Abstract
The aim of the study was to investigate Simo decoction–induced contractions of antral smooth muscles of rats and its mechanisms. The contractile responses of longitudinal strips to consecutive concentrations of Simo decoction were characterized by atropine, gallamine, 4-diphenylacetoxy-N-methylpiperidine methiodide, and adrenaline, hexamethonium, L-arginine, and nifedipine and compared with Krebs solution (control) and acetylcholine-induced contractions. Simo decoction dose-dependently increased contractions of antral strips (P = .000 vs control); its maximal effect was higher than acetylcholine (10−3 mol L−1; P < .05); Simo decoction–induced contractions were completely inhibited by atropine, 4-diphenylacetoxy-N-methylpiperidine methiodide, or 4-diphenylacetoxy-N-methylpiperidine methiodide + gallamine (P = .000 for all) but were partly suppressed by gallamine, adrenaline, hexamethonium, L-arginine, and nifedipine (P = .000 for all). Simo decoction promotes the contractions of antral strips mainly through activation of muscarinic M3 receptor, while partly through activation of M2 receptor, Ca2+ channel, nicotinic receptor, and inhibition of adrenergic receptor as well as release of nitric oxide.
Functional dyspepsia and gastroparesis usually present delayed gastric emptying. 1 –4 Traditional prokinetic agents, such as metoclopramide and erythromycin, 5 can relieve upper gastrointestinal symptoms of these diseases in the short term. However, tolerance of these drugs can be developed because of the downregulation of gastrointestinal hormone receptors, 6 which makes the long-term efficacy of these drugs uncertain.
Herbs, as a new therapeutic strategy, have been used for improvement of gastric dysmotility. Chaihu-shugan-san, a traditional Chinese medicine, has been shown to be effective for functional dyspepsia in China. 7 Iberogast, a herb extract from Germany, can relieve symptoms of functional gastrointestinal diseases, 8,9 which manifests that the effects of herbs are comparable with traditional prokinetic drugs. Simo decoction is considered as a prokinetic drug in traditional Chinese medicine, which has been used to improve gastrointestinal dysmotility for a long time in China, but the direct effect of Simo decoction on gastrointestinal smooth muscle has not been investigated.
The contractile activity of gastrointestinal smooth muscles is controlled by some receptors, ion channels, and gastrointestinal hormones, such as muscarinic, adrenergic, and nicotinic receptors, as well as Ca2+ channel and nitric oxide. In an earlier study, the improvement of gastrointestinal motility by Simo decoction was considered to be dependent on the regulation of motilin and cholecystokinin in serum and local tissue. 10 But so far there is no proof that Simo decoction can act on gastrointestinal smooth muscles via the aforementioned solutions.
In this study, we assumed that Simo decoction could promote the contractions of antral longitudinal strips. Spontaneous contractions of strips induced by Simo decoction were noted in the organ bath, and the roles of muscarinic, adrenergic, and nicotinic receptors and Ca2+ channel and nitric oxide in Simo decoction–induced contractions were also investigated in our study to ascertain its detailed mechanisms.
Materials and Methods
Animals
Male Sprague-Dawley rats (300-350 g) were purchased from the Experiment Animal Center of Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China. All animals were housed under standard laboratory conditions with free access to food (standard pellets, Tongji Medical College, Wuhan, China) and water. All animals in our study were treated strictly in accordance with the National Institute of Health Guide for the Care and Use of Laboratory Animals, and the work was approved by the Ethics Committee of Laboratory Animals.
Experimental Protocols
To investigate the contractive responses of antral longitudinal strips to Simo decoction, 7 concentration gradients of Simo decoction (1 μL, 5 μL, 25 μL, 50 μL, 100 μL, 150 μL, and 200 μL) were cumulatively administrated into the organ bath at 5-minute intervals and the dose–response curve obtained. As a control, equal volumes of Krebs solution as Simo decoction were also added to the organ bath at 5-minute intervals. The contractions induced by 5 concentration gradients of acetylcholine (10−7-10−3 mol L−1) were also noted in the organ bath to compare the contractions induced by Simo decoction. Simo decoction–induced contractions were characterized by using antagonists, such as nonselective muscarinic receptor antagonist atropine (10−6 mol L−1), selective muscarinic M3 and M2 receptors antagonists 4-diphenylacetoxy-N-methylpiperidine methiodide (0.4 × 10−6 mol L−1) and gallamine (10−6 mol L−1), nicotinic receptor antagonist hexamethonium (10−4 mol L−1), and L-type Ca2+ channel antagonist nifedipine (30 nmol L−1), as well as nitric oxide donor L-arginine (10−4 mol L−1) and adrenergic agonist adrenaline (10−7 mol L−1).
Tissues Preparation and Contractility Studies
All animals in this study were fasted overnight with free access to water before experiment and then sacrificed by cervical dislocation. The stomach was dissected out and cut along the greater curvature and rinsed with 4°C Krebs solution, continuously gassed with 95% O2/5% CO2 (vol/vol). The tissue was pinned on a silica dish and continuously gassed with 95% O2/5% CO2 (vol/vol) and then the mucosal layer was gently removed. An 8 mm × 2 mm longitudinal strip was cut parallel to the longitudinal fibers and ligated with 5-0 silk thread at the ends of the strip. The strips were placed into organ baths containing 25 mL Krebs solution (37°C, HAAKE DC10 immersion circulator, Thermo Scientific, USA) and continuously gassed with 95% O2/5% CO2 (vol/vol). One end of the strip was attached to an isometric force transducer (Fort-10, World Precision Instruments Inc, USA) and the other end was fixed on the armature of the bath. After stabilization for 60 minutes, the experiment started. Spontaneous contractile activity of gastric longitudinal strips under the optimal tension of 1g was recorded by an MP100 system (Biopac Systems Inc, USA) through a TBM4 M amplifier (Watson Precision Instruments Inc). The responses of strips to Simo decoction, Krebs solution, and acetylcholine based on baseline were noted in the organ bath through cumulative administration every 5 minutes. In subsequent steps, the strips were pre-incubated with the above-mentioned agents for 5 minutes and then Simo decoction was added and the cumulative concentration–response curves were obtained.
Drugs and Solutions
Simo decoction (specification: 10 mL/division; pH = 4.0-6.0) was purchased from Hunan Hansen Pharmaceutical Co, Ltd (Hunan, China), which was composed of Fructus aurantii, Radix aucklandiae, Semen arecae, and Radix linderae. Simo decoction was identified by Hunan Provincial Institute for Medicine according to National Standards of Proprietary Medicine.
The following agents were used: acetylcholine (Sigma-Aldrich, St Louis, MO, 10−7-10−3 mol L−1), atropine (Sigma-Aldrich, 10−6 mol L−1), gallamine (US Pharmacopeia, Rockville, MD, 10−6 mol L−1), L-arginine (Sigma-Aldrich, 10−4 mol L−1), hexamethonium chloride (Sigma-Aldrich, 10−4 mol L−1), adrenaline (Tianjin Jinyao Amino Acid Co Ltd, Tianjin, China, 10−7 mol L−1), nifedipine (Sigma-Aldrich, 30 nmol L−1), and 4-diphenylacetoxy-N-methylpiperidine methiodide (Fluorochem, Derbyshire, England, 0.4 × 10−6 mol L−1). All reagents were dissolved in distilled water except for 4-diphenylacetoxy-N-methylpiperidine methiodide and nifedipine, which were dissolved in dimethyl sulfoxide, and then further dissolved in Krebs solution in a 25-mL organ bath to obtain the above-mentioned final concentrations. All agents were added to the bath in volumes less than 1% of the total bath volume. Krebs solution contained (mmol L−1): NaCl, 119; KCl, 4.7; CaCl2, 2.5; MgSO4·7H2O, 1.2; NaHCO3, 25; NaH2PO4, 1.2; and C6H12O6, 11 (pH = 7.2-7.4). The solution was gassed continuously with 95% O2/5% CO2 (vol/vol).
Data Expression
The areas under contractive curve were analyzed by Acknowledge software (Biopac Systems Inc). Five-minute periods were analyzed at baseline or after each concentration or volume of Simo decoction, Krebs solution, and acetylcholine were added to the organ bath. The variation percentage of area under contractive curve was expressed as percentage variation of the changes of the effect–concentration curves. Percentage variation was calculated according to the following formula:
where X is the area under contractive curve at baseline and Y is the area under contractive curve after administration with Simo decoction, Krebs solution, or acetylcholine.
Statistical Analysis
Data were expressed as means ± standard error of means. At least 7 strips were studied in each group. One-way analysis of variance was performed among contractions induced by different doses of Simo decoction or acetylcholine; and contractions induced by Simo decoction in the presence or absence of atropine, gallamine, 4-diphenyl-acetoxy-N-methylpiperidine methiodide, 4-diphenylacetoxy-N-methylpiperidine methiodide + gallamine, adrenaline, L-arginine, hexamethonium, and nifedipine were evaluated by 2-way analysis of variance, and 2-way analysis of variance was also performed between contractions induced by Simo decoction or equal volumes of Krebs solution, followed by post hoc analysis and LSD or Dunnett’s T3 multiple comparison according to Levene’s test of equality of error variance. If Levene’s test showed homogeneity of variance, LSD multiple comparison was done for multiple comparison, whereas if Levene’s test indicated heterogeneity of variance, Dunnett’s T3 multiple comparison was performed. Nonpaired Student’s t test was performed between the maximal contractile effect induced by Simo decoction and acetylcholine. Values of P < .05 were considered statistically significant. Statistical analysis was performed with SPSS version 17.0.
Results
Effects of Simo Decoction on Contractions of Antral Longitudinal Strips at Baseline
Compared with control (Krebs solution), the dose–response curve of Simo decoction was significantly elevated (Figure 1A, P = .000 by 2-way analysis of variance): 1 μL Simo decoction (in 25 mL krebs solution) induced a contractile response (6.24 ± 0.64%). With the increase of concentrations of Simo decoction, the contractile activity of longitudinal strips also gradually increased (Figure 1A, P = .000 by 1-way analysis of variance), and the maximal effect was at 200 μL (128.84 ± 7.47%), but no significant differences were observed at 100 μL, 150 μL, and 200 μL (P > .05).

Effects of Simo decoction (SMD) on contractions of antral longitudinal strips. (A) Effects of Simo decoction on contractions of antral longitudinal strips at baseline. Two-way analysis of variance was performed, followed by post hoc analysis. #Significant difference compared with 1 μL (P < .05), *Significant difference compared with control (Krebs solution) (P < .05). (B) Simo decoction dose-dependently promoted contractions of antral longitudinal strips. (C) Effects of acetylcholine on contractions of antral longitudinal strips at baseline. One-way analysis of variance was performed followed by Dunnett’s T3 multiple comparison. *Significant difference compared with 10−7 mol L−1 (P < .05). (D) The maximal effect of contraction induced by acetylcholine and Simo decoction. Nonpaired Student’s t test showed that the maximal effect of Simo decoction (100 μL, 150 μL, and 200 μL) was higher than that of acetylcholine (10−3 mol L−1) (P < .05). Values are expressed as means ± standard errors of mean. N = 8 for all groups.
The contractile responses of longitudinal strips to acetylcholine (10−7-10−3 mol L−1) were also increased in a dose-dependent manner (Figure 1C, P = .000 by 1-way analysis of variance). The maximal effect was at 10−3 mol L−1 (97.50 ± 4.37%); no significant difference was observed at 10−4 and 10−3 mol L−1 (P > .05).
When 100 μL Simo decoction and 10−4 mol L−1 acetylcholine were administrated, the contractile responses reached a plateau (Figure 1A and B), but the maximal effect of acetylcholine (10−3 mol L−1) was significantly less than that of Simo decoction (100 μL, 150 μL, 200 μL) (Figure 1D, P < .05 by nonpaired Student’s t test).
Effects of Muscarinic Receptor on Simo Decoction–Induced Contractions of Antral Longitudinal Strips
Nonselective muscarinic receptor antagonist atropine (10−6 mol L−1) completely inhibited Simo decoction–induced contractions (Figure 2A, P = .000 by 2-way analysis of variance); the dose–response curve of longitudinal strips induced by Simo decoction was absolutely eliminated by atropine with no significance after the administration of each concentration of Simo decoction (P > .05).
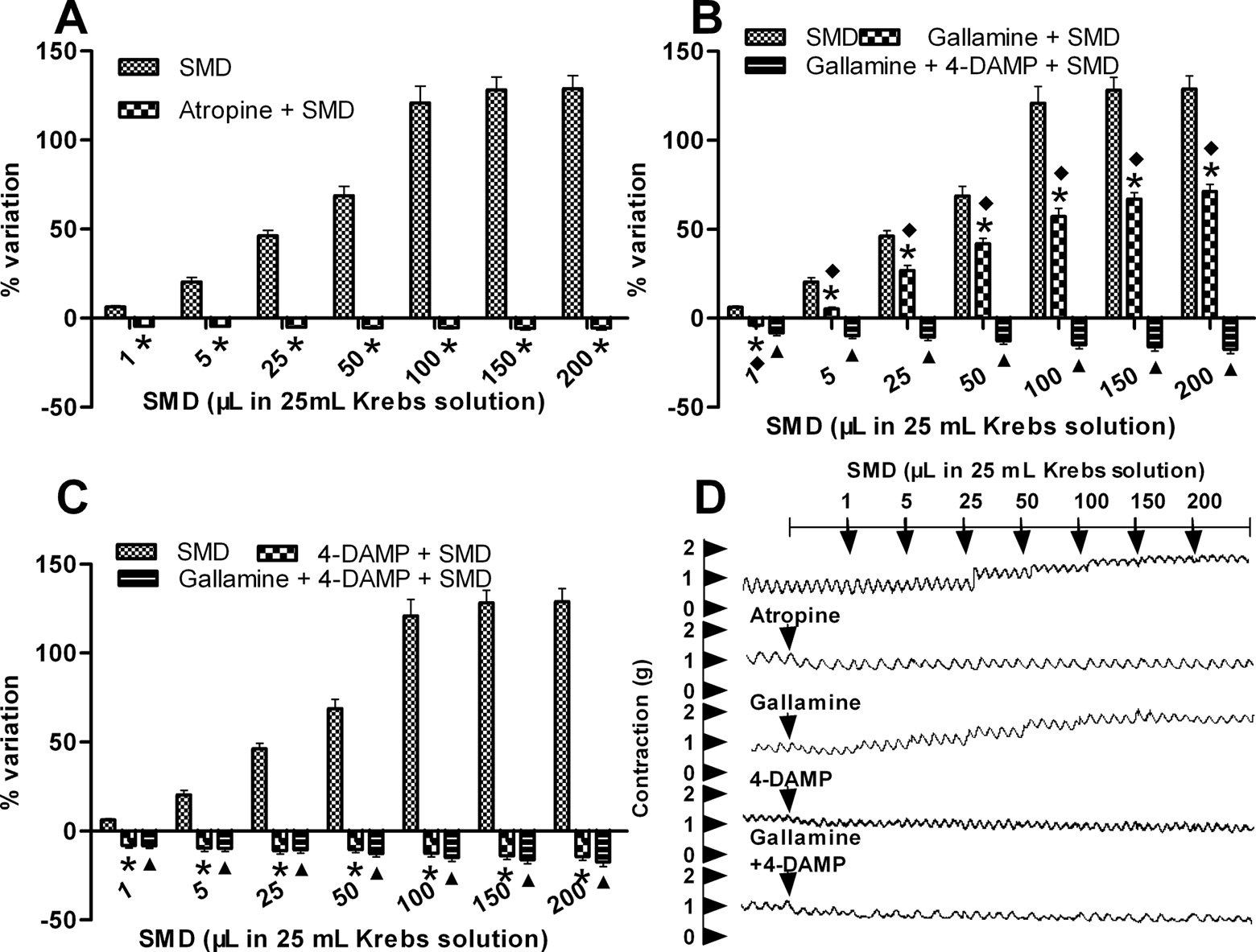
Effects of muscarinic receptor antagonist on Simo decoction (SMD)–induced contractions of antral longitudinal strips. (A) Effects of nonselective muscarinic receptor inhibitor atropine on Simo decoction–induced contractions of antral longitudinal strips. (B) Effects of selective muscarinic M2 receptor inhibitor gallamine or gallamine + 4-diphenylacetoxy-N-methylpiperidine methiodide (selective muscarinic M3 receptor inhibitor) on Simo decoction–induced contractions of antral longitudinal strips. (C) Effects of 4-diphenylacetoxy-N-methylpiperidine methiodide or gallamine + 4-diphenylacetoxy-N-methylpiperidine methiodide on Simo decoction–induced contractions of antral longitudinal strips. Values are expressed as means ± standard errors of mean. N = 8 for all groups. Two-way analysis of variance was performed followed by post hoc analysis. *Significant difference compared with Simo decoction (P < .05); ▴Significant difference compared with Simo decoction (P < .05). ♦ Significant difference compared with gallamine + 4-diphenylacetoxy-N-methylpiperidine methiodide + Simo decoction (P < .05). (D) Simo decoction–induced contractions were completely inhibited by atropine, 4-diphenylacetoxy-N-methylpiperidine methiodide, or gallamine + 4-diphenylacetoxy-N-methylpiperidine methiodide and partly inhibited by gallamine.
The contractions induced by Simo decoction were also completely suppressed by selective muscarinic M3 receptor antagonist 4-diphenylacetoxy-N-methylpiperidine methiodide in the presence or absence of selective muscarinic M2 receptor antagonist gallamine (Figure 2C, P = .000 by 2-way analysis of variance). However, gallamine showed partly inhibitory effect on Simo decoction–induced contractions (Figure 2B, P = .000 by 2-way analysis of variance), and the concentration–effect curve shifted to right slightly.
Effects of Adrenergic Receptor on Simo Decoction–Induced Contractions of Antral Longitudinal Strips
Adrenergic receptor agonist adrenaline partly inhibited Simo decoction–induced contractions (Figure 3A, P = .000 by 2-way analysis of variance); the Simo decoction–induced curve shifted to right, and adrenaline restrained contractions of longitudinal strips induced by Simo decoction at each concentration (P < .05).

Effects of adrenergic receptor agonist on Simo decoction (SMD)–induced contractions of antral longitudinal strips. (A) Effects of adrenaline on Simo decoction–induced contractions of antral longitudinal strips. Values are expressed as means ± standard errors of mean N = 8 for all groups. Two-way analysis of variance was performed followed by post hoc analysis. *Significant difference compared with Simo decoction (P < .05). (B) Simo decoction–induced contractions of longitudinal strips were partly inhibited by adrenaline.
Effects of Nicotinic Receptor on Simo Decoction–Induced Contractions of Antral Longitudinal Strips
Pretreated with nicotinic receptor antagonist hexamethonium, the contractile activity of antral longitudinal strips induced by Simo decoction was partly restrained (Figure 4A, P = .000 by 2-way analysis of variance); hexamethonium partly limited contractions evoked by Simo decoction at each concentration (P < .05) except for 1 μL (P > .05).

Effects of nicotinic receptor antagonist on Simo decoction (SMD)–induced contractions of antral longitudinal strips. (A) Effects of hexamethonium on Simo decoction–induced contractions of antral longitudinal strips. Values are expressed as means ± standard errors of mean. N = 8 for all groups. Two-way analysis of variance was performed followed by post hoc analysis. *Significant difference compared with Simo decoction (P < .05). (B) Simo decoction–induced contractions of antral longitudinal strips were partly inhibited by hexamethonium.
Effects of Exogenous Nitric Oxide on Simo Decoction–Induced Contractions of Antral Longitudinal Strips
After incubation with nitric oxide donor L-arginine, the contractile activity of antral longitudinal strips induced by Simo decoction was partly inhibited (Figure 5A, P = .000 by 2-way analysis of variance); the Simo decoction–induced curve also shifted to the right, and L-arginine showed significant inhibitory effect on Simo decoction–induced contractions after the administration of each concentration of Simo decoction (P < .05).

Effects of exogenous nitric oxide on Simo decoction (SMD)–induced contractions of antral longitudinal strips. (A) Effects of L-arginine on Simo decoction–induced contractions of antral longitudinal strips. Values are expressed as means ± standard errors of mean. N = 7 for L-arginine + Simo decoction and N = 8 for Simo decoction. Two-way analysis of variance was performed followed by post hoc analysis. *Significant difference compared with Simo decoction (P < .05). (B) Simo decoction–induced contractions of antral longitudinal strips were partly inhibited by L-arginine.
Effects of L-Type Ca2+ Channel on Simo Decoction–induced Contractions of Antral Longitudinal Strips
As shown in Figure 6A, pretreatment with L-type Ca2+ channel inhibitor nifedipine (30 nmol L−1) produced a shift to the right of the dose–effect curve of longitudinal strips induced by Simo decoction (P = .000 by 2-way analysis of variation); nifedipine attenuated Simo decoction–induced contractions at each concentration (P < .05).

Effects of inhibition of exogenous Ca2+ influx on Simo decoction (SMD)–induced contractions of antral longitudinal strips. (A) Effects of nifedipine on Simo decoction–induced contractions of antral longitudinal strips. Values are expressed as means ± standard errors of mean. N = 8 for all groups. Two-way analysis of variance was performed followed by post hoc analysis. *Significant difference compared with Simo decoction (P < .05). (B) Simo decoction–induced contractions of antral longitudinal strips were partly inhibited by nifedipine.
Discussion
Our results indicate that Simo decoction can regulate the antral motility through promoting contractions of antral strips, which is mainly due to activation of muscarinic M3 receptor. In addition, the activation of muscarinic M2 receptor, nicotinic receptor, and Ca2+ channel and the inhibition of adrenergic receptor and release of nitric oxide are all also involved in the action of Simo decoction on antral strips.
Simo decoction is composed of 4 herbs, Fructus aurantii, Radix aucklandiae, Semen arecae, and Radix linderae. Naringin, an extract from Fructus aurantii, is an active ingredient of Simo decoction, which can stimulate contractions induced by noradrenaline through inhibition of postsynaptic alpha-2 adrenergic receptor 11 and mainly distributes in stomach and small intestine, 12 which indicates that Simo decoction can act on the gastrointestinal tract directly. Except for Naringin, Semen arecae or its extract arecoline and Fructus aurantii have potential to promote gastrointestinal motility via activation of M3 and H1 receptors, respectively, 13 –16 and the effect of arecoline on colonic contractions is considered related to M3 receptor-extracellular Ca2+ influx-Ca2+ store release pathway. 14,15 All these studies confirm that Simo decoction can act on smooth muscle directly and suggest that multitudinous mechanisms should be responsible for the action of Simo decoction on contractions of strips.
In our study, we have observed that Simo decoction dose-dependently evokes excitation of gastric antral longitudinal smooth muscle in vitro. Furthermore, after administration with Simo decoction in vivo, accelerated gastric emptying has also been observed in chronically stressed mice, 10 and gastrointestinal hormones including motilin and cholecystokinin are considered involved in the action of Simo decoction on gastrointestinal motility. 10 Our study further suggests that Simo decoction can produce direct stimulation on gastric antral smooth muscle except for regulations of gastrointestinal hormones.
Simo decoction presents the similar effects as muscarinic receptor stimulant, 17 which mainly promotes contractility of antral longitudinal strips via activation of muscarinic M3 receptor. The muscarinic receptor includes 5 subtypes in stomach smooth muscle, 18 and the muscarinic M2 and M3 receptors are principally responsible for the contractions of gastrointestinal smooth muscles and abundantly express in the muscle layer of gastrointestinal tracts. 19 –22 Furthermore, the interaction between muscarinic receptors has already been reported in a previous study. 23 The action of muscarinic M3 receptor to regulate contractility of gastrointestinal smooth muscle is predominant, whereas M2 receptor can contribute, but not play a dominant role, in regulation of contractility of gastrointestinal smooth muscle. 22 Our results show that the activation of muscarinic M3 receptor is mainly responsible for the action of Simo decoction. It is likely to indicate that the Simo decoction has the same way and effect as muscarinic receptor stimulants, but the maximal effect of Simo decoction is stronger than traditional muscarinic receptor stimulant acetylcholine, which further indicates that some other solutions should be involved in Simo decoction’s action on smooth muscle.
Our results also show that the blockade of nicotinic receptor and excitation of adrenergic receptor and exogenous nitric oxide will suppress the contractile responses of antral longitudinal strips to Simo decoction. Excitatory response of gastrointestinal smooth muscles can be elicited by activation of presynaptic nicotinic receptor on nerve terminal, which induces acetylcholine release. 24 Presynaptic nicotinic acetylcholine receptors in guinea pig ileum mediate the release of substance P and neurokinin A from myenteric neurons, 25 which indicates that multitudinous pathways after activation of nicotinic receptor may be involved in the regulation of Simo decoction on antral strips contractions. The blockade of β-adrenergic receptor can increase the contractility of antral strips. 26 Nitric oxide is a traditional inhibitory neurotransmitter for gastrointestinal motility, and areca, as the most important component of Simo decoction, exhibits significant inhibition of nitric oxide synthase. 27 This likely suggests that all these solutions should be responsible for Simo decoction–induced contractility of antral smooth muscle.
The blockade of Ca2+ channel will lead to hyporesponsiveness of gastrointestinal smooth muscle, and the L-type Ca2+ channel can inhibit the exogenous Ca2+-influx. Simo decoction–induced contraction is also weakened by L-type Ca2+ channel antagonist nifedipine, which suggests that the exogenous Ca2+-influx should be involved in the action of Simo decoction on antral smooth muscle, and this result is in accord with Li’s study of arecaline. 14 It also suggests that areca and its active component arecoline are important constituents in Simo decoction.
In conclusion, our study suggests that Simo decoction is an effective prokinetic drug to promote contractility of antral smooth muscle directly, which is related to activation of muscarinic receptor, and activation of muscarinic M3 receptor is the main determinant, whereas the activation of muscarinic M2 receptor, nicotinic receptor, L-type Ca2+ channel and the inhibition of adrenergic receptor and release of nitric oxide can contribute to but are not the main determinants for Simo decoction–induced contractions.
Footnotes
Acknowledgments
The authors would like to thank Hunan Hansen Pharmaceutical Co Ltd for support of this study.
Author Contributions
Chibing Dai and Wei Qian were involved in planning and conducting the study, acquisition of data, interpreting data, and drafting the article. Na Liu, Jing Gong, and Wenmei Chen were involved in conducting the study. Xiaohua Hou was involved in planning and interpreting data, drafting and revising the article, and procuring the financial aid. The final draft was approved by all authors.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by a grant from the National Basic Research Program of China (973 Program) (Project No. 2009CB523002).
Ethical Approval
All animals in our study were treated strictly in accordance with the National Institute of Health Guide for the Care and Use of Laboratory Animals, and the work was approved by the Ethics Committee of Laboratory Animals (NO. 2010-71).

