Abstract
Allium ampeloprasum subsp iranicum (Leek) has been traditionally used in antihemorrhoidal topical herbal formulations. This study aimed to evaluate its safety and efficacy in a pilot randomized controlled clinical trial. Twenty patients with symptomatic hemorrhoids were randomly allocated to receive the topical leek extract cream or standard antihemorrhoid cream for 3 weeks. The patients were evaluated before and after the intervention in terms of pain, defecation discomfort, bleeding severity, anal itching severity, and reported adverse events. A significant decrease was observed in the grade of bleeding severity and defecation discomfort in both the leek and antihemorrhoid cream groups after the intervention, while no significant change was observed in pain scores. There was no significant difference between the leek and antihemorrhoid cream groups with regard to mean changes in outcome measures. This pilot study showed that the topical use of leek cream can be as effective as a standard antihemorrhoid cream.
Hemorrhoidal disease is one of the most common causes of colorectal surgery consultations. 1 The exact prevalence of hemorrhoidal disease is very hard to establish, because many patients do not seek care. 2 Thus, epidemiologic data can vary widely due to methodological issues; however, the prevalence of hemorrhoidal disease is estimated to be between 4.4% and 12.8% in normal adult populations and about 40% in patients with symptoms of anal diseases.3,4 A total of 3.2 million ambulatory patients, 306 000 hospitalizations, and 2 million prescriptions are estimated for hemorrhoidal diseases in the United States. 5
The disease is classified as internal and external hemorrhoids; internal is divided into 4 grades according to the level of hemorrhoidal vein prolapse. 6 Near half of all patients with hemorrhoidal disease are asymptomatic; the most prevalent complaints are rectal bleeding, pain associated with thrombosed hemorrhoids, and perianal pruritus in symptomatic patients. 4
Only a small minority of patients with hemorrhoidal disease need surgical intervention. 7 Conservative management strategies are widely used successfully for most patients with symptomatic hemorrhoids. These include high-fiber diets or supplements, topical analgesics, warm sitz baths, and stool softeners.8,9
Topical herbal formulations were popular in treatment of different diseases in traditional medicine.10,11 Different topical herbal formulations have been evaluated for their beneficial effects in patients with hemorrhoidal diseases.12–14 Anti-inflammatory, analgesic, and venotonic forms of activity are considered to comprise the main mechanisms of action in these herbs. Flavonoids, terpenoids, triterpenes, and tannins are the major chemical constituents that are considered to be responsible for these forms of pharmacological activity. 12
Allium ampeloprasum subsp iranicum (leek) is one medicinal plant that has traditionally been used in antihemorrhoid topical herbal formulations in Iran. Its most important active constituents are flavonoids and saponins. Flavonoids have shown beneficial effects in patients with hemorrhoidal diseases in multiple randomized clinical trials.15–17 Its saponins have also shown steroidal, anti-inflammatory, and antiulcerogenic activity in previous studies. 18
Due to the above-mentioned pharmacologic properties of Allium ampeloprasum subsp iranicum and its traditional use in patients with hemorrhoids, we decided to evaluate its safety and efficacy in a pilot randomized and controlled study.
Methods
Trial Design
The study was designed as a 2-arm, double-blind randomized and controlled clinical trial using a parallel design with a 1:1 allocation ratio. There was no change in methods after the trial’s commencement.
Participants
Thirty-four patients attending the colorectal clinic of the Shiraz University of Medical Sciences between September 2014 and October 2014 with symptoms of hemorrhoidal diseases were evaluated for inclusion in the study. The clinical diagnosis was made by physical examination in patients with compatible complaints by digital rectal exam and colonoscopy. Participants were required to be older than 18 years for inclusion in the study. The exclusion criteria were history of contact allergic reactions to leek, pregnancy, breast-feeding, and previous history of anorectal surgery, concomitant fissure, ascites, malignancy, and inflammatory bowel diseases. Finally, 20 patients were allocated to intervention and control groups.
Preparation of Creams
For the preparation of creams, liquid paraffin (12% w/w), white petrolatum (7% w/w), and cetostearyl alcohol (11% w/w) were selected for the oily phase. The mixture was heated to 70°C. Fresh aqueous liquid obtained from cutting Allium ampeloprasum subsp iranicum was filtered for the aqueous phase of the cream. A total of 69 mL of this extract, containing sodium louryl sulfate as surfactant and methyl and propyl paraben as microbial preservatives, was heated to 70°C. Then, the aqueous phase was mixed with the oily phase continuously while being cooled. After cooling, 30 g of the uniform cream was placed in an aluminum tube. For the control group, an established antihemorrhoid cream (Behvazan Pharmaceutical Co, Rasht, Iran) was used. This cream consists of lidocaine (5% w/w), hydrocortisone acetate (7.5% w/w), aluminum subacetate (3.5% w/w), and zinc oxide (18% w/w). Thirty grams of this cream was packed in tubes similar to the test group.
Interventions
The patients recruited were randomly assigned to receive leek cream (n = 10) as part of the intervention group or antihemorrhoid cream (n = 10) as part of the control group for a period of 3 weeks. Patients received 2 cc of the leek cream or standard topical antihemorrhoid cream twice daily for 3 weeks. The topical agent was applied on the anus with an applicator. The patients in both groups received a high-fiber diet without any laxative or stool softener.
Outcomes
Patients were evaluated prior to and following 3 weeks of the intervention in terms of the following items: Pain (11-grade visual analogue scale, 0-10) Defecation discomfort (11-grade visual analogue scale, 0-10) Bleeding severity (5-grade scoring system, 0-4) Anal itching severity (5-grade scoring system, 0-4) Overall subjective improvement (%) Number of participants with any observed or reported adverse events
Randomization, Blinding, and Allocation Concealment
Twenty eligible patients were randomly allocated to 2 parallel groups (the leek cream as trial drug and antihemorrhoid cream as standard drug) by the secretary of the clinic, who had been instructed to use a randomized list. The randomized list was generated using Microsoft Excel with a block randomization method, as previously described. 19 The physicians, researchers, and statisticians were blind to the allocation of patients. As the leek and antihemorrhoid cream tubes looked identical, the patients were also blind to the drug allocation.
Statistical Methods
A Wilcoxon–Mann–Whitney test and a χ2 were used for statistical comparison of primary characteristics and outcomes between the intervention and control groups. A Wilcoxon sign-ranked test was used for statistical comparison of values obtained before and after the intervention. A P value of less than .05 was considered significant.
Ethical Considerations
The study protocol was in compliance with the Declaration of Helsinki (1989 revision) and was approved by the Local Medical Ethics Committee of Shiraz University of Medical Sciences (SUMS; reference number: CT-9377-7127).
Results
From September 2014 to October 2014, a total of 34 patients were assessed for eligibility and 20 of them were randomized to receive the trial drug or standard topical antihemorrhoid cream. All allocated patients completed the study. Figure 1 shows a flow chart that reveals detailed descriptions of patients’ enrolment, randomization, and outcomes.

Flowchart of study inclusion, allocation, and follow-up.
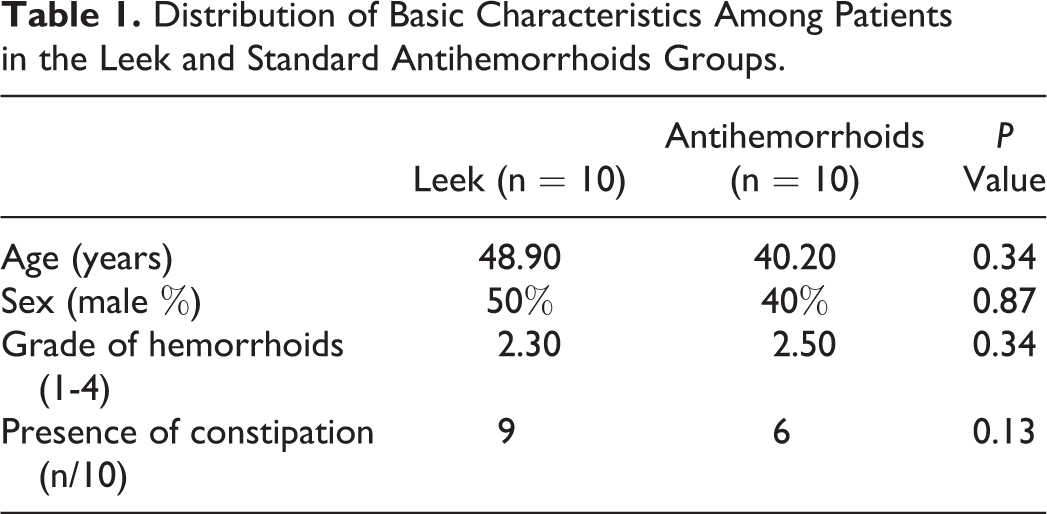
The baseline characteristics (age, sex, grade of hemorrhoidal diseases, and presence of constipation) of the patients in the 2 groups are shown in Table 1. There were no significant differences in the basic characteristics of the 2 groups.
Distribution of Basic Characteristics Among Patients in the Leek and Standard Antihemorrhoids Groups.
A significant decrease was observed in the grade of bleeding, severity, and defecation discomfort in both the leek and antihemorrhoid cream groups after the intervention, while no significant change was observed in pain scores (Table 2).
Comparison of Outcomes Among Patients in Leek and Standard Antihemorrhoid Groups.
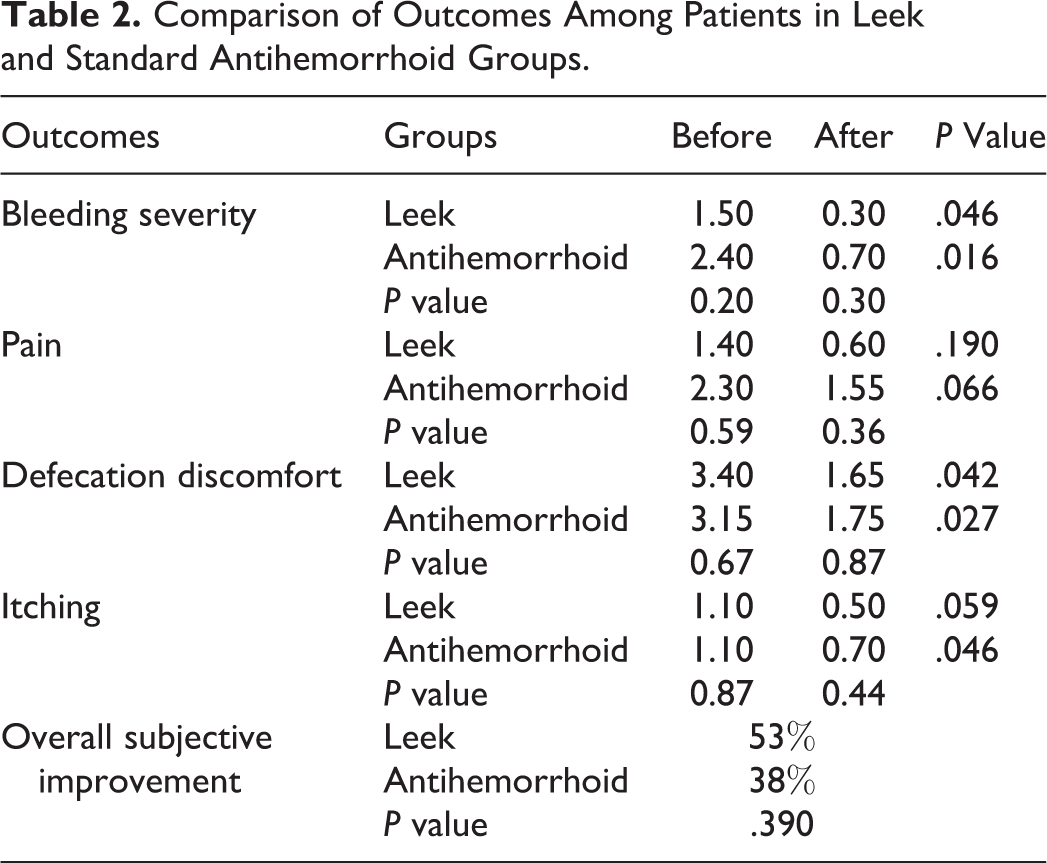
No significant difference was observed between the mean subjective improvement of leek and antihemorrhoid cream groups (53% vs 38%, respectively, P = 0.39). There was also no significant difference between the leek and antihemorrhoid cream groups with regard to mean changes in the grades of bleeding severity (1.20 vs 1.70, respectively, P = 0.16), anal-itching severity (0.60 vs 0.40, respectively, P = 0.86), and defecation discomfort (1.75 vs 1.40, respectively, P = 0.84). One patient in the leek cream group complained of irritation on the application site. No patient reported any other systemic or local adverse events during the follow-up period in the intervention group or in the control group.
Discussion
This pilot study has shown that topical use of leek extract cream can be as effective as an established antihemorrhoid cream in the alleviation of hemorrhoidal diseases. The observed beneficial effects can be explained by the previously described antihemorrhoidal, steroidal, anti-inflammatory, and antiulcerogenic activity of active constituents of leek, such as flavonoids and saponins.16–18
The measurement in clinical parameters of hemorrhoidal disease is difficult. The self-assessed symptom-scoring system we used is popular in research protocols on hemorrhoids. 20 However, such methods are subjective and may be biased. Another important consideration is that hemorrhoidal disease is associated with dietary behavior and lifestyle factors that cannot be directly controlled, so there are a variety of behaviors in each patient that have unavoidable effects on observed outcomes.
The short duration of patient follow-up was an important limitation of this study. A 3-week follow-up of patients does not allow for evaluation of following symptomatic relapses of hemorrhoidal diseases and the preventive role of the intervention. However, the 3-week duration of follow-up can be considered adequate for evaluation of the symptomatic control of acute hemorrhoids.21,22 The lack of a placebo group was another important limitation of this study. However, it is routine protocol to compare topical agents with standard antihemorrhoidal creams.23,24 The ingredients of the antihemorrhoid cream used as a topical standard treatment have shown their benefits compared with placebos in previous studies.25–27 In fact, routine prescription of antihemorrhoid creams beside other conservative approaches in our clinical setting did not ethically allow us to use placebo as a control group, leading us to use this formulation as a standard intervention for the pilot study. The low number of patients was another limitation, as this was a pilot evaluation of this herbal formulation.
In conclusion, this pilot study showed the topical use of Allium ampeloprasum subsp iranicum (leek) cream can be as effective as a standard antihemorrhoid cream. This potential effect should be further investigated in studies with larger sample size, longer follow-up duration, and placebo control.
Footnotes
Acknowledgments
The authors would like to express their thanks to Dr Ali Soleimani Eslami for his statistical analysis assistance and Miss Puralifard for help to preparation of drugs and all the study participants for their participation.
Author Contributions
The work presented in this article was carried out through collaboration between all authors. SHM and MH made the initial hypothesis. All authors participate in defining the research theme and providing the proposal. LG, SHM, and ERH visited the patients, enrolled them, and followed them. ZS prepared the leek cream. MH wrote the first draft of the article. All authors have contributed to, edited, and approved the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was a part of a PhD thesis by Dr Seyed Hamdollah Mosavat, which was supported by
Ethical Approval
This study was approved by the Ethics Committee of the Shiraz University of Medical Sciences (Approval Code: CT-9377-7127). Protocols were in accordance with the Declaration of Helsinki.
