Abstract
Our study aims to conduct an evaluation and comparison between the leaflets of local and imported oral solid over-the-counter medications (OTC) found in the Palestinian markets. Forty-nine leaflets were evaluated using a scoring method and depending on the appearance of 31 headings, along with the use of manual word counting to assess the amount of information within 23 topics of all 31. None of the evaluated PIs, local or imported, completed all 31 criteria. Remarkably, the imported product leaflets achieved higher scores than their local leaflets in 19 out of 31 topics, whereas the local products achieved better scores than the imported agents in only 2 items, and 10 criteria were equally scored between them. None of the PIs mentioned pharmacokinetic information, instructions for converting tablets or capsules into liquid forms, or shelf life. In general, imported leaflets give more detailed information than their local counterparts. The lowest value of the fold difference in mean rank was 0.85 times for the direction of use, and the highest value was 2.43-fold for adverse drug reactions. The results showed a deficiency in the information provided in the local package inserts compared to the imported versions of oral solid OTC medications available in Palestine. These findings encourage the Palestinian Ministry of Health to provide full criteria for pharmaceutical companies to ensure that the leaflets contain sufficient and updated information and ensure that the patients use their drugs safely and effectively.
Keywords
Background
Being among the most valuable tools for patients, a drug leaflet (DL) or package insert (PI) is an instrument that provides information to patients about the medication (Thürmann, 2006), which is an essential reference for understanding the medications, especially when the connection between patients and their health care providers cannot be achieved (Nabors et al., 2004). In particular, many patients and healthcare professionals read the leaflets to find their inquiries. This information helps practitioners to provide proper counseling to patients regarding their drugs (Al-Ramahi et al., 2012; W. M. Sweileh et al., 2015). Therefore, the written information in the leaflets must be readable and comprehensible to users, particularly the information that watches for appropriate drug use (Al-Ramahi et al., 2012; Van Haecht et al., 1991). In addition, pharmaceutical companies must continually update this information to help physicians and pharmacists trust in the information provided in the DL (Al-Ramahi et al., 2012; W. M. Sweileh et al., 2015).
Over-the-counter (OTC) medications are prevalent drugs that are readily available to the public without a medical prescription (Chan & Tran, 2016). This means that patients can obtain drugs without the role of practitioners (Chan & Tran, 2016). Therefore, OTC medications should contain useful and easily understood information as their uses have increased in recent years (Marathe et al., 2020).
DLs are constantly reviewed worldwide (Food and Drug Administration, HHS, 2014). Institutes such as the Food and Drug Administration (FDA) occasionally determine the overall design of the DL and watch for updating of its content (Food and Drug Administration, HHS, 2014). Although the Palestinian Ministry of Health (MOH) requires pharmaceutical companies to design a DL that is written in two languages (English and Arabic), it does not monitor the quality of the leaflet content (Palestinian Ministry of Health, 2007).
Examining the quality of the information provided in the DL is substantial, as the presence of obvious details in the leaflet will help the patient use the medication correctly (Gillies et al., 2014). Evaluation of the information content of the DLs was carried out in many previous studies (Al-Aqeel, 2012; Arandy et al., 2019; Chhaya, 2017; Eshtayeh et al., 2019; Khamas et al., 2019; Qatmosh et al., 2017; Rahim & Rafiq, 2013; A. Sawalha et al., 2008; Shivkar, 2009). Some of these depended on the presence of particular topics in the PIs (Al-Aqeel, 2012; Chhaya, 2017; Khamas et al., 2019; Rahim & Rafiq, 2013; Shivkar, 2009), while other studies built their assessment on both the presence of certain headings and the amount of information provided in the PIs (Arandy et al., 2019; Eshtayeh et al., 2019; Qatmosh et al., 2017; A. Sawalha et al., 2008). In Palestine, many studies were conducted using similar principles to assess the quality and quantity of information provided in the PIs (Arandy et al., 2019; Eshtayeh et al., 2019; Qatmosh et al., 2017; A. Sawalha et al., 2008). These studies showed that local drug leaflets lacked much information compared to imported agent leaflets (Arandy et al., 2019; Eshtayeh et al., 2019; Qatmosh et al., 2017; A. Sawalha et al., 2008).
We conducted this research to evaluate the leaflets of OTC medications. This category is among the most widely sold drugs overall in the world (Cooper, 2013; Taybeh et al., 2020), and the prevalence of its use in Palestine is as high as 60% (P. W. Sweileh, 2004), reaching 98% among university students (A. F. Sawalha, 2008). Additionally, patients may not be provided with adequate information from their physicians due to workload and may find retaining verbal information for a long time challenging. Therefore, the quality evaluation of OTC leaflets becomes more critical. OTC agents are prescribed by pharmacists or sold to patients without a prescription, so instructions from the health care provider may be absent. Furthermore, many patients believe that OTC medications are safe and without harm (Ylä-Rautio et al., 2020). Patients with multiple medical conditions may have serious side effects when using a specific class of OTC medications; for example, patients with chronic kidney disease may experience deterioration in their clinical condition when using nonsteroidal anti-inflammatory drugs (NSAIDs) (Baker & Perazella, 2020).
In this study, our objective was to evaluate the quality of the information provided in the leaflets of both local OTC products and their imported competitors. The study tends to highlight some important findings with the leaflet content of information. Moreover, we attempt to determine the main problems of the information provided in the DLs that should be resolved. In Palestine, this is the first study to highlight the challenges and problems of OTC medications. We hope that our findings serve as a reference for pharmaceutical companies in developing high-quality DL.
Methods
Study Design
This cross-sectional study was carried out to evaluate and compare the leaflets of locally manufactured OTC drugs and their imported competitors. This work was achieved by assessing the presence of specific topics in the leaflets and counting the words within each topic. The study was completed in 8 months between September 2020 and April 2021.
Inclusion and Exclusion Criteria
Initially, we reviewed the list of over-the-counter medications that were approved by the Palestinian Ministry of Health (MOH) and available on its website (Palestinian Ministry of Health, 2020). The selection of PIs was based on the following: (1) available in the Palestinian market during the study period, (2) registered in the Palestinian MOH, (3) listed as an OTC drug in Palestine (Palestinian Ministry of Health), and (4) available in an oral solid dosage form. Imported agents are defined as any medications imported from any country outside Palestine. On the other hand, we excluded dietary supplements, drugs that were withdrawn from the market (i.e., ranitidine), combination products, and drugs that do not have at least one equivalent imported product. Therefore, 11 different active ingredients were chosen according to the selected criteria, which included loperamide, famotidine, omeprazole, bisacodyl, cetirizine, acetylsalicylic acid, naproxen sodium, paracetamol, ibuprofen, loratadine, and bismuth subsalicylate. Forty-nine PIs represented these ingredients; 26 were Palestinian products, and the other 23 PIs were imported agents (Table 1).
Trade Names of OTC Medications (Local and Imported) That Were Included in the Study.

Collection of PIs
We conveniently selected 15 pharmacies located in the largest cities in the West Bank: Nablus, Ramallah, and Hebron. Three pharmacists visited these pharmacies to ask about the availability of the selected OTC products. After that, each responsible pharmacist was asked about the possibility of obtaining the leaflets, and there was great cooperation with us.
Evaluation of the Information Content of PIs
PIs were evaluated using two methods. The first was the scoring method. We extensively reviewed the literature with regard to the evaluation of package insert information and included all information statements that were previously described (31 headings) (Al-Aqeel, 2012; Arandy et al., 2019; Eshtayeh et al., 2019; Phueanpinit et al., 2016; Qatmosh et al., 2017; Rahim & Rafiq, 2013; Ramdas et al., 2013; A. Sawalha et al., 2008; W. M. Sweileh et al., 2015). The headings included: brand name; active ingredient; inactive ingredients; indications; therapeutic class; mechanism of action; drug dose; duration of use; missing dose; maximum dose; directions for use; overdose and management; warnings and precautions; effects on the ability to drive and use machinery; contraindications; adverse drug reactions; drugã drug reactions; drug-food interactions; pregnancy considerations; lactation considerations; pediatric considerations; geriatric considerations; possibility of crushing and mixing with food or beverages; the possibility of tablet splitting or opening the capsule; name and address of manufactures; instructions to convert tablet or capsule into liquid forms; pharmacokinetic information; storage; shelf-life; date of the last version; and source of information. For each topic, the leaflet was given 1 point if mentioned and 0 points if missing. Subsequently, the total scores of each company for the local products, the total scores of all local products, the total scores of all imported products, and the total scores of all selected PIs were calculated for each information statement. The second method was a simple word count, which was carried out by manually counting the number of words within the 23 selected headings. Eight headings were excluded in this method either because the count was not valuable (brand name; active ingredients; name and address of manufactures; date of last version; and shelf life) or because the information statements were not mentioned in all local PIs or their imported counterparties (geriatric considerations; instructions to convert tablets or capsules into liquid dosage forms; and pharmacokinetic information).
All data were evaluated according to the selected criteria by three independent pharmacists. Reviewers had to refer to the principal investigator (AK) for discrepancies. There have been no critical disagreements between them.
Ethical Concerns
The Institutional Review Board (IRB) of An-Najah National University determined that this study was exempted from review. All pharmacists gave their informed consent to share their own information related to the current study.
Statistical Analysis
All data were introduced in version 21 of the Statistical Package for Social Sciences program (SPSS). The results of categorical data were presented as percentages and frequencies, while continuous data were described as median, quartile 1 (Q1), and quartile 3 (Q3). The Mann-Whitney U test was performed to check for differences in median word count between local and imported PIs. This test was applied for 23 headings and subheadings. In addition, the fold differences in mean rank were calculated for each topic by dividing the mean rank of the higher value by the mean rank of the lower value in both imported and local PIs. The p-value was considered statistically significant whenever its value <.05.
Results
The analysis was carried out on 49 OTC PIs for 11 active ingredients available in the Palestinian market; 26 of them were locally manufactured, and the remaining 23 PIs were produced abroad. Local agents were produced by five Palestinian pharmaceutical companies, as shown in Table 2. All selected medications were also classified as the following: 25 analgesic drugs, 14 gastrointestinal drugs, and 10 antihistamine drugs.
Trade Names of OTC Medications for Each Local Company That Was Included in the Study.

A: Birzeit Pharmaceutical Company; B: Sama Pharmaceuticals Manufacturing; C: Jerusalem Pharmaceuticals; D: Pharmacare PLC; E: Beit Jala Pharmaceutical Company.
None of the PIs, either local or imported, met complete adherence with all selected headings and subheadings. In particular, the DLs of the imported agents achieved higher scores than their local counterparts on 19 out of 31 criteria, whereas the local products achieved better scores than the imported agents in only two items, and the remaining 10 criteria were scored equally between them (Table 3).
Total Scores of Each Statement Mentioned in the Leaflets of Local and Imported OTC Medications.
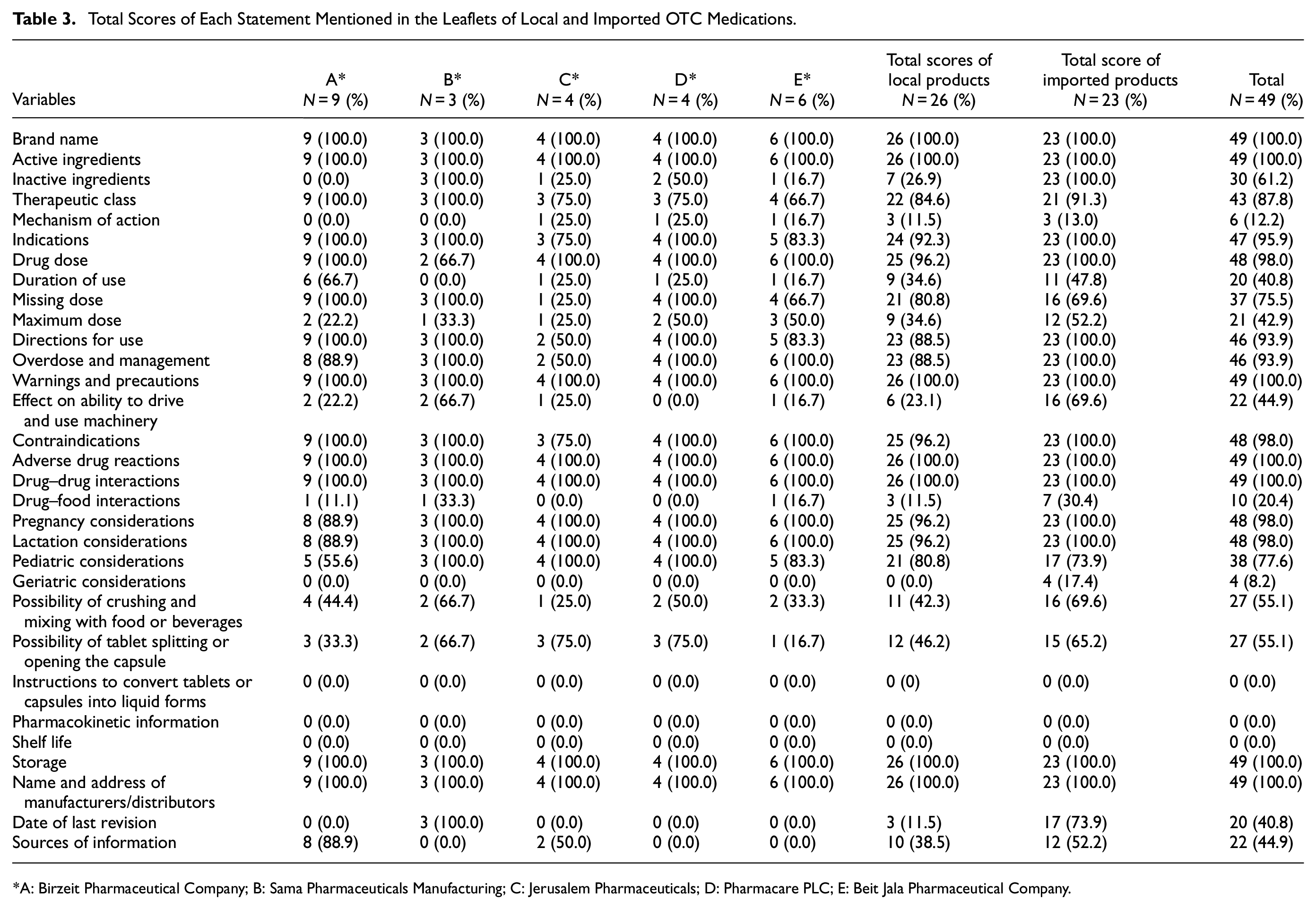
A: Birzeit Pharmaceutical Company; B: Sama Pharmaceuticals Manufacturing; C: Jerusalem Pharmaceuticals; D: Pharmacare PLC; E: Beit Jala Pharmaceutical Company.
Our analysis found that all OTC PIs mentioned brand name (100.0%), active ingredient (100.0%), warnings and precautions (100.0%), adverse drug reactions (100.0%), drugã drug interactions (100.0%), name and address (100.0%), and storage (100.0%), while none of the PIs included instructions to convert tablets or capsules into liquid forms (0.0%), pharmacokinetic information (0.0%), and shelf life (0.0%). Approximately 45% of the PIs did not cover the two main pharmaceutical considerations: “possibility of opening the capsule or tablet splitting” and “possibility of crushing and mixing with food or beverages,” and higher scores were recorded in favor of imported products. Only 21 products (9 local and 12 imported, 42.9%) included some notes about the maximum dose in their leaflets. Furthermore, the PIs provided information on the mechanism of action (12.2%), drug-food interactions (20.4%), and geriatric considerations (8.2%). However, all imported PIs contained the following headings: inactive ingredients (100.0%), indications (100.0%), drug dose (100.0%), directions for use (100.0%), overdose and management (100.0%), contraindications (100.0%), pregnancy considerations (100.0%), and lactation considerations (100.0%). Comparably, most local PIs included these headings, except for inactive ingredients mentioned in only 7 (26.9%) out of 26 PIs. It was also found that 76.9% of local DLs did not include information on the effect on driving and using machinery compared to 30.4% of the imported DLs. Similarly, imported products (73.9% and 52.2%, respectively) were more focused on information on the last revision date and the sources than local products (11.5% and 38.5%, respectively). Concerning the item “duration of use,” we noticed that 65.4% of the local PIs and 52.2% of the imported PIs did not contain any information.
Our results also showed that geriatric considerations were not found in any PI of local products. Local agents had strong points in implementing information on missing doses and pediatric considerations in more than 80% of their PIs. In general, no apparent differences were found in the recorded scores between local and imported PIs regarding the therapeutic class and mechanism of action.
A simple word count was completed for each topic in the 23 topics to compare local and imported PIs. The imported PIs had a higher median word count than their local counterparts in all 23 criteria, except for direction of use, mechanism of action, duration of use, missing dose, and drug-food interactions, where either the local PIs achieved higher scores than the imported ones or both groups recorded the same scores. Significant differences between the word count of local and imported DLs were observed in 11 out of 23 topics as follows: inactive ingredients (p < .001), therapeutic class (p = .036), warnings and precautions (p = .005), effect on the ability to drive and use machinery (p = .001), contraindications (p = .007), adverse drug reactions (p < .001), pregnancy considerations (p < .001), lactation considerations (p < .001), the possibility of crushing and mixing with food or beverages (p = .003), the possibility of tablet splitting or opening the capsule (p = .026), and storage (p = .003). The lowest value of the difference in mean rank was 0.85-fold for the direction of use, and the highest value was 2.43-fold for adverse drug reactions (Table 4).
Comparison Between Word Count for Local and Imported OTC Medications.
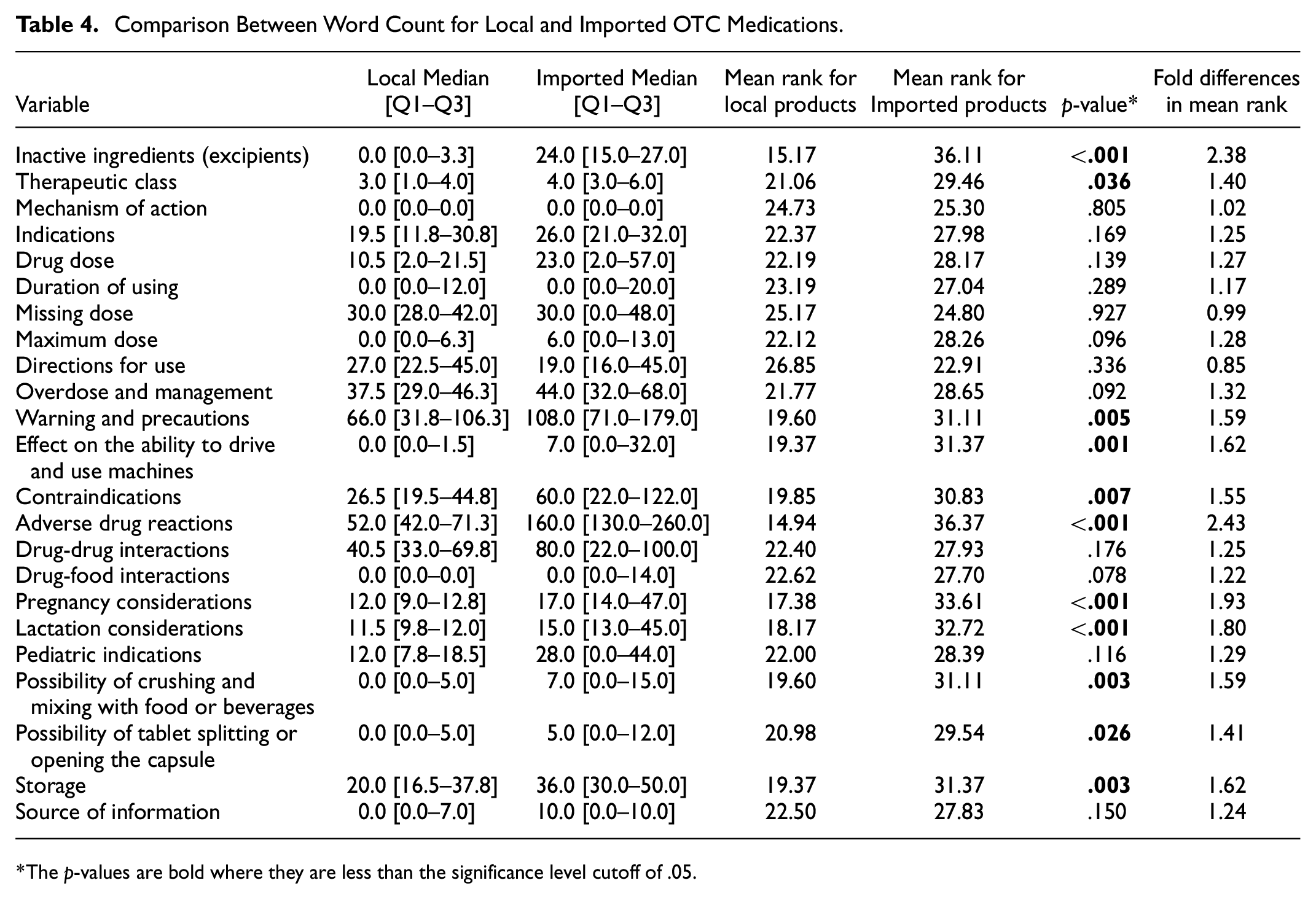
The p-values are bold where they are less than the significance level cutoff of .05.
Discussion
Leaflets of OTC medications are one of the important sources of information for users. It must contain appropriate and clear information. Our analysis showed significant differences between local and imported agents in terms of the amount of information provided in their DLs. These variations were in favor of imported products. In general, imported DLs mentioned a higher number of topics and wrote more details than local DLs. The same results were found in many previous studies conducted in Palestine (Arandy et al., 2019; Eshtayeh et al., 2019; Qatmosh et al., 2017; A. Sawalha et al., 2008) and abroad (Munsour et al., 2019).
Safety is an important concern that can affect the therapeutic plan. One of the important topics that should be found in the leaflets is “inactive ingredients.” As mentioned in the results, this topic was found in all imported DLs (100.0%) and only 26.9% of the local DLs. The median word count, which represented a statistically significant difference (p < .001), was 0.0 [0.0–3.3] in local leaflets compared to 24.0 [15.0–27.0] in imported leaflets. The danger of dismissing this topic increases in patients who had an allergy to one or more of the inactive ingredients. For example, polyethylene glycol (PEG) is commonly used in the pharmaceutical industry for tablets and capsules (Dave, 2008). PEG can cause an allergic reaction that can sometimes be serious for some people (Stone et al., 2019; Wenande & Garvey, 2016). On the other hand, the maximum dose was listed in 34.6% and 52.2% of the local and imported DLs, respectively. The difference in the median word count between the two groups was not significant. The importance of this topic arises not only from the risk of toxicity but also from the ignorance of the public to this essential information.
We also noticed that almost all leaflets (98.0%) in our study mentioned contraindications in terms of safety. This result is somewhat superior to a Malaysian study that found that contraindications were mentioned in 78 leaflets of 92 (84.8%) (Mittal et al., 2018). Contraindications were found to be read by 60% of Egyptian patients who routinely use the leaflets (Amin et al., 2011). Providing clear points about this heading is essential, as it increases the patient’s role in helping healthcare providers prevent serious problems from happening. Missing the dose is another issue that can sometimes be harmful to patients if left without education on how to handle this problem. Missing doses were mentioned in 69.6% of the imported leaflets and, interestingly, in 80.8% of the local leaflets. A systematic review in the United Kingdom found that only half of the products included some instructions for missing doses in their leaflets (Albassam & Hughes, 2021). Detailed information on this topic helps patients with memory deficits, especially elderly patients (Fialova & Onder, 2009). Another aspect related to safety is adverse drug reactions (ADRs). This topic was found in the 49 selected leaflets. However, the difference in word count between local and imported DLs was statistically significant (p < .001). The median word count [Q1–Q3] was 52.0 [42.0–71.3] and 160.0 [130.0–260.0] for the local and imported leaflets, respectively. Similar results were also found in many other Palestinian studies (Arandy et al., 2019; Eshtayeh et al., 2019; Qatmosh et al., 2017; A. Sawalha et al., 2008). Notes about adverse drug reactions should be sufficient, accurate, clear, and at the same time, have not to worry the patients. A Danish study showed that one-third of patients stopped taking their medications because the information provided in the PI regarding side effects raised their concerns about drug use (Horwitz et al., 2009).
Antihistamines are known to be among the most commonly used OTC drugs, and many products on the market contain this ingredient. This class of medications affects the ability of its users to drive or use machines (Popescu, 2008). Therefore, the leaflets on such drugs must contain detailed information regarding the drug’s effect on driving skills. This item was mentioned in 69.6% of imported DLs compared to 23.1% of their local counterparts in our study. The median word count was 7.0 [0.0–32.0] for imported products and 0.0 [0.0–1.5] for local agents, which represented a significant difference between the two groups (p = .001). Adding such information to the leaflets is substantial, as not all pharmacists provide sufficient information on this issue. In a Saudi study, approximately 74% of community pharmacists did not provide spontaneous counseling to patients about the effect of antihistamines on driving skills (Khojah, 2019).
Paracetamol is considered the most commonly used antipyretic and analgesic medication in the world (Lau et al., 2016). Approximately two-thirds of athletes in Palestine use analgesics (paracetamol or aspirin) (Qasrawi et al., 2021). Half of the Palestinian parents did not know that paracetamol could cause severe harm to their children when administered overdose (Daifallah et al., 2021). The low awareness regarding this problem requires manufacturers to add more information on overdose and its management in their leaflets, especially for paracetamol. It is the most available OTC drug and among the most common agents that cause poisoning in Palestine.
Leaflets are an important source of information, not only for patients but also for healthcare professionals, as doctors and pharmacists can refer to this tool to obtain some notes and recommendations on the drug. Interestingly, we noticed that none of the leaflets mentioned pharmacokinetic information and instructions to convert the tablet and capsule to liquid form. The lack of pharmacokinetic information in DL can deprive healthcare providers of the information they need to treat a certain age group, such as children, who have different kinetic properties compared to adults (Fernandez et al., 2011). Furthermore, instructions to convert tablets and capsules into liquid form can help patients who have difficulty swallowing to take their medication easily, especially when the active ingredient is only available in one pharmaceutical form. Another interesting point was that 17.4% of imported products mentioned geriatric considerations in their leaflets, while none of the local leaflets included this information. Unfortunately, the absence of this information may pose a risk for elderly individuals, especially for drugs that need dose adjustment when used for geriatric patients (i.e., anticholinergic drugs) (Glass et al., 2008; Kose et al., 2020). A similar defect was found in an American study. This study evaluated the information on topical agents and found that only one in 15 leaflets mentioned geriatric considerations (Zaghi & Maibach, 2007).
Citing the source of information in DL improves its quality (Moult et al., 2004). In our study, 38.5% of the local leaflets and 52.2% of their imported counterparts mentioned their information sources. Unfortunately, most of the information sources for local products were from the 90s editions, and clearly, they were clearly not updated. Referring to updated sources of information and adding them to leaflets can reinforce users’ confidence in DL (Moult et al., 2004).
Clearly, information regarding the duration of use was absent in the majority of PIs. In Pakistan, a study found a defect in mentioning the drug-food interaction and duration of use. This study recommended that the content of the leaflets be designed according to international standards (Rahim et al., 2016). Providing information on this concept helps patients avoid the dependence problem that arises from prolonged use of stimulant laxatives (i.e., bisacodyl) (Kamm et al., 2011). It can also prevent serious problems such as renal toxicity as a result of the long-term use of NSAIDs (Naughton, 2008).
In Palestine, there are no definite standards for designing DL. A previous study suggested developing global guidelines and stand-alone guidelines to make DL more friendly to patients (Munsour et al., 2019). These recommendations were also seen in a recent major review study conducted in Asia and Africa. This review showed great defects in the leaflets of many products and recommended that the World Health Organization (WHO) continue to work with regulatory and authority bodies in Africa and Asia to provide the best sharing of practical information for designing high-quality written medical information. Moreover, the study noted that DL should meet the needs of users and must be appropriate for them (Nualdaisri et al., 2021).
Strengths and Limitations
A well-designated PI should be written with understandable language and contain accurate, clear, and complete information. This is important because OTC drugs are commonly used and dispensed by users without the need for a doctor’s prescription. Therefore, the right use of the information content of PIs will improve patient compliance and reduce harm due to the inappropriate use of these drugs. This is especially important, as patients may lose counseling or forget some information provided by their healthcare providers.
This is the first study in Palestine to evaluate and compare the information content of PIs between local and imported OTC products. This study does not include all OTC products available in Palestine, as dietary supplements, combination products and liquid dosage forms were excluded. These agents are commonly used and administered by patients. The evaluation criteria used in the present study might differ from other studies, as there are no global consensus criteria that were made for this purpose. Another issue that should be considered when evaluating the information content of the PIs is to test the readability of the text that was not addressed in the current study. Furthermore, pictograms, which were not evaluated in our study, are one of the most important tools that were used to improve patient understanding.
Conclusions
In this study, it was clear that there are significant differences between the local and imported PIs of the oral solid OTC medications available in Palestine. These differences were found in the quality and quantity of the information provided. Incomplete information can unfavorably affect the use of these medications. These findings encourage the Palestinian MOH to provide full criteria for pharmaceutical companies to ensure that the leaflets contain sufficient and updated information and ensure that the patients use their drugs safely and effectively. It is worth mentioning that current research has certain social dimensions that might impact the ability to comprehend medical information provided in leaflets. Patient literacy level, health literacy, and access to healthcare are examples of these social factors. Specifically, patients with low literacy levels/health literacy may not fully understand all medical information provided by drug package inserts, which eventually affects their ability to use medications safely and effectively.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
