Abstract
Recently, pharmaceutical companies have been striving to occupy a greater percentage of the global market with clinical research actively being conducted through mutual collaboration networks. In particular, the international Botulinum toxin (BT) market is characterized by intensifying competition. The majority of BT research has been conducted in the United States and Europe, whilst various countries have started their regional research through international collaboration networks. With this trend, this study aimed to examine the current collaboration trend and network of clinical trial research in the field of BT by employing both bibliometric and collaboration network analyses with one of the global academic and research databases. Based on the collaboration network, which is organized by 8,296 articles and 10,298 institutes, four centrality measures successfully present several international and regional clinical BT research leading institutes. Moreover, there are some hidden active institutes in this area, which are not highlighted in this paper’s bibliometric analysis. This study contributes to improving the understanding of clinical trials in BT research, a relevant medical topic. Moreover, collaboration network analysis is one of the most valuable approaches of examining the international and regional clinical research.
Introduction
Botulinum toxin (BT) plays an important role in various medical conditions (Kasyanju Carrero et al., 2019), because it can be employed as a therapeutic and esthetic indicator and allows the global BT market to be extensively expanded (Dressler, 2016). Therefore, a number of companies have invested in and conducted large-scale clinical trials with rapidly increasing R&D initiatives and the global BT market has consistently changed accordingly.
As started in the United States and Europe with few neurological indications, a number of scholars have conducted clinical trials using BT through therapeutic and esthetic approaches, as well as medical treatments (Zargaran et al., 2022). For instance, after the merger and acquisition between Abbive and Allergan, one of the largest BT manufacturers in the world, the company actively shed light on the therapeutic diversity and global distribution of BT (Mcneil, 2019). With this effort, the global market was estimated to have annual sales of USD 3.2 billion in 2020, 1 whilst approximately 50% of this market is split evenly between therapeutic and cosmetic purposes (Chiu et al., 2021; Dressler, 2020).
BT was originally developed by Botulinum Toxin Type A and its clinical trials were mainly conducted in the United States, whilst in other countries, only a few BT products have received national/regional medical approvals (Bach & Simman, 2022) despite the great demand for BT products (Saeed et al., 2022). For instance, only one BT product manufactured in Asia received approval from the United States’ Food and Drug Administration. 2
Considering the high demand for BT products, a number of organizations and companies are likely to conduct clinical trials on their BT products. However, because the majority of clinical trials are considered impractical and expensive activities, organizations and companies have primarily focused on finding suitable collaboration partners and sponsors. In addition, the institutions make groups to do the research of global clinical trials. To accelerate academically driven clinical research and to increase collaboration between international clinical trial groups, international collaboration has been proposed. The global partnership has been built in several groups, such as pediatric cancer and cooperative clinical trial execution, which may further improve patient outcomes (Major et al., 2022; Rodger et al., 2018). This implies that exploring appropriate affiliations and collaboration networks for BT clinical trials is one of the highest priority tasks for successful BT research (Dressler, 2012).
Thus, this study aims to present a collaborative network of BT clinical research that considers affiliations and sponsors. The research questions (RQs) were as follows:
• RQ 1: How does collaboration work in clinical trials in BT?
• RQ 2: Can the key affiliations for clinical trials of BT through collaboration network analysis be presented?
To address these RQs effectively, this study collected a dataset of publications on clinical trials in SCOPUS, one of the largest bibliometric databases in the world (Choi et al., 2021). The remainder of the paper is organized as follows: Section 2 presents an overview of prior research, and Section 3 explains the collected datasets. The network modeling approaches and results are summarized in Sections 4 and 5 respectively.
Literature Review
Botulinum Toxin Trend Research
After esthetic indications were approved by the United States Food and Drug Administration in 2002 for temporary improvement of glabellar lines in patients aged 65 years and younger, BT has been used effectively in recent decades and researched via clinical trials (Arora, 2022). Moreover, other symptoms, including depression and chronic migraine, have been highlighted (Dressler & Johnson, 2022; Matta et al., 2022; Wollmer et al., 2022).
As a representative example, Şenel and Muslu (2020) presented global trends in the BT market between 1975 and 2017 by conducting a check of the total publications on BT and ranked the countries that were interested in BT based on publication productivity. They also suggested a reference network visualization approach examined by keywords focusing on neurological terms in publications.
Related Network Analysis
The majority of prior research on medical and clinical trials has focused on individual research activities or gene/chemical network analysis (Gilbert et al., 2022; Selby et al., 2013). However, considering that the collaborative activities of diverse institutes can accelerate the efficiency and effectiveness of clinical and academic research (Aldieri et al., 2018; Rey-Rocha et al., 2015), several simultaneous collaboration network analyses have been examined (Funderburk et al., 2019).
Khalagi et al. (2021) investigated the pattern and characteristics of collaboration networks among Iranian osteoporosis researchers through co-authorship social network analysis. They collected all articles published by Iranian researchers from 2009 to 2019 from three well-known academic databases, namely PubMed, SCOPUS, and Web of Science. After considering Iranian researchers with more than five articles in the last 10 years, they provided several academic guidelines and potential collaborators in the field of osteoporosis through the authorship graph, co-authorship, and organizational/national networks.
Bian et al. (2014) investigated the collaboration network of the Clinical and Translational Science Award operated by the National Institute of Health (NIH) and the National Center for Advancing Translational Sciences and proposed a link prediction model for potentially successful collaborations. By computing several network centrality measurements with the relative influences of each researcher with well-known prediction models (e.g., PageRank and Markov Model),
Similar to other research topics, the coronavirus disease 2019 (COVID-19) had notable impacts on BT usage and cosmetic surgery. For instance, Ramirez et al. (2022) reported that the cosmetic use of BT is one of the most common esthetic procedures worldwide.
Considering the rapid interest in BT usage, a number of researchers, organizations, and nations have made notable efforts to conduct clinical trials and research on BT. However, given that a large number of recent studies have considered overall clinical trials and specific diseases at the institutional, national, or regional levels, it is difficult to identify the global collaboration network of BT and find potential successful collaborators for the next level of BT (Sethi et al., 2021).
The importance of international collaboration of surgical clinical trials has been raised, and organizations conducting clinical trials continue to use global networks to reduce costs and accelerate recruitment (Drain et al., 2014; Huamaní & González-Alcaide, 2013). This study provides new analysis and insight into how to find active groups in developing new indications and ingredients, using global clinical trials. The target is the institute having difficulty getting information on global clinical trials. Nowadays, international collaboration is being conducted by signing agreements between the state, university hospitals, or companies, by creating research groups (Major et al., 2022). Because of the ability to reduce operational costs while recruiting a large number of patients in a timely manner and increase in geographic dispersion of drug development operations, it was anticipated that more active research and trials would be possible if the network with the groups which actively conducted studies was already known (Thiers et al., 2008). According to the above literature review of previous studies, collaboration is underway to review the advantages of clinical cooperation networks and open innovation. This study aimed to further analyze this and help the affiliations to find a network to collaborate with. Thus, this study is expected to provide one of the cornerstones for the global network analysis of BT clinical trials in the last 10 years (2013–2022).
Subjects and Methods
Data Collection
In this analysis, articles published from January 2013 to January 2022 from five sections (Health Professions, Pharmacology, Toxicology, Pharmaceutics, and Nursing) were collected from the SCOPUS database, which included “Botulinum Toxin” in “Article title, abstract, and keywords.” The list of journals indexed in Scopus from the Elsevier website was also collected, and is provided directly in a spreadsheet format. The exported search results were obtained in a csv format with 14 fields, with the following field names: “Authors, Author(s) ID, Title, Year, Cited by, DOI, Link, Affiliations, Authors with affiliations, Abstract, Funding Details, Publisher, PubMed ID, Open Access.” Because the funding information column means the supporting agency, this column was retitled as Sponsor. After this, the articles that were not primarily related to BT and clinical trials were reviewed and excluded.
Preprocessing
The data in the Scopus datasets was extracted and unified using Python Pandas. Affiliations and Sponsors’ name of research is separated by the delimiter “;” and each country was separated by the delimiter “,”. Each institution’s name was stored in the individual column. The sponsors simply list the name of the agency, while the affiliations consist of the name, country, and address of the agency, so the country could be extracted separately. The abbreviations of affiliation and sponsor were unified into one and the data was then filtered and only affiliations and sponsors were selected to tailor the data for checking collaborations of research. Moreover, hospital selection is important in clinical trials, and in most countries, important hospitals are often owned by universities, meaning that, in these instances, the hospital was renamed the name of the university (Kim et al., 2021). Because there are several different ways to present the same affiliation (e.g., Harvard University Medical School/Harvard Medical School), all affiliations were revised and rechecked manually in the datasets. Moreover, several affiliation names that were not presented in English were revised as per their English affiliation names. Using these procedures, this study found 8,296 articles and 10,298 unique institutions (affiliations and sponsors).
Network Construction
Network Construction and Generation
The affiliations or sponsors have connections with those institutions that have published articles with the clinical trials. Thus, the research collaboration network consists of affiliations and sponsors. Based on the available publication datasets of affiliations and sponsors, a network matrix, which represents the collaboration between research institutions, was built. This study proposes a cumulative-weighted graph network, an undirected collaboration network, for observing a clinical trial collaboration network. Figure 1 shows an example of the collaboration network used in this study. One institution could do the research with several research institutions and each affiliation could make deep collaborations with other affiliations. To take that into account, this study used a cumulative-weighted graph network. This network was generated using NetworkX (2022). NetworkX library using Python helps to understand the collaboration network of research institutions and is capable of utilizing computational network modeling and implementing a highly flexible graph (Hagberg & Conway, 2020).

Example of a network model.
The properties of the network were defined as follows:
•
•
•
Node Centrality Measurements
To identify influential affiliations, the hub nodes in the network need to be highlighted. Thus, this study employed several centrality measures that are widely used in social network analysis: degree, closeness, betweenness, and eigenvector centrality measures. Considering a series of centrality measures, a set of influential nodes are presented.
•
•
•
•
Results
Bibliometric Analysis
Publication Distribution
Figure 2 shows publication growth from 2013 to 2021, where 865 articles were published in 2013, compared to 1,176 articles in 2021.

Number of articles from 2013 to February 2022.
Publication Sources
Table 1 lists the top publications on BT. Here, “Toxins” and “Neurourology and Urodynamics” are presented as the top publication sources with 1,040 (16.4%) and 701 (11.1%) articles, respectively.
Top 10 Sources Publishing Botulinum Toxin Publications, 2013 to 2022.02 (n = 6,334).

Productive Countries
This study identified 110 countries for BT research in SCOPUS. Table 2 lists the most productive nations based on the number of articles and it was determined that the United States (n = 2,220, 35%) and China (n = 525, 8.3%) were the most productive nations.
Top 10 Productive Countries (2013–2022. 2; n = 6,334).

Network Analysis
Overall Research Collaboration Network in BT
Among the 10,298 affiliations in 8,296 articles, only 127 and 352 affiliations appeared in over 20 and 10 articles, respectively. Among the 8,296 articles, 1,962 were examined with a single affiliation. Thus, this study considered 6,334 articles in the network analysis. Figure 3 shows a representative collaboration network.

Graph network of the top 20 affiliations and those that have more than 10 research collaborations.
Table 3 lists the leading affiliations. Among the top affiliations, six were from the United States, of which three, Allergan, the NIH, and Mayo Clinic, were the leading organizations for clinical BT research. Ipsen from France and the National Natural Science Foundation of China (NSFC) were also included.
Top 10 Productive Affiliations (2013–2022.02; n = 6,334).

Degree Centrality
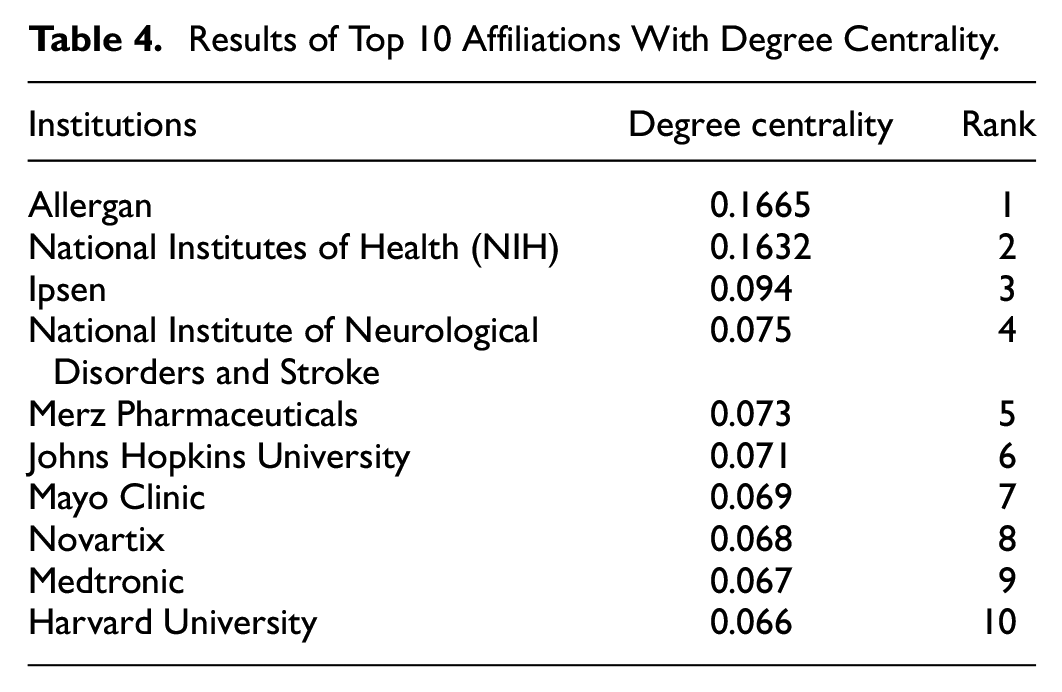
Table 4 lists the results of degree centrality in collaborative BT clinical research. The degree centrality simply counts the connected total number of nodes and institutions, which means that the higher level of degree centrality indicates the greater number of clinical trials in BT research. Allergan was shown to be the most central affiliation in BT research, meaning it was the most productive collaborative affiliation for the research. The NIH, Ipsen, National Institute of Neurological Disorders and Stroke, and Merz Pharmaceutical were also listed.
Results of Top 10 Affiliations With Degree Centrality.
Betweenness Centrality
Table 5 shows the results for betweenness centrality in collaborative BT clinical research. Betweenness centrality is the computational distance of each node and the institution which is in the shortest path means the institution is needed to be connected to the important affiliations and sponsors. The high level of betweenness centrality in specific nodes indicates that the nodes play key and responsible roles in mutual collaboration. Consistent with the results of degree centrality, Allergan is the top affiliation in clinical network collaboration, followed by NIH, NSFC, Ipsen, and Mayo Clinic.
Results of the Top 10 Affiliations With Betweenness Centrality.

Closeness Centrality
Table 6 shows the results for closeness centrality in collaborative BT clinical research and was determined by how close a specific affiliation is to all the nodes in the network. A smaller distance to all nodes indicates a higher level of closeness centrality. This study determined that the institution plays a central role in the network by calculating closeness centrality, which was correlated with the distance from the center in the network. As it tends toward the center of all connections, the closeness centrality value decreases, and based on this, it can be seen where the center value corresponds. The NIH is the top affiliation on closeness centrality, followed by Allergan, Ipsen, and the National Institute of Neurological Disorders and Stroke.
Results of the Top 10 Affiliations With Closeness Centrality.

Eigenvector Centrality
Table 7 shows the results of eigenvector centrality in collaborative BT clinical research. Eigenvector centrality considers not only the total adjacent nodes also the importance of the adjacent node. If one institution connects to the institutions that did lots of research, it has a high eigenvector centrality. The NIH has the highest eigenvector centrality, followed by the National Institute of Neurological Disorders and Stroke, Johns Hopkins University, Harvard University, and Cambridge University.
Results of the Top 10 Affiliations With Eigenvector Centrality.

Discussion and Conclusion
This study examined the collaboration trend and network analysis of clinical trials in BT research by considering SCOPUS. Considering 6,334 articles published between 2013 and 2022, a collaboration network for clinical trials in BT research and examined several recent bibliometric results is presented.
The dataset utilized was posted on the GitHub repository for universal use and for practical use in the field and is available at our open repository website. 3 There were many cases where clinical trials were conducted on a whim, however, this study tried to contribute to the academic side by adding scientific methods using neural network. The advantage of the proposed methods is that clinical trials are possible without complex agents because the institution could know where they should conduct clinical trials collaboration through the algorithm.
The purpose of this study is to employ both bibliometric and collaboration network analyses and sharing methods to explore partnerships in the pharmaceutical industry. Studies such as Yang and Lee (2018) have shown similar results, namely that university hospitals can be intermediators in the clinical trials partnership and this shows in our study via eigenvector centrality. However, the difference between this study and the prior study is that collaboration was analyzed focusing on the individual market situation of BT not an overall view. Because the clinical status is different for each market, and active collaborative affiliations are identified differently, this study focused more on these areas to help in selecting institutions more clearly that could help collaboration.
To answer the first research question, a collaboration network with several centrality measures was constructed. As presented in Table 4, in the case of degree centrality, all the top 10 affiliations are located in the United States. Considering the meaning of degree centrality, it can be concluded that the leading nation in collaborative BT clinical research is the United States, which is also examined in the bibliometric analysis (Table 2).
As presented in Tables 5 and 6, this study examined both closeness and betweenness centrality measures to effectively address the second research question. Although the majority of top institutes for both measures came from the United States, several different institutional types were observed, namely: global companies, national laboratories, hospitals, and universities. Thus, diverse stakeholders are required to conduct BT clinical research. Therefore, it can be an excellent opportunity for “newbies,” who want to find their active collaborators in the world. Contrary to the results of degree centrality, several regional institutes are presented in the top 10 affiliations in betweenness (e.g., Yonsei University (Republic of Korea), Deutsche Forschungsgemeinschaft (Germany)) and closeness centrality measures (e.g., Merz Pharmaceuticals (Germany)). This indicates that regional collaborations are actively examined in BT research clinical trials. For instance, 105 articles were introduced by collaboration activities between Merz Pharmaceuticals and German companies, while the collaboration of Deutsche Forschungsgemeinschaft and German companies introduced 65 articles as their regional research.
Based on eigenvector centrality, specific affiliations, which show a high level of eigenvector centrality, represent a number of connections with important affiliations. As shown in Table 7, several affiliations not presented in the lists of other centrality measures were observed. For instance, various universities and national affiliations have been presented (e.g., Harvard, Cambridge, and Stanford). As expected, Allergan ranks as the top institution in all employed centrality measures, simply because it is the first company to introduce BT for medical purposes in society.
In the case of several pharmaceutical companies, it could be a problem if they could not find a partner when they started to focus on the global market and needed clinical approval overseas. Thus, it is believed that the proposed collaboration network analysis can be a valuable approach for searching for research-critical institutions in specific countries or regions. It can contribute to the collaboration of researchers, organizations, and countries by disclosing collaborative structures combined with information about the centrality of network participants.
Figure 2 shows that the degree of interest in Botulinum Toxin increases and about more than 2000 collaborations are done in the United States. Compared to the findings in the map of collaboration networks in clinical trials (Yang & Lee, 2018), the size of collaborative groups has increased not only using Botulinum Toxin but also in whole parts and it implies that partnerships have increased among agencies participating in clinical trials.
In medicine, in particular, it is important for the industry to create close interactions between clinicians in medical schools and their hospitals or academic medical centers. In the case of clinical trials in other countries, this should be carried out through collaborations with various institutions, which will reduce notable efforts it takes to find and search for clinical research institutes.
In addition, recently, with a notable interest in beauty and plastic surgeries, several developing and developed countries are considered emerging markets. Thus, the Botox market is growing rapidly, which presents an excellent opportunity to expand into other countries’ markets by identifying major research institutes. In particular, in the case of BT, there are few FDA-approved products, meaning that it will have a pre-emptive effect in markets.
This study can contribute to improving the understanding of clinical trials in BT research, which is a relevant medical topic. It is also shown that a set of network analyses can be effective for presenting collaboration activities of the topics as well as finding potential collaborators.
This analysis has the potential to facilitate collaboration in clinical trials with other affiliations, one of the recent research trends, and is of great significance in that it can be intensively targeted in the clinical setting of a specific country in consideration of market expansion.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Research Foundation of Korea funded by the Korean Government (NRF-2023K2A9A1A01098773). This research was also supported by the MSIT (Ministry of Science and ICT), Korea, under the ICAN (ICT Challenge and Advanced Network of HRD) program (IITP-2020-0-01816) supervised by the IITP (Institute of Information & Communications Technology Planning & Evaluation).
Ethics Statement
Not applicable.
Data Availability Statement
Data will be made available on request
