Abstract
Developing countries like India, Thailand, and Malaysia are promoting medical tourism as a commodity, generating income through their economic trade contributing to their national revenue. However, there is no legal framework formulated within these countries to regulate its medical tourism industry. In Malaysia, the current legislation that regulates the private health care facilities is not enough to cover all avenues of medical tourism, especially since the industry largely implicates the private sector. Hence, we aim to explore the consequences of the pro-medical tourism stance of Malaysia and its impact toward stem cell tourism. The in-depth interviews of Malaysian policymakers and the systematic review of academic articles and government documents revealed that the current legislation in Malaysia neglects many areas within medical tourism namely medical visa, insurance, and medical extradition that protects the wellbeing of tourists. The deficiency in the absence of a specific law or policy cultivates stem cell tourism that remains generally unregulated plagued with many ethical exploitations, judging by the pro-medical tourism stance.
Introduction
In the age of globalization with an increase in consumer culture, to live healthy, people are traveling across national borders, to make full use of the available alternatives fulfilling their every need. Apart from the conventional traveling that involves visiting destinations for recreational and leisure purposes maintaining mental health, it is evident that people are also traveling seeking possible healthcare while they are vacationing in these unique destinations (Uygun & Ekiz, 2018). The phenomenon of people worldwide traveling across international borders for the purpose of maintaining, restoring, and enhancing their mind and body is known as medical tourism (Al-Lamki, 2011; Hanefeld et al., 2014; Lunt et al., 2011). The rising medical cost and the lengthy waiting period for clinical trials within their home-countries are persuading people to travel to countries with wider medical options for a cheaper cost (Altın et al., 2012; Lunt et al., 2010). The search for quality and affordable healthcare, accessible clinical trials including availability of experimental procedures and treatments are identified as factors that will continue to drive the medical tourism industry (Al-Lamki, 2011; Lunt et al., 2011; Turner, 2012; Wendt, 2012). Developing countries such as India, Malaysia, and Thailand are widely identified as top medical tourism destinations with fairly reasonable medical costs, as presented in Table 1 (Wong Kee Mun, 2012).
Cost Comparison of Selective Procedures in Thailand, India and Malaysia.

Source. Jose and Sachdeva (2010).
Developed Asian countries such as Singapore, Taiwan, and South Korea were also identified as possible choices whose costs are still considerably low compared to the western developed countries such as the United States (US) and United Kingdom (UK) (Stephano, 2018). A study by John Connell (2006) verified that the medical tourism industry will grow as long as cost differentiation and privatization of medical care continues. In Canada, the expanding medical tourism industry is said to have raised concerns such as health equity, the discontinuity of care, and the difference between the balanced local regulation compared to those of medical tourism countries (Turner, 2007). The study by Horowitz et al. (2007) supported future expansion prospect of medical tourism indicating the low medical cost as the major factor based on the identified medical tourism destinations. They pointed out that the quality and risks of treatments in those destinations compelled the US Senate to set-up a task-force in 2006, to study the safety and consequences of medical tourism. An empirical data presented by Brent Lovelock and Kirsten Lovelock (2018) supported a study by John Connell (2013), proving variability between conventional tourists and medical tourists. They explained that leisure may easily be a part of medical tourism and by putting the “hotel” touch to the hospitals adds a sense of leisure into their medical travels. Their study identified key factors such as the treatments, the pre- and post-conditions and the caregivers accompanying them with possibility to facilitate the level of the holiday. Majority of these studies focused on factors that generally promotes medical tourism and its implication on tourist welfare. However, studies by Rupa Chinai and Goswami (2007) and Smitha Mathew and Brindha (2014) concentrated specifically on attracting medical tourists to Chennai, India, turning it into a medical tourism spot. The study also highlighted the medical tourist visa concerns within India.
Previous studies have all highlighted factors that urge medical tourism in many countries. As an industry that equally thrives in Malaysia, this study is significant to demonstrate whether the factors that encourage medical tourism globally also corresponds here.
Stem Cell Tourism
Medical tourism also implicates other areas of technology such as stem cell and aesthetic medicine. As an emerging technology, stem cell is popular for its therapeutic potential owed to its unique regenerative quality. Its ability to self-renew, replicate, and differentiate into any cell or tissues has opened doors to many promising revolutionary therapies and experimental studies that normally are not available in many countries around the world (MacReady, 2009; Mummery et al., 2014). It persuades people to travel abroad seeking these interventions, expecting to cure or treat their diseases and conditions, despite the risk of exploitation by unproven therapies (Zakrzewski et al., 2019). The practice of traveling to other countries with liberal or lack of regulation in search of experimental stem cell therapy that are sometimes unproven is known as “stem cell tourism” and is considered as a sub-set of medical tourism in this study. Hematopoietic stem cell such as adult stem cell extracted from bone morrow (including peripheral blood) are well characterized and common in treating several blood-based disorders for example beta-thalassemia and leukemia. However, other derivatives such as human embryonic, mesenchymal, and induced pluripotent stem cells require exploration to understand how it can offer therapeutic solution consistently for any specific diseases without side effects or risks. Unfortunately, despite the lack of evidence, these stem cell therapies are still offered as experimental treatment which are sought after by many whom have depleted all possible treatment options (Fung et al., 2017; Master & Resnik, 2011).
A study by Eliza Barclay (2009) reported that stem cell experts around the world are concerned that developing countries are using the medical tourism industry to market their unproven stem cell therapies without undergoing proper safety testing or risk assessment. The providers of these therapies overpromise the benefit with very little or no emphasis of its risks, persuading the terminally ill and those that have exhausted all other treatment options to travel across continents to undergo the experimental procedures that may or may not have preclinical backing (Lindvall & Hyun, 2009). Charles E. Murdoch and Scott (2010) examined the concept of “hope” and its influence toward patients’ autonomy in seeking such experimental intervention opening doors to stem cell tourism. While their discussion was not meant to endorse stem cell tourism, it identified the need to have a balanced viewpoint to preserve scientific rigor toward research advancement in the area of stem cell that is widely unexplored. The fact that stem cell therapies are mostly still under experimental stage and carried out as clinical trial without consistent success rate is noteworthy. While medical tourism does have its merit, promoting various legitimate and authorized treatments available globally but in the absence of regulation and oversight, unproven stem cell therapies are integrated by irresponsible parties to appeal to those who are desperate. The pro-medical tourism stand therefore induces tourist surge to Malaysia for stem cell-based intervention that are not accessible in some countries of the world but run the risk of being unsafe and unethical even. It is one of the reasons why stem cell tourism is considered a sub-set of medical tourism by this study. While private health care providers stand to profit by the pro medical tourism, it is important to review regulatory matters when expensive revolutionary and experimental intervention involving stem cell and aesthetic medicine are likely offered by the same private health care facilities exploiting those desperate.
Nowadays, the stem cell-based interventions are not only offered as a treatment for medical conditions and diseases alone but it has also crawled its way into aesthetic medicine as cosmetic therapy. You are now presented with “stem cell facelifts” for rejuvenation and anti-aging purposes within aesthetic medical providers, which are also promoted as a part of medical tourism (McArdle et al., 2014). It is clear that medical tourism industry unintentionally harbors immoral private health care providers offering all types of intervention in the name of medical tourism (including stem cell based) with ethical consequences that this study hopes to reveal.
Medical Tourism in Malaysia
In Malaysia, medical tourism went unnoticed until the 1990s, without emphasis on medical tourism or allocation of national budget for the purpose of improving health care for tourists in Malaysia. Tourism and the health care industry remained unrelated, progressing distinctly on its own up until then. In 1997, the rising number of tourists seeking medical treatments in Malaysia triggered the formation of the National Committee on Health Tourism by the Ministry of Health. It was an effort to expedite the progress of health tourism in Malaysia explicitly involving the private health care providers. The committee was one of the first step taken in support of the medical tourism. It comprised representatives from the Ministry of Health, Ministry of Tourism, the Association of Private Hospitals of Malaysia (APHM), and others (Malaysia Health care Travel Council [MHTC], 2018b).
The study by Noor Hazilah Manaf (2010) reported that the foreign exchange earned, based on the surge of tourists to Malaysia, has expanded at an average annual rate of 12.4%, with 17.3 billion tourists in 2000 to 50 billion in 2008. In 2005, the noteworthy rise in tourist visits increased Malaysia’s national income, equally boosting medical tourism as an industry. The Malaysia Health care Travel Council (MHTC, 2018b) was formed in 2009 as the official agency within the Ministry of Health to promote medical tourism in Malaysia. In the last 4 years between 2015 and September 2018, annually there have been some fluctuation in tourist visits as presented in Table 2. Although 2018 data are incomplete, the total number is expected to surpass the previous year judging by statistics.
Annual Tourist Arrivals Between 2015 and 2018.

Source. Industry performance report Tourism Malaysia (2018).
With that, Malaysia began to actively promote medical tourism with allocated budget addressing the matter first in their Eight Malaysian Plan (2001–2005), which is a 5-year national plan that comprehensively outlines the development policies and strategies of the Malaysian government (Malaysian Government, 2001). It continued to receive attention and budget in the 9th (2006–2020), 10th (2011–2015), and the current 11th (2016–2020) Malaysian Plan. In the 2018 budget tabling, the prime minister announced a Malaysian Ringgit 30 million as incentive to enhance the medical tourism industry indicating continual support (Malaysian Government, 2006, 2010).
A study by Neil Lunt and colleague (2014) verified that a majority of countries that involved in medical tourism has not regulated the industry with laws or policies. While that is true, Thailand and India have begun addressing visa concern, unlike Malaysia. The 90-day non-immigration “O-A” visa allows tourists to stay in Thailand for as long as the duration of their medical procedure and the medical e-visa that is specifically for tourists from more than 150 countries seeking medical procedure in India for a duration of 120 days, which are examples of small regulatory step in protecting medical tourists’ rights (Indian Ministry of Home Affairs, 2018; Thai Ministry of Foreign Affairs, 2018). Despite that, currently Thailand and India do not have legal framework meant to specifically regulate medical tourism or their stem cell technology. This is very similar to the practices in Malaysia.
Since the medical tourism in Malaysia involves the private health care providers, the Private Health care Facilities and Services (PHFS) Act (1998) is the only legislation that regulates these facilities (i.e., hospitals and clinics), which includes stem cell and aesthetic medical providers. The act focuses on the providers’ approval, license, registration, and inspection. It includes restriction and prohibition involving the premises, donor consent, advertisements, and penalty for non-compliances, but it does not explicitly disclose the procedure and practices involving medical services offered to tourists, stem cell or even aesthetic medicine (Ministry of Health [MOH], 1998). Generally, stem cell and aesthetic medicine in Malaysia are dealt as separate area of technology with only guidelines such as the Guideline for Stem Cell Research and Therapy (2009), National Guideline for Hematopoietic Stem Cell Therapy (2009), and Guideline on Aesthetic Medical Practice (2013) (MOH, 2009a, 2009b, 2013; Parliament of Malaysia, 2015). Currently, the wellbeing of medical tourist, medical visas, medical insurances, medical extradition, and others pertaining to medical tourism are not covered in Malaysia’s existing laws or policies. Any stem-cell-related therapies are carried out as clinical trials and approved by the National Stem Cell Research and Ethics (NSCERT) subcommittee based on the guidelines. Both stem cell and aesthetic medical providers (i.e., clinics and hospitals) are expected to conform to the Private Health care Facilities and Services (PHFS) Act (1998), while their physicians adhere to the Medical Act (1971). The Medical Act (1971) is meant to govern the medical practitioners conducts, regardless of the procedure they are offering and implicates both public and private sectors (Parliament of Malaysia, 2015). However, the Private Health care Facilities and Services (PHFS) Act (1998) and the Medical Act (1971) have no distinct provision on the matters involving medical tourism, stem cell, and aesthetic medicine. In the absence of specific regulatory framework and a transparent standard operating procedure (SOP), exploitations are potential and may complicate in the case of death of a tourist during a medical procedure. Although it is rare, it is not uncommon or impossible; hence, precautions are imperative.
In Malaysia, the potential of medical tourism industry is well documented. The destination competitiveness and quality service are identified as the main challenges of medical tourism industry (Sarwar, 2013). Some even examined the Muslim friendly market and the religious commitment in Islamic medical tourism industry (Iranmanesh et al., 2018; Rahman & Zailani, 2017; Sarwar, 2013). However, studies that documents the regulation and ethical issues involving medical tourism industry are exceptionally limited. The article “Medical Tourism. Concerns, Benefits, and the American Legal Perspective” by Burkett in 2007 is one such study, discussing the regulative concerns of medical tourism in the United States. However, the variability of how the issue impacts and dealt with in different countries urges for more comprehensive study involving them, such as this. (Burkett, 2007). Hence, this study examines the Malaysian medical tourism industry. It mainly aims to highlight the ethical and regulatory misconducts and exploitations involving the industry and also to demonstrate how the pro-medical tourism stance impacts stem cell tourism, particularly in the absence of a clear oversight, which no other paper has documented thus far. It hopes to draw attention to the fact that, although, stem cell tourism is not an explicit sub-set of medical tourism with completely different area of concerns, but since they are both services offered by the private health care providers in Malaysia, under similar regulative pattern (i.e., absence of specific regulatory framework), medical tourism does affect stem cell tourism.
Methodology
This is a qualitative study that incorporated both primary and secondary data analysis. The primary data were retrieved by conducting in-depth interviews of Malaysian policymakers within the different divisions (i.e., Medical Practicing Division, Stem Cell Research and Ethics subcommittee [NSCERT], and National Pharmaceutical Regulatory Agency [NPRA]) of the Ministry of Health using semi-structured questionnaires. A total of four (2 from the Stem Cell Research and Ethics [NSCERT] subcommittee, one from the Medical Practicing Division, and one from the National Pharmaceutical Regulatory Agency [NPRA]) participants were identified using purposeful sampling (Palinkas et al., 2015). The participants’ experiences and access to information pertaining to medical and stem cell technology practices in Malaysia facilitated a greater understanding of the medical tourism and stem cell technology that requires documentation. It is not uncommon for qualitative studies to employ smaller sample size to gather in-depth understanding through probing, and it is for this very reason, the study utilized a smaller sample size (Dworkin, 2012).
The secondary data consist of systematic literature review involving journal articles that was accessed through mainstream database and existing government documents. The journal articles were retrieved by executing a two-part keyword search, whereby the first search was conducted using the keyword “medical tourism” at the title section through Web of Science (WoS) for the period of 1997–2017. The search period chosen was to capture the point when medical tourism began receiving focus in Malaysia with the set-up of National Committee on Health Tourism in 1997 and up to the point the search was carried out which was 2017. The WoS database was considered sufficient based on its social science citation index (SSCI) to capture social science subject categories such as anthropology, sociology, bioethics and others (Web of Science, 2019). Although there are other social science databases such as ProQuest, WoS was considered sufficient to include all possible writings pertaining to this topic, published in medical, science and health care journals including social sciences. However, the accessibility of the database be one of the limitations of this study.
The resulting articles were then refined based on a second keyword using “stem cell” at the topic section. It was mostly to demonstrate that medical tourism and stem cell tourism are somewhat linked and addressed together, as it was highlighted previously by Edna F. Einsiedel and Hannah Adamson (2012). The relevant government documents were retrieved from the official portals and websites of various ministries in Malaysia (i.e., Ministry of Health, Ministry of Tourism, and others). The analysis of the primary and secondary data proved evident in answering key question, ‘does Malaysia’s medical tourism strategy promote stem cell tourism?’ which fulfills one of the objectives of this study. Although the literature reviews were adequate in integrating critical information regarding medical tourism and stem cell technology, the in-depth interviews proved necessary to verify some of the basic assumptions concerning Malaysia’s stem cell technology and its medical tourism strategy. The incorporation of mixed-method (i.e., in-depth interview and literature review) in this study served as triangulation of different sets of data to maximize convergence and validity of the research findings (Carter et al., 2014; Flick, 2018).
Results
The analysis of the in-depth interviews (primary data) and literature review (secondary data) revealed many aspects of medical tourism industry in Malaysia. It not only discovered the ethical and regulatory concerns but also presented how the medical tourism industry impacts stem cell technology. The primary data proved crucial to dismiss misconceptions regarding medical service and stem cell regulation. The secondary data were key in gathering the regulative conduct of medical industry in Malaysia and demonstrating the merits of examining medical tourism and stem cell tourism as interrelated concerns. Together, the data enabled a comprehensive understanding of the medical tourism phenomenon in Malaysia (Patton, 1999).
Database Keyword Search
The two-part keyword search was carried out using WoS database. The first search that employed the key word, “medical tourism,” resulted in a total of 210 articles. Many of the inquiries among the resulting article (from the first search) was very significant among authors based in the United States and may well coincide with their people traveling abroad seeking medical services and health care. Only 17 were written by Malaysian-based authors as presented in Figure 1, indicating limited enquiry despite the clear effort by the Malaysian government toward medical tourism. Among the resulting articles from the first search, two articles included “stem cell tourism” within their discussion (retrieved by the second keyword search), proving there is merit to address the two industry (medical and stem cell tourism) together. Although Google Scholar database using the same the keywords resulted in a total of 1,600 publications, but its extreme limitation was the reason it was not used.
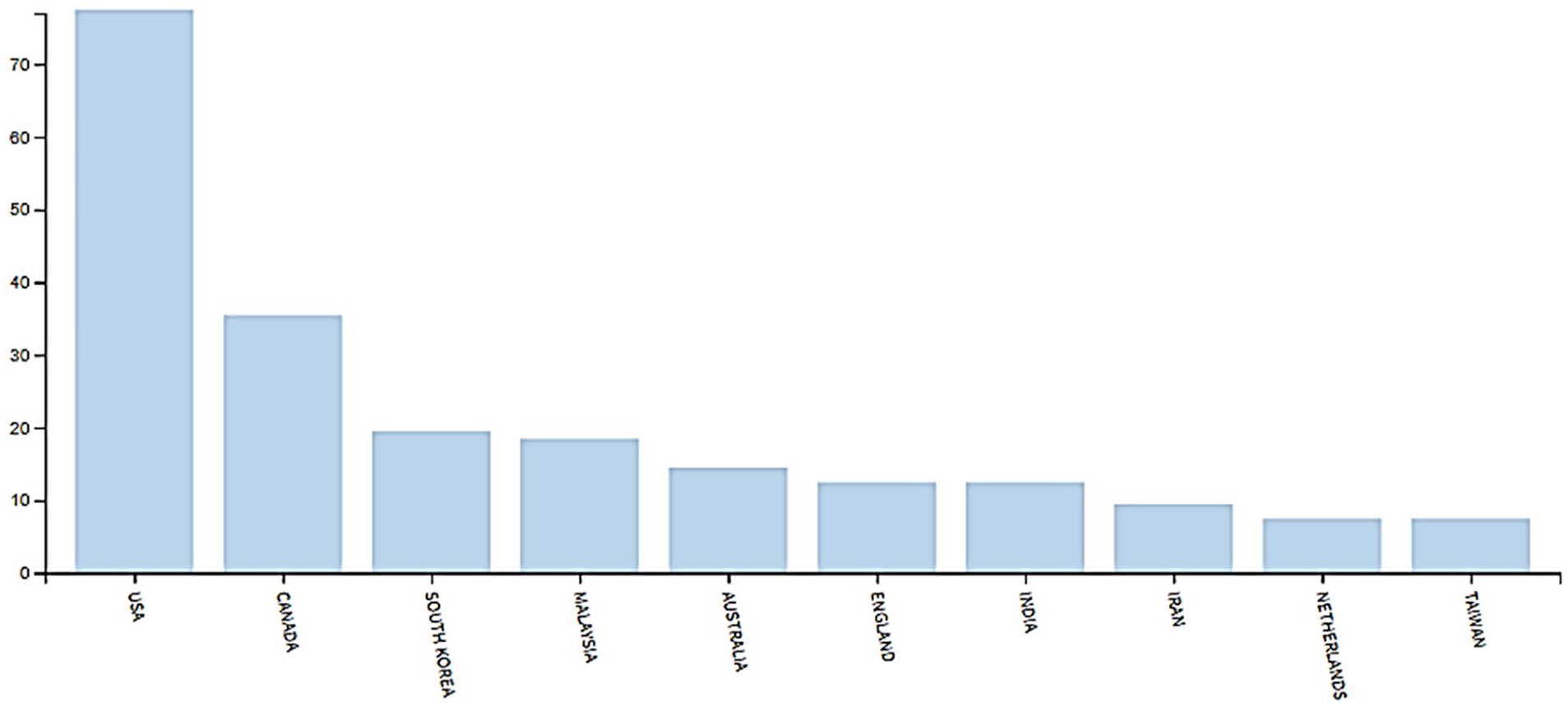
Articles written on medical tourism (1997–2017).
While studies continue to identify Malaysia as a top developing Asian destination for medical tourism, there are troubling aspects of the industry such as ethical consequences and regulation, which is not fully understood, divulged, or documented. While the question of how “touristy” medical tourism is and the pleasurable nature of the trip are all relevant inquiries, the implications of medical tourism in the absence of a law or policy that incorporates oversight measures in relation to health care services offered to tourists is an inquiry long overdue. Because without regulation, tourists’ welfare is constantly at risk and in the case of violation, their rights are left unprotected. Separately, the controversial nature of stem cell equipped with many unknown mechanisms often results in the tight regulation of their clinical trials in most developed Western countries such as the United States, the United Kingdom, and Australia. But in countries without oversight or legal framework, the medical tourism industry run the risk of promoting unproven stem cell therapy, whereby the participating private health care providers can make these experimental procedures equally accessible to tourists besides the common procedures promoted under medical tourism packages.
In Asia, the developing countries’ widespread privatized hospitals and clinics enable health care to be marketed as a commodity (“Minister,” 2018). Generally, a commodity is defined as valuable goods that can be traded, bought, or sold such as oil, wood, and even meat (Oxford, 2019). However, in developing countries, the income generated by the number of tourists visiting these countries seeking medical treatments are staggering to millions which governments and ministries are working toward establishing the medical tourism industry as a sustainable national revenue. The practice has turned health care and medical services within these countries from being a basic human right to being sold as a commodity-generating income.
Medical Tourism in Malaysia
Tourist arrival in Malaysia, since 2015, has been rather inconsistent as presented in Table 2. Despite the 2018 data being incomplete (up until September), the number is expected to surpass in 2019 compared to the previous year, based on the statistics. With the unsteady growth, there is a decline in tourists from many developed non-Asian countries visiting Malaysia and Table 3 presents the decrease for the United States, the United Kingdom, Canada, France, and Germany. However, surge of tourists from Asian countries namely China, Brunei, and South Korea to Malaysia has gradually increased between these years. Although tourist number from Singapore fluctuates, it is still identified as the country with the highest number of tourists visiting Malaysia annually.
Annual Tourist Arrivals of Selected Developed Non-Asian Countries Between 2015 and 2018 (January–September).

Source. Industry performance report Tourism Malaysia (2018).
Regardless of the fluctuation of tourists arriving in Malaysia, the statistics of tourists spending for medical and health care during their visits and those identifying medical or health care as the main purpose of their visits reported growth as shown in Table 4.
The Medical & Health Treatment Component in Tourism Malaysia.

Source. Industry performance report Tourism Malaysia (2018).
The expected turnover reports in the Ninth Malaysian Plan (2006–2010) was achieved and medical tourism was accepted as a new driver in service sector (Economic Planning Unit, 2005). While the 10th Malaysian Plan (2011–2015) listed tourism and private health care as number 5 and 10, respectively, within the National Key Economy Area (NKEA), it aimed to improve the revenue of medical tourism by 10% every year and eventually making Malaysia as the top destination in Asia (Malaysian Government, 2010). The current Deputy Finance Minister shared his conviction that the ministry’s policy of not charging the Sales and Service Tax (SST) for medical bills as a positive effort in bringing tourists into Malaysia compared to Singapore’s premium health care. As an effort to secure tourists from Indonesia, the Malaysian Health care Travel Council (MHTC) signed a memorandum of understanding (MoU) with the AirAsia Indonesia as the preferred airline providing extra service to those traveling, including arranging medical tourism trips and pick up services from the airport for doctors’ appointment and treatments (Sivanandam, 2018).
The efforts and initiatives supported by the Malaysian government, in 2018 the “International Living” magazine listed Malaysia as the fourth best health care in the world and the only Asian country (International Living, 2018). Malaysia also won the “Health and Medical Tourism: Destination of the Year” award three consecutive years between 2015 and 2017 (International Medical Travel Journal [IMTJ], 2018). In 2016, the medical tourism industry generated about MYR1billion with about 921,000 medical tourists, while in 2015 it generated about MYR900million with about 859,000 tourists (MHTC, 2018a). This indicates a 10% increase in medical tourism revenue between 2015 and 2016 fulfilling its national economic plan. In 2017, the industry is said have grown 14% bringing in MYR1.5billion with more than 1.2 million medical tourists. In 2019, a 25% growth which is equivalent to MYR1.8billion is expected with the anticipated increase of international tourists drawn in by the wide range of medical experts (i.e., cardiology, fertility, and cosmetic surgery) within the Malaysian private hospitals (Yunus, 2019). The medical tourism industry is expected to grow in 2020 with MYR2.8billion based on the large incentive allocated for Malaysia Health care Travel Council (MTHC) for its strategy. (“2018 Budget,” 2018; Rusman & Fiona, 2018). The overwhelming Malaysian ringgit undeniably supports that the medical tourism is in fact considered as Malaysia’s national commodity, just as it is in majority of developing countries, that promotes travel health care such as India and Thailand with limited gross national income. In these countries, medical tourism’s high turnover is essential to improve their economic diversity and development status by bringing in foreign trade and increasing job market (Turner, 2007). Judging by the ministries’ efforts, Malaysia seem to have adopted a rather supportive strategy making medical and health care fairly accessible and appealing to the target population.
The Dilemmas of Malaysia’s Pro-Medical Tourism Stance
Despite the profitable market gains and the economic impact of medical tourism, the debate if health care is a commodity or basic human right continues mostly in the developed countries especially in the United States. Ben Shapiro explains that health care is a commodity judging by the tight regulation of health care in the United States. He argued that regulation especially health insurance is what leads health care to being a commodity instead of basic human right (Shapiro, 2017a). He pointed out the two main qualities that represent something as public goods are non-excludable and non-rivalrous, which basically means that everyone is allowed to the service and that there is enough to go around which does not describe health care in the United States at all (Shapiro, 2017b).
Medical tourism, an inequitable choice
In Malaysia and other developing countries, the issue of health care becoming a commodity brings similar inequality concerns, but not just between the native and the medical tourists but among natives themselves. While the government continues to pump in billions to improve their health care system, they depend largely on private health care providers to sustain the medical tourism industry equipped with their luxurious health care facilities and spa-like service, somewhat disregarding the public sector. The Malaysian government is collaborating with the private sector and governmental agencies within Malaysia and abroad in marketing Malaysia as a medical destination. This diverts resources to the most profit-making industry involving tourists than the natives.
While the medical tourism initiative does revolve around tourists as its target consumer, the pursuit has caused the private health care treatments a privilege to middle and low-income Malaysian since it has caused price inflation based on general surge of tourists seeking health care in Malaysia. Generally, the expensive state-of-the-art equipment available within private health care facilities, employment of qualified experts needed to monitor and operate these equipment, the growing population demand for more resources with increased life expectancy in Malaysia have all been identified as factors that have contributed to the rise in medical cost in Malaysian private health care sector. Although public health care sector in Malaysia is satisfactory, the limited equipment, large crowds and extensive waiting period make public health care a formidable experience compared to private (Bernama, 2011). It is still the only available alternative for the average Malaysian without a medical insurance. A local insurer reported that the insurance penetration rate in Malaysia has not changed that much in the last 30 years with only 3.5 to every 10 Malaysians having some form of insurance, compared to one in every 10 Malaysian previously. It is said that only one-third of the Malaysian population have medical coverage, while close to 90% are underinsured (Insurance, 2018).
The private health care providers also blame the insurers for charging “administrative fee” apart from the hospital bills, causing a price inflation. According to these private health care providers, for a hospital bill of MYR2,000, the insurers impose MYR200 administrative fee that the providers are expected to bare themselves. While the insurers deny the claim, they blame the providers for the increase in hospital charges which evidently have resulted in higher insurance premium. They urged the government’s involvement in managing the cost of health care, instructing the private health care providers to be more transparent concerning the price of their product and services (FocusM Team, 2017). The blame between the insurer and the providers is a cruel cycle that the consumers are stuck in. In the last 4–5 years since 2016, the medical inflation has risen about 14% annually. Malaysia is said to have the third highest medical inflation rate among Asian countries based on a survey in 2017 (Wong, 2017). Hence, the private health care in Malaysia is quickly becoming inequitable among its population not only due to the substantial cost of medical treatments in private hospital compared to public as presented in Table 5 but as a result of the high medical insurance premiums in addition to weak regulation. The significant difference makes public hospitals and clinics the most practical choice for the average Malaysians.
Cost of Medical Treatments in Public and Private Hospitals in Malaysia.

Source. The RinggitPlus Team (2018).
While those in the developed countries have the option of traveling abroad for “affordable” treatment options, the poor (an average) within the developing countries continue to be marginalized with expensive choices. While Malaysians can travel to India for certain treatments (since they have the lowest price range as presented in Table 1), this is without factoring in the travel and accommodation expenses which many cannot afford. A study by Glenn Cohen (2015) on medical tourism identified that the health care experience of medical tourists and that of the general population of that country are not the same. The inequality that he pointed out was obscuring the term medical tourism in its relation to sustainability (Cohen, 2015). The governments of developing countries involved in medical tourism need to address the inequality of medical access to equally protect its population and medical tourist, disregarding profit-gains.
Stem cell tourism
While the official Malaysian medical tourism website is currently promoting the “Wellness Package,” a health screening test but any subsequent procedure or treatment that is necessary are at their very disposal within the private health care providers. This may easily include stem cell therapies which are widely theorized for its ability to treat diverse medical condition including those incurable, due to stem cell’s unique regenerative quality (Trounson & McDonald, 2015). Table 6 presents the number of private health care facilities offering stem cell therapies in Malaysia.
Private Health care Facilities Offering Stem Cell Therapies in Malaysia.

While there are 16 clinics and four hospitals that are licensed (based on the Medical Practicing Division’s “List of Licensed Private Health care Facilities and Services” as of 31st December 2017), their stem cell therapies are not. There are six clinics (including clinical centers) that are unlicensed but offering unproven stem cell therapies for a number of conditions based on the search through the National Medical Research Registry (National Medical Research Register [NMRR]). Stem cell therapies in Malaysia are considered proven, only if they are listed in the National Medical Research Registry (National Medical Research Register [NMRR]) particularly since they are only offered as clinical trials at the moment. According to the standard of Ministry of Health, all clinical trials are required to register within the National Medical Research Registry (National Medical Research Register [NMRR]), that enables public verification, before they can apply for the ethical approvals from the Medical Research and Ethics Committee (MREC) (2012). On top of that, clinical trials involving stem cell will require the approval of the National Stem Cell Research and Ethics (NSCERT) Subcommittee. Although it is difficult to view approval records of the National Stem Cell Research and Ethics (NSCERT) Subcommittee, the National Medical Research Registry (National Medical Research Register [NMRR]) is the best platform to verify.
Globally stem cell technology is controversial. The use of human embryos, creation of cells through cloning, and human experimentations have all triggered major ethical concerns worldwide (Foohey, 2010; Lo & Parham, 2009; Schmidt, 2013). Its clinical trials are expensive and are not easily come by causing those diagnosed with terminal conditions to seek experimental treatments abroad where stem cell technology is largely permissive or unregulated. According to Carolyn Brown (2012), many clinics in China, India, and Thailand are actively promoting their stem cell therapies to tourist. While Prasanna Kumar Patra and Sleeboom-Faulkner (2009) reported that tourists that travel to India for stem cell therapies are mostly from the upper-middle or middle-income category, there are diverse selection of clinics and doctors in Thailand advertising stem cell treatment making them available for all category of people. However, the cost-benefit ratio favors the providers than the patients. In low- and middle-income countries like Thailand, India, and Brazil, resources are limited and this enable operation without fretting malpractice suit or the need for follow-ups especially involving emerging technologies with unclear regulation (Finch, 2014).
These days, the medical tourists from developed countries with strict stem cell regulation can also consider Malaysia as a destination apart from India and Thailand, in the pretext of medical screening. They can easily seek out stem cell therapies considering their affordable cost and unclear regulation, although stem cell is not a part of the promoted package. This phenomenon is known as stem cell tourism, which is becoming a sub-set of medical tourism (at least in Malaysia) that equally attracts foreign tourists from countries where stem cell technology is extremely costly and strictly regulated to developing countries with weak oversight. A Malaysian business weekly wrote a piece on the medical tourism in Malaysia that identified a specific private health care provider and the stem cell therapy it offers as one of the main factors tourist surge to Malaysia (Yap, 2014). An article published in Malaysia Health care Travel Council (MHTC) about the development of stem cell treatment centers by Bioscience Americas in Central America especially Costa Rica, a destination widely known for medical tourism. The project is evident, how stem cell research and transplantation often cultivate in countries where regulation of the technology is either absent or rather laxed (MHTC, 2016). In Malaysia, the policymakers at the Ministry of Health verified that stem cell technology is currently remaining unregulated, like many developing countries promoting medical tourism such as India and Thailand: Stem cell technology is still unregulated, because we do not have laws. Regulations in Malaysia, you need an act or law—Medical Development Division (Ministry of Health).
This position has triggered many ethical concerns and exploitations that is apparent in Malaysia, Thailand, and India. The primary concern is the false-advertising and direct marketing of the stem cell therapies by some private health care providers which public fall prey to. Currently, there are several private health care providers in Malaysia that are marketing their stem cell therapies directly within their official websites, news portals, magazines, and social media, but based on these advertisements, it is difficult to verify if they have acquired proper approval not only for their therapies but their advertisements too (Ung, 2012). These private health care providers are marketing stem cell not as therapies or clinical trials, but under a different branding, allowing these treatments to go undetected without alerting the right authorities, exploiting the technicalities. Up until now, there are close to 30 private clinics and hospitals in Malaysia identified offering some form of stem cell therapy that are not listed in the National Medical Research Register (NMRR, 2017) despite involving human subjects. A particular private hospital only registered its clinical trial beginning 2018, although it was identified to have been offering stem cell therapies since 2012. There is also aesthetic medical providers (i.e., clinics and hospitals) that have incorporated stem cell–based procedures within their aesthetic product and services which not only the National Stem Cell Research and Ethics (NSCERT) sub-committee are not aware of but remains unregulated as it is not covered within the stem cell guideline nor listed in the National Medical Research Registry (National Medical Research Register [NMRR]). Due to the unclear regulation and weak oversight, a foreign stem cell entity successfully signed a memorandum of understanding (MoU) with the Malaysian state official to establish a rabbit breeding farm without the knowledge or approval of the Ministry of Health. The entity was selling the notion that their fetal precursor cell from rabbits were successful in treating Down syndrome (Mohamad, 2008). However, once the Ministry of Health came to know, their sample request for verification purpose was not entertained. The project did not advance and the entity left seeking business elsewhere, but the fact they succeeded in such partnership is not only concerning, but it is alarming knowing there is always a possibility (Mohamad, 2008). This is the second concern which the policymakers agreed: If private hospital wants to offer stem cell therapy, they need to attain approval from the Ministry of Health. The Ministry of Health need to go inspect (or audit) to ensure everything is ok before approval. But sometimes there are others who do not follow, obey or comply (for example, the private hospital that offered Down Syndrome therapy). Medical Development Division (Ministry of Health)
There are also concerns of premature publicizing of unproven stem cell products and therapies by private health care providers whom neglect the standard operating procedure (SOP) pertaining to stem cell technology that require pre-clinical evidence using a range of animals proving their efficacy and risk. This is identified as the third concern. While these profit-seeking entities falsely oversell their treatments as effective and successful, it is generally not the case, since majority of stem cell research and clinical trials still have inconsistent outcome (Tiwari & Raman, 2014). In India, some are even presenting bogus government approvals and declaration just to appear legit which is clearly a human rights violation. While the dubious business partnership between the Malaysian state officials and the foreign stem cell entity without attaining authorization is alarming, the radical technology that uses animal-based cells and tissues for human administration is known as xenotransplantation, that the entity was marketing, is actually prohibited in Malaysia. The prohibition of the technology is made by the Malaysian Fatwa, also known as the Islamic religious committee that offers advices and recommendation to the government regarding Islamic practices toward emerging technologies (MOH, 2009a). There are other entities that also market a diverse range of non-invasive stem cell wellness products imported from Japan, South Korea, and China such as face masks, lotions, creams, wellness drinks, without attaining approval from the National Pharmaceutical Regulatory Agency (NPRA). A product search through the Quest3+ search engine within the National Pharmaceutical Regulatory Agency (NPRA)’s official portal, verifies that many are not within the list of registered products which identified as the forth concern (National Pharmaceutical Regulatory Agency [NPRA], 2017)
While aesthetic medicine appears to be a separate issue not relating to stem cell, in Malaysia they are somewhat interrelated. This is because many aesthetic medical providers (i.e., clinics and hospitals) are incorporating stem cell in their aesthetic procedures to treat a range of conditions, from autoimmune disease, loss of stamina, healing after surgery or heart failure, reduce hyperpigmentation, hormone therapy to even hair loss. While aesthetic medicine similar to stem cell is only overlooked by a guideline, no legal framework bestows it mandate leaving the technology similar to stem cell, unregulated. The stem cell guidelines do not address the overlap issue (between aesthetic medicine and stem cell) nor is it captured by the National Stem Cell Research and Ethics (NSCERT) sub-committee. There is no review by National Pharmaceutical Regulatory Agency (NPRA) of these procedures or products leaving aesthetic medicine a gray area within stem cell technology. This is the fifth concern.
Unlike private sectors, stem cell research and clinical trials in the public sector (hospitals and research laboratories) have limited area for exploitation especially since their standard operating procedure (SOP) is more complexed with redundant bureaucracy requiring constant documentation and consent of the minister. There are even government circulars that are legally binding with clear instructions: Guideline is not binding, circulars are. In the circular by Dato Hassan, it is quite clear, as to what it permissible and what is not. (National Stem Cell Research and Ethics (NSCERT) Sub-committee member)
The ethical issues mainly implicate the private sector. In the absence of regulation and poor oversight, the pro-medical tourism stance of Malaysia and the incentives it actively backs for that purpose unintentionally fosters stem cell tourism too.
Legal Position of Medical and Stem Cell Tourism
Although medical tourism is marketed globally, there is no definitive regulation in that area in countries such as India, Thailand, and Malaysia. This puts medical tourists’ safety at risk undergoing experimental and unproven procedures within these countries (Crooks & Snyder, 2011). In Malaysia, despite the government’s broad effort in marketing medical tourism as a commodity with the involvement of different agencies, it has not begun to enact laws or exclusively regulate the industry. Studies have highlighted the lack of regulation as the major concern of medical tourism such as Thinakorn Noree et al. (2016) in Thailand and Indrajit Hazarika (2010) and Priya Shetty (2010) in India. A letter written by a Malaysian physician to a local news, expressing disappointment concerning the death of Leigh Aiple in 2014, an Australian whose expensive cosmetic procedures at a private hospital in Malaysia proved catastrophic. Although the letter was not written explicitly to call on the lack of regulation but to explain the gravity of the case from a medical viewpoint. The physician focused on the unacceptable number of surgeries and the lack of follow-up and emphasized the benefit of a regulatory policy that may have protected the stakeholders’ rights (Physician, 2018).
Regulation in Malaysia
Since the medical tourism largely implicates the private medical sector, in Malaysia the law or legislation that is in focus is the Private Health care Facilities and Services (PHFS) Act 1998. It was enacted by Ministry of Health to regulate and oversee the private health care providers including other health-based establishments concerning the issue of registration, licensing, violation, and non-compliance to the law and others (MOH, 1998). The Medical Practicing Division within the Ministry of Health verified that the Private Health care Facilities and Services (PHFS) Act 1998 is not meant to address stem cell–related matters exclusively: Because we are not going specifically on stem cell, we are going into the renewal of license. (Medical Practicing Division)
They also verified that it is within their responsibility to license the private health care providers (i.e., hospitals and clinics). Their duties include auditing the entities and their facilities, reviewing their treatment procedures, licensing the physicians and doctors, medical malpractice oversight, and ensuring everything conforms to the Private Health care Facilities and Services (PHFS) Act 1998, the Code of Professional Conduct, and the Good Clinical Practice (GCP) (MOH, 2009c). However, since there is no law or legislation exclusive on the matter concerning medical tourism, it is hard to verify how the regulators address the welfare of the medical tourists or protect their rights based on existing legislation. In the absence of such laws, medical malpractice involving tourists can be challenging and complexed with jurisdiction concerns despite balance regulation within the tourists own countries.
Currently, the stem cell technology in Malaysia is only overlooked by the Guideline for Stem Cell Research and Therapy (2009) that does not have legal mandate. In the absence of legislation and policy, stem cell technology remains unregulated and promote stem cell tourism in Malaysia allowing it to sustain without legal intervention. According to the guideline, any stem cell research or therapy (i.e., clinical trial) are required to follow the procedure in phases as presented in Figure 2. There is a slight difference between seeking approval for research for academic purpose and approval for clinical trials. Academic institutions, both private and public, are required to attain institutional level approval (i.e., institutional review board [IRB] and institutional ethics committee (IEC)) before securing the National Stem Cell Research and Ethics (NSCERT) sub-committee’s approval. While research within the private and public research laboratories (non-academic) require National Stem Cell Research and Ethics (NSCERT) sub-committee’s approval directly. The clinical trials and research involving human subjects within both private and public facilities will require a registration in National Medical Research Registry (National Medical Research Register [NMRR]) before securing National Stem Cell Research and Ethics (NSCERT) sub-committee’s approval. The National Medical Research Registry (National Medical Research Register [NMRR]) serves as a verification platform, which public can access for information. The ethical concerns identified earlier can either be due to unclear regulative protocol or simply exploited for personal gains since there are many isolated areas urging for improvements such as the use of stem cell in aesthetic medicine, the unproven stem cell therapies conducted without gaining the National Stem Cell Research and Ethics (NSCERT) sub-committee’s approvals and the issue of accountability which allow perpetrators to go unpunished since the guideline is not integrated to existing legislation like the Private Health care Facilities and Services (PHFS) act (1998) or new laws. The policymakers verified that the National Stem Cell Research and Ethics (NSCERT) sub-committee is unable to act without a formal complaint from the public, which is a serious matter and the sixth concern:
We do not have any statute or any aspect of our law that actually handles the stem cell research or transplantation. (NSCERT Sub-committee member)
As far as I know, there is no specific bill called stem cell bill. (Medical Practicing Division)
. . .but without any complaints from the public, the Ministry of Health cannot do anything. . . (Medical Development Division [Ministry of Health])

The protocol of stem cell research and clinical trial in Malaysia.
Our study proves that the pro-medical tourism stance of Malaysia does in fact foster stem cell tourism especially considering the regulatory deficiency that exists. If Malaysia continues to promote medical tourism disregarding regulatory control that laws and policies offer, there is nothing safeguarding the rights and wellbeing of the various stakeholders in the case of violation, misconduct, and even death. It is time, Malaysia dismiss the idea that regulation is only necessary when an industry intensifies because medical tourism and stem cell technology is no longer at its infancy. Increasing business or demand should not be the point when regulation is needed, instead it should be the primary focus and the initial design of any project, and not profit gain alone. The basic rights of the tourists whom contribute significantly to Malaysia’s national revenue should be safeguarded, either by making travel insurance compulsory to enter Malaysia especially for medical visits protecting their rights or to compel tourists to obtain temporary medical insurance for the duration of their visit. Since Malaysia does not actually require visa for visits, their visa on arrival is convenient for the tourists, but does not protects them during catastrophic events. However, in countries like Germany, Poland, Spain, Portugal and even several others, visa is a requirement and to attain the visa, tourists are expected to show proof of international medical insurance as a part of their application, especially coverage including medical evacuation and repatriation (Migration and Home Affairs, 2016). Health care and emerging technology such as stem cell in Malaysia have many issues that pleads for effective regulation. In the last two decades, while the Private Health care Facilities and Services (PHFS) act (1998) and stem cell guidelines was in place, exploitation continued. It is affecting the native population as well as medical tourists that this study has shown.
A national regulatory solution may be timely, but since medical tourism is a transnational industry operating across national borders, a transnational regulative effort between Malaysia and the governments of major tourist population visiting Malaysia is most appropriate. A study by Marie-Laure Djelic and Kerstin Sahlin-Andersson (2006) have defined transnational regulation as “a mode of governance in the sense that it structures, guides and controls human and social activities and interactions beyond, across and within national territories.” With the increasingly global medical tourism market, the regulatory task is also expected to adopt governance that is a co-operation between the involved governments also known as “inter-governmental co-operation.” Since majority of tourists visiting Malaysia are from Southeast Asian countries (i.e., Singapore, China, Brunei and South Korea and others) it is recommended that Malaysia initiate a transnational regulative effort through its ties with the Association of Southeast Asian Nations, also known as ASEAN which coinciding explicitly with its original aims and purposes. The traditional regulation has its deficiencies, not to mention the “difficulties to co-ordinate inconsistencies between standards,” to have transnational regulators such as ASEAN and the International Society of Stem Cell Research (ISSCR) (2016) to undertake the effort is crucial to initiate an inter-governmental co-operation. The Organization for Economic Co-operation and Development (OECD) have been tasked to “promote policies that will improve the economic and social well-being of people around the world” and this includes medical tourism. As case study by Fabrizio Cafaggi et al. (2013) on transnational regulation and water management published by OECD in the book International Regulatory Co-operation: Case Studies, Vol. 3 was quite informative and relevant in medical tourism as well.
Before that, the Malaysian ministries and agencies involved in medical tourism industry need to first work together to address the many areas within its current regulation that require substantial improvement to effectively regulate medical tourism industry and stem cell technology. The recommendations include that the Malaysian Ministry of Home Affairs to work closely with the Ministry of Health to evaluate the medical visa issue in relation to tourists’ wellbeing, similar to those exercised in Thailand and India. Currently tourists from the United States and Europe do not need visa to enter Malaysia. The stamp upon arrival allows them to stay for 90 days, which also apply to tourists from other countries. Although this is convenient for the tourists, over-stayers and international emergency services including extradition may not be accessible. The Ministry of Health also need to address the issue of inequity of medical access preventing price inflation in private sector that will be celebrated by the local population.
At the moment, the National Pharmaceutical Regulatory Agency (NPRA) have released the Guidance Document and Guidelines for Registration of Cell and Gene Therapy Products (CGTPs) in Malaysia (2016) which classifies stem cell therapy as a drug, a practice similar to India, whom adopted the “Guidance Document for Regulatory Approvals of Stem Cell and Cell Based Products (SCCPs)” (2014). The CGTP guideline integrates many existing legislations such as the Private Health care Facilities and Services (PHFS) act (1998), Medical Devise act (2012), and Control of Drugs and Cosmetic Regulations 1984 (CDCR 1984) and currently it is on voluntary basis. While it will bestow the stem cell guideline with legal mandate, it will come into full effect beginning 2021 and until then Malaysia is relying on professional integrity calling upon self-regulation which have proven futile all this while. While the CGTP (2016) addresses emerging technology, the Ministry of Health should place medical tourism regulation within the Malaysian Health care and Tourism Council (MHTC) oversight urging them to work closely with various local and international government, ministries, and agencies to establish transnational relation between Malaysia and countries with high tourist surge to Malaysia.
The tourists traveling to Malaysia and other developing countries ought to thoroughly research not only of the treatment they are seeking or the costs but the many risks involved in traveling abroad for any medical treatments such as the concern of its authorization, its legitimacy, its side-effects, and above all the standard protocol in the case of exploitation and emergency. Everyone hopes for a safe and successful trip, but when traveling to countries where regulation is nonexistent puts tourists at risk and in harm’s way without protection during medical complications (Killalea, 2016). Similar to Malaysia, many of these counties have yet to figure out the impact of weak regulation, nor have they undertaken any effort to resolve them. Therefore, while medical tourists exercise vigilance, a global intervention is necessary.
Conclusion
Medical tourism is a lucrative commodity in many developing Asian countries, including Malaysia. While the government is pleased with the boost in their national revenue, their continual financial support to improve private medical health care for the purpose of medical tourism continue to drive a wedge between average-income Malaysian and the above average. The inequality of medical access between these two groups is a serious concern. While those privileged can afford the treatment and spa-like service, the average Malaysian find it costly unless they have medical insurance. While medical insurance offered by insurance companies are paid monthly or annually, their range of packages is not a bargain nor afforded by all. Although public health care is satisfactory, their limited doctors, facilities, and lengthy waiting period makes private health care a better option for a faster diagnosis. The surge of medical tourists has also promoted price inflation among health care treatments and services at private health care facilities which the natives find extremely pricy.
Despite the inequality, the major concern of medical tourism in Malaysia is unregulated health care and medical procedures which is unintentionally promoting exploitative activities involving stem cell technology and aesthetic medicine which this study found. In the absence of laws and policies, it is apparent that the stem cell therapies are marketed under different branding allowing it to go undetected by the enforcers and business ventures for profit gain without proper licensing. While the Private Health care Facilities and Services (PHFS) act (1998) covers almost everything concerning private health care, it does not explicitly address types of treatments or services, nor protect medical tourists or address the challenges of inter-country complication should any unwanted event takes place. Based on our study, currently the available law proofs are deficient with many areas with possible exploitation. Hence, to effectively protect medical tourists and native populations from not only the neglects of medical tourism but also of stem cell tourism, the current laws in respective countries not only need revising but new laws or stipulations are necessary to address controversial issue involving stem cell technology, aesthetic medicine, and the medical tourism as a whole. This effort should tie in transnational missions to address the issue that has global risk factor, despite the profit gains.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
