Abstract
Objectives:
Sepsis is a serious medical condition caused by the body’s systemic inflammatory response to infections. The antimicrobial peptides, human beta-defensins, play a key role in modulating host immune responses, and aberrant expression of human beta-defensins has been implicated in many infections and inflammatory diseases. However, little is known about the expression of human beta-defensin-3 in systemic infectious diseases.
Methods:
We investigated the gene expression and protein level of human beta-defensin-3 in peripheral whole blood from 107 participants—67 patients with sepsis and 40 healthy controls—and evaluated the feasibility of human beta-defensin-3 as an indicator for sepsis. Total RNA was extracted from peripheral blood samples, and relative mRNA expression of human beta-defensin-3 was determined by reverse transcription-quantitative polymerase chain reaction. Plasma concentration of human beta-defensin-3 was measured by enzyme-linked immunosorbent assay. Pearson’s correlation analysis was performed to assess the relationship between human beta-defensin-3 mRNA and protein levels. Receiver operating characteristic analysis was performed to evaluate the value of human beta-defensin-3 as a biomarker for sepsis.
Results:
Human beta-defensin-3 mRNA expression was significantly downregulated in sepsis patients compared to controls (p = 0.001). The mean fold change of mRNA expression (±standard error) was 0.82 ± 0.63 in sepsis patients and 1.39 ± 1.09 in controls. Plasma concentration of human beta-defensin-3 (pg/mL) was significantly lower in sepsis patients compared to healthy controls (p = 0.039). The mean protein concentration (±standard error) was 539.6 ± 39.4 in sepsis patients and 715.5 ± 53 in controls. There was a significant correlation between human beta-defensin-3 mRNA expression and the corresponding protein level in sepsis patients (r = 0.358, p = 0.04), but not in healthy controls (r = 0.124, p = 0.51). For discriminating sepsis patients from healthy controls, the area under the receiver operating characteristic curve was 0.722 (95% confidence interval: 0.597–0.847, p = 0.002) for human beta-defensin-3 mRNA and 0.689 (95% confidence interval: 0.557–0.827, p = 0.009) for human beta-defensin-3 protein.
Conclusion:
This is the first study to show the downregulation of human beta-defensin-3 gene expression and protein level in sepsis, which may contribute to the complex immunological imbalance in sepsis. The significant correlation between human beta-defensin-3 mRNA expression and protein concentration suggests that mRNA expression could be used to predict protein level. Our study also showed a potential role of human beta-defensin-3 as a blood-based biomarker for sepsis. More studies on the clinical significance of human beta-defensin-3 in sepsis could further support a biomarker development.
Introduction
Sepsis is defined as a life-threatening organ dysfunction caused by the dysregulated host response to infection.1–3 It is characterized by a systemic inflammatory response syndrome (SRIS) in addition to a documented or presumed infection.4,5 Sepsis is among the most common causes of death in hospitals and may progress to severe sepsis and septic shock if the inflammatory process is left untreated.4,5 Despite its fatal consequences, the use of appropriate antibiotics, immediate fluid resuscitation, and intensive care are not sufficient, and the incidence of sepsis is strikingly on the rise.6,7 Antimicrobial-resistant, which has become an alarming threat to public health, contributes to the significant increase in sepsis mortality.8,9
Antimicrobial peptides (AMPs) are small cationic class of naturally occurring amphiphilic peptides and are considered an essential component of the host defense in all multicellular organisms. 10 These peptides have been recognized for their function as new antibiotics due to their antimicrobial, anti-inflammatory, and immunostimulatory qualities.10,11 One of the major groups of AMPs is the defensin family, which vitally contributes to the host immune response by providing a first-line host defense against pathogenic microorganisms. 12 In humans, defensins are divided into alpha and beta-defensins according to their molecular structure and the difference in the position and connectivity of cysteine disulfide bonds. Human alpha-defensins are found in neutrophils, macrophages, and Paneth cells in the intestine, whereas human beta-defensins (hBDs) are secreted by most leukocytes and epithelial cells. 12 Eight members of the beta-defensin family occur in a cluster on chromosome 8p23.1. 13 Human beta-defensin-3 (hBD3) is encoded by the DEFB103A gene and is highly expressed in many human epithelial tissues. The expression hBD3 can be also induced by cytokines or microbial stimuli. 14 hBD3 was first isolated as a 5-kDa nonhemolytic AMP from human psoriatic scale lesions. 15 In recent years, hBD3 has been recognized as an important part of innate immunity due to its antimicrobial activity toward gram-negative and gram-positive bacteria as well as fungi and some viruses.14,15 In addition, hBD3 contributes to the adaptive immune response through its immunomodulatory properties such as chemoattraction of T-lymphocytes and immature dendritic cells. 16 Compared to other beta-defensins, hBD3 is unique in its salt-insensitive broad spectrum activity against several pathogenic microorganisms at physiologic salt concentrations, which makes this peptide of biological significance and particularly relevant in diseases. 17 Abnormal expression of beta-defensins has been associated with sepsis and various infectious diseases. For instance, the levels of beta-defensins are elevated in plasma and bronchoalveolar lavage fluid in patients with pulmonary infections.18–20 Whereas human beta-defensin-2 (hBD2) expression is decreased in burn wound, 21 hBD2 transcripts is suppressed in peripheral blood cells of patients with sepsis. 22 In recent studies, induced hBD3 expression has been reported in the intestinal tract of children with chronic inflammatory bowel disease, 23 and increased hBD3 peptide has been described in biopsies from terminal ileum of patients with Crohn’s disease. 24 Moreover, a great reduction in hBD3 level has been found in the gingival crevicular fluid of patients with periodontitis. 25 These studies highlight the involvement of hBD3 in systemic diseases. However, no previous studies have investigated the role of hBD3 in sepsis. Moreover, the key role of host defense peptides in innate and adaptive immunity, and their differential regulation in infectious and inflammatory conditions make them attractive disease biomarkers.
Therefore, the aim of the current study was to examine the gene expression and protein level of hBD3 in peripheral blood samples from patients with sepsis and healthy control individuals, and to evaluate the feasibility of hBD3 as a biomarker for sepsis.
Materials and methods
Study participants
This case-control study included 107 participants divided into the following groups:
Patient group, which included 67 patients with sepsis, was diagnosed and treated in the intensive care unit (ICU) at Salmaniya Medical Complex in the Kingdom of Bahrain. The diagnosis of sepsis was performed based on the criteria of the American College of Chest Physicians/Society of Critical Care Medicine Consensus Conference Committee (ACCP/SCCM).1–3
Control group, which included 40 individuals with no history of infections and inflammatory diseases, was enrolled from Salmaniya Medical Complex Blood Bank, Kingdom of Bahrain.
Calculation of sample size was made as the minimum number of participants in each group with sufficient statistical power (two-sided significance at 0.05 with a power of 80%) to detect a 20% difference between the mean hBD3 mRNA expression and protein level in the two groups.
All participants were informed clearly of the nature of this study. Informed consent was obtained from all patients and control participants under a protocol that was approved by the Research and Ethics Committee of the College of Medicine and Medical Sciences, Arabian Gulf University, Kingdom of Bahrain.
Blood sampling
Peripheral whole blood samples were collected from the participants by venipuncture in ethylenediaminetetraacetic acid (EDTA) tubes. For patients with sepsis syndrome, blood samples were drawn on the day of diagnosis, before any kind of available treatment. Plasma was separated by centrifugation for 15 min at 1000× g (at 2°C–8°C) within 30 min of blood collection. Plasma was aliquoted and stored at −80°C until use.
RNA extraction
The QIAamp RNA Blood Mini Kit (Qiagen, MD, USA) was used for RNA extraction following the manufacturer’s procedures. All RNA samples were treated with DNase (RNase-free DNase Set; Qiagen) to remove traces of genomic DNA. The concentration of the RNA was measured using a NanoDrop™ 1000 Spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA), and optical density (OD) 260/280 and 260/230 ratios were obtained to determine the purity of the RNA. The integrity of the RNA was assessed using agarose gel electrophoresis.
cDNA preparation
Total RNA (2 µg) was reversed transcribed into cDNA using a high capacity cDNA reverse transcription (RT) kit (Applied Biosystems, CA, USA) following the manufacturer’s instructions. Briefly, 10 µL of RNA was mixed with 10 µL of 2× RT Master Mix (containing 2 µL RT Buffer, 0.8 µL deoxyribonucleotide triphosphate (dNTP) Mix, 2 µL RT random primers, 1 µL MultiScribe™ reverse transcriptase, 1 µL RNase inhibitor, and 3.2 µL nuclease-free water) to a total volume of 20 µL. The RT reaction was incubated for 10 min at 25°C, followed by 2 h at 37°C and 5 min at 85°C. All cDNA samples were stored at −20°C for further analysis.
Reverse transcription quantitative polymerase chain reaction
The expression of the target gene (hBD3) was measured relative to the expression of a reference gene (human beta-actin (hB-actin)) in the blood of sepsis patients and healthy controls using reverse transcription quantitative polymerase chain reaction (RT-qPCR). Primers for the hBD3 gene (DEFB103B) and for a reference gene (ACTB) were designed based on sequence information in GenBank using NCBI primer design (Table 1). cDNA (30 ng) was mixed with SYBR Green 1 probe, forward and reverse primers (50 ng/µL), and nuclease-free water to a total volume of 10 µL. RT-qPCR was carried out using the 7900HT Fast Real-Time PCR system (Thermo Fisher Scientific). All experiments were run in duplicate under identical experimental conditions with the following cycling conditions: 95°C for 10 min, followed by 95°C for 15 s and 60°C for 60 s for a total of 40 cycles. A no-template control was included in each polymerase chain reaction (PCR) run. Results were analyzed using Sequence Detection Software, version 1.7 (Applied Biosystems; Thermo Fisher Scientific). Crossing of threshold (Ct) values obtained for the target gene were normalized against the reference gene values. Relative quantification of PCR was determined using the 2–ΔCT method. 26
Primers used in RT-qPCR.

RT-qPCR: reverse transcription quantitative polymerase chain reaction; hB-actin: human beta-actin.
Enzyme-linked immunosorbent assay
The protein concentration of hBD3 was measured in sepsis patients and control participants using commercial DEFB103A ELISA (enzyme-linked immunosorbent assay) kit purchased from MyBioSourse (CA, USA,), according to the manufacturer’s instructions.
Statistical analysis
The SPSS software (version 23; IBM Corp., Armonk, NY, USA) was used to analyze the data. Differences in hBD3 mRNA and protein levels as well as clinical data between cases and controls were conducted by χ2 or Student’s unpaired t-test, and presented as the mean ± standard error (SE) or the mean ± standard deviation (SD). The relationship between hBD3 mRNA expression and protein level was assessed using Pearson’s correlation analysis. The value of hBD3 as a disease biomarker was evaluated using receiver operating characteristic (ROC) analysis. The area under the ROC curve (AUC) and 95% confidence intervals (CIs) were obtained; p < 0.05 was considered to indicate a statistically significant difference.
Results
Characteristics of study participants
This study included 107 individuals, 67 patients with sepsis and 40 healthy controls. The basic characteristics of study participants are shown in Table 2. The group of sepsis patients consisted of 30 males and 37 females with a mean age of 35 (SD = 19), whereas the group of healthy controls consisted of 35 males and 35 females with a mean age of 33 (SD = 9.2). There were significant differences in sex distribution and mean age between the two groups (p < 0.05). According to the criteria of ACCP/SCCM, the clinical data of 14 out of 67 sepsis patients revealed septic shock, with persisting hypotension requiring vasopressors to maintain mean arterial pressure (MAP) ⩾65 mm Hg and having a serum lactate level >4 mmol/L despite adequate volume resuscitation. The most observed types of microorganisms were Klebsiella spp. followed by Candida spp. and Staphylococcus spp. The most observed pathogenic condition in sepsis patients was chronic renal diseases (n = 10), followed by pneumonia (n = 7), acute respiratory distress syndrome (n = 4), and neurological complication (n = 4). The death rate due to sepsis syndrome was up to 17.5%.
Basic characteristics of study participants.

Data are presented as numbers (n) for categorical data or mean ± standard deviation for parametrically distributed data.
p < 0.05 compared to the healthy controls.
mRNA expression analysis
Using RT-qPCR, the expression of hBD3 gene was determined relative to the expression of a reference hB-actin gene in peripheral blood of patients with sepsis and healthy controls. As can be seen in Figure 1, the mRNA expression of hBD3 was significantly lower in sepsis patients as compared to healthy controls (p = 0.001). Sepsis patients exhibited a reduction in hBD3 mRNA expression by a 1.7-fold compared to healthy controls. The mean ± SE was 0.82 ± 0.63 in sepsis patients and 1.39 ± 1.08 in controls.

Relative expression of human beta-defensin-3 (hBD3) mRNA in sepsis patients and healthy controls. The expression of hBD3 mRNA was quantified relative to human beta-actin (hB-actin) by reverse transcription quantitative polymerase chain reaction (RT-qPCR) in the peripheral blood of patients with sepsis (n = 67) and healthy controls (n = 40). The boxplot data are presented as the mean fold change of hBD3 mRNA expression ± standard error (SE).
Protein concentration analysis
Using ELISA, hBD3 plasma protein concentration was measured in the peripheral blood of sepsis patients and healthy controls. As shown in Figure 2, hBD3 protein concentration (pg/mL) was significantly lower in sepsis patients compared to healthy controls (p = 0.039). Sepsis patients displayed a reduction in hBD3 protein level by a 1.32-fold compared to healthy controls. The mean ± SE was 539.6 ± 39.4 in sepsis patients and 715.5 ± 53 in controls.

Protein concentration of human beta-defensin-3 (hBD3) in plasma of sepsis patients and healthy controls. hBD3 protein concentration (pg/mL) was measured by enzyme-linked immunosorbent assay (ELISA) in the plasma of sepsis patients (n = 67) and healthy controls (n = 40). The boxplot data are presented as the mean ± standard error (SE).
Correlation analysis
We next compared the mRNA expression of hBD3 with the corresponding protein level in the peripheral blood of sepsis patients and healthy controls using Pearson’s correlation analysis. The results showed a significant correlation between mRNA expression and protein concentration of hBD3 in sepsis patients (r = 0.358, p = 0.04), whereas no significant correlation was found between mRNA and protein levels of hBD3 in the healthy controls (r = 0.124, p = 0.51).
ROC analysis
ROC analysis was performed to evaluate the value of hBD3 as a biomarker for sepsis. From the ROC analysis, the AUC and 95% CIs were obtained. The results showed that the AUC of hBD3 mRNA was 0.722 (95% CI: 0.597–0.847, p = 0.002; Figure 3). Moreover, the AUC of hBD3 protein was 0.689 (95% CI: 0.557–0.827, p = 0.009; Figure 3).
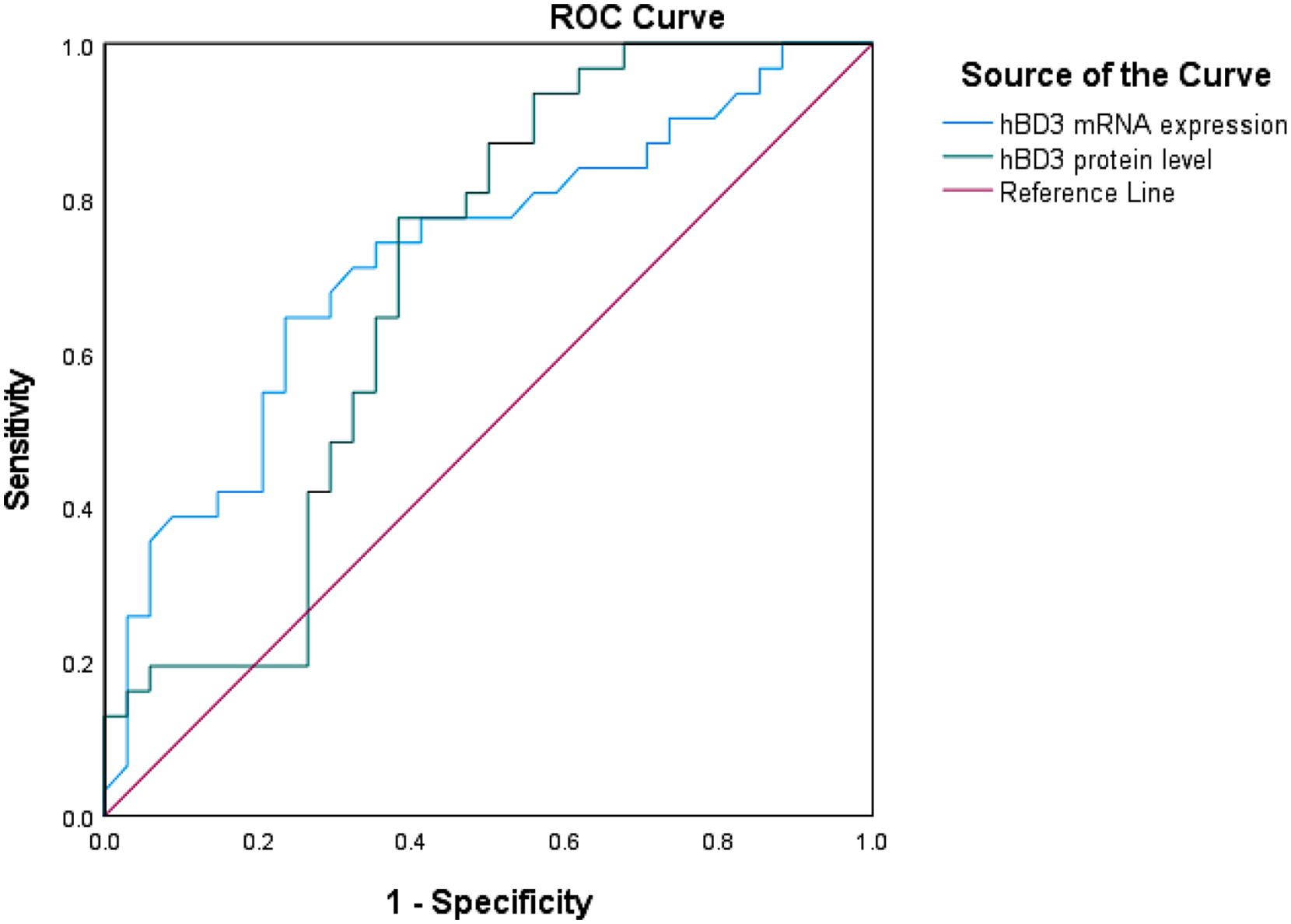
Receiver operating characteristic (ROC) curves of human beta-defensin-3 (hBD3) mRNA expression and protein concentration. The area under the ROC curve (AUC) of hBD3 mRNA expression to distinguish between sepsis patients and healthy controls (blue line) was 0.722 (95% confidence interval (CI): 0.597–0.847, p = 0.002). The AUC of hBD3 protein concentration to distinguish between sepsis patients and healthy controls (green line) was 0.689 (95% CI: 0.557–0.827, p = 0.009).
Discussion
The host defense hBDs are a group of AMPs that contribute to the innate immunity via their antimicrobial activities toward many microorganisms.14,15 With their chemotactic for immature dendritic cells and memory T cells, hBDs also participate in the adaptive immune response. 16 hBDs are among the natural endogenous AMPs, which are rapidly released in many infectious and inflammatory diseases,11,12,14 and altered their expression is implicated in the pathogenicity of many diseases. Among several members of hBD family, hBD3 has attracted recent attention in relevance to diseases due to its significant salt-independent potency toward several pathogenic microorganisms at physiologic salt concentrations. 17
However, little is known about the expression of hBD3 in systemic infectious diseases such as sepsis. Therefore, in this study, we investigated the gene expression and protein level of hBD3 in the peripheral blood of patients with sepsis and healthy control individuals, and evaluated its potential role as a biomarker for sepsis.
We found a significant reduction in hBD3 mRNA expression and protein level in sepsis patients compared to controls, suggesting an important effect of systemic infections on hBD3 gene expression and protein level. When we analyzed the relationship between hBD3 mRNA and protein level in sepsis patients and healthy controls independently, we observed a significant correlation between hBD3 mRNA expression and protein concentration in sepsis patients. However, we did not find a significant correlation between hBD3 mRNA protein levels in the healthy controls. These results suggest that the transcript level of hBD3 gene can be used as a representation for the corresponding protein level.
Changes in hBD3 have been reported in several infectious and inflammatory conditions. For instance, altered hBD3 expression has been found in children with chronic inflammatory bowel disease23,24 and in gingival crevicular fluid of patients with periodontitis. 25 Moreover, hBD3 serum concentration has been suggested to have an immunomodulatory role during chronic inflammation. 24 In recent studies, overexpression of hBD3 has been also observed in oral squamous cell carcinomas, Kaposi’s sarcoma, and cervical cancer,27–30 indicating a role of this peptide in human cancers.
In sepsis, only few studies have examined the role of some hBDs. Book et al. 22 reported a significant reduction in hBD2 mRNA expression in patients with severe sepsis compared to healthy controls and nonseptic critically ill patients. In addition, high hBD2 plasma levels were reported in children with severe sepsis, 31 whereas lower hBD2 serum levels in cord blood of preterm neonates were found to be associated with late-onset sepsis. 32 However, there are no previous studies on the role of hBD3 in sepsis, to the best of our knowledge, and the present study is the first to examine the gene expression of hBD3 in this complex pathological condition. In our study, the downregulation of hBD3 may imply that the innate immunity is impaired due to decreased antimicrobial activity and immunomodulating effects of hBD3.
The variability of signs and symptoms of sepsis which are influenced by many factors such as virulence and bioburden of pathogens and host susceptibility contributes to the complication of sepsis diagnosis and estimation of its severity. 7 This life-threatening disease requires immediate treatment, and early identification of high-risk patients is important to initiate the treatment strategies. Biomarkers are highly desirable for early detection, which may allow timely and early intervention to reduce the risk of death.33,34 In this regard, several biomarkers have been evaluated in sepsis such as procalcitonin (PCT) and C-reactive protein (CRP). However, none of the evaluated biomarkers have sufficient ability to distinguish sepsis from other inflammatory conditions or to predict the disease outcome.33,34
The host defense peptides can be attractive disease biomarkers, due to their vital roles in the innate and adaptive immunity as well as their differential regulation in infectious and inflammatory conditions. In this context, saliva AMPs have been proposed as potential biomarkers for periodontitis. 35 Recent studies have also investigated serum hBD2 level as a biomarker for disease activity and monitoring treatment response of psoriasis.36,37 Furthermore, human hBDs which are differentially expressed in tumors may serve as cancer biomarkers.30,38 In the present study, ROC curve analysis showed significant and fairly good values of hBD3 mRNA expression and protein level to distinguish between sepsis patients and healthy controls, suggesting the potential use of hBD3 as a biomarker for sepsis. One of the main limitations in our study is the small sample size, which may limit the statistical power, and a larger prospective studies are required to confirm these results. In addition, the present study focused exclusively on gene expression and protein level of hBD3 and did not investigate other defensins. Nevertheless, studies in our laboratory are underway to examine the significance of hBD1 (human beta-defensin-1), hBD2, and other beta-defensins as well as cytokines in sepsis patients. Finally, although the results of the present study demonstrated that peripheral blood hBD3 is differentially expressed between sepsis patients and healthy controls, the clinical application of hBD3 in sepsis requires further investigation and optimization.
Conclusion
The present study showed for the first time the downregulation of hBD3 gene expression and protein level in peripheral blood of patients with sepsis. The downregulation of hBD3 may contribute to the complex immunological imbalance in sepsis patients. The significant correlation between hBD3 mRNA and protein levels suggests that mRNA expression could be used to predict protein level. Our study also showed a potential role of hBD3 as a biomarker for sepsis. Further validation of these results may open a new door toward the development of hBD3 as a novel blood-based biomarker for sepsis.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from Research and Ethics Committee of the Arabian Gulf University, Bahrain (R1-E26-PI-01/15).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This is a self-financed work as a part of an MSc thesis.
Informed consent
Written informed consent was obtained from all participants before the study.
