Abstract
The present study aimed to examine the immunomodulatory effect of ethanolic extract of
Some chemical constituents have been identified from
In recent years, natural products have become a vital part of complementary and alternative medicine. Natural products that show antitumor activity have been gaining popularity among those faced with the deleterious side effects of treatment using chemotherapeutic drugs or synthetic compounds for cancer treatment. The resulting immunosuppression induced by chemotherapeutic has been suggested to be a major contributing factor to exacerbate in cancer mortality. The recovery from immunodeficiency induced by chemotherapy remains a major clinical problem in cancer patients. Typically, hematological recovery is medically considered to have occurred when the number of neutrophils and platelets have been restored to the normal level. However, molecular modulations and cellular interactions in lymphoid and cytokine system are required for recovery of the immune system function and should also be considered. 7 Therefore, it is interesting to design therapeutic approaches to improving anticancer treatments in order to reduce the side effects.
In the present study, the immunomodulatory effects of ethanolic extract of
Materials and Methods
Plant Materials
Fresh tuber of
Plant Preparation and Ethanolic Extraction
Fresh plants were harvested and washed thoroughly with running tap water and then with distilled water. The tubers were air dried and then oven dried under reduced temperature. The fully dried plants were powdered and weighed before cold maceration. The powder of
Animals
Healthy Wistar rats were obtained from the Animal Experimental Unit, Animal Research and Development Centre, Gadjah Mada University, Yogyakarta. The animals were housed and maintained under the standard conditions of 12-hour light/dark cycle, 25° ± 2°C and 60% to 70% humidity and were fed with standard rat chow and water ad libitum. The experimental protocol was conducted in accordance with the Guideline for Care and Use of Animals Laboratory and approved by the Institutional Animal Ethics Committee of Universitas Gadjah Mada.
Experimental Animals
Thirty-six normal rats were divided randomly into 6 groups of 6 rats each group and used in the experiments. Group I served as normal rats received vehicle (carboxymethyl cellulose Na 0.5%), group II served as cyclophosphamide-treated rats (150 and 110 mg/kg body weight on day 1 and 4) and were administered carboxymethyl cellulose Na 0.5% orally once daily for 7 consecutive days; groups III, IV, and V received variation of doses of 250, 500, and 1000 mg/kg body weight of the ethanolic extract of
Proliferation of Lymphocytes
On day 7, the rats were sacrificed and a surgery was performed to take out the spleen and centrifuged (1200 rpm, 4°C, 10 minutes). The pellets obtained were suspended in 0.01 M Tris-buffered ammonium chloride, pH 7.65, and allowed to stand at room temperature for 2 minutes. Then, on the bottom of the tube was added fetal bovine serum (Gibco BRL, Grand Island, NY, USA). The suspension was then centrifuged (1200 rpm, 4°C, 5 minutes), and the supernatant was discarded. The pellets were washed with RPMI (Roswell Park Memorial Institute) medium twice and centrifuged. Supernatant was discarded and the cell resuspended in a complete medium. Cells were cultured with a density of 106 cells/mL in a complete medium and then incubated in 5% CO2 incubator at 37°C for 48 hours. Proliferative activity of culture supernatant was measured by a MTT (3-(4, 5-dimethylthiazolyl-2)-2,5-diphenyltetrazolium bromide) test assay.
Phagocytic Activity of Macrophages
On day 7, rats were sacrificed with a light anesthetic. The abdomen was opened and cleared from the peritoneal sheath with alcohol 70% (v/v) and cold RPMI-1640 (Sigma Chem, St Louis, MO, USA) was injected into the peritoneal cavity. The animals were allowed to stand for 3 minutes until macrophages attached to the peritoneal cavity and around the intestine can be detached and suspended in the RPMI medium. Peritoneal fluid was removed from the peritoneal cavity and aspirated with a syringe. Peritoneal fluid was aspirated and centrifuged at 1200 rpm, 40°C for 10 minutes. Supernatant was removed and then added 3 mL of complete RPMI medium containing fetal bovine serum.
The phagocytic activity was performed in vitro according to the previous method using a diameter of 3.0 μm latex beads (Sigma Chem). 8 The beads were resuspended in phosphate-buffered saline (Sigma Chem) in order to get concentrations of 2.5 × 107/mL. Peritoneal macrophages were cultured previously washed twice with RPMI-1640, and add 200 mL of suspension of beads in each wells and incubated in a 5% CO2 incubator, 37°C for 60 minutes. The cells then washed with phosphate-buffered saline 3 times, then dried at room temperature and fixed with methanol. Methanol was then removed and allowed to stand until dry cover slips. After drying, cover slips were stained with Giemsa (Merck, Darmstadt, Germany) 20% (v/v) for 30 minutes and washed with distilled water, removed from culture wells and dried at room temperature. The macrophage cells (100 cells) were observed using a light microscope and counted the number of phagocytic activity of macrophages to latex bead particles and the number of latex beads that uptake by macrophages.
Determination of CD8 Profiles by Flow Cytometry
Blood samples were collected from treated rats on days 0 and 7 under anesthetized condition and kept in a vacutainer containing ethylenediamine tetra-acetic acid. Sample preparation was performed by mixing 5 μL of whole blood and 10 μL was rat antibody CD3 antigen fluorescein isothiocyanate and PE anti-rat CD8a and then was vortexed gently and allowed to stand in a dark room for 15 minutes. For dilution, lysing reagent was added, then allowed to stand in a dark room for 15 minutes. After immunolabeling, cells were analyzed on a FACS Calibur Flow Cytometer and CellQuest Pro IVD software (Becton Dickenson, Mountain View, CA, USA).
Detection of Cytokines Levels
Tumor necrosis factor-α, IL-1α, and IL-10 levels were measured by enzyme-lnked immunosorbent assay using the commercially available kits listed (Table 1), as previously described. 9 One hundred milliliters of standard (diluted in the lysis buffer) or each properly diluted sample of plasma was analyzed in triplicate, and a portion of the sample was assayed for protein content. Data were expressed as pg/mg of protein.
Primary Antisera Used in This Study.

Abbreviations: ELISA, enzyme-linked immunosorbent assay; FITC, fluorescien isothiocyanate; IL, interleukin; TNF-α, tumor necrosis factor-α.
aAntisera were raised in rat.
Statistical Analysis
Data from all experiments were presented as mean values with ± standard error of mean (SEM). Statistical analysis was performed by 1-way analysis of variance with Bonferroni post hoc test (2-tailed).
Results
Proliferation of Lymphocytes and Phagocytic Activity of Macrofages
The ethanolic extract of
Effect of Ethanolic Extract of
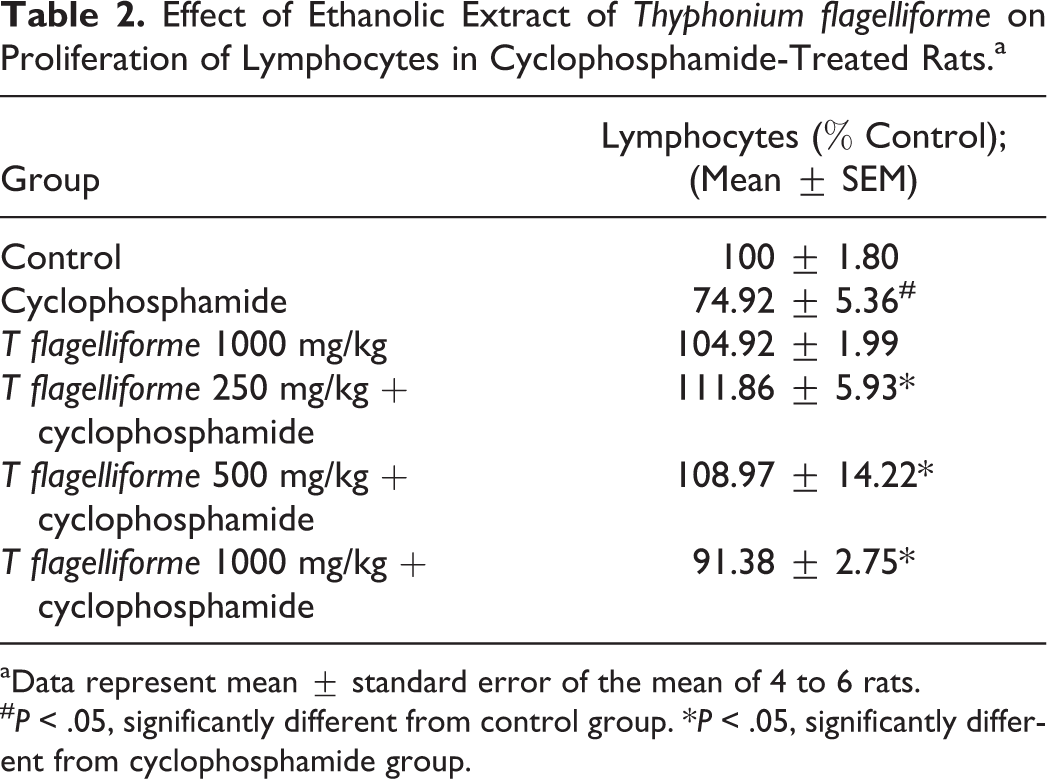
aData represent mean ± standard error of the mean of 4 to 6 rats.
#
Effect of Ethanolic Extract of

aData represent mean ± standard error of the mean of 4 to 6 rats.
#
Determination of CD8+T cells by Flow Cytometry
The results demonstrated that ethanolic extract of
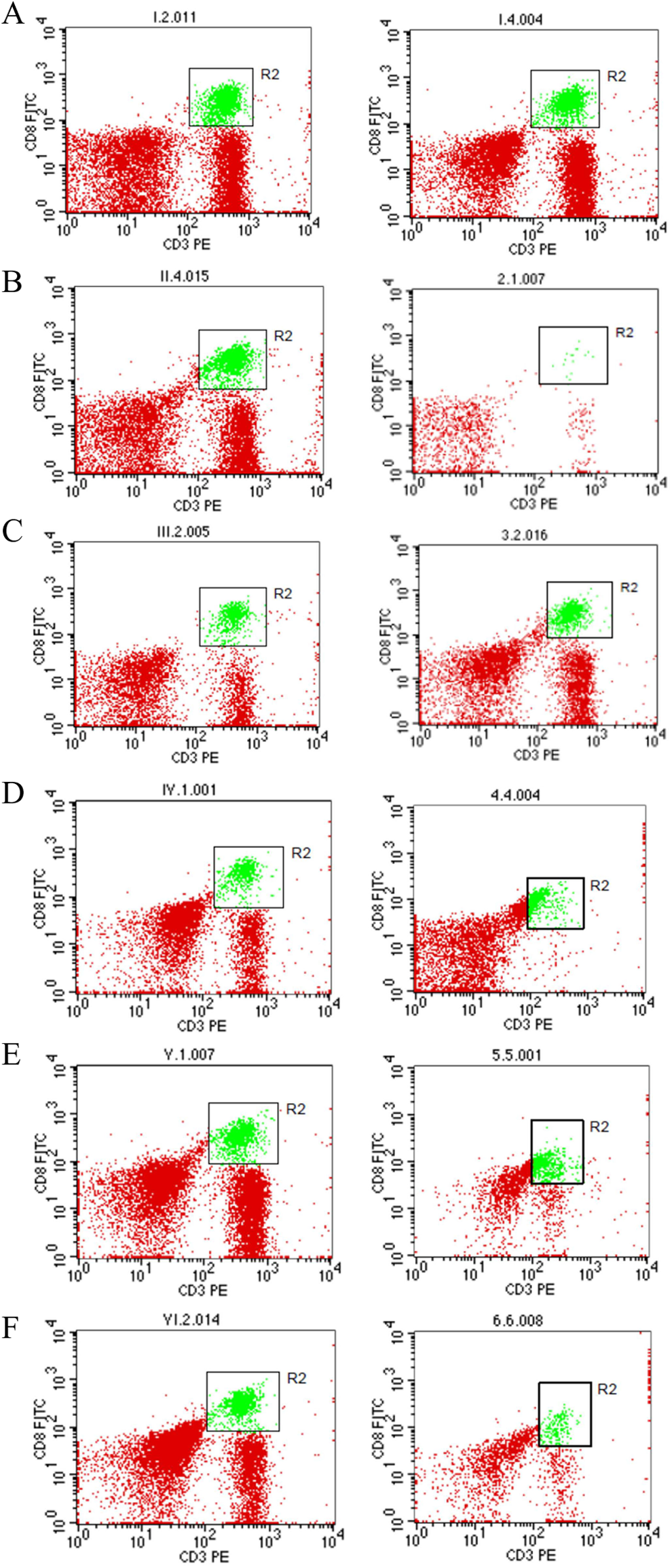
Effect of ethanolic extract of
Effect of Ethanolic Extract of

aData of CD8+ T cells were measured on day 0 (pretreatment) and day 7 (posttreatment), which represent mean ± standard error of the mean of 4 to 6 rats.
#
Detection of Cytokines
Furthermore, immunomodulatory effect of ethanolic extract of
Effect of Ethanolic Extract of

Abbreviations: IL, interleukin; TNF-α, tumor necrosis factor-α.
aData represent mean ± standard error of the mean of 4 to 6 rats.
#
Discussion
We have demonstrated in the present study that the oral administration of ethanolic extract of
Cyclophosphamide is an antineoplastic drug that is widely used, in single or combination with other products. 10 Cyclophosphamide, is known to be biologically active after biotransformed by microsomal enzymes, leading to the production of a number of active metabolites capable of alkylating nucleic acid and chromosome damage through the generation of free radicals, thus promoting mutations. 11,12 Previous reports suggests that the cyclophosphamide can be used as an immunosupressive agent. Several lines of evidence demonstrated that cyclophosphamide was able to induce immunosuppression and or mutagenicity in mice and has been used as a valuable animal model to detect the effects of immunomodulatory or antimutagenic. 13,14
Lymphocytes have the ability to distinguish foreign objects from its own network, as it has on the cell surface receptor, Toll cell receptor. T lymphocytes (T cells) function in helping B cells produce antibodies, which recognize and destroy infected cells, activate macrophages in phagocytosis and the control threshold and the quality of the immune system. Macrophages are one of the effector cells to eliminate the parasite through the mechanism of phagocytosis in the immune system is not specific. The activity of macrophages can be enhanced by immunostimulant agents, either vaccine or chemical compounds, including compounds from natural products. T cell antigen receptors are always membrane bound and noncovalently associated with a set of four invariant glycoproteins collectively called CD3. Thus, CD3 has been regarded as a marker for T cells. Generally, T helper cells are also known as CD4+ T cells, cytotoxic T cells are known as CD8+ T cells. 15 Cytotoxic T cells CD8+ acts as a factor of antitumor adaptive immune system of the host and become an important prognostic factor in cancer treatment, for example, in cases of colorectal cancer. 16,17
Tumor necrosis factor-α may enhance the immune response in vivo to enhance proliferation of B and T cells and stimulate the formation of cytotoxic T cells. In addition, TNF-α increases IL-2 level, which induces the production of immunoglobulins and an increase in IL-2-stimulated natural killer cell activity and proliferation of monocytes. 18 IL-1 is another cytokine released by various cell types including macrophages, dendritic cells, lymphocytes, endothelial cells, fibroblasts, and keratocytes. Interleukin exists in 2 forms, IL-1α and IL-1β, both 17-kDa glycoproteins, the latter is secreted by cells and membrane bound forms of the former. IL-1 is required for the successful initiation of some forms of immune response. 19 Forms of other cytokines, IL-10 is an immunosuppressive glycoprotein 19 to 21 kDa in size that is secreted by Th2 cells, some B cells, and macrophages are activated. Now, it is clear that IL-10 primarily acts on activated macrophages to suppress the secretion of IL-1, IL-12, TNF-α, and reactive oxygen radicals. 18
In the present study, we found that treatment with ethanolic extract of
In conclusion, immunosuppressive effect induced by cyclophosphamide in rats can be effectively reduced by the oral administration of ethanolic extract of
Footnotes
Author Contributions
AN, MI, and RM were involved in plant collection, processing, and carrying out the experimental work. IPS and AEN supervised the overall study. AN and MI drafted the article. All authors read and approved the final version of the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We are thankful to the Ministry of Education of Indonesia for providing funding for the study via the National Strategic Research Competition Grant (Grant Letter No. 389/SP2H/PL/Dit.Litabmas/IV/2011).
Ethical Approval
The experimental protocol was conducted in accordance with the Guideline for Care and Use of Animals Laboratory and approved by the Institutional Animal Ethics Committee (IAEC) of Universitas Gadjah Mada, Yogyakarta, Indonesia.
