Abstract
Herbal mannas are the sweet secretions usually created by insects’ activities on some plant species. Several types of herbal manna have been administered by herbalists for different therapeutic properties in Persian traditional medicine. The most repeatedly described manna in mediaeval Persian literatures are
It is a resinous, sweet, yellow-brown substance with a tear-like shape (1-3 mm) that is produced by the nutrition–secretion activity of
Taranjebin has been mainly used as a mild laxative and in the treatment of neonatal jaundice in Persian ethnomedicine. There are several clinical and in vivo studies on its therapeutic effects on neonatal jaundice. 7 –9 It has been used as a sweetener, mild laxative, antitussive, and demulcent in sore throat, mild cholagogue, and antispasmodic, diuretic, detersive for bladder, motive for humors, and as a body warmer. It was suggested in traditional Persian literature for health maintenance and to treat mild fever. In Iranian ethnomedicine, this manna has been used for treatment of rubella. On the other hand, it was contraindicated in acute fever, measles, smallpox, dysentery, hematuria, and piles. The dosage of the manna varied from 20 to 150 g, which depends on ailment and patient conditions. 10 –12
Controversial indications and contraindications could be observed for fever, infectious, and viral diseases such as measles in old literatures and today’s herbalist prescriptions. It seems that antipyretic and antiviral properties of the manna are possible because of the immunomodulatory properties of
Materials and Methods
Materials
Human Jurkat E6.1 cells (NCBI code: C121, Jurkat-FHCRC cell line) were purchased from Pasteur Institute of Iran. The cell proliferation reagent WST-1 was purchased from Roche Applied Sciences (Germany). Other materials, reagents, and solvents were of the highest available chemical purity and were purchased from Merck or Sigma-Aldrich.
Separation of Soluble and Insoluble Fractions
Isolation and Characterization of Water-Soluble Carbohydrate Biopolymers
To isolate water-soluble biopolymers, ethanol (4 volumes) was added to the soluble part of
Structural Characterization of Biopolymers
Total sugar content of polysaccharides was determined spectrophotometrically by phenol–sulfuric acid assay with glucose as a standard. The protein content of the carbohydrate macromolecules was evaluated by Bradford assay, and the uronic content was determined spectrophotometrically against standard galacturonic acid. 16 The presence of starch backbone for the polysaccharides was tested by adding 2 drops of an aqueous iodine–potassium iodide solution to the samples. Starch was used as a positive control, which gives a dark bluish color with the reagent. 17
Cell Viability by WST-1 Assay
Human Jurkat E6.1 cells were cultured in complete media (CM10 containing RPMI 1640 supplemented with 10% fetal calf serum, 100 U/mL penicillin, and 100μg/mL streptomycin) at 37°C in 5% CO2 at 90% humidity. The cells were then seeded in 96-well flat-bottom plates at a density of 2 × 104 cells per well and 100μL medium containing different concentrations of
Statistics
Analysis of variance was used to analyze the data followed by the LSD-
Results and Discussion
Characterization of Polysaccharide and Carbohydrate Macromolecules
As shown in Figure 1, 91.74% of
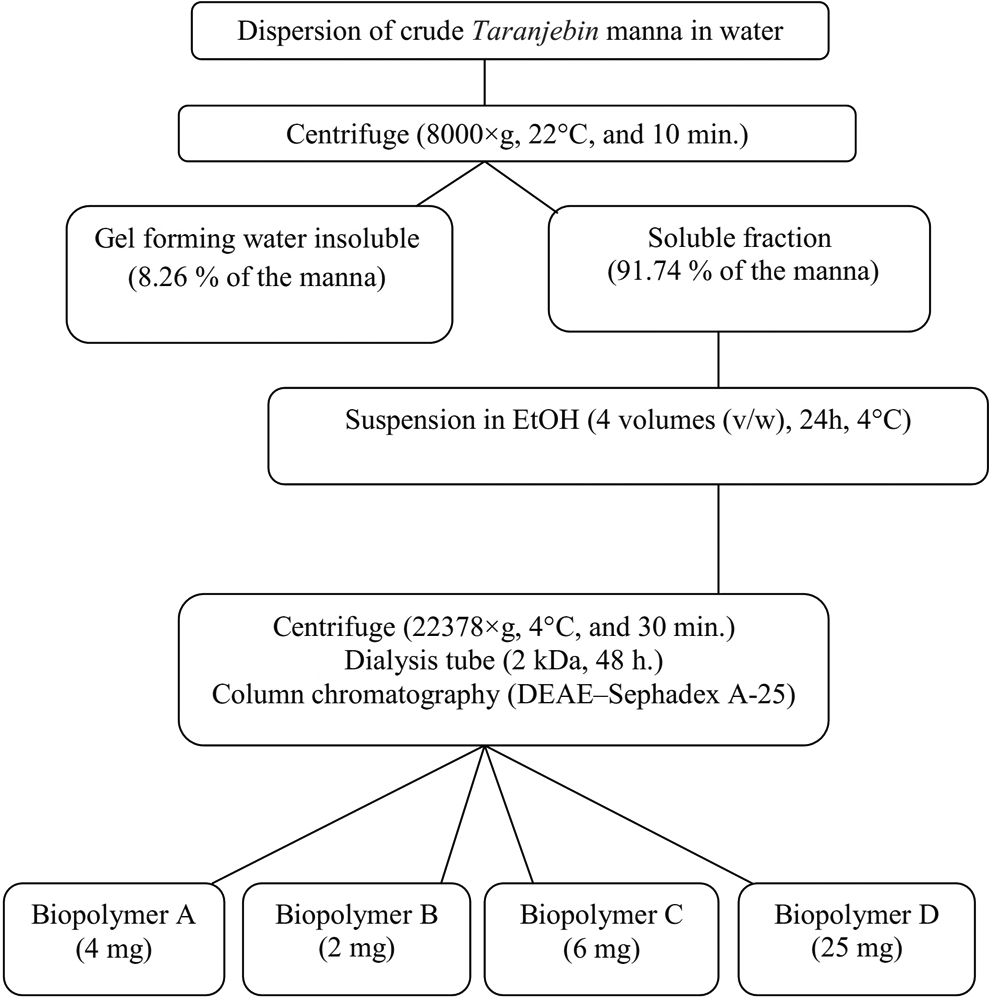
Procedure for biopolymer isolation from
The water-soluble portion of
Since water solubility has an important role on bioavailability and biological properties of biopolymers and macromolecules,
19
only the water-soluble macromolecules of
Among the water-soluble macromolecules, macromolecule D had the highest yield.
No protein content was detected in the isolated biopolymers. The highest uronic acid content was detected in A and D. The ratio of glucose to galacturonic acid was 2.43 for A, 9.57 for B, 2.39 for C, and 2.50 for D. Using iodine–potassium iodide reagent revealed that none of the isolated macromolecules has a starch backbone. All the isolated macromolecules had galacturono-glycan (acidic heteroglycan containing galacturonic acid) structure.
Immunomodulatory Activities of Taranjebin Macromolecules
In this study, Jurkat cells were used to study the immunomodulatory activities of the isolated macromolecules of

The effects of isolated macromolecules (A, B, C, and D) and crude water-soluble fraction of
According to the IC50 value, the order of sensitivity of the Jurkat cell line to the isolated macromolecules was C > A > B > D (Table 1).
IC50 Values of Isolated Macromolecules of

aData are expressed as the mean ± standard deviation.
In this study, etoposide was used as the positive control, and even at the lowest concentration, the cytotoxicity was higher than 50% (IC50 < 3.91 μg/mL).
According to the IC50 value, macromolecule D showed no significant cytotoxicity (Table 1). In contrast, the crude water-soluble fraction of
Since this study was designed to investigate the immunomodulatory effects of
As we know, there is no other report on the immunomodulatory or cytotoxic properties of
On the other hand, Etebari et al evaluated the genotoxicity of aqueous extract of
In another study by Upur et al, the effects of a traditional polyherbal formulation (Abnormal Savda Munziq), which contained
All the isolated macromolecules (A, B, C, and D) in this study were acidic heteroglucan containing galacturonic acid with different ratios. Isolation of acidic polysaccharides was reported from
Goncharov et al isolated a polysaccharide fraction from
We could not find any report on the immunomodulatory or cytotoxic properties of this manna but acidic glucan from other sources such as
On the other hand, the cell line that was applied in this study (Jurkat cells) is an immortalized human T lymphocyte that phenotypically resembles resting human T lymphocytes. This cell line not only has been used in many reports as a target for testing the effects of new antitumor compounds but also is usually used to investigate the physiology and stimulation of T cells. 21,38,39
Considering the nature of Jurkat cells, the high IC50 (in comparison to etoposide) and the proliferative properties of the crude water-soluble fraction of
Conclusion
The results of present study showed that the total aqueous fraction of
Footnotes
Authors’ Note
This study was a part of the PharmD thesis project of Mohammad Reza Karami.
Author Contributions
AH wrote the draft and contributed in guidance and data collection. SH contributed in the guidance and revisions of final version of the article. MRK contributed in data collection and analyzing data.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financed by Shiraz University of Medical Sciences (Grant No. 93-01-70-7984).
Ethical Approval
This study was an experimental and laboratorial work and did not require ethical approval.
