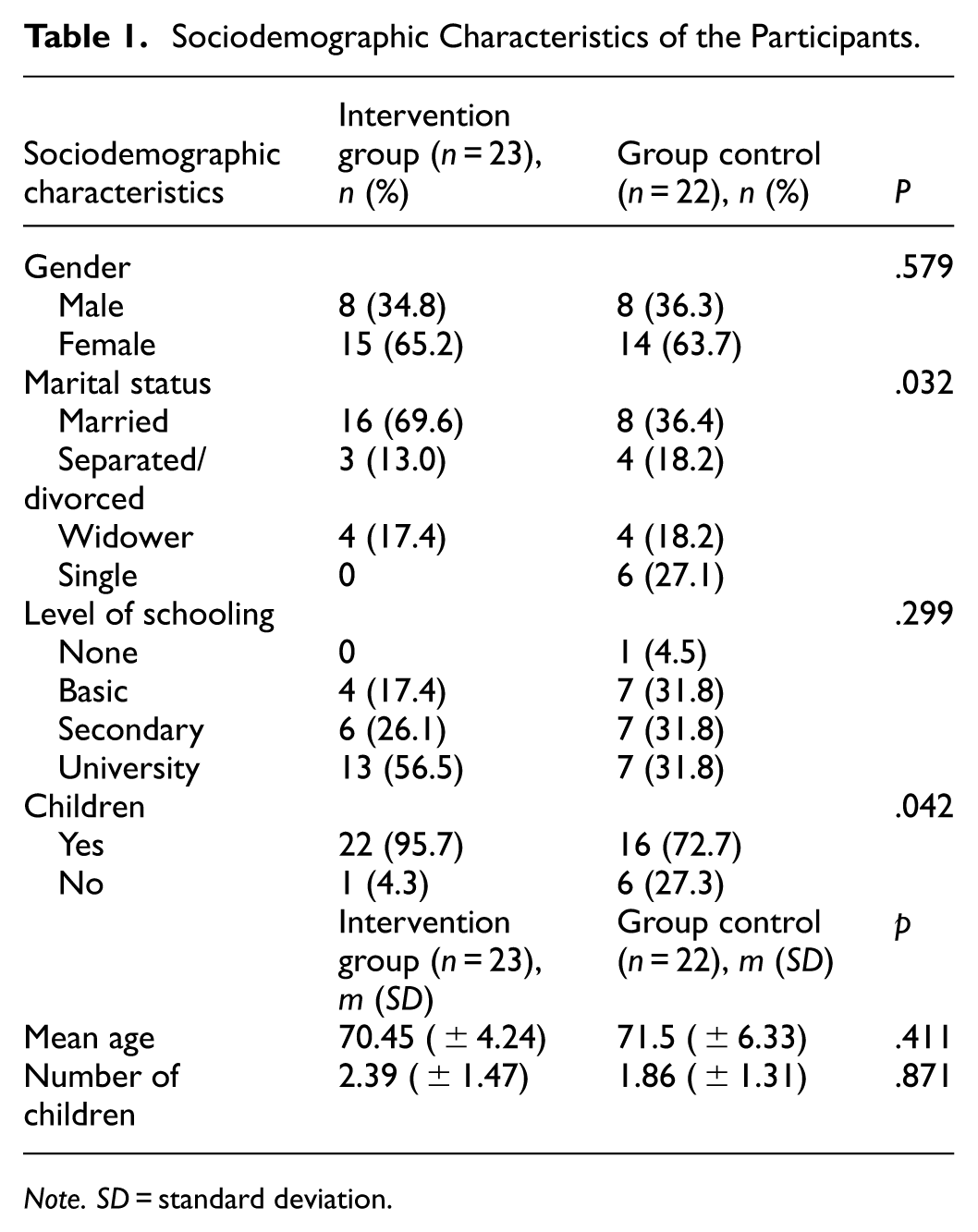
Note. SD = standard deviation.
Cognitive Outcomes
A significant difference was observed in the mean global cognitive score improvement by the CogniFit® assessment from the beginning to the end of the intervention, with differences of 132 points (SD ± 98.55) in the intervention group versus 18 points (SD ± 71.53) in the control group (p < .001; Figure 2). All participants in the intervention group performed at least 36 trainings throughout the 12 weeks of the intervention, with a mean of 46.87 sessions (±13.775).
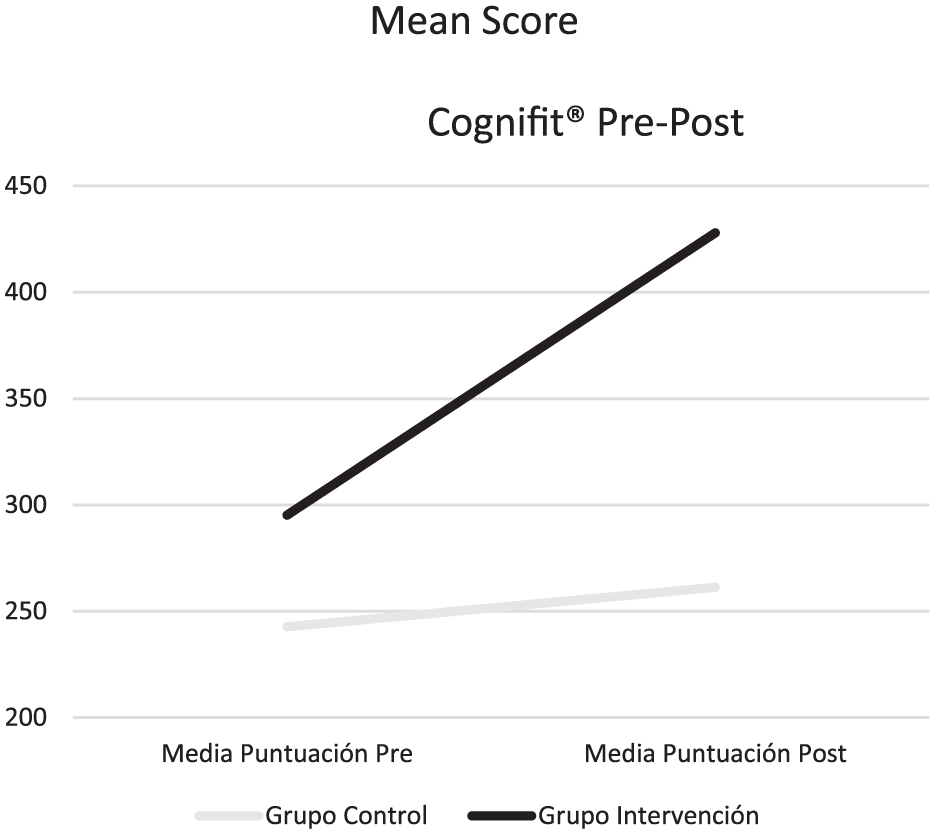
Mean difference in the CogniFit® Global Assessment Scale score from the beginning to the end of the intervention in the intervention group and the control group.
The specific score of each domain of the cognitive sphere (z score; Table 2) revealed that, between groups, significant differences were found for 13 domains of the cognitive sphere, improving in these domain intervention groups: phonological short-term memory (p = .032), visual short-term memory (p = .017), naming (p < .001), working memory (p = .007), visual perception (p < .001), hand–eye coordination (p = .005), response time (p < .001), contextual memory (p < .001), inhibition (p = .014), nonverbal memory (p = .002), planning (p = .020), short-term memory (p < .001), and monitoring (p < .001).
Results of the General CogniFit® Evaluation of the People in the Intervention and Control Groups Who Completed the Pre- and Posttests.

p < .05; **p < .001; ***p, t test for the difference in means between the control group and the intervention group for each of the components of the Cognifit® assessment.
The intervention group with cognitive training had significant improvements in 16 domains of the cognitive sphere: auditory perception (p < .001), contextual memory (p < .001), divided attention (p = .002), hand–eye coordination (p < .001), naming (p < .001), nonverbal memory (p < .001), phonological short-term memory (p = .004), planning (p = .012), recognition (p = .007), response time (p < .001), cognitive flexibility (p = .004), short-term memory (p = .002), monitoring (p < .001), visual perception (p < .001), visual short-term memory (p = .004), and working memory (p < .001). A significant effect was also observed within the control group in four domains: divided attention (p = .031), estimation (p = .022), focus (p = .033), and cognitive flexibility (p = .031). Significant differences in the MoCA score were also found between the groups (p = .044; Table 3).
Baseline and Posttraining Mean Differences in Montreal Cognitive Assessment (MoCA) Scores.

Note. SD = standard deviation.
Significant value of the paired Wilcoxon test.
p* differences between the intervention and control groups according to the Mann–Whitney U test.
Memory Failure and Health-Related Variables
The pre-post MFE score ranged from 11.9 points (SD ± 6.69) to 11.4 points (SD ± 6.7) in the intervention group (p = .336), whereas it ranged from 15.59 points (SD ± 7.12) to 18.68 points (SD ± 9.29) in the control group (p = .003; Figure 3).

Mean difference in the daily life memory failure score from the beginning to the end of the intervention according to group.
With respect to the variables related to health status (Table 4), there were significant differences only in heart rate (lower in the intervention group) and insomnia score; the control group had a decreased heart rate, which equates to a decreased severity of insomnia.
Pretest and Posttest Scores for the Variables Related to Health Status, Quality of Life, Insomnia and Depression.

Note. SD, standard deviation.
Significant value of the χ2 test.
Significant value of the paired Wilcoxon test.
p < .05; **p < .001; p***, significance for differences in means between the intervention and control groups for the different variables.
Correlations Between the Different Scales
Table 5 shows the correlations between the different scales in the posttest phase among all the participants. The general CogniFit® score was positively correlated with the number of trainings and the total MoCA score. However, it was negatively correlated with the total score on the MFE questionnaire, insomnia, quality of life, and depression. When we performed a partial correlation adjusted for age and schooling, the results were similar, although the correlation became stronger.
Correlations Between the General CogniFit® Evaluation Score, z Scores in the Different Domains, Number of Trainings with the CogniFit® app, MoCA Score, MFE Score, Insomnia, Quality of Life, and Depression Postintervention.

Note. The data are presented as the correlation coefficient rho. Adjusted for level of education and age. MoCA = Montreal cognitive assessment; MFE = memory failures of everyday.
p < .05; **p < .001.
Discussion
The intervention group improved their cognitive sphere both according to the general evaluation of CogniFit® and according to the MoCA, and they also improved their memory lapses in everyday life. Therefore, it seems that cognitive training through the CogniFit App® may be beneficial in improving specific domains of the cognitive sphere, as measured by the tools used in this study.
The training and evaluation of cognitive skills in older adults as a prevention of cognitive deterioration and/or part of active aging programs using the CogniFit® app also improved the cognitive skills under study compared with those of the control groups, which performed a different intervention (Embon-Magal et al., 2022; Gigle et al., 2013; Peretz et al., 2011; Shatil, 2013). Other studies have also shown improvements in sleep quality (Haimov & Shatil, 2013), in walking by sedentary elderly people (Verghese et al., 2010), and in the control and improvement of chronic diseases such as diabetes and dyslexia (Bloom et al., 2017; Bahar-Fuchs et al., 2020; Horowitz-Kraus & Breznitz, 2009).
All the studies concluded that through the use of cognitive training, the participants achieved significant improvements in the parameters under study, as in our study results (Bahar-Fuchs et al., 2020; Embon-Magal et al., 2022; Gigle et al., 2013; Haimov & Shatil, 2013; Horowitz-Kraus & Breznitz, 2009; Peretz et al., 2011; Shatil, 2013; Verghese et al., 2010). In addition, there are clinical trial protocols that propose the use of CogniFit® as a treatment or therapy for other pathologies, such as schizophrenia (Välimäki et al., 2021) and bipolar syndrome (Preiss et al., 2013), or in patients with intellectual disabilities (Siberski et al., 2015). Therefore, the benefits of this battery of individualized training go beyond cognitive improvements. Even so, relatively few studies exist on this topic and the care and treatments with which cognitive training is related.
Compared with the control group, the training with CogniFit group demonstrated improvements in several cognitive abilities. In addition, other digital cognitive training programs have also demonstrated positive outcomes in older adults. A study by Smith et al. (2009) using the BrainHQ platform revealed significant improvements in processing speed and memory among older adults after just 10 hr of training. Similarly, randomized controlled trials using Lumosity have reported modest but significant gains in cognitive flexibility and reasoning abilities (Hardy et al., 2015). These findings suggest that digital cognitive training, when properly structured and adapted, can be an effective tool for maintaining or enhancing cognitive functioning in aging populations.
We found that not only did the intervention group improve in score after the intervention, but the control group also improved. These improvements may be due to the memory questions in the CogniFit battery of questions and to the self-interest and initiative of the participants to work on these aspects individually in their day-to-day lives after the initial evaluation. Significant differences were detected in the CogniFit scores from before to after the intervention, as well as after the assessment with the MoCA, although there were differences between the results of the two instruments. This difference may be because the MoCA assesses eight cognitive abilities—executive, visual-constructive, attention, abstraction, memory, language, calculation, and orientation functions—in a survey that takes approximately 10 min and serves as a screening tool for the early detection of mild cognitive impairment (Delgado et al., 2019). The CogniFit tool measures 23 cognitive abilities and takes approximately 40 min per participant; it relates the score obtained with the cognitive capacity of each age group and allows comprehensive monitoring at the individual level (Yaneva et al., 2022). For this reason, while the MoCA is a useful tool for screening for cognitive impairment, the CogniFit tool is much more effective and complete in terms of tracking changes and progress at the cognitive level.
Memory failure became less common in the intervention group after training. In addition, this decrease in memory lapses was significantly correlated with a better overall score obtained with the CogniFit® and with the number of trainings performed. Other studies have shown a decrease in memory failure after individuals perform cognitive training (Heo & Park, 2021; J. Lee et al., 2023), although these improvements are more significant when specific types of memory training are added (Li et al., 2016).
With respect to the decrease in the pre- to postoperative scores of the intervention group in the MFE questionnaire, we reported findings similar to those of Montejo, Montenegro, Fernández-Blázquez, et al. (2014), Montejo, Montenegro, Sueiro, and Huertas (2014), and Montenegro-Peña et al. (2025), who reported significant relationships between the MFE questionnaire score and perceived health status and mood (mainly measured through depression and anxiety rating scales), revealing a positive correlation between subjective memory and perceived quality of life, in line with our results. Their study also revealed positive associations of memory failure, especially activity memory and communication memory failure, as well as complaints about one’s own memory, with anxiety and depression, which also coincides with our findings (Embon-Magal et al., 2022; Halloway et al., 2018).
With respect to sleep, a statistically significant negative correlation between insomnia score and the number of training sessions completed via the CogniFit® app was observed. This suggests that participants who completed more training sessions tended to report fewer symptoms of insomnia, according to the Oviedo Sleep Questionnaire. These results agree with those of Haimov and Shatil’s (2013) study on improving the quality of sleep in people with insomnia through cognitive training. Notably, the use of devices with electronic screens affects the conciliation and quality of sleep, and if cognitive training is carried out shortly before sleeping, it may not generate the desired effects on insomnia (Walsh et al., 2020). The improvement in the severity of insomnia in the control group may be due to the use of drugs not recorded during the intervention.
In addition, both groups deteriorated in quality of life, possibly because they were older and began to have more health problems, more frailty, and less personal satisfaction, resulting in a lower quality of life (Vinsalia & Handajani, 2021; Zúñiga et al., 2019).
Limitations
This study has several limitations. First, although both groups had similar characteristics, there was no randomization in the allocation of the groups. Similarly, at the beginning of the intervention, the mean CogniFit® score was greater in the intervention group than in the control group. Although the quasiexperimental design presents potential threats to internal validity, such as nonrandom selection or maturation effects, limitations were mitigated by the application of validated instruments, the use of appropriate statistical analyses, and the inclusion of a nonequivalent control group. In summary, the quasiexperimental design was the most appropriate for the objectives of the study, allowing for a rigorous and ethical evaluation of the intervention in a vulnerable population.
Another limitation could be the loss of some subjects after the first baseline evaluation, and the voluntary participation of subjects may represent bias in relation to the general population. The adaptive skills of the participants may have influenced the CogniFit® score, and a slight improvement was observed in the control group. However, our results are encouraging, and most studies have shown beneficial effects after the use of CogniFit®. On the other hand, several variables, such as sleep quality and quality of life, were self-reported by the participants, so there may be limitations in the data provided.
Another limitation of this study is the internal consistency reliability coefficients, such as Cronbach’s alpha, which can be affected by sample size and is not large enough to adequately calculate for the instruments used. Estimating Cronbach’s alpha requires a sufficiently large sample to provide stable and generalizable results. In small samples, the value of alpha can be biased or imprecise, potentially overestimating or underestimating the true internal consistency of a scale.
The study has several strengths, such as the inclusion of a representative sample, the demonstration of the benefits of the intervention for people older than 60 years of age, the adherence of participants in completing the intervention, the significant relationship of cognitive training with decreased memory failure in daily life, and the expansion of existing knowledge about cognitive training and, specifically, about the CogniFit® program, on which all the studies obtained significantly positive results. Another strength is the maintenance of the benefits of the intervention at least 8 weeks after the final evaluation.
Conclusions
Cognitive training through the CogniFit® program in people older than 60 years significantly improves skills and cognitive abilities and reduces memory failure, greatly influencing other spheres of life, for example, improving sleep and decreasing levels of depression. This study revealed the importance of carrying out cognitive training to improve and prevent memory failure.
It is important to highlight the importance of cognitive training as a component of active aging to maintain and improve individual capacities as individuals grow older. In addition, research into the benefits of cognitive training for older people should be further conducted. Future research should focus on improving randomization, examining long-term effects, including diverse populations, comparing multiple interventions, and utilizing more precise measurement tools to better assess the effectiveness and sustainability of cognitive training.
Implications for Health Care Practice
The results of this study suggest that cognitive training through digital platforms such as CogniFit® may constitute a complementary and effective tool in the comprehensive approach to cognitive aging. Its regular application was associated with improvements in key variables such as quality of life, sleep quality, depressive symptoms and daily memory function, which reinforces its value as a nonpharmacological intervention in older adults.
From a clinical perspective, these findings support the incorporation of structured cognitive stimulation programs in primary care, geriatric, psychology and neuropsychology settings. The digital nature of the intervention facilitates its remote or semipresential implementation, which is especially relevant for patients with reduced mobility or limited access to face-to-face services.
In addition, cognitive training could act as a protective factor against mild cognitive impairment by fostering cognitive reserve and promoting the maintenance of functional skills in daily life. Although it does not replace other clinical interventions, its inclusion in personalized treatment plans may improve the emotional well-being and functional autonomy of older adults.
Through the CogniFit® app, cognitive programs can also be implemented for the prevention and treatment of related variables such as insomnia, depression, and especially memory failure. Similarly, cognitive training could be used as a health promotion tool for active aging.
It is important that health professionals, specifically nurses, know the benefits of these programs. Nursing professionals are in direct contact with elderly people through different health services, especially primary care. This population group requires the greatest number of home visits and has the greatest health demands; thus, they can be helped the most by preventing cognitive problems, detecting their memory failures early, and working on their improvement/prevention. The high rate of demographic aging across the world makes it important to maintain the autonomy and health of elderly people, not only on a physical level but also on a mental level.
Footnotes
Acknowledgements
The authors would like to thank the Vicerrectorado de Internacionalización and the Ayuntamiento de Toledo for their help. They also thank all the people who participated in the data collection.
List of Abbreviations
World Health Organization (WHO)
EuroQol (EQ-5D)
Oviedo Sleep Questionnaire (COS)
Visual Analog Scale (VAS)
Memory Failures of Everyday (MFE)
Montreal Cognitive Assessment (MoCA)
ORCID iDs
Ethical Considerations
This study was approved by the Social Research Ethics Committee of the University of Castilla-La Mancha (reference number CEIS-735725-R2P4). The research adhered to the fundamental principles of the Declaration of Helsinki and the European Council Convention on Human Rights and Biomedicine. All participants' data were handled in accordance with Organic Law 3/2018 of December 5, on the Protection of Personal Data and Guarantee of Digital Rights, ensuring strict confidentiality and preventing access by unauthorized third parties.
Consent to Participate
All participants received an information sheet explaining the purpose and procedures of the study, and each provided informed consent prior to participation.
Authors’ Contributions
Study design: JMCT, VMP, ESS, AICC, EBC, SRC, JALA
Data collection: JMCT, VMP, ESS, AICC, EBC, SRC, JALA
Data analysis: JMCT, JALA
Study supervision: JMCT, VMP, ESS, AICC, EBC, SRC, JALA
Manuscript writing: JMCT, EBC
Critical revisions for important intellectual content: JMCT, VMP, ESS, AICC, EBC, SRC, JALA
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by a grant from the European Regional Development Fund (ERDF) (Fondo Europeo de Desarrollo Regional [FEDER], [DOCM 27/01/2021]). In addition, it has also been partially financed through the collaboration agreement between the City Council of Toledo and the Universidad de Castilla-La Mancha, for the promotion of academic and cultural activities during the year 2022. Esperanza Barroso-Corroto is supported by a grant (SBPLY/23/180502/000002) from the Junta de Comunidades de Castilla-La Mancha (Spain) and cofinanced by the Fondo Social Europeo Plus (ESF+ 2021-2027) Program.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
