Abstract
This study aimed to investigate the effect of violet syrup on cough alleviation in children with intermittent asthma. In a parallel, double-blind, randomized controlled trial, 182 children aged 2 to 12 years with intermittent asthma were randomly assigned 1:1 to receive violet syrup or placebo along with the common standard treatments in both groups (short-acting β-agonist). Both groups were evaluated in terms of the duration until cough suppression was achieved. No significant difference was observed in basic characteristics. The duration lasting to yield more than 50% cough reduction and 100% cough suppression was significantly less in the violet syrup group compared to placebo (P = .001, P < .001, respectively). There was no significant difference in therapeutic effects between boys and girls. There was a significant inverse correlation between the age of children and rate of cough alleviation and suppression by violet syrup. This study showed that the adjuvant use of violet syrup with short-acting β-agonist can enhance the cough suppression in children with intermittent asthma.
The prevalence of allergic diseases differs in different parts of the world. Among them, asthma is the most predominant chronic disease, and the most common cause of cough in children, which is diagnosed with periodic and reversible obstruction of the respiratory tracts and can be widely divergent. Coughing, wheezing, shortness of breath or rapid breathing, and a feeling of pressure in the chest are seen in children with asthma. 1,2 According to the World Health Organization, more than 300 million people worldwide have asthma, and it is predicted that this rate will increase by 20% over the next 20 years. 3 In the United States, about 9.6 million children (13.7%) have asthma, among whom almost 14% are boys and 10% girls. 1 In Iran, the prevalence of wheezing in 6- to 7-year-old children and in 13- to 14-year-old children is 6.7% and 7.10%, respectively. 3 Due to the chronic nature of asthma, it is a reason for several absences from school and is the most common diagnosis for hospitalization in pediatric hospitals. 1 Asthma is divided into 4 levels of severity: mild intermittent, mild persistent, moderate persistent, and severe persistent. The most common type of this disorder in children is intermittent asthma (with daily symptoms occurring less than 2 times per week and nocturnal symptoms happening less than twice a month, not resulting in activity limitation). 4 Although coughing is a significant sign of asthma, it may just be a pretension by children suffering from mild asthma. 5,6 These patients have dry cough with low amounts of phlegm, which usually tends to intensify at night. 5 The diagnosis of clinical asthma is based on its associated clinical patterns and symptoms. The history of coughing associated with wheezing, family history of asthma, eczema, or allergic rhinitis, and response to the treatment with bronchodilators, especially in younger children using spirometry, is not possible, and these factors are strongly in favor of diagnosis of asthma. Repeated episodes of wheezing, specifically following viral infections of the upper respiratory tract, confirm the diagnosis of asthma. 6
Different types of drugs have been used for symptomatic treatment of cough, including antihistamines (diphenhydramine, promethazine, cetirizine), decongestants (pseudoephedrine), expectorants and mucolytics (expectorants guaifenesin and bromhexine), bronchodilators (salbutamol), and narcotic and nonnarcotic drugs that affect the cough center (such as codeine and dextromethorphan). 5,6 Several kinds of herbs have been used for curing asthma since ancient times.
Viola is a plant with about 400 species and is used for treating respiratory problems. This plant (scientifically known as Viola odorata) grows in the wild and is known as Viola odorata or blue violet family, with fragrant dark-purple flowers. All parts of the plant can be used for medical purposes. 7 Viola odorata is are native to Asia, North Africa, and Europe and belongs to the violet family Violaceae. It contains different compounds such as alkaloids, glycosides, saponins, tannins, methyl salicylate, mucilage, vitamin C, coumarin, and flavonoids. 7 Viola has expectorant agents, contributing to reduction of pulmonary inflammation and edema of the mucous membrane. Its anticough feature is suitable for asthmatic patients by easing their respiration. To some, viola is effective in sore throat, throat and chest discomfort, dyspnea, acute bronchitis, whooping cough, and pneumonia. 8 Its aqueous extract contains a combination of different essential oils, methyl esters, salicylic acid, flavonoids, anthocyanins, saponins, pollen, and coumarin, causing good and strong anti-inflammatory and antibacterial effects. The most significant active substances of Viola odorata are plant peptides (cyclotides) including cycloviolacin. 9,10 This plant has antihypertensive effects, causing weight loss and antilipid impacts, as it prohibits synthesis and absorption of lipids. According to several studies, it also has antioxidant, antifungal, and antibacterial properties. 7,11,12 Viola flower syrup was also suggested by traditional Persian healers such as Avicenna (980-1037 AD) and Haly Abbas (930-994 AD) 13,14 for cough control and asthma. Regarding the traditional use of violet syrup in the treatment of asthma and cough, we intended to examine its efficacy in cough suppression in 2- to 12-year-old children with intermittent asthma.
Materials and Methods
This double-blind, randomized controlled clinical trial was conducted on children (2-12 years old) with intermittent asthma, visiting outpatient clinics in Qom from October to December 2014. Children aged between 2 and 12 years with the clinical diagnosis of intermittent asthma were enrolled in the study. Children who need or take treatments other than short-acting β-agonists, having a family member who smokes, and suffering from other chronic diseases were excluded from the study (Figure 1). Demographic information of the patients including the age, sex, place of living, and educational level of parents were recorded.
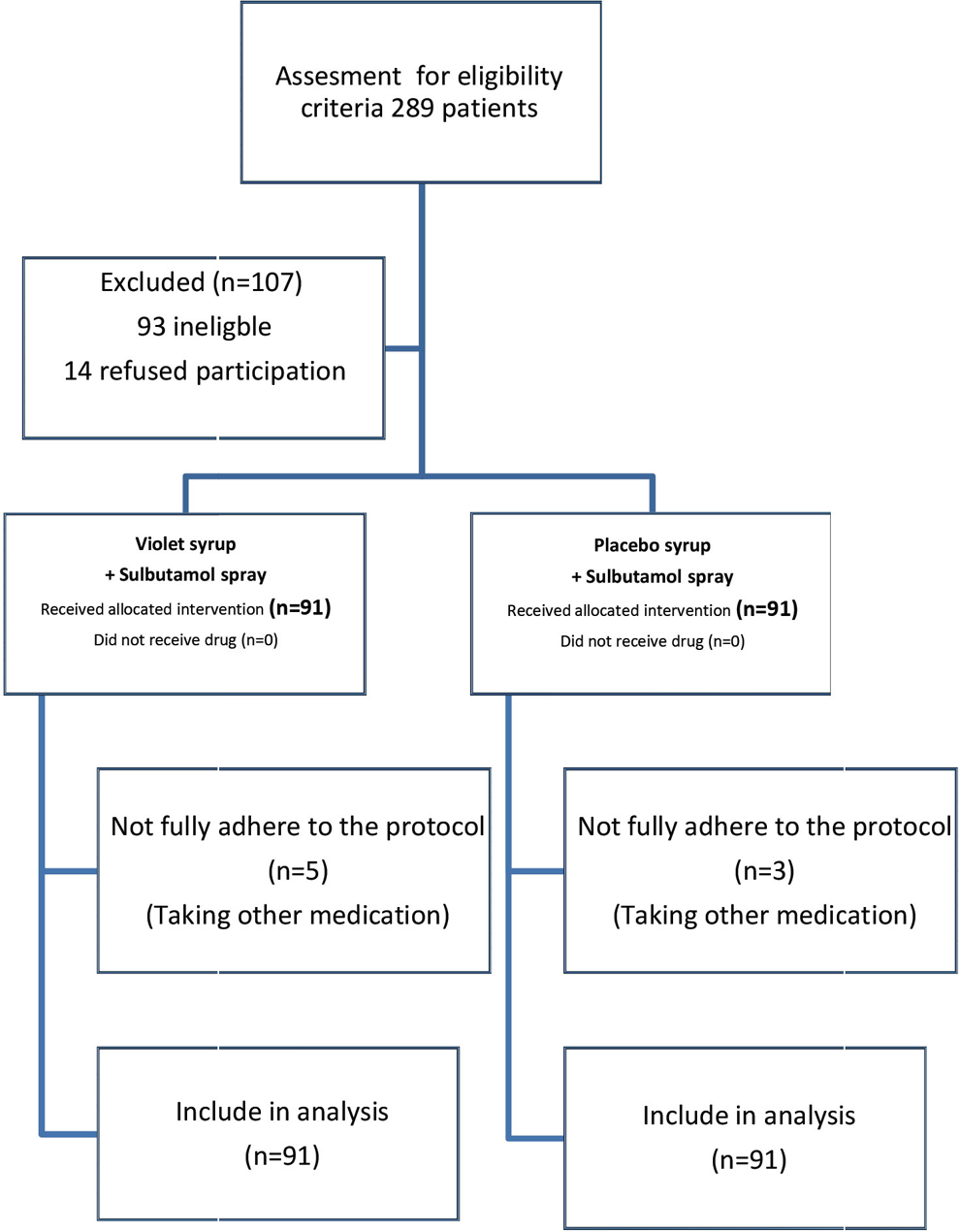
Flowchart of study inclusion, allocation, and follow-up.
The children’s parents completed and signed the informed written consent forms for conducting the study and the relevant experiments before enrolment in the study. The participants (182 children) were randomly assigned, following block randomization with 1:1 allocation method. Participants, their parents and those assessing the outcomes were blinded to group assignment. Children received Viola flower syrup or placebo syrup along with the common standard treatment for intermittent asthma (salbutamol spray as short-acting β-agonist 2 puff 4 times a day) in both groups. Both violet and placebo syrups were administered for 5 days with a dose of 2.5 cc or 5 cc 3 times per day for 2 to 5 and 5 years and older children, respectively. Twelve grams of dry viola flower was used in each 100 cc of viola syrup.
The patients’ parents were asked about the duration required to yield more than 50% cough reduction and 100% cough suppression after 5 days of intervention.
The data were analyzed using SPSS, version 17. In this study, frequency, mean, and standard deviation of the groups were obtained. In addition, χ2, independent t test, Mann–Whitney test, and Spearman’s correlation test were performed to analyze the significance of differences between trial groups. Differences with P < .05 were considered as significant.
The study protocol was in compliance with the Declaration of Helsinki (1989 revision) and was approved by the Local Medical Ethics Committee of Islamic Azad University in Iran (ID Number: 10264713-Q). This study was also registered in AEARCTR (The American Economic Association’s Registry for Randomized Controlled Trials; RCT ID: AEARCTR-0000580).
Results
In this study, a total of 289 patients were assessed for eligibility, and finally, 182 patients were randomized to receive the trial drug or placebo from August 2013 to February 2014. Ninety-one patients in each group completed the study. Figure 1 is a flow chart that reveals detailed descriptions of patients’ enrolment, randomization, and outcomes. Five patients in the violet syrup group and 3 in the placebo group did not have complete adherence to the study protocol (all of them receiving other asthma or cough medications). However, all of these patients were included in the final analysis according to the intention-to-treat analysis protocol.
Distribution of patients in both violet syrup and placebo groups was not significantly different in terms of sex (P = .292), age (P = .914), place of living (P = .773), and educational status of parents (P = .144, .25; Table 1).
Basic Demographic Characteristics of Participants Included in the Trial.

The duration required to yield more than 50% cough reduction and 100% cough suppression was significantly less in the violet syrup group compared to the placebo group (P = .001, P < .001, respectively; Table 2). There was no significant difference in therapeutic response between boys and girls (P = .337). In the violet syrup group, there was a significant inverse correlation between the age of children and rate of cough alleviation and suppression (r = −.259 and P = .013, r = −.223 and P = .33, respectively).
Duration Lasting to Yield More Than 50% and 100% Cough Suppression in the Violet Syrup and Placebo Group.

Sixty children in the violet syrup group and 53 in the placebo group had wheezing in pulmonary examination before the intervention. The number of children with wheeze in pulmonary examination after the intervention in the violet syrup group was significantly less than in the placebo group (4 vs 25, respectively, P < .001).
Discussion
Asthma is a chronic disease of the respiratory tract and is classically diagnosed by symptoms such as coughing, wheezing, and shortness of breath. According to the World Health Organization, more than 300 million people worldwide have asthma, and it is predicted that this rate will increase by 20% over the next 20 years. 3 Cough is a very common symptom of asthma in childhood that causes discomfort and concern in patients and their families. The majority of cough symptoms are due to acute viral infection (either benign or self-limiting) of the upper respiratory system. 15 The common over-the-counter drugs for acute cough in children and adults include liquidator expectorant and anticough drugs that are widely prescribed. 16
Viola odorata has been used traditionally in different forms for curing different medical conditions. In Iranian traditional medicine, it was known as a plant with cold and wet temperament and has been used in hot and dry temperament diseases such as fever, excessive thirst, and uremic pruritus. 17,18 It was also widely recommended in Iranian traditional medicine for pulmonary diseases such as cough, pneumonia, and pleurisy. 17 The viola also was recommended for children in 3 different indications (febrile convulsion, rectal prolapse, and cough) in Iranian traditional medicine. The maximum allowed dose was about 20 grams (about 250 mg/kg) of dry flower per dose in Iranian traditional medicine. (In this study, we have used about 30 mg/kg/dose.) Nausea and vomiting are considered as the most common adverse effects in the case of overdose in Iranian traditional medicine. 17 There is no reported adverse effect in the evidence-based literature for Viola odorata. 19,20 There was no reported adverse event in this study that can be explained by the low dose of the herb that was taken.
In current scientific literature, Viola odorata has been shown to have analgesic, sedative, antihyperlipidemic, antihypertensive, and lung tissue protecting effects in animal studies. 7,21 –23 It is also used to treat chronic insomnia with positive effects in an uncontrolled human study. 24 In textbooks of phytotherapy, its antitussive effect for asthmatic children is mentioned as unproven use. 20 According to the American Herbal Products Association’s Botanical Safety Handbook, no human or animal study has shown Viola odorata to have any drug or supplement interaction. 19 Although the information on herb–drug interactions is not sufficient to rule out any potential interaction between Viola odorata and β-agonists, such an interaction seems to be less likely with the inhalation dosage form of β-agonists, as used in this study.
In this study, the highest rate of cough reduction in both groups was seen over 1 to 3 days. This rate was statistically significant in both groups (P = .001). Although no study has been done on the effects of viola, the impacts of ivy syrup, which has similar properties as viola, on cough have been examined. Hecker et al, 25 in a study on the effect of ivy on clinical symptoms of 1350 patients with chronic bronchitis, showed that using this medicine can stifle cough in 92.2% of the patients. In addition, Büechi et al 26 studied the effect of a plant extract combination consisting of dry leaf extract of ivy as the main active ingredient. Results indicated the reduction of respiratory symptoms including cough and phlegm in 62 patients by the end of treatment. In a study by Schmidt et al, 27 the consumption of ivy alleviated or stifled couch in 94.2% of children. Investigating the effect of dry leaf of ivy on treatment of chronic airway obstruction in children with asthma suggests that this syrup reduces airway resistance more than placebo and improves the respiratory function of children suffering from bronchial asthma. 28 Clinical reviews showed that the extract of ivy leaves, such as viola, contains saponins, which is a liquidator expectorant, antispasm, bronchodilator, and antibacterial substance, mainly attributed to its triterpenoid saponins content. 29,30 In 2007, more than 80% of herbal expectorants were prescribed in Germany comprising ivy extract and amounted to nearly 2 million prescriptions nationwide and a volume of sales exceeding 13 million Euros. 31 Studies have shown that cyclotides extracted from Iranian Viola odorata have antibacterial impact on gram-negative bacteria as well as good antimicrobial effects. 32 In a study by Lee et al, 33 anti-inflammatory and anti-asthmatic effects of viola on the inflammation of respiratory tracts of allergic mice were examined. Results indicated that viola considerably inhibited the total serum level of IgE and cytokines such as IL-3 and IL-4. It also effectively decreased overresponse of the airways and eosinophilia and excessive secretion of mucus. This investigation recommends the use of this herb for treating asthma. 33 Adverse effects, poor taste, high costs, and use of preservative and flavoring agents in synthetic chemical compounds are increasing the interest toward using herbs. 34 Furthermore, anticough drugs such as dextromethorphan, codeine, and diphenhydramine are associated with several complications like dizziness, drowsiness, respiratory inhibition, tachycardia, and even death. In this regard, the US Food and Drug Administration has warned against prescribing these herbs to children less than 2 years of age, and the American Academy of Pediatrics has not confirmed its usage. 35 –37 Regarding the observed positive effects of violet syrup on cough, it can be suggested as a potential cough suppressing natural treatment for children with intermittent asthma. It is recommended that studies with larger sample size and other dosages of this drug should be performed to confirm the results of this study and select the best dosage.
Conclusion
This study showed that violet syrup (as adjuvant of short-acting β-agonist) is significantly more effective than placebo in reduction and suppression of intermittent asthma caused cough in children aged between 2 and 12 years.
Footnotes
Acknowledgments
The authors would like to express the deepest appreciation to Deputy of Research and Technology of Islamic Azad University, Qom Branch, specially Dr Gholamreza Najafi and the Medical Students Research Committee for all of their support.
Author Contributions
The work presented in this article was carried out through collaboration between all authors. MJQ and HS made the initial hypothesis. All authors participate in defining the research theme and providing the proposal. MJQ, MHA, and MG visited the patients, enrolled them, and followed them. MHE, MS, and MA interpreted the data and wrote and edited the article. MBM supervised the work. All authors have contributed to, edited, and approved the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Islamic Azad University.
Ethical Approval
This trial was approved by Medical Ethics Committee of Islamic Azad University in Iran (ID Number: 10264713-Q). This study was also registered in AEARCTR (The American Economic Association’s Registry for Randomized Controlled Trials; RCT ID: AEARCTR-0000580).
