Abstract
HIV incidence among young adult men-who-have-sex-with-men (YMSM) is among the highest in the United States (US), yet YMSM have lower rates of HIV testing than most other MSM. Among 1,835 U.S. Black, Hispanic, and White YMSM who reported condomless anal intercourse (CAI) in the prior year, 30% (95% confidence interval [CI]: [28%, 32%]) had not been tested for HIV in the past year as recommended by national guidelines, and 19% (95% CI: [17%, 21%]) had never been tested. Factors associated with not being tested in the past year included not having a primary care provider (PCP)) (odds ratio [OR]: 2.00, 95% CI: [1.53, 2.60]), discomfort with asking a PCP for an HIV test (OR: 2.66, 95% CI: [2.05, 3.44]), living in a smaller community (OR: 1.71, 95% CI: [1.35, 2.18]), younger age (OR: 2.00, 95% CI: [1.53, 2.60]), and greater self-perception of having an undiagnosed HIV infection (OR: 1.46, 95% CI: [1.07, 1.99]). YMSM who had not been tested in the prior year were less likely to know where to get tested, yet were interested in trying home-based HIV self-testing. This study shows that knowledge, clinician-relationship, geographic, and perceptional barriers must be overcome to improve HIV testing among YMSM. Home-based HIV self-testing may address some of these barriers, particularly for YMSM living in smaller communities.
Keywords
Introduction
In the United States (US), more than 25% of new HIV infections are diagnosed among adolescent and young adult (13- to 24-year-old) men-who-have-sex-with-men (YMSM; Centers for Disease Control and Prevention [CDC], 2017). Testing is necessary to identify those with an undiagnosed infection and is thus the first step in the HIV care continuum (Doll et al., 2018; Gardner et al., 2011; Johnson et al., 2008). Infrequent testing has been associated with late HIV diagnosis, which can lead to increased morbidity and mortality for those affected, and an increased risk of transmission to others (Cohen et al., 2011; Mannheimer et al., 2014). Despite a national emphasis on HIV testing among MSM (CDC, 2015; Cooley et al., 2014; Seth et al., 2017), 52% of newly diagnosed US MSM reported that they did not know or believe they had HIV (Stein, 2017). The CDC recommends HIV testing for MSM at least annually and more frequent testing based on individual risk, local HIV epidemiology, and individual and institutional testing policies (DiNenno, 2017). However, it is unclear if US YMSM at higher risk for HIV infection are being tested in accordance with these national guidelines.
Previous investigations estimating the frequency of prior year HIV testing among YMSM have several limitations, which have contributed to a wide range of estimates (20%–75%), partially attributable to variations in the age of participants included and differences in risk factors across studies (Cooley et al., 2014; Pierce et al., 2018; Seth et al., 2017; Veinot et al., 2016). Other limitations of prior studies include lacking a national scope (Daniels et al., 2018; Reilly et al., 2014; Tieu et al., 2016; Washington et al., 2015) and not accounting for HIV risk (McKenney et al., 2017; Ohl & Perencevich, 2011). CDC’s National HIV Behavioral Surveillance (NHBS) monitors sexual risk-taking and HIV testing behaviors among YMSM to obtain national testing estimates, but uses venue-based sampling in fewer than 25 major urban areas (Cooley et al., 2014; Gallagher et al., 2007; Whitham et al., 2018). As a result, data on HIV testing behaviors (including facilitators and barriers) and HIV risk among YMSM are lacking on a national scale, particularly for those living outside major urban areas.
Despite previous work suggesting the need for expanded testing initiatives (CDC, 2015), research examining possible methods to increase HIV testing among YMSM has been limited. Oral fluid home-based HIV self-testing is commercially available and approved for use in the US (Figueroa et al., 2018), and has been suggested as a way to increase testing rates and reduce barriers (Campbell et al., 2018; Hall et al., 2017; Maksut et al., 2016; Merchant et al., 2017a; Merchant et al., 2017b). Home-based HIV self-testing might improve testing access by decreasing geographical, logistical, and psychological barriers to HIV testing (Chiu & Young, 2016; Koblin et al., 2017; Maksut et al., 2016), and more effectively reach MSM who do not wish to disclose their sexuality (Koblin et al., 2017). Thus, home-based HIV self-testing, with ready access to test kits from stores and the internet, might be an appealing method to increase HIV testing among YMSM who live in smaller US communities who have reduced access to traditional HIV testing venues. In this investigation, we (a) examined how well HIV testing aligned with national CDC guidelines in a national sample of Black, Hispanic, and White YMSM from across the US who were at greater risk for an HIV infection from having condomless anal intercourse (CAI) within the previous year; (b) identified potential barriers to HIV testing among this higher HIV-risk population; and (c) measured interest in home-based HIV self-testing as one possible means of increasing HIV testing among this population.
Method
Study Sample
Between August and December 2014, we used social media to recruit English- or Spanish-speaking 18- to 24-year-old Black, Hispanic, and White YMSM living in the US who reported any previous CAI, self-identified as HIV negative, and provided consent to complete an anonymous online survey. This investigation was part of a larger study on HIV testing; the full survey has been previously published (Merchant et al., 2017b). Participants were recruited via multiple social media platforms, including OkCupid, Manhunt, Grindr, Bender, Facebook, Reddit, BlackGayChat, Growlr, and Pinterest (Merchant et al., 2017c). We limited this analysis to YMSM who reported CAI within the past year because they constitute a higher HIV-risk population for whom HIV testing is recommended at least annually by CDC guidelines (DiNenno, 2017). Rhode Island Hospital’s Institutional Review Board approved this study.
Measures
In addition to collecting demographic information, including age, race/ethnicity, educational attainment, and community size where residing (“large city or surrounding suburbs,” “medium city or surrounding suburb,” “small city,” “town,” or “rural area”), participants were asked when they last had CAI. Answer choices for recency of CAI were “less than a month ago,” “between 1 and 6 months,” “between 6 months and 1 year ago,” “between 1 year and 2 years ago,” “more than 2 years ago,” and “never.” Participants also were asked about the recency of their last HIV test (not including blood donation) using the same time frames. Other questions included primary care provider (PCP) access, comfort asking a PCP for an HIV test (responses collected on a 4-point scale as
Statistical Analyses
Spearman’s correlation was used to examine the relationship between HIV testing status and most recent CAI. Demographic characteristics, health access, and sexual history factors were compared by HIV testing status (tested within the past year, tested over a year ago, and never tested) using percentages and frequencies. Multivariable logistic regression was used to identify potential barriers to HIV testing, examining timing of last HIV test using three different criteria: (a) tested in past year versus those who were never tested or were tested over a year ago, (b) ever tested versus never tested, and (c) tested in past year versus tested over a year ago. The associations of the variables of race/ethnicity, age, community size, insurance status, primary care access, comfort with asking for testing from a PCP, and self-perceived risk of having an undiagnosed HIV infection with these outcomes were assessed.
The size of community of residence was dichotomized as large urban areas (large cities/surrounding suburbs) versus smaller communities (medium sized cities/surrounding suburb, small cities, towns, and rural areas). Participants who did not want to respond or answered “do not know” for any of the items were excluded from the multivariable models. Only 9% of participants were excluded in these analyses.
Using cross-tabulations, we also examined the relationships among HIV testing status, size of community of residence, knowledge of testing locations, and beliefs and preferences regarding home-based HIV self-testing, examining both the relationship between HIV testing status and testing location knowledge and preferences among the entire sample, as well as by size of community. We used Cochrane–Mantel–Haenszel tests to examine differences in the association between testing preferences and community size by HIV testing status (“never tested,” “tested within the past year” and “tested over a year ago”). Clopper Pearson exact 95% confidence intervals (CIs) were computed for all percentages and Fisher’s exact tests were used for comparisons with an alpha of 0.05. SAS 9.4 (SAS Institute, Cary, NC) was used for all analyses.
Results
HIV Testing History and Participant Demographic Characteristics
Among the YMSM who accessed the website and underwent eligibility assessment, 1,835 met criteria for this analysis, which was having had CAI within the prior year (Figure 1). Among these participants, 30% had not been tested for HIV in the past year as recommended by CDC guidelines, and 19% (345) had never been tested. There was no relationship between time of last CAI and most recent HIV test (

Participant enrollment.
Timing of Last HIV Test Versus Timing of Last CAI.
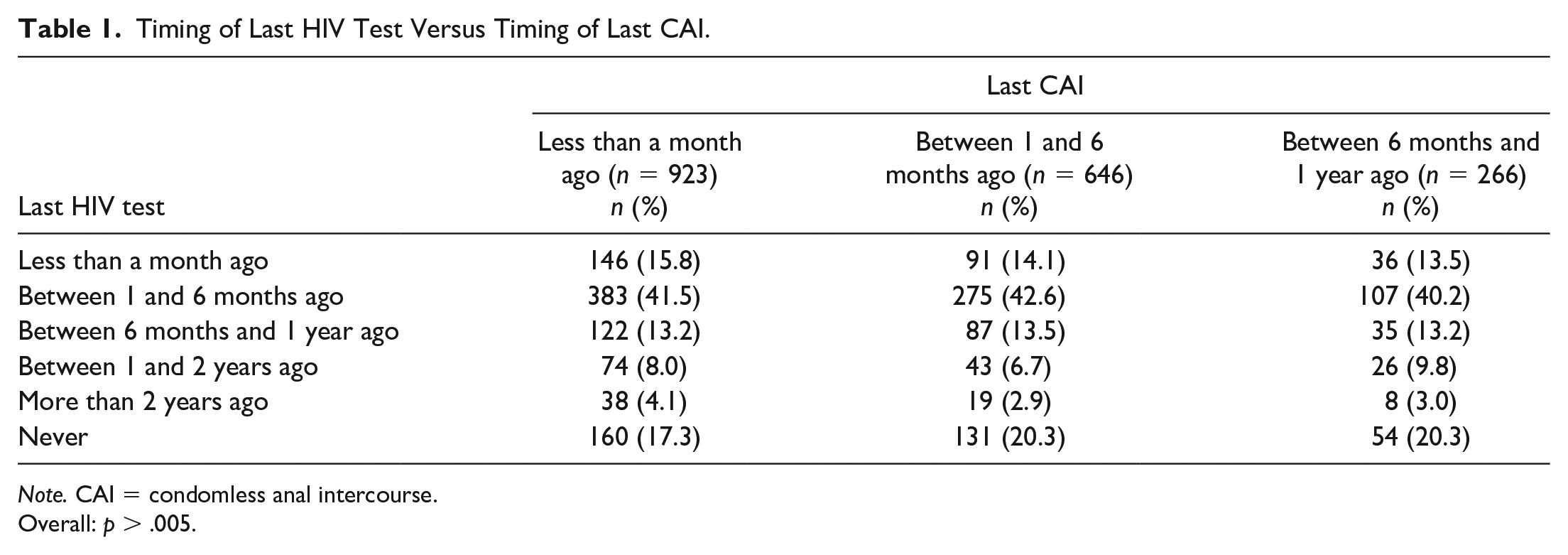
Overall:
Demographic, Health Access, and Sexual History Factors by HIV Testing Status.

Barriers to HIV Testing
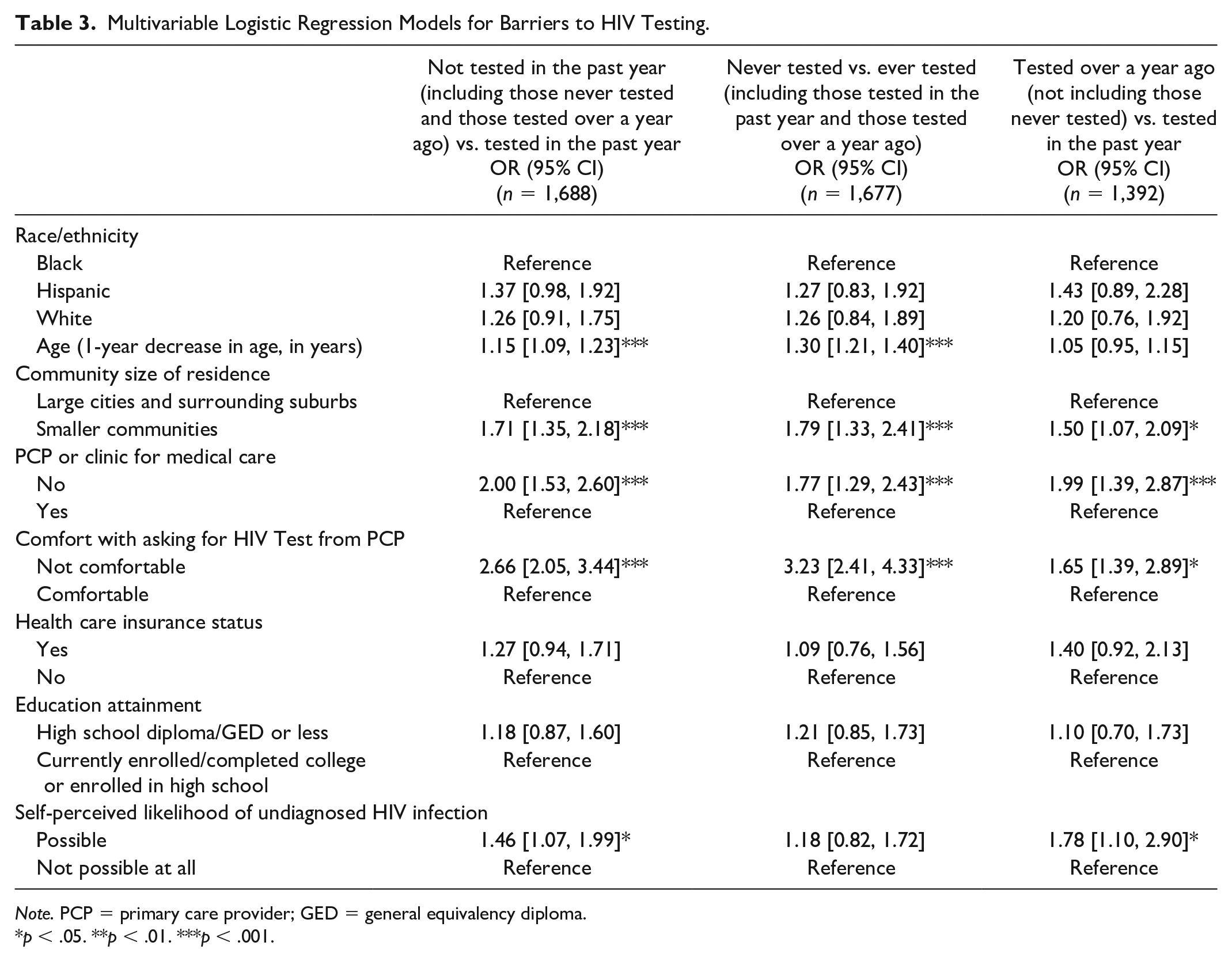
To understand the barriers to HIV testing in accordance with CDC guidelines, we first examined differences between those not tested versus tested in the past year. To better understand the impact of past testing experiences, we also examined differences between YMSM who had never been tested versus those who had ever tested; and between those tested over a year ago versus tested in the past year. Not having a PCP (odds ratio [OR]: 2.00, 95% CI: [1.53, 2.60]), discomfort with asking a PCP for an HIV test (OR: 2.66, 95% CI: [2.05, 3.44]), living in a smaller community (OR: 1.71, 95% CI: [1.35, 2.18]), younger age (OR: 2.00, 95% CI: [1.53, 2.60]), and greater self-perception of having an undiagnosed HIV infection (OR: 1.46, 95% CI: [1.07, 1.99]) were associated with not having been tested within the past year (Table 3). These findings were similar to those found in the comparison of YMSM who had never versus those who had ever been tested, with the exception there being no association between self-perception of undiagnosed HIV infection and HIV testing status (OR: 1.18, 95% CI: [0.82, 1.72]). The comparison of YMSM who had been tested more than a year ago versus those who had been tested in the past year yielded similar results.
Multivariable Logistic Regression Models for Barriers to HIV Testing.
Knowledge of HIV Testing Locations and Beliefs and Preferences for Home-Based HIV Self-Testing
We examined knowledge of HIV testing locations and beliefs about and preferences for home-based HIV self-testing as a function of testing status and residence, specifically comparing those living in large urban areas versus smaller communities (Table 4 and Supplemental Tables 1A, B, and C). Regardless of residence, YMSM were more likely to know of medical facilities (86%) rather than community-based organizations (65%;
HIV Testing Location Knowledge and Beliefs and Preferences for Home-Based HIV Self-Testing by Time of Last HIV Test and Community of Residence.

Tests comparing tested within past year, tested over a year ago, never tested: *
Tests between larger urban areas and smaller communities: †
Home-based HIV self-testing was a stronger motivator for testing both sooner and more often for those not tested within the prior year (sooner: 46%; more often 49%) and for those who had never been tested (sooner: 51%; more often 46%), when compared with those tested within the prior year (sooner: 30%,
Discussion
This evaluation of HIV testing practices and preferences of a racially and ethnically diverse national sample of higher HIV-risk US YMSM highlights the need for greater efforts to promote HIV testing among this population. Among the participants in this study, who were recruited nationally from primarily gay social networking sites/apps and were at higher risk for HIV acquisition due to CAI within the past year, only 70% had been tested for HIV within the prior year. This percentage is lower than the 75% reported by the NHBS in 2014 for 18- to 24-year-old YMSM (CDC, 2016). Unlike our study, the NHBS estimates testing rates using venue-based sampling in fewer than 25 major urban centers and includes MSM potentially at lower risk for HIV (CDC, 2016). The proportion of men ever tested in our study was higher than the 53% reported by YMSM in the American Men’s internet survey in 2014; however, their estimates included YMSM over a larger age range (15–24 years old) regardless of risk, recruited primarily from general social networking sites (Sanchez et al., 2016). Our relatively higher testing proportion may in part be due to greater recruitment through MSM-centric social networking sites, the exclusion of adolescents 15 to 18 years old, and inclusion of only those at higher risk for HIV acquisition due to CAI. Regardless of these differences between our study and other contemporaneous investigations, these studies indicate more work is necessary to improve HIV testing among YMSM in the US. Such work should include further education aimed at increasing testing knowledge while reducing testing stigma and increased access to testing options, including home-based self-testing (Bauermeister et al., 2018; Lelutiu-Weinberger et al., 2015).
Although we did not observe disparities in HIV testing status by race/ethnicity or health care insurance when controlling for other factors, we did uncover notable barriers to HIV testing. These barriers included smaller size of community where YMSM lived (possibly attributable to problems with testing access), not having an identified PCP/clinic for medical care, discomfort with asking PCP/clinician for an HIV test, and lack of knowledge of available testing locations. Health care access and comfort with providers are well known as factors influencing HIV testing frequency (Phillips et al., 2013). Geographic disparities in testing between rural and urban residents have been reported previously across all age groups of MSM (McKenney et al., 2017; Traynor et al., 2018). Our results also show that within the 18- to 24-year-old age group, testing behaviors vary considerably; younger MSM are much less likely to be tested. In addition, we found a troubling association of a lack of HIV testing being associated with greater self-perceived likelihood of a possible undiagnosed HIV infection. This association might reflect a complex problem of denial, lack of knowledge and insight, fear of testing results, alienation from the health care system in anticipation of bias (homophobia, racism, or both), or other factors (Arnold et al., 2014; Pharr et al., 2015; Pyun et al., 2014). In any case, further research and effective interventions are needed to address and overcome these barriers.
These results show variations in barriers to HIV testing among those never tested versus those tested over a year ago, suggesting the need for targeted interventions for these sub-populations, particularly for younger men who have never been tested for HIV. For the not recently HIV tested, interventions that address fears of being tested in light of self-perceived risk for a positive test appear necessary, which also has been suggested in other studies (Pharr et al., 2015). These interventions could include information dissemination campaigns, online or in person (Brennan et al., 2018; Hightow-Weidman et al., 2018). For both the never HIV tested and not recently tested, geographic constraints (living in smaller communities), having access to a PCP/clinic, and having the comfort level to ask for a test appear to be critical factors. Training clinicians and health care facilities to provide culturally competent care for YMSM and encourage HIV testing also are important parts of the solution to engaging this population (Campbell et al., 2018; Wilson & Moore, 2009).
Our results also suggest that increased awareness of local and other HIV testing options is needed. While we cannot discern from this study if lack of testing was primarily due to lack of knowledge of testing venues, the association between lack of knowledge of testing locations and lack of testing is concerning. As in previous studies, we did observe greater enthusiasm for home-based HIV self-testing among those not tested in accordance with national guidelines (Katz et al., 2018; Lightfoot et al., 2018; Sharma et al., 2014; Zhang et al., 2017). In addition to not necessitating access to traditional HIV testing venues, home-based self-testing does not require having a clinician nor asking a clinician for a test, which is one of the major barriers to testing (Branham et al., 2017; Frye et al., 2018; Levy et al., 2014; Qin et al., 2018). Both promotion and distribution of home-based self-testing can be facilitated online, eliminating geographic barriers (Wesolowski et al., 2018). If stated preferences match behavior, then home-based HIV self-testing might be helpful in reaching YMSM who have not been tested within the past year, have never been tested, or live outside of large urban areas.
We do recognize the limitations to the study. These limitations include concerns about the representativeness of this or any other sample of YMSM obtained through social media websites, and the inherent problems and ambiguity of self-report data. The responses obtained through social media might not be representative of all YMSM who need HIV testing in the US. Most of our participants were in college and had access to a PCP as well as health insurance; individuals lacking social media access may have different attitudes and access to care. Our sample size did not permit us to identify unique challenges faced by YMSM in small towns and rural areas, which could differ from those in medium-sized cities. Due to the focus in the investigation on Black, Hispanic, and White YMSM, the findings might not be applicable to YMSM of other racial/ethnic backgrounds or those living outside the US. Other barriers to HIV testing not explored in this investigation could also play important roles. Nevertheless, the findings support previous research indicating deficits in HIV testing among US Black, Hispanic, and White YMSM, and the need to overcome knowledge, clinician-relationship, geographic, and self-perception of HIV infection testing barriers, perhaps through home testing (McKenney et al., 2017; Reilly et al., 2014). As also would be expected, expressed preferences for type of testing might not match actual behavior; it is possible that those interested in home-based testing would not actually use it.
Conclusion
Almost one third of Black, Hispanic, and White US YMSM at higher risk for HIV due to CAI had not been tested for HIV in accordance with national guidelines. Knowledge, clinician-relationship, geographic, and risk perception barriers must be overcome to improve HIV testing in this higher risk population. Home-based HIV self-testing could help address some of these barriers, particularly for YMSM living in smaller US communities and those who are not being tested in accordance with national guidelines.
Supplemental Material
sj-docx-1-sgo-10.1177_21582440211016835 – Supplemental material for Barriers to HIV Testing and Opportunities for Expansion Using Home-Based HIV Self-Testing: Results of a National Study of Higher HIV Risk Young Men Who Have Sex With Men
Supplemental material, sj-docx-1-sgo-10.1177_21582440211016835 for Barriers to HIV Testing and Opportunities for Expansion Using Home-Based HIV Self-Testing: Results of a National Study of Higher HIV Risk Young Men Who Have Sex With Men by Sarah J. Marks, Roland C. Merchant, Melissa A. Clark, Tao Liu, Joshua G. Rosenberger, Jose A. Bauermeister and Kenneth H. Mayer in SAGE Open
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by a grant from the National Institute on Nursing Research, R21NR013869. Dr. Liu’s work on this project was partially funded by the Providence/Boston Center for AIDS Research (P30AI042853).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
