Abstract
Children with spastic cerebral palsy experience difficulty with ambulation. Structural changes in muscle and fascia may play a role in abnormal gait. Myofascial structural integration (Rolfing) is a manual therapy that manipulates muscle and soft tissues to loosen fascia layers, reposition muscles, and facilitate alignment. This study aimed to document (1) gait characteristics of 2 children with cerebral palsy and (2) effects of myofascial structural integration on their gait. Children received 3 months of weekly therapy sessions by an experienced practitioner. Gait parameters were recorded at baseline and after treatment using an electronic walkway. Children with cerebral palsy demonstrated abnormal velocity and cadence, decreased step length and single support times, and increased double support time. After treatment, both children demonstrated improvement for 3 months in cadence and double support time. The objective gait analyses demonstrated temporary improvements after myofascial structural integration in children with spastic cerebral palsy.
Cerebral palsy is a disorder of movement, tone, and posture. Children with cerebral palsy who are able to ambulate often show unsteadiness and clumsiness. The primary cause of cerebral palsy is a neurological injury to the developing brain. However, recent research findings have shown that the effects observed in cerebral palsy cannot be solely explained by neural changes. Secondary structural changes in the muscle and extracellular matrix tissue, or fascia, contribute to the increased muscle tone and stiffness.1–3 Treatments targeting muscle structure and soft tissue organization, such as stretching and massage, may be beneficial in improving gait and motor function in children with cerebral palsy. The efficacy of these methods, however, has not been widely studied.
Myofascial structural integration, or Rolfing, is a specific manipulative technique developed by Ida P. Rolf, PhD, and classified as a complementary practice. 4 A structured 10-session protocol focuses on a different anatomical area in each session to treat the entire body. Therapists use manual pressure on muscles and extracellular matrix to loosen and reposition the muscles and fascia. This pressure serves to restore, to the extent possible, the natural structural alignment of the bones and muscles.
The results of a recent pilot study of myofascial structural integration for children with cerebral palsy showed that the treatment led to overall positive change in gross motor abilities as measured by the Gross Motor Function Measure. 5 Parents observed that several children had improved gait patterns after treatment. A previous study of Rolfing in participants 10 to 42 years of age found that participants with mild cerebral palsy showed gains in velocity, stride length, and cadence and that those with moderate cerebral palsy made gains in velocity. 6
Gait characteristics can be assessed objectively and accurately using a portable electronic gait analysis system. The GAITRite® system (CIR Systems Inc, Sparta, New Jersey, USA) is a walkway that records various spatial-temporal gait parameters. It has been primarily used to assess older patients. Its ease of operation makes it suitable for use with children. Several recent studies have documented the reliability of the measures in children. 7,8
This exploratory case series answers the following questions: (1) Which gait parameters differed between children with mild cerebral palsy and norms in the literature for measures from the GAITRite system? (2) How did gait change after myofascial structural integration treatment in 2 children with spastic cerebral palsy? The findings would be useful for generating hypotheses about gait change after this or other interventions.
Case Summaries
Subject A was a 7-year-old male with right hemiplegic cerebral palsy; Subject B was a 6-year-old male with diplegic cerebral palsy. Both were ambulatory and classified as level II according to the Gross Motor Function Classification System. Parents gave informed consent prior to their child’s participation. All procedures were approved by the Stanford Institutional Review Board.
The children underwent a full course of myofascial structural integration therapy (ten 75-minute sessions) over a 3-month period. The same practitioner, a certified therapist with 35 years of experience and special expertise in children, treated both subjects. During the intervention, the children continued to receive physical and occupational therapies and medications but did not add any additional therapies.
The children were assessed before treatment, after treatment, and 3 and 6 months after treatment. The assessment used the GAITRite system, a 14-foot-long carpeted electronic walkway with embedded sensors that allow accurate measurement of the location and pressure of individual steps. Participants walked the length of the mat barefoot until they completed at least 4 walks along the full length of the carpet with a self-paced, relatively consistent, and natural walking speed. We analyzed walks that continued in 1 direction with consistent velocity and had at least 4 footfalls completely within the sensor area. At each assessment, at least 6 walks met these criteria and were combined for data analysis.
Multiple measures were exported from the GAITRite software. We considered several outcome parameters: mean velocity, cadence, step length left and right, stride length, single support time left and right (percentage of gait cycle bearing weight on 1 limb), and double support time (percentage of gait cycle bearing weight on 2 limbs). An additional measure of step length symmetry was calculated as the difference between right and left step length divided by the total stride length. Velocity, cadence, stride length, step length, single support, and double support have been shown to be reliable measures in children. 9 –11
To establish whether our methods would result in similar findings to those in the literature, we followed the same protocol with 2 typically developing children, ages 6 and 7 years, who had no motor impairments. The mean spatial and temporal gait characteristics of their walks were compared to published norms for children 4 to 8 years of age. 8 All measures of these controls fell within the standard deviation of the published norms for their age, increasing confidence in our methodology.
The baseline spatial and temporal gait characteristics of each child with cerebral palsy were compared to the same published norms 8 to establish which parameters differed significantly from those of 4- to 8-year-old children with no motor disability. The mean values from the pretreatment walks were compared to the normative values 8 using a t test to determine if differences between each subject’s data and the normative data reached statistical significance.
Table 1 shows the gait parameters of each child with cerebral palsy in relation to published norms. Compared to normative data, subject A showed lower velocity and cadence. He also showed significant, shorter step length on the right and stride length, decreased single support on the right and double support, and significant asymmetry of step length. Compared to normative data, subject B showed higher velocity and cadence that did not reach statistical significance. He also demonstrated decreased single support on the left and right and increased double support and step length symmetry.
Baseline Spatial and Temporal Gait Characteristics in 1 Child With Hemiplegic Cerebral Palsy (Subject A) and 1 Child With Diplegic Cerebral Palsy (Subject B) Compared to Normative Values for Children Ages 4 to 8 Years With No Disability.a
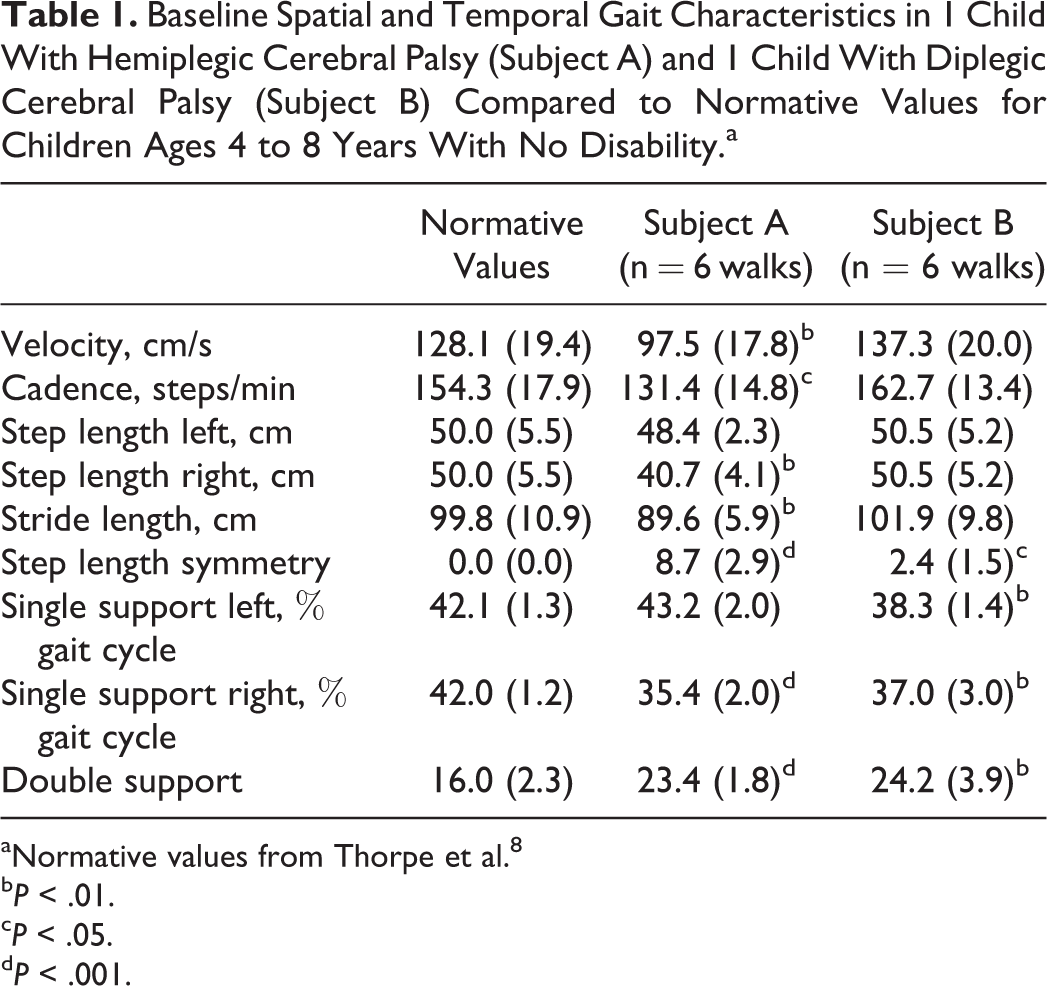
aNormative values from Thorpe et al. 8
b P < .01.
c P < .05.
d P < .001.
To establish whether subjects experienced a significant change in gait parameters as measured in the after-treatment phase, we plotted cadence and double support as a function of session in relation to treatment (Figure 1). Visual analysis of trend lines revealed that the children showed movement toward the normative values immediately after treatment and 3 months after treatment with a return toward baseline 6 months after treatment. Figure 1A shows the changes in cadence for the 2 children from baseline through 6 months after treatment in relation to the normal cadence for this age range. Figure 1B shows changes in double support time over the same period.

Gait characteristics in a child with hemiplegic cerebral palsy (A) and a child with diplegic cerebral palsy (B) compared to normative values for children ages 4 to 8 years with no disability at baseline and 3 time points after treatment. Figure 1A shows changes in mean cadence in steps/minute. Figure 1B shows changes in mean double support time as a percentage of the gait cycle.
Discussion
Children with cerebral palsy commonly have difficulty with ambulation secondary to weakness and spasticity in their lower limbs. In this study, using the portable, electronic gait analysis system, we demonstrated abnormalities in the spatial and temporal characteristics of gait in 2 children with cerebral palsy. We were able to document modest improvements in gait immediately after myofascial structural integration treatment and for 3 additional months using the GAITRite system.
At baseline, both children with cerebral palsy demonstrated decreased single support times in 1 or both legs and correspondingly longer double support times than seen in typically developing children of the same age. Decreased single support and increased time in double limb support represent a compensation for poor balance and motor control in children with cerebral palsy. It is not surprising that the child with hemiplegia also showed asymmetry in step length. In addition, subject A showed statistically significant lower velocity and cadence, whereas subject B showed higher velocity that did not reach statistical significance. Lower velocity in subject A likely indicates increased effort and energy expenditure in walk. The higher velocity observed in subject B appeared to be his compensation for a general lack of control in walking. By speeding up, he managed to dart across the mat without stumbling or falling.
After myofascial structural integration treatment, we saw improvements in both subjects. Both children showed changes posttreatment in mean cadence that moved in the direction of the normative levels. Subject A showed increased cadence whereas subject B showed decreased cadence. Subject A also showed increased step length on the affected side, resulting in increased stride length. Visual inspection of the data also found decreased double support time after treatment. Visual evaluation of the gait measures over time suggested that improvements persisted through 3 months after treatment and then began to return to baseline. This finding suggests that myofascial structural integration may have a direct but transient effect on the muscles, similar to other treatments for cerebral palsy, such as botulinum toxin A. 12,13
This study was a small case series. It needs to be replicated in a larger sample. The limited experience with the GAITRite system in children with cerebral palsy complicates interpretations of the data. In this study, we found very different baseline velocities in the 2 children with cerebral palsy. We think that the increased velocity in subject B was not a positive sign but rather a compensation for his poor control in walking. Additional studies of children with cerebral palsy on the GaitRite electronic walkway system may help clarify the interpretation of the various temporal and spatial characteristics of gait in children with cerebral palsy.
Conclusion
This study adds evidence that myofascial structural integration, a specific manipulative and movement-based complementary practice, used as a complementary treatment in the comprehensive management of young children with cerebral palsy, may lead to modest, temporary changes in gait. Following 2 children with diagnoses of spastic cerebral palsy, we were able to document improvements in gait using an objective gait measurement tool, the GAITRite system. In an ongoing study of myofascial structural integration for young children with cerebral palsy, we will increase the sample size and the number of walks gathered at each stage of the study to replicate and expand these findings. We will also consider the mechanisms by which manipulative techniques may improve gait in cerebral palsy.
Footnotes
Acknowledgments
All data were collected at Stanford University and the treatment sessions were completed at the private studio of Karen Price in Palo Alto, California. We thank Lynne C. Huffman, MD, Irene M. Loe, MD, and John Tamaresis, PhD, for assistance with statistical analyses and for comments on earlier versions of the article. We thank Jessica Rose, PhD, for the use of the GAITRite® Electronic Walkway System and the children and their families for participation.
Author Contributions
ABH participated in the design of the study; participated in the acquisition, analysis, and interpretation of data; and drafted the manuscript. KSP participated in the acquisition of data, participated in the analysis of data, and critically reviewed the manuscript. ECL participated in the acquisition of data, participated in the analysis and interpretation of data, and critically reviewed the manuscript. CAB participated in the analysis and interpretation of data and critically reviewed the manuscript. TMJ acquired and participated in the analysis and interpretation of data and critically reviewed the manuscript. ELP participated in the interpretation of data, critically reviewed the manuscript, and provided technical support. HMF conceived of the design of the study, drafted and reviewed the manuscript, obtained funding, and offered administrative and material support.
Declaration of Conflicting Interests
The authors certify that no party having a direct interest in the results of the research supporting this article has or will confer a benefit on them or on any organization with which they are associated.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support was provided by a research grant from the Gerber Foundation (11PH-010-1210-2936), a grant from the Maternal Child Health Bureau (T-77MC090796), and a National Institutes of Health Clinical and Translational Science Award (UL1 TR001085).
Ethical Approval
All procedures were approved by the Stanford Institutional Review Board (Protocol 16807). Parents provided written informed consent.
