Abstract
Mango ginger (Curcuma amada Roxb.) is among the less-investigated species of Curcuma for anticancer properties. We have investigated the anticancer potential and the mechanism of action of a supercritical CO2 extract of mango ginger (CA) in the U-87MG human glioblastoma cell line. CA demonstrated higher cytotoxicity than temozolomide, etoposide, curcumin, and turmeric force with IC50, IC75, and IC90 values of 4.92 μg/mL, 12.87 μg/mL, and 21.30 μg/mL, respectively. Inhibitory concentration values of CA for normal embryonic mouse hypothalamus cell line (mHypoE-N1) is significantly higher than glioblastoma cell line, indicating the specificity of CA against brain tumor cells. CompuSyn analysis indicates that CA acts synergistically with temozolomide and etoposide for the cytotoxicity with combination index values of <1. CA treatment also induces apoptosis in glioblastoma cells in a dose-dependent manner and downregulates genes associated with apoptosis, cell proliferation, telomerase activity, oncogenesis, and drug resistance in glioblastoma cells.
Introduction
The genus Curcuma contains more than 80 species of rhizomatous herbs of which many have significant value as medicines, dyes, and spices. 1 Curcuma amada Roxb. is an important member of the genus commonly known as mango ginger due to the raw mango–like aroma of the rhizome. It has a morphological and phylogenic resemblance with ginger (Zingiber officinale) but imparts mango (Mangifera indica) flavor. Mango flavor is in part attributed to cis-ocimene among the 68 volatile aroma components present in the essential oil of mango ginger rhizome.2 –5 Ethnobotanically, it is used for treating stomachaches, itching, skin diseases, bronchitis, asthma, hiccough, and inflammation due to injuries.6,7 It is also described as a useful agent against inflammation of the mouth, ulcers of the male genitalia, scabies, lumbago, and stomatitis.6 –8 The ethanol extract of Curcuma amada displayed antifungal activity and broad spectrum of antibacterial activity against several strains. 9 In addition to a sesquiterpene dimer “difurocumenonol” 10 and a substituted sesquiterpene “amadannulen,” 11 6 diterpinoids have been isolated so far from the rhizomes of mango ginger, of which the (E)-labda-8(17),12-diene-15,16-dial is the major ingredient.12,13The (E)-labda-8(17),12-diene-15,16-dial, a novel dialdehyde isolated from the chloroform extract of rhizomes of Curcuma amada has antitubercular activity against Mycobacterium tuberculosis H37Rv strain in BACTEC-460 assay. 12 This compound is also reported to have antifungal activity against several Candida species, mosquitocidal activity against Aedes aegyptii larvae, and cytotoxicity against KB cervical carcinoma cells.13,14
Malignant brain tumors are among the most aggressive forms of human cancer with a high degree of morbidity and mortality. In the United States, more than 190 000 people are diagnosed with brain tumors every year, of which 40 000 are primary. 15 Gliomas, especially glioblastoma multiforme, are the deadliest and the most common form of malignant brain cancer with the survival in most cases limited to a few months after diagnosis. Chemotherapy options for brain cancer, especially high-grade gliomas, are significantly limited due to the poor penetration of the blood–brain barrier and/or drug resistance of cancer cells.15,16 It has been reported that curcumin, the natural polyphenol from Curcuma longa (turmeric) rhizomes, can cross the blood–brain barrier. 17 However, mango ginger (Curcuma amada) is a less-investigated species for anticancer properties. In a recent investigation, we have found that a supercritical CO2 extract (CA) prepared from mango ginger rhizomes has superior anticancer potential than Curcuma longa (turmeric) and Curcuma xanthorrhiza supercritical extracts against human rhabdomyosarcoma cell lines; however, whether bioactives in CA also cross the blood–brain barrier requires further research. Additionally, CA has synergistic cytotoxicity with vinblastine and cyclophosphamide, 18 commonly used for treatment of rhabdomyosarcoma patients. In this investigation, we have analyzed the anticancer potential and the mechanisms of action of CA in human glioblastoma cells in vitro.
Materials and Methods
Cell Line and Cell Culture
Human glioblastoma cell line (U-87MG) was purchased from American Type Culture Collection, Manassas, VA, and the cells were grown in RPMI medium supplemented with 10% fetal bovine serum and antibiotics in a humidified 5% CO2 incubator maintained at 37°C. The normal mouse embryonic hypothalamus cell line (mHypoE-N1) was purchased from CELLutions Biosystems, Inc, Burlington, Ontario, Canada, and grown in Dulbecco’s Modified Eagle Medium, supplemented with 10% fetal bovine serum and antibiotics in the CO2 incubator.
Drugs and Curcuma Extract
Supercritical CO2 extract of Curcuma amada was prepared by Flavex Naturextrakte GmbH, Rehlingen, Germany. The supercritical CO2 extraction is designed to extract all the potential compounds in a uniform product without any residue of organic solvents using high-pressure CO2 unlike other solvent extraction methods. Moreover, the extracted product, usually in a liquefied form, maintains almost similar quality from batch to batch. Temozolomide, etoposide, and curcumin were purchased for the investigation from Sigma-Aldrich, St Louis, MO, and Turmeric Force was obtained from New Chapter Inc, Brattleboro, VT.
Cytotoxicity
Glioblastoma (U-87MG) cells were treated with increasing concentrations of CA, temozolomide, etoposide, or their combination for 72 hours in 96-well plates. MTT [3-(4,5-dimethyl thiazol-2-yl)-2,5-diphenyltetrazolium bromide] assay performed with the Cell Proliferation Kit I (Roche Biochemicals, Indianapolis, IN) was used to analyze cytotoxicity of individual drugs/extracts and their combinations. The experiments were repeated 4 times with 3 replications for each treatment and the IC50, IC75, and IC90 values were calculated.19,20 The relative cytotoxicity of CA in normal mouse hypothalamus cell line (mHypoE-N1) was also analyzed to compare the toxicity and specificity of CA in normal and brain tumor cells.
CompuSyn Analysis
To determine the synergistic/additive/antagonistic effect among temozolomide, etoposide, and CA, cytotoxicity data were analyzed further using CompuSyn software (ComboSyn, Inc, Paramus, NJ). This program is based on Chou and Talalay’s 21 multiple drug effect equations and it defines synergism as more-than-expected additive effect and antagonism as a less-than-expected additive effect.19,20 The combination index was calculated by the Chou–Talalay equations for multiple drug effects, which take into account both potency (inhibitory concentration values) and shape (slope, m) of dose–effect curve.
Apoptosis Assay
U-87MG cells (106/2 mL) were treated with increasing concentrations of CA in a CO2 incubator at 37°C for 48 hours. The drug and/or extract-treated cells were stained with Annexin V-FLUOS kit (Roche Biochemicals) for 15 minutes and analyzed in a Beckman Coulter Elite flow cytometer. The percentages of apoptotic, necrotic, and live cells were estimated from gated flow cytometric scatter diagrams, and the percentage of apoptotic cells were plotted against CA concentrations. 19
Gene Expression Studies by Reverse Transcriptase-Polymerase Chain Reaction Assay
U-87MG cells (2 × 106) were plated in 5 mL of RPMI medium and treated with increasing concentrations of CA (0-100 μg/mL) for 72 hours after cell attachment. Tumor cells treated with camptothecin (5 μg/mL), which induces apoptosis, was used as a positive control. Total RNA was extracted from treated cells and 5 μg RNA was reverse transcribed to synthesize cDNAs. The mRNA expression of genes associated with apoptosis (APOBEC3B, Bax, Bak, Bcl-2, p10, p21, p53, and STAT3), cell proliferation (Ki67, PCNA, and Cyclin B2-CCNB2), drug resistance (MDR-1, MRP, LRP, and DRP), oncogenesis (C-myc, N-myc, and V-jun) and telomerase activity (hTERT and hTER) were analyzed by reverse transcriptase-polymerase chain reaction using 0.4 μg of cDNA using gene-specific primers. 18 The expression of a housekeeping gene, glyceraldehyde 3-phosphate dehydrogenase (GAPDH), was used as a control. The expression of gene was quantified using gel pictures from 3 separate experiments by the UNSCAN-IT Gel software (Silk Scientific, Inc, Orem, UT).
Western Blot Analysis
U-87MG cells (5 × 106/5 mL) were treated with increasing concentration of CA (0-100 μg/mL) for 72 hours, and total cellular protein was extracted with 0.5 mL of Invitrogen’s protein extraction buffer (Invitrogen, Frederick, CA) according to the manufacturer’s instructions. The protein concentration was determined and 100 μg protein was separated on 7.5% sodium dodecyl sulfate polyacrylamide gel electrophoresis. The separated protein was blotted on a nitrocellulose filter. The filters were hybridized with antihuman monoclonal/polyclonal antibodies specific for each protein (Bax, Bak, Bad, Bcl-2, Bcl-X, p53, and Caspase 3) in a western blot procedure and detected using the horseradish peroxidase or alkaline phosphatase color detection kit (Bio Rad Laboratories, Hercules, CA). The relative expression of proteins compared to untreated control sample was quantified using UNSCAN-IT Gel software (Silk Scientific, Inc), and the fold-level changes in protein expression were plotted against CA concentrations.
Statistical Analysis
Mean inhibitory concentrations and standard deviation estimates were calculated using Microsoft Excel software using data from 3 separate experiments. The fraction of surviving cells at each concentration of drugs/combinations was used for the analysis of synergism/additiveness/antagonism between drugs/extracts by the CompuSyn software (ComboSyn, Inc, Paramus, NJ).
The relative mRNA expressions (average pixel units) at different CA concentrations were statistically analyzed by 1-way analysis of variance, and the treatments were compared using Dunnett’s multiple comparison tests (GraphPad Prism software, La Jolla, CA).
Results
Cytotoxicity of CA, Etoposide, and Temozolomide
CA demonstrates significant cytotoxicity to U-87MG cell line compared with the normal mHypoE-NI cell line, as indicated by the cytotoxicity curves presented in Figure 1. The IC50, IC75, and IC90 values of CA are 4.92 ± 0.81, 12.87 ± 0.85, and 21.30 ± 1.13 μg/mL, respectively, which is much lower than the values for curcumin (IC50 = 37.3 ± 4.04; IC75 = 51.00 ± 7.07; IC90 >200 μg/mL) and Turmeric Force (38.51 ± 7.07; IC75 = 40.40 ± 2.97; IC90 = 48.50 ± 0.58 μg/mL). The CA IC50, IC75, and IC90 values for normal brain cell line (mHypoE-N1) are 40.57 ± 0.06, 62.60 ± 1.55, and 85.43 ± 5.03 μg/mL, respectively, which are significantly higher than that for U-87MG cell line, indicating the specificity of CA toward brain tumor cells.
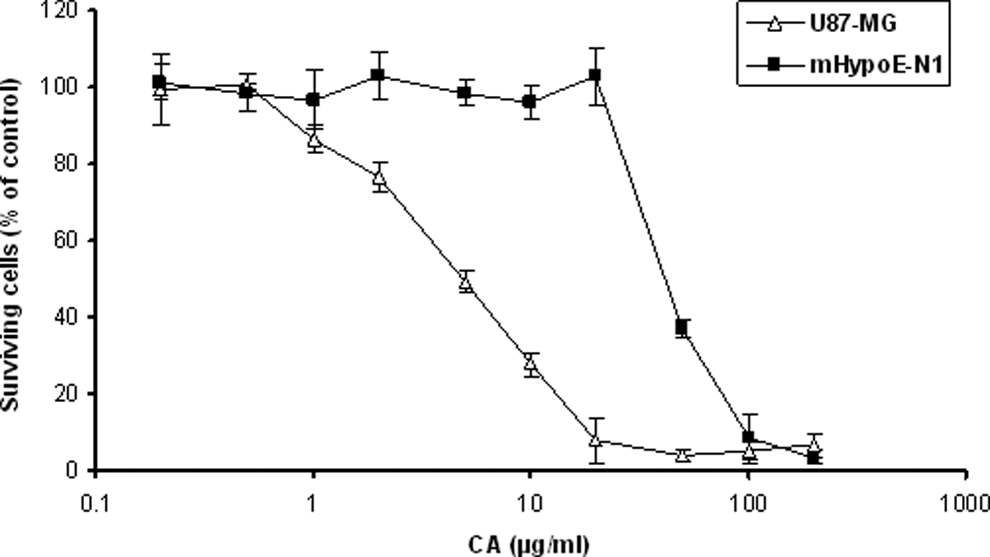
Cytotoxicity of Curcuma amada supercritical CO2 extract (CA) in U-87MG human glioblastoma and normal mouse embryonic hypothalamus (mHypoE-N1) cell lines. Tumor cells were treated with CA for 72 hours and cytotoxicity analyzed by MTT assay. The mean percentage of surviving cells (n=4) and standard deviation estimates were plotted against CA concentrations.
The cytotoxicity values for temozolomide and etoposide in U-87MG cell line (Table 1) indicates that both these agents, unlike CA, have limited efficacy for inducing high percentage of cell death even at high doses. However, CA alone and in combination with temozolomide and etoposide is more effective than single agents in inducing >90% cell death.
Cytotoxicity of Temozolomide (TMZ), Etoposide (ETP), Supercritical CO2 Extract of Mango Ginger (Curcuma Amada) (CA), Curcumin (CUR), Turmeric Force (TF) in U-87MG Human Glioblastoma Cell Line

Synergistic Effect of CA With Temozolomide and Etoposide
The dose–effect plots and medium–effect plots of combinations of temozolomide and etoposide with CA are presented in Figure 2, and the combination index values are given in Table 2. Temozolomide and etoposide with CA show synergistic cytotoxicity as indicated by combination index values of <1 (Table 2).

CompuSyn analysis of cytotoxicity data to determine synergism/additiveness/antagonism between cancer drugs and CA in U-87MG cell line. Dose–effect plots of single agents (A) (temozolomide [TEM], etoposide [ETP], and supercritical CO2 extract of Curcuma amada [CA]) and drug and/or extract combinations (B) (T + E = temozolomide + etoposide; T + C = temozolomide + CA; E + C = etoposide + CA; T + E + C = temozolomide + etoposide + CA) in U-87MG cell line. Medium–effect plots of single agents (C) and drug and/or extract combinations (D).
Combination Index (CI) Values for Drug/Extract Combinations in U-7MG Cell Line.

Abbreviations: ETP, etoposide; TEM, temozolomide; CA, supercritical CO2 extract of mango ginger (Curcuma amada).
a CI value: <0.1 = very strong synergism; 0.1-0.3 = strong synergism; 0.3-0.7 = synergism; 0.8-0.9 = moderate to slight synergism; 0.9-1.1 = nearly additive; 1.1-1.45 = moderate to slight antagonism; 1.45-3.3 = antagonism; >3.3 = strong antagonism.
CA-Induced Apoptosis
The results of Annexin-V-FLUOS staining indicated that CA induces apoptosis in a dose-dependent manner (Figure 3). CA induced 8.7%, 11.4%, and 23.7% apoptosis at 10, 50, and 100 μg/mL concentrations, respectively, in 48 hours.

Analysis of apoptosis induced by CA in U-87MG cell line. Cells were treated with CA for 48 hours and stained with Annexin-V-FLUOS kit (Roche Biochemicals). The percentage of apoptotic cells were plotted against CA concentrations (0-100 μg/mL) and the Camptothecin (5 μg/mL) treated positive control.
Effect of CA on Gene-Specific mRNA Expression
The expression of mRNAs of genes associated with apoptosis, cell proliferation, telomerase activity, drug resistance, and oncogenesis were analyzed by reverse transcriptase-polymerase chain reaction, and the results are presented in Figures 4A to 8A. The relative changes in mRNA expression induced by increasing concentrations of CA are given in graphs (Figures 4B–8B). Among the genes associated with apoptosis, CA treatment induces downregulation of STAT3, Bcl-2, and mutant p53 expression. The ratio of Bax/Bcl-2 was also increased by CA, indicating the level of apoptosis (Figure 4A and B).

(A) Reverse transcriptase-polymerase chain reaction assay of apoptotic gene expression (STAT3, p10, p21, Bax, APOBEC 3B, p53, and Bcl-2) along with housekeeping gene GAPDH in CA-treated U-87MG cells. (B) Quantification of expression of apoptotic genes by UNSCAN-IT gel software. The relative expression of genes (average pixel units) as well as Bax/Bcl-2 ratio is plotted against CA concentrations. The significant difference between treatments was compared by 1-way analysis of variance with Dunnett’s multiple comparison test (GraphPad Prism Software, La Jolla, CA) (*P < .05; **P < .01; ***P < .001).

(A) Reverse transcriptase-polymerase chain reaction assay of cell proliferation gene expression (Ki67, PCNA, and CCNB2-cyclin B2) along with housekeeping gene (GAPDH) in CA-treated U-87MG cell line. (B) Quantification of expression of cell proliferation genes by UNSCAN-IT gel software. The relative expression of gene (average pixel units) was plotted against CA concentrations. The significant difference between treatments was compared by 1-way analysis of variance with Dunnett’s multiple comparison test (GraphPad Prism Software, La Jolla, CA) (**P < .01; ***P < .001).
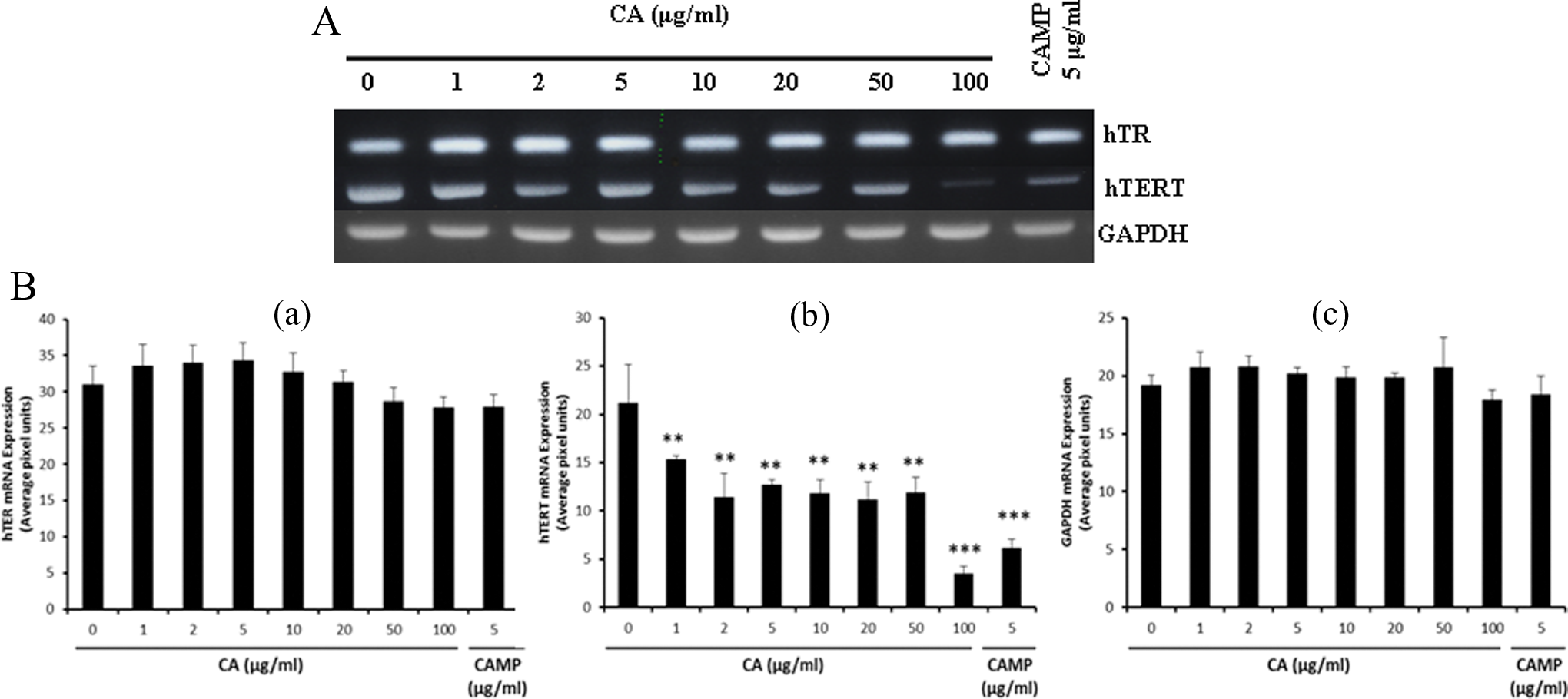
(A) Reverse transcriptase-polymerase chain reaction assay of gene expression associated with telomerase activity (hTER and hTERT) along with GAPDH in U-87MG cell line. (B) Quantification of expression of genes associated with telomerase activity by UNSCAN-IT gel software. The relative expression of gene (average pixel units) was plotted against CA concentrations. The significant difference between treatments was compared by 1-way analysis of variance with Dunnett’s multiple comparison test (GraphPad Prism Software, La Jolla, CA) (**P < .01; ***P < .001).

(A) Reverse transcriptase-polymerase chain reaction assay of expression of genes associated with drug resistance (MDR-1, MRP, LRP, and DRP) along with housekeeping gene GAPDH in U87MG cell line. (B) Quantification of expression of drug resistance associated genes by UNSCAN-IT gel software. The relative expression of gene (average pixel units) was plotted against CA concentrations. The significant difference between treatments was compared by 1-way analysis of variance with Dunnett’s multiple comparison test (GraphPad Prism Software, La Jolla, CA) (*P < .1).

(A) Reverse transcriptase-polymerase chain reaction assay of expression of genes associated with oncogenesis (N-myc, C-myc, and V-jun) along with housekeeping gene GAPDH in U87MG cell line. (B) Quantification of expression of genes associated with oncogenesis by UNSCAN-IT gel software. The relative expression of genes (average pixel units) is plotted against CA concentrations. The significant difference between treatments was compared by 1-way analysis of variance with Dunnett’s multiple comparison test (GraphPad Prism Software, La Jolla, CA) (*P < .05; **P < .01).
CA treatment of U87-MG cells also downregulates the transcription of genes associated with cell proliferation (proliferating cell nuclear antigen [PCNA] and cyclin B2 [CCNB2]), although Ki67 is unchanged (Figure 5A and B). We have analyzed the expression of 2 important genes associated with telomerase elongation, human telomerase reverse transcriptase (hTERT) and human telomerase genes (hTER), in CA treated U-87MG cells. CA treatment significantly downregulates the expression of hTERT mRNAs among the 2 important genes associated with telomerase elongation (Figure 6A and B). Among the drug resistance genes, low level downregulation is observed in LRP and MRP genes (Figure 7A and B). CA treatment also downregulates the expression of oncogenes like N-myc to a large extent and C-myc to a small extent (Figure 8A and B).
Effect of CA on Protein Expression
The results of protein expression analyzed by western blot hybridization are presented in Figure 9A and B. CA treatment of U-87MG cells induced an increase in the expression of pro-apoptotic proteins (Bax, Bak, Bad, and Caspase 3) with increase in CA doses. On the other hand, the anti-apoptotic proteins (mutant p53, Bcl-2, and Bcl-X) show an overall downregulation with CA treatment.

(A) Western blot analysis of apoptotic proteins (Bax, Bak, Bad, Bcl-2, Bcl-X, p53, and Caspase 3). Total proteins (100 μg) were separated on 7.5% polyacrylamide gels, transferred to nitrocellulose filter, hybridized with gene specific antibodies, and detected using Bio-Rad horseradish peroxidase or alkaline phosphatase coloring reagent. (B) Quantification of protein expression associated with apoptosis by UNSCAN-IT gel software. The relative protein expression (fold change based on untreated control) was plotted against CA concentrations.
Discussion
In the genus Curcuma, mango ginger (Curcuma amada Roxb.) is among the less-investigated species especially for its anticancer and other medicinal properties. However, it is a well-described herb in the Indian Ayuvedic system of medicine.6 –8 Recently, we reported that the supercritical CO2 extract of mango ginger (CA) is superior in terms of its cytotoxicity in human rhabdomyosarcoma cell lines than Curcuma longa and Curcuma xanthorrhiza supercritical extracts. 18 Furthermore, CA acts synergistically with anticancer drugs such as vinblastine and cyclophosphamide in terms of cytotoxicity. In the present study, we have investigated the anticancer properties of CA in human U-87MG glioblastoma cells and explored the molecular pathways underlying the substantial cytotoxicity demonstrated by CA. The results showed that CA is 8.5 times more cytotoxic to glioblastoma cell line (U-87MG) than normal hypothalamus cell line (mHypoE-N1), which indicates the specificity of CA toward brain tumor cells.
Pharmacologically, mango ginger has been used against a variety of human ailments. Traditionally, it is used for treating skin allergies, 22 stomach problems, 8 and hypercholesterolemia. 23 It has also been shown to possess antioxidant24,25 and antibacterial activity.26,27 More than 130 chemical constituents have been reported in Curcuma amada rhizomes, of which 121 have been identified. 28 Policegoudra et al10,29 have identified difurocumenonol, a new antimicrobial compound from mango ginger rhizome. The major chemical components of mango ginger rhizome that have been reported to include (E)-labda-8(17),12-diene-15,16-dial,12,14 starch, phenolic acids, volatile oils, curcuminoids, and terpenoids like difurocumenonol, amadannulen, and amadaldehyde. 9 However, it is not clear whether curcuminoids are in fact a constituent of mango ginger rhizome, which has a pale yellow color similar to ginger and not orange as in turmeric. In our experience, curcuminoids are not a component of mango ginger. Such discrepancies in the literature may in part be attributed to misidentification of raw materials. For example, species such as Curcoma mangga referred to as mango-like turmeric contains curcuminoids and (E)-labda-8(17),12-diene-15,16-dial. 30 The compounds like difurocumenonol and amadaldehyde have been demonstrated to possess anticancer activity.29,31
To understand the molecular changes induced by CA treatment, we analyzed the expression of genes associated with apoptosis (STAT3, P10, p21, Bax, APOBEC3B, P53, and Bcl-2), cell proliferation (Ki67 and PCNA), telomerase activity (hTER and hTERT), drug resistance (MDR-1, MRP, LRP, and DRP), and oncogenesis (C-myc, N-myc, and V-jun). Increasing concentrations of CA downregulate the mRNA expression of genes such as STAT3, Bcl-2, p53 and increases the Bax/Bcl-2 ratio, which could be a favoring factor for apoptosis. We also noticed an increase in the percentage of cells undergoing apoptosis with increasing CA concentrations. This would indicate that the cytotoxicity of CA in U-87MG cells may be contributed predominantly by the induction of apoptosis. The changes in the Bax and Bcl-2 expression with CA treatment of U-87MG cells is noteworthy since it has a direct relationship on apoptosis. 32 Oltivai et al 33 reported that Bax accelerates apoptotic cell death, without any alteration of cell division and viability. According to them, Bax synthesis does not appear to be a de novo response following a death stimulus and Bax by itself does not cause cell death. Bax also counters the death repressor activity of Bcl-2 by Bax/Bcl-2 heterodimerization. The discovery that B-cell lymphoma 2 (Bcl-2), a protein overexpressed in many types of cancer cells, promotes cell survival but not cell proliferation 34 has led to the view that impaired apoptosis is a critical step in tumor development. 35 The central mechanisms in the mitochondrial pathway related to programmed cell death is the extended Bcl-2 family of proteins. In mammalian cells, 5 pro-survival proteins like Bcl-2 and Bcl-X antagonize the pro-apoptotic function of Bak and Bax. 36
In the pathway associated with cell proliferation, CA treatment downregulates PCNA and cyclin B2 (CCNB2) mRNA expression in U-87MG cells. Since PCNA is actively involved in DNA synthesis and cell proliferation of tumor cells, inhibition of the process by CA would be advantageous for its therapeutic development. It is also reported that PCNA can form a complex with p21 resulting in the inhibition of DNA replication. 37 The cyclin B2 is associated with p34cdc2 and both are considered to be essential components of the cell cycle regulatory machinery and primarily associated with the Golgi region. 38 CA treatment also appears to downregulate the expression of drug-resistant genes like LRP and MRP to a low extent. U-87MG cells have low MDR-1 expression and CA treatment did not cause any change in it. However, the downregulation of MRP and LRP mRNAs with CA treatment would be a desirable attribute.
Telomerase has been frequently described as an ideal cancer target mainly because it is activated in approximately 85% of human cancers.39,40 On the contrary, telomerase activity tends to be very low in normal somatic cells except stem cells. In the present study, we demonstrated that CA inhibits the expression of hTERT mRNAs and not hTER in human glioblastoma cells. It is argued that the downregulation of hTERT will lead to the inhibition to telomerase activity, followed by the shortening of telomeres and a final pushing of cells into a crisis state leading to apoptosis. Therefore, telomerase inhibition can be used as a good indicator for evaluating the efficiency of chemotherapeutic and chemopreventive agents. Downregulation of hTERT expression has been reported with anti-neoplastic agents like vincristine, VP-16, and cisplatin. 41 The inhibition of hTERT expression adds to the anticancer potential of CA in brain cancer cells, in addition to the inhibition of other genes associated with cell proliferation, drug resistance, and promotion of apoptosis. CA treatment also has an inhibitory effect on the mRNA expression of oncogenes like N-myc and C-myc. In the regulatory pathway, C-myc is reported to be an upstream event in the regulation of hTERT. 42 The inhibition of oncogenes by CA would be a desirable attribute for its further therapeutic development. To conclude, the findings in this study suggest the anticancer potential of CA, directly or in synergistic combination with conventional chemotherapeutic drugs. Additional in vitro and animal studies are required to further explore this potential.
Footnotes
Author Contributions
CR, IVL, and SJM were responsible for study design, experimentation, and manuscript preparation. K-WQ formulated and prepared the supercritical CO2 extract of mango ginger for the investigation. All authors have read and approved the article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Steven J. Melnick is the founder of Dharma Biomedical LLC, which is an evidence-based ethnobotanical and evochemical drug discovery and nutraceutical company operating on for-profit basis. Dr Karl-Werner Quirin is the Chief Executive Officer of Flavex Naturextrakte GmbH, a company producing specialty botanical extracts for cosmetics and food supplements on the basis of supercritical CO2 extraction. Dr Cheppail Ramachandran and Ms Ivonne V. Lollett are also employees of Dharma Biomedical LLC.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This investigation was partially funded by STOP! Children’s Cancer of Palm Beach County, Inc, Florida.
Ethical Approval
Since this investigation did not involve human subjects or animal species, approvals from the institutional committees were not required.
