Abstract
Interest in vitamin D and the
Keywords
Vitamin D: Origins of an Archaic Biomolecule
The light-dependent vitamin D endocrine system is critical for maintaining calcium homeostasis, with cells sustaining a stable Sub-micromolar level of free intracellular calcium (iCa2+). Calcium in turn exhibits a reciprocal effect on the production of calcitriol, the biologically active form of vitamin D. Vitamin D signaling via the vitamin D receptor (VDR) likely evolved to facilitate regulation of calcium flux/equilibrium, calcium storage, and calcium signaling within neuronal and muscle tissue. It has been suggested that such regulation is critical for the evolutionary process 1 ; we argue in this article that vitamin D may indeed have an evolutionary link to regulation of calcium, but that this relationship is additionally critical over an individual lifecycle, and is closely dependent on VDR-related nutrigenetic associations, some of which add in environmental exposure (ultraviolet radiation [UV-R]) as a covariable.
The article explores how vitamin D interacts with another light sensitive vitamin, folic acid, and how these 2 vitamins might influence long-term biological effects (the evolution of skin pigmentation), 2,3 as well as shorter “lifecycle” timeframe effects on clinical phenotypes of relevance to human health and well-being. 4
That vitamin D is an important regulator of many processes within the cell may well be reflected in its ancient origins. It is certainly interesting to speculate that its physiologic role has evolved to counter the challenges faced by evolutionary advancement, particularly of homeostatic iCa2+ control in organisms that left ancient, calcium-replete oceans to reestablish their lineage in calcium-deplete terrestrial ecosystems. 1 It has been estimated that vitamin D photosynthesis has occurred in marine phytoplankton for 500 million years and that terrestrial vertebrates have been generating de novo vitamin D for the latter 70% of this same time frame. 5 A full vitamin D endocrine system that deploys a specific VDR nuclear receptor and cytochrome P450 enzymes, and which is regulated by calciotropic hormones and vitamin D binding protein, is only found in vertebrates. The origins of this area of metabolism may therefore relate to archaic xenobiotic P450-related detoxification pathways and have been driven by the need to handle the challenges of higher gravity in a calcium-deplete terrestrial environment. 6
Vitamin D: Sunlight Meets Skin
Figure 1 shows relevant aspects of vitamin D photosynthesis, a process that utilizes UV-B radiation in the wavelength 280 to 320 nm, with a maximal effect between 295 and 297 nm. Since vitamin D can only be produced via photochemistry, terrestrial vertebrates need to consume foods that contain vitamin D, or need be exposed to UV-R to meet their vitamin D requirement. UV-B transforms 7-dehydrocholesterol into previtamin D3 in the skin (stratum basale and stratum spinosum). This vitamin precursor then photoconverts into either inactive metabolites (lumisterol or tachysterol) or undergoes a slow temperature-dependent isomerization reaction to yield vitamin D3 (calciol), which is metabolized on to calcidiol [25(OH)D3] in the liver and, subsequently, on to the active vitamin form, calcitriol [1,25(OH)2D3] in the proximal tubules of the kidney. 7

Simple schematic of vitamin D photosynthesis and distribution.
Some terrestrial vertebrates provide adjustments to this process to counter the effects of fur or feathers. Often, vitamin D is generated from 7-dehydrocholesterol in oils secreted onto fur or feathers by the skin. The 7-dehydrocholesterol is spread during preening, allowing vitamin D photosynthesis. The micronutrient is then consumed during grooming.
8
Interestingly, one mammal that has no source of vitamin D available to it is the subterranean naked mole rat (
Clothing, altitude, latitude, time of day, and weather conditions, including pollution levels, all influence previtamin D3 synthesis. In humans, individuals with lightly pigmented skin produce higher amounts of previtamin D3 than individuals with darkly melanized skin. As a consequence of this, deeply pigmented skin might be considered nonadaptive for de novo 1,25(OH)2D3 synthesis under conditions of limited UV-R.
It was a lack of sunlight that was first attributed to the deficiency syndrome of vitamin D in children. Inadequate bone calcium causes rachitic deformities, 9 and as early as the mid-17th century rickets was recognized as a discrete phenomenon arising due to urbanization of England’s population, and the associated atmospheric pollution (smoke and smog) that hampered seasonal vitamin D synthesis at northerly latitudes. 10 By the 20th century, further industrialization and migration to high latitudes both in Western Europe and the United States saw the creation and proliferation of urban slums and burgeoning atmospheric pollution with overcrowding and impoverished lifestyles adding to UV-R deprivation. As a consequence of this anthropogenic vitamin D–restricting environment, a high prevalence of rickets (under-mineralization of bone) developed among infants.
So how does the atmosphere influence the quality of light/UV-R needed for vitamin D synthesis, and does it have adverse effects on related vitamins? UV-A is more efficient at penetrating both ozone and human skin than UV-B or UV-C. 2,11 –14 Additionally, factors such as photoperiod (day length) and total solar irradiance (TSI) are important factors to consider, along with those alluded to above. It is particularly interesting to recognize the impact that solar eruptions can have, and their contribution to TSI. These phenomena lead to ionized particles (solar storms) affecting power grids, satellites, and communications. Understandably, much work has focused on the impact of these dramatic solar events on human infrastructure, but very little research has looked at the effects on human biology.
TSI, which increases with sunspot activity, 11 is a balance between sunspot-related magnetic influences that shield the solar plasma and highly energetic faculae that surround sunspots. UV-A and to a lesser extent UV-B are likely to be a highly relevant component of the TSI. This is important not only in the context of vitamin D stability and photodegradation but also in maintaining folate stability: Folate has been shown to exhibit an important relationship with UV-R. It is now believed that dermal exposure to UV-A radiation promotes photolytic degradation of folate, lowering systemic vitamin status, 2,15,16 work now strongly supported by a recent Australian study. 17 While vitamin D is usually discussed in terms of photosynthesis, it is also degraded by UV-R. Vitamin D3 synthesized by the action of UV-B can be degraded by UV-A after as little as 10 minutes of nontropical sun, although the rate of loss is lower in winter. 2,7 This previtamin D3 photo-isomerization prevents vitamin D toxicity, but means that UV-A degrades vitamin D3 at times of the year when there is inadequate UV-B to photosynthesize calciol in the skin 7 and hence generate biologically active 1,25(OH)2D3. 1,25(OH)2D3, the main form of vitamin D, is itself UV-A labile and has a short half-life of 15 hours in the circulation. 7,18 –20 By contrast, 25(OH)D3 has a half-life of around 14 days in the circulation, although limited fat and muscle storage also occurs (half-life 60 days). 20 Despite these additional stores, they may be inadequate for lean people when UV-B photosynthesis of the vitamin is absent. 21
Recent research suggests that TSI and photoperiod may influence human biology both in the short term and in the longer term, possibly via folate- and vitamin D–related phenomena,
22
–24
and this is discussed in detail below. Although a paradigm will be developed that frames both vitamin D– and folate-related nutritional genetics within a molecular mechanism that could explain aspects of the evolution of skin pigmentation, other genetic factors are known to be important in hominid adaptive melanization: the multifactorial involvement of the following key genes is critical in skin pigmentation:
Recent research highlights an interesting biological contradiction. UV light is a well-established carcinogen, yet Fell and colleagues 29 have provided evidence that UV-seeking behavior is addictive and that UV addiction is mediated by the hedonic action of beta-endorphin and anhedonic effects of withdrawal. They suggest this biological/behavioral mechanism may well have enhanced evolutionary vitamin D biosynthesis in ancient human populations, but that in contemporary society it has led to a rise in skin cancer incidence.
Vitamin D Metabolism and Bioactivity in Brief
The major elements of vitamin D metabolism beyond its photosynthesis are shown in Figure 2. The vitamin enters the circulatory system via the diet or following cutaneous photosynthesis. It is incorporated into chylomicrons and transported via the lymphatic system into the blood plasma. It is carried around the body bound to vitamin D binding protein (DBP) in the form of calcidiol and is ultimately metabolized to the active hormone calcitriol/1,25(OH)2D3.

Simple overview of the vitamin D endocrine system.
Synthesis is highly regulated: calciol undergoes 2 consecutive hydroxylation reactions that act to regulate both calcitriol synthesis and iCa2+ levels. In the liver, calciol 25-hydroxlase converts calciol into calcidiol, and in the kidney, calcidiol 1-hydroxylase converts calcidiol into calcitriol. Both these enzymes are in the cytochrome family and are encoded by
These biochemical mechanisms underpin physiological regulatory processes in which vitamin D maintains calcium homeostasis. There are 3 main processes: (
The Vitamin D Receptor Is Ubiquitous
1,25(OH)2D3 is a conformationally flexible secosteroid that elicits its biological function via binding to the VDR, which is a member of the steroid hormone receptor superfamily.
30
–34
Ligand binding leads to transcriptional regulation of a variety of target genes.
35
Many of these such as
The VDR has 2 overlapping ligand binding sites; these are represented by genomic and alternative pockets, which respectively bind a bowl-like ligand configuration for gene transcription or a planar-like ligand shape for rapid biologic responses. VDRs exhibit narrow ligand selectivity. They bind calcitriol/1,25(OH)2D3 with high affinity and are capable of distinguishing between 1,25(OH)2D3 and other vitamin D3 precursors or metabolites. 37,38 When the VDR genomic pocket binds 1,25(OH)2D3, it can then interact with the retinoid X receptor to form a heterodimer that allows the nuclear receptor to bind with vitamin D responsive elements that control gene expression. Activated VDR can recruit co-activators or co-repressors to modulate gene transcription, which is most often examined in relation to the regulation of intestinal calcium and phosphate absorption. This influences skeletal calcium as a mechanism to maintain calcium homeostasis within an acceptable physiological range. However, it is increasingly being recognized that 1,25(OH)2D3-related VDR control of gene expression, and rapid biologic responses can also mediate in the pathoetiology of chronic diseases of aging, including cancer, type 2 diabetes, vascular disease, infection, and osteoporosis. 38
VDR is ubiquitous, and it is found in cells of the intestinal epithelium, breast epithelium, kidney tubules, pituitary gland, parathyroid gland, reproductive tissue, keratinocytes, pancreas, skeletal osteoblasts and chondrocytes, and immune cells including T-lymphocytes, macrophages, and monocytes. Of these, the greatest VDR content is to be found in bone, the parathyroid gland, kidney, and intestine. 36
Steroidal King of Light-Sensitive Vitamins
Vitamin D does not fit the classic definition of a vitamin, as the most important source of this steroid pro-hormone is via endogenous synthesis in the skin following UV exposure. However, it is not the only light-sensitive vitamin, although arguably it is one of the most important. Other vitamins that are also sensitive to and/or transduce light signals include folic acid, vitamin A, riboflavin (vitamin B2), and niacin (vitamin B3). In some cases light transduction by these vitamins plays a major role in regulating circadian rhythm. 16,39
Cryptochromes are blue-light photoreceptors that are central to generating circadian oscillations in animals and plants. They occur in the ganglion cell layer of the retina and transmit/transduce light stimuli to the master circadian clock, which in humans is located in the suprachiasmatic nucleus. It has been shown that purified human cryptochrome 2 (hCRY2) exhibits fluorescence properties consistent with the presence of both flavin (vitamin B2) and folate cofactors, 40 although evidence of photoreception in mammalian cryptochromes remains indirect. 41 CRY1 and CRY2 are 73% homologous in all organisms and absorb light in the 350 to 450 nm wavelength range. Most often, the folate cofactor is 5,10-methenyl-H4folate, and the flavin vitamer is flavin adenine dinucleotide (FAD). In this relationship, the folate vitamer functions as a light-catching antenna whereas FAD facilitates the subsequent redox reaction. Basically, exposure to blue-light photons excites 5,10-methenyl-H4folate, and an electron is then transferred to the reduced catalytic flavin (FADH−) and then on to CRY1 or CRY2. 39,42 Interestingly, in plants, folate vitamer–containing cryptochromes regulate blue-light dependent growth, while in bacteria, insects, and amphibians they stimulate enzymes that repair UV-induced DNA damage. In mammals, they regulate the circadian clock. Although vitamin A interacts with opsin proteins in circadian photoreception, 39 and is obviously critical in vision, it is now thought that cryptochromes are the major mammalian circadian photoreception system. Additionally, while systemic folate is sensitive to UV exposure on the skin, and is easily degraded, the opposite is true of vitamin D, which requires UV for its synthesis. This diametrically opposed effect of UV light may have played a significant role in evolutionary processes (see below).
While vitamin D is the first vitamin that most people bring to mind when nutrients are discussed in the context of sunlight, and for good reason, it is not the only nutrient of significance. Niacin as NAD(P)(H) is also closely linked to human biology via UV exposure. A niacin deficiency or poor NAD status leads to dermal sun sensitivity, reflecting a poor cellular response to UV exposure (ie, sunlight dermatitis in the deficiency disease, pellagra). Indeed, NAD deficiency leads to genomic instability and may enhance cancer development. In keratinocytes, NAD deficiency promotes photodamage. This stems from both poly(ADP-ribose) polymerases and sirtuins being inhibited by a lack of NAD+. A lack of this important vitamin-related substrate for these enzymes leads to unrepaired photolytic damage to DNA and promotes cell death. Furthermore, the rapid depletion in NAD due to increased poly(ADP-ribose) polymerase activity following genomic damage (UV damage/mycotoxins, etc) has long been seen as one of the potential mechanisms of apoptosis, 43 with a variety of deleterious effects in play such as delayed DNA excision repair, a build-up of single and double strand breaks, chromosome breakage, telomere erosion, and malignancy. 44
After vitamin D, vitamin A is perhaps the second vitamin people tend to associate with light. However, in truth both preformed and pro-vitamin A carotenoids fall into this category. The role of 11-
Of course, vitamin A and vitamin D interact as nuclear cofactors (heterodimerization of VDR-RXR) making it difficult to consider either vitamin in isolation. In fact, the best way to summarize vitamin D as being the most significant of all light-sensitive vitamins is to draw attention to the fact that vitamin D signaling is so fundamentally important that irrespective of whether deficiency stems from inherited defects, nutritional deficits, a lack of sunlight, malabsorption, or covert disease, the consequences can be significant and variable across the lifecycle. This is largely because vitamin D signaling is regulated at several levels and is far more complex than a simplistic ligand–receptor–DNA interaction. 49
Earlier it was suggested that folic acid and vitamin B2 might play a key role in the generation of circadian oscillations, but vitamin D may also be important. The current paradigm for the mammalian circadian clock involves interplay between 2 transcription–translation feedback loops consisting of “clock” genes and their expression products. Feedback loop one involves positive elements, including the transcription factors CLOCK and BMAL1. Following heterodimerization, these transcription factors enter the nucleus and bind the circadian E-box promoter, enhancing transcription of expression products PER1 and PER2 and both CRY1 and CRY2. These products negatively feedback to inhibit their source genes by arresting CLOCK/BMAL1-mediated activation. The second feedback loop initiates Rev-Erba and Rora genes via CLOCK/BMAL1. The expressed protein products of these genes compete for binding at the
From a more mundane food perspective, many micronutrients are light sensitive. Apart from folate, 2 good examples are thiamin (vitamin B1), which can exhibit total loss from bread when freshly baked produce is exposed to light in shop windows, and vitamin B2. When vitamin B2 in milk is exposed to sunlight or even fluorescent light, photolysis leads to lumiflavin under alkaline conditions or lumichrome under neutral or acidic conditions. Neither has vitamin activity and they can catalyze peroxidation of lipids and conversion of methionine to methional. Methional confers an off taste or “sunlight” flavor to milk. As a consequence, cartons now have a protective lining to stop B2 photolysis. It would be wrong to omit the many antioxidant vitamins that undoubtedly also help mop up free radicals and mediate cell repair processes following UV-induced DNA damage.
Vitamin D Health Correlates Including a Perspective on Seasonality
There is now overwhelming evidence that seasonality derived from planetary rhythm influences all organisms via recognized, putative, and as yet undiscovered cellular and genetic mechanisms. The sun in particular plays a pivotal role in orchestration of the human lifecycle.
The human exposome, which includes all wavelengths of UV-R, photoperiod, diet, and temperature, as well as an almost incalculable number of other environmental factors, has contributed to the evolution of our species. Some of these have operated recently and at a cultural level. For example, cultural practices related to food production, powerful selection pressures, and genetic drift have conspired to rapidly alter around 700 regions within our genome in our recent past (between 5000 and 15 000 years BP). 52 –55 Examples include evolved traits for lactose tolerance and starch digestion. 56 Many genes have adapted less well to our contemporary Western diet and hasten the onset of common chronic degenerative disorders—diabetes, cardiovascular diseases, and certain cancers. Despite this, some environmental phenomena are still in tune with our genes and affect us in fascinating ways. As an example, the genome and environment are tightly interactive soon after conception. We have previously demonstrated this for both folate and vitamin D genes, 16,22 –24 but it is recognized that UV-R can also attenuate the maternal immune system leading to cytokine production that influences the fetal genome. 57
Geophysical cycles impart a rhythm to life and are critical to our biology; accumulating evidence indicates season of birth modifies disease risk and life span. 57 Two vitamins for which this may most easily be demonstrated are the 2 light-sensitive micronutrients—vitamin D and folate. In the context of folate, schizophrenia and neural tube defects have a similar rhythm of seasonal conception, 58 with schizophrenia exhibiting a 5% to 8% bias toward late winter/early spring conception. 59 We know that pharmacologic folic acid prevents neural tube defects (NTD), and the key C677T-MTHFR folate-related gene polymorphism has greater prevalence in both NTD 60 and schizophrenia. 61 Furthermore, maternal oxidant stress inhibits neural tube closure and alters left–right embryonic asymmetry as might occur in the schizophrenic brain. 62 Both clinical phenotypes involve concurrent fourth-embryonic week processes sensitive to folate degrading oxidant stress, a likely action of pro-oxidant sunlight action, which has coined the “solstitial” hypothesis, a paradigm linking month of birth to left-handedness and other markers of lateralization. 63 However, one of the most interesting phenomena linking folate to seasonality, and one that might be critical in both schizophrenia and neural tube defects, is the recent finding that the day length a woman experiences during the periconceptional period predicts the C677T-MTHFR genotype of her child. 22 The biologic mechanism for this is unclear, although UV-A dermal destruction of 5-methyl-H4folate leading to a lower cellular 5,10-methylene-H4folate status 15,64 might increase the viability of TT genotype embryos and hence population mutant T-allele frequency.
A similar story exists for vitamin D, helping account for environment-related disease phenomena. Seasonality at conception is linked to multiple sclerosis, 65,66 and month of birth influences immune-mediated disease implicating UV-B and vitamin D as risk factors. 67 In this latter report, the risk of immune-mediated disease (Crohn’s disease, rheumatoid arthritis, ulcerative colitis, and lupus) was inversely correlated to second trimester UV-B exposure and third trimester vitamin D status. 67 Interestingly, seasonality at conception is also associated with brain tumors in adults, 68 expression of biogenic amine-related genes in psychiatric patients, 69 and in the development of specific behavioral traits. 70 The present authors demonstrate that the longer the photoperiod at conception, the less the likelihood of depression in adulthood, 24 a relationship that might potentially be linked to vitamin D.
A recent large-scale Norwegian study indicates that solar activity at birth predicts infant survival and women’s fertility. 71 The authors relate this phenomenon to possible UV-R-related effects on vitamin D and folate and discuss the possible role of vitamin-related gene variants. This study builds on the work of Lucock and colleagues 22 –24 and is supported by similar earlier large studies that indicate that month of birth 72 and the solar cycle 73 actually influence longevity, with Marzullo and Fraser 58 reporting that the human embryo responds to both photoperiod and oxidant stress and suggest that later life disease correlates could be a consequence of this. Another recent, supportive large population study concluded that life expectancy at age 50 years depends on specific embryonic or in utero events. 74
The most logical basis for seasonal effects at conception would seem to relate to photoperiod; however, as has been shown, the quanta of solar radiation received could also be a factor: radiation intensity is linked to the solar cycle, with individuals born at the peak of such cycles living 1.5 years less than those born in nonpeak years. 57 Additionally, the solar cycle and hence solar irradiance seems to have a direct effect on gene occurrence. 23,24
Foster and Roenneberg 75 describe how geophysical cycles influence temporal biology, and how despite modern man’s apparent isolation from seasonal influences such as temperature, photoperiod, and nutrient availability in industrialized nations, seasonality still has a significant impact on human developmental processes and our subsequent health. Since seasonality is closely linked to agriculture/diet, Disanto and colleagues 67 have suggested that seasonal effects can act before birth via the “fetal origins of adult disease hypothesis,” something the present authors have also suggested in the context of geophysical events. 24 This may translate into season of birth (more correctly the prenatal timing of specific developmental processes) influencing susceptibility to disease. This has been shown to occur for multiple sclerosis and type 2 diabetes, 2 conditions closely linked to vitamin D. Foster and Roenneberg describe 28 conditions (taking account of hemispheric differences) that link general pathologies, psychiatric disorders, and neurological illness to month of birth. 75 Many of these have folate or folate genes, or related epigenetic phenomena implicated in their etiology. 76 This supports an argument for environmental modulators of photolabile or photosynthetic vitamins mediating disease risk via a complex downstream interaction of genetic/epigenetic phenomena, thus providing a plausible explanation for seasonality in developmentally originated disorders. 76
Of course, vitamin D status per se is also increasingly correlated to chronic degenerative diseases, including hypertension, cardiovascular disease, diabetes, stroke, osteoporosis, multiple sclerosis, obesity, and several cancers including colorectal, breast, pancreatic, and prostate cancer. Individual studies have been discussed in a recent review that additionally explores the role of VDR nutrient–gene interactions in respect of a range of clinical correlates. 77
Vitamin D–Folic Acid Relationship: A Novel Evo-Devo Paradigm Linking Light-Sensitive Vitamins to Human Phenotype Within and Across the Lifecycle
It is well established that the vitamin D endocrine system plays a critical regulatory role in maintaining health. In particular, it controls calcium homeostasis and hence bone integrity, cell growth/differentiation, and immune response.
4
Increasingly, this vitamin, which is better classified as a steroid hormone, is being associated with an ever wider range of disorders: from cancers to vascular disease and diabetes.
78
Such a large sphere of influence becomes less surprising when one recognizes that the
Vitamin D is a light-sensitive hormone, its synthesis involves the UV-B (295 nm) catalyzed epidermal conversion of 7-dehydrocholesterol into previtamin D3. This intermediate isomerizes into calciol, which is then metabolized to calcidiol, and subsequently on to the final active metabolite, calcitriol. Folate is another light-sensitive vitamin and may have a synergy with vitamin D in the context of defining phenotype within and across the lifecycle. While vitamin D is synthesized by UV radiation, folate is readily degraded by it. Folate is as important to biological processes as vitamin D, and is necessary for both de novo biosynthesis of DNA-thymidylate (dTMP) and methionine derived methyl groups (genomic [CpG] and nongenomic methylation reactions). It is also required for purine synthesis and serine–glycine interconversion as well as histidine catabolism. 79 This means a shortage of folate can lead to uracil being misincorporated into the primary base sequence in place of thymine, and hence it can promote DNA fragility. As half our methionine is provided de novo, a folate shortage can also adversely influence the methylome and hence disrupt epigenetic control. 80 Dysregulated folate nutrition and/or genetics are now unequivocally associated with neural tube defects (NTD) and many other developmental and degenerative disorders, including cancers, vascular disease, and neuropsychiatric disorders. 81
As discussed above, there is increasing evidence that geophysical cycles (particularly during the earliest phase of the lifecycle) influence human biology and clinical phenotype. Given the critical role that these 2 light-sensitive vitamins play in cell metabolism, recent research opens up some interesting ideas on how environment (UV-R) might alter/modulate the systemic level of these vitamins that are required as cofactors/ligands for key proteins that have variable activity depending on genotype. Where such polymorphic proteins are absolutely critical for early embryo development, it is conceivable that certain “UV-R–vitamin–genotype” combinations might lead to embryo loss. For example, low systemic levels of folate or vitamin D might favor selection of embryos with specific vitamin-related gene variants that have expression products that are more effective at utilizing low vitamin levels. If selected, such variants might alter disease risk later in life according to an individual’s long-term nutritional habits. 22 This argument has been developed for the folate-related C677T-MTHFR variant and seems plausible given that an estimated 70% to 80% of pregnancies are lost after conception.
With the above in mind, evidence is now pointing to these environmental and nutritional agents interacting to modify genotype–phenotype relationships across the lifecycle in a way that fits the “developmental origins of adult disease” paradigm, but that also provides a molecular explanation for the current hypothesis that UV-R photosynthesis of vitamin D and photodegradation of folate directed the evolution of parallel but opposing phenotypic clines of skin pigmentation. 24 The premise being that the aberrant effects of folate degradation on fecundity promotes protective melanization toward equatorial latitudes, while the need for vitamin D photosynthesis and calcium balance facilitates epidermal depigmentation moving away from equatorial latitudes.
Research findings justify further study, and cannot fail to pique interest into this area of human ecology. As alluded to earlier, increasing evidence indicates that elements of our biology and phenotype are tied to the geophysical cycles we are subjected to early in life and involve seasonality including photoperiod, UV-R exposure, temperature, and so on. Given Darwinian thinking, it should not be any surprise that these types of environmental phenomena are tuned into our genome and are likely to be tightly interactive during the periconceptional period.
The arguments that link folate to such events have been reported by several groups and are summarized in a recent review article by one of the present authors. 16 Two roughly concurrent articles explain how both photoperiod and total solar irradiance at conception influence the occurrence of specific folate genotypes, most notably the C677T-MTHFR, and a case is made to implicate UV-R degraded folate as a pressure that offers a selective advantage to embryos with the 677T allele, as this likely favors dTMP synthesis. 22,23
However, the main purpose of the present article is to draw attention to our recent 2014 finding
24
that shows solar irradiance and photoperiod does indeed influence vitamin D and folate genotype. The folate genotypes studied (C1420T-SHMT, 19 bp del-DHFR, 2R3R-TS) encoded expression products critical for the elaboration of DNA, while all main
Identifying environmental and nutritional agents that interact to modify gene–phenotype relationships across the lifecycle is important, but how this paradigm might have influenced the evolution of skin pigmentation is an equally fascinating area to consider.
A Paradigm to Potentially Explain a Folate- and Vitamin D–Mediated Molecular Mechanism by Which UV-R Selects for Skin Pigmentation
The role of photosensitive vitamins in the evolution of skin pigmentation is fascinating; however, as alluded to earlier, many genes are likely to have been important in this process. One of the most significant was
Detailed molecular mechanisms for the role of vitamin D in human evolution are lacking; however, vitamin D per se is clearly important as a potential target for putative selection processes. Higher follicular fluid and serum calcidiol predicts success of in vitro fertilization,
83
while during pregnancy a woman’s calcidiol level jumps 4- to 5-fold to sequester the additional calcium needed for fetal skeletal growth raising potential issues at higher latitudes where maladapted pigmentation profile, sunscreen use, or cultural issues might promote insufficiency.
84
Furthermore, vitamin D inadequacy in early life can lead to pelvis malformation and an inability to facilitate normal childbirth.
85
The real question has to be, “How does the
Previous research has shown that both
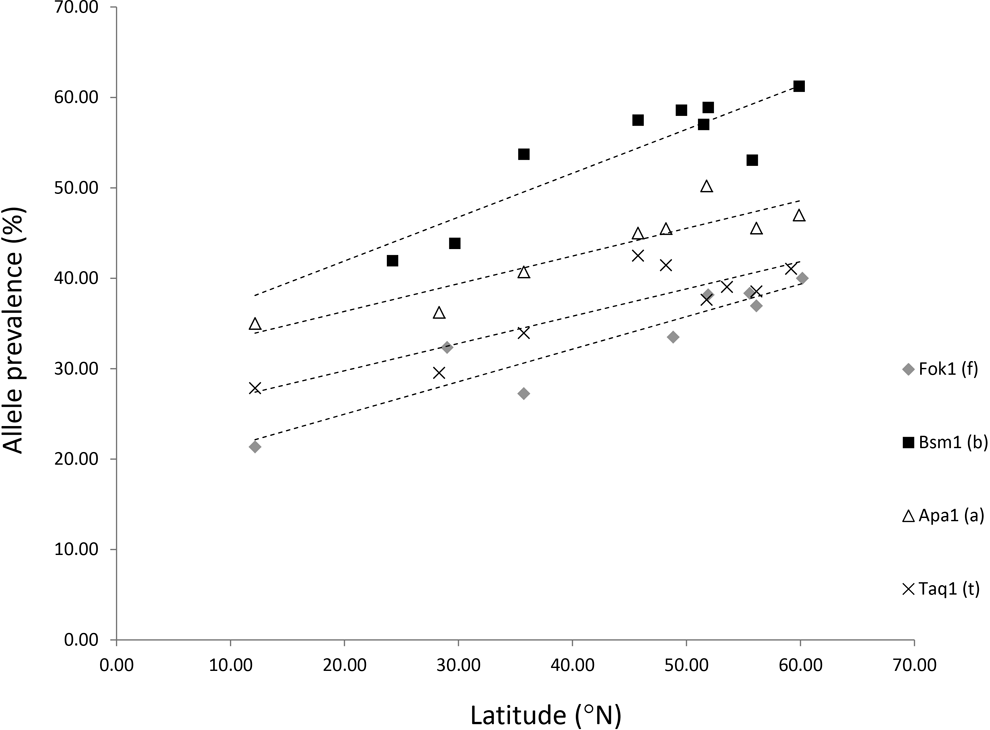
Relationship between the frequency of 4
One hypothesis that would explain this apparent paradox relates to the fact that while UV-B is responsible for the synthesis of vitamin D in the skin, UV-A can actually destroy vitamin D. Since UV-A has better penetration of both ozone and human skin than does UV-B/C (NASA SAGE III-SOLVE II), 2,22 it is likely to be a highly relevant component of the TSI in the context of the balance between vitamin D stability and photodegradation: As alluded to earlier, calciol synthesized in the skin by the action of UV-B can be degraded by UV-A after only 10 minutes of nontropical sun, 7,18 although this degradation becomes less relevant in winter. While this photoisomerization prevents hypervitaminosis D, the implication is that UV-A degrades vitamin D3 at times of the year when there is inadequate UV-B to photosynthesize cholecalciferol in the skin 7 and hence generate biologically active 1,25(OH)2D3. Calcitriol, the biologically active form of vitamin D, is itself UV-A unstable and has a short half-life of 15 hours in the circulation. 7,18 –20 By contrast, calciol has a half-life of around 2 weeks in the circulation. Some fat storage does occur.
Despite these vitamin stores, they may be inadequate for lean people when UV-B photosynthesis of the vitamin is absent. 21 Despite current ideas on depigmentation, UV-B synthesis of vitamin D will become increasingly marginal as one moves further north from the equator due to greater seasonality and increasing levels of destructive UV-A. To emphasize this point, the annual fluence of UV-B varies more with latitude than does UV-A. This is due to greater absorption of UV-B by the ozone layer. Moan has quantified this difference: The annual fluence of UV-B radiation at 310 nm at 60°, 45°, and 30° latitude are, respectively, 20%, 40%, and 65% of the annual fluence at the Equator. However, the corresponding figures for 60°, 45°, and 30° latitude for UV-A at 360 nm are 60%, 80%, and 92%, respectively. 105
To explain the paradox alluded to above, our study
24
shows that predominantly UV-A-related radiation around conception increases the BsmI b allele, while the increase in the prevalence of the b allele with increasing latitude (Figure 3) might also reflect increasing levels of UV-A (perhaps in combination with overall lower UV-B levels and stronger seasonality). While a latitudinal relationship for
It is recognized that in northern Scandinavia/Lapland, skin pigmentation is darker than would be predicted.
3
The UV-R regime of these latitudes is almost exclusively UV-A throughout the year, with virtually no UV-B apart from a low summer exposure. Thus, habitation at these latitudes without reliance on a vitamin D–rich diet of marine animals would be impossible. Much of the dietary vitamin D stores are found in body fat.
109
This supports a possible evolutionary connection between the development of generous subcutaneous fat stores and vitamin D storage in these populations. This may further help explain a link to insulin as alluded to earlier. Ultimately, the selection pressure for depigmentation in these populations is relaxed because of a high vitamin D diet, with darker skin enabling protection from high levels of UV-A as a result of not just direct solar irradiation but also UV reflection from snow and ice.
2
Given the global influence of VDR on homeostasis, other, yet to be discovered, critical early life events and evolutionary processes may also be modulated by vitamin D responsive elements. Effects are likely to relate to the functional consequences of different
We have also speculated on a mechanism by which folate might be acted on by UV as a selection pressure for melanization
24
: Periconceptional exposure to UV-R may play a role in defining critical folate-related genotypes (
Relevance at a Population Level
There has been significant debate on excessive versus inadequate UV-R exposure, much of it arising from the need to synthesize calciol in the skin without risking the development of skin cancers. This is quite a challenge in health promotion given that 2 to 3 million non–melanoma skin cancers and 132 000 malignant melanomas occur globally each year. 77 Martin and colleagues 77 draw attention to the fact that although health professionals advise the use of protective clothing and SPF sunscreen, this can prevent the photoconversion of 7-dehydrocholesterol to previtamin D3. However, they also list other studies where the opposite has been shown to be true.
The concentration of blood calcidiol is the best measure of vitamin D status. There has been conjecture on levels of adequacy, a minimal level of 75 nmol/L per day has been recommended for both skeletal and general health. However, more recently, a level of 50 nmol/L per day has been established as being adequate. At a level of <25 nmol/L per day, mild to severe vitamin D deficiency is likely. 85,110
Ascertaining a safe level of sun exposure has led to models based on skin types and erythemal dose, which is a measure of the time taken for UV-R to cause a slight reddening of the skin. In Australia, the Cancer Council has released broad recommendations on achieving adequate vitamin D synthesis while remaining “sun safe.” For a typical light-skinned individual, exposure should be limited to a few minutes of sunlight either side of the diurnal sunlight peak (ie, 10.00
Footnotes
Acknowledgements
The authors wish to thank all former staff and students who have contributed to our improved knowledge of vitamin D and folate-related biology.
Author Contributions
Idea researched and developed by ML;
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
