Abstract
Objective
The pathomechanism of chronic fatigue syndrome/myalgic encephalomyelitis (CFS/ME) is unknown; however, a small subgroup of patients has shown muscarinic antibody positivity and reduced symptom presentation following anti-CD20 intervention. Given the important roles of calcium (Ca2+) and acetylcholine (ACh) signalling in B cell activation and potential antibody development, we aimed to identify relevant single nucleotide polymorphisms (SNPs) and genotypes in isolated B cells from CFS/ME patients.
Methods
A total of 11 CFS/ME patients (aged 31.82 ± 5.50 years) and 11 non-fatigued controls (aged 33.91 ± 5.06 years) were included. Flow cytometric protocols were used to determine B cell purity, followed by SNP and genotype analysis for 21 mammalian TRP ion channel genes and nine mammalian ACh receptor genes. SNP association and genotyping analysis were performed using ANOVA and PLINK analysis software.
Results
Seventy-eight SNPs were identified in nicotinic and muscarinic acetylcholine receptor genes in the CFS/ME group, of which 35 were in mAChM3. The remaining SNPs were identified in nAChR delta (n = 12), nAChR alpha 9 (n = 5), TRPV2 (n = 7), TRPM3 (n = 4), TRPM4 (n = 1) mAChRM3 2 (n = 2), and mAChRM5 (n = 3) genes. Nine genotypes were identified from SNPs in TRPM3 (n = 1), TRPC6 (n = 1), mAChRM3 (n = 2), nAChR alpha 4 (n = 1), and nAChR beta 1 (n = 4) genes, and were located in introns and 3′ untranslated regions. Odds ratios for these specific genotypes ranged between 7.11 and 26.67 for CFS/ME compared with the non-fatigued control group.
Conclusion
This preliminary investigation identified a number of SNPs and genotypes in genes encoding TRP ion channels and AChRs from B cells in patients with CFS/ME. These may be involved in B cell functional changes, and suggest a role for Ca2+ dysregulation in AChR and TRP ion channel signalling in the pathomechanism of CFS/ME.
Keywords
Introduction
Acetylcholine (Ach) is a neuronal cholinergic neurotransmitter that transmits activation signals to receptors located in the central nervous system (CNS), skeletal and smooth muscle, preganglionic autonomic nerve fibres, postganglionic autonomic parasympathetic nerves, immune cells, and other tissues of the non-neuronal cholinergic system (NNCS).1–4
Two types of membrane proteins bind Ach, muscarinic receptors (mAChRs) and nicotinic receptors (nAChRs), both of which have multiple isoforms. mAChRs are metabotropic receptors classified as M1 – M5, while nAChRs are ion channels that are typically heteromers of two subunits (selected from nine alpha and three beta subunits), with the exception of homomeric nicotinic alpha 7. 5 The ratio of subtypes affects the signal conducting speed through the receptor. 6 Importantly, one receptor subtype may impact on the receptor function of the other linked subtype.
In the NNCS, ACh binds AChRs on immune and other cell types. ACh is produced by lymphocytes expressing nAChRs that influence B lymphocyte function, including bone marrow development, B lymphocyte activation, and the autoantibody response.7–9 Within the NNCS, ACh also performs endocrine and paracrine functions in tissues such as smooth muscle, beta pancreatic cells, glial cells, lymphocytes, ocular lens cells, and brain vascular endothelium.10–14 These ACh functions are mediated by calcium signalling, which is particularly important for the activation of immune cell surface receptors.
mAChRs are inhibited by a type of calcium channel known as mammalian transient receptor potential (TRP) ion channels. 15 These are Ca2+-permeable cation channels that act as an excitatory signal when opened to induce cell depolarisation and cause a Ca2+ influx, which plays a role in intracellular signalling pathways. TRPs are comprised of six main groups including TRPA (ankyrin), TRPC (canonical), TRPM (melastatin), TRPML (mucolipin), TRPP (polycystin), and TRPV (vanilloid). 16 TRPs are present on almost all cell types, and their dysregulation has been associated with pathological conditions and disease.17–22
Chronic fatigue syndrome (CFS) is also often referred to as myalgic encephalomyelitis (ME). To acknowledge previous work that refers to either terminology, this study refers to the hybrid acronym CFS/ME. CFS/ME is characterised by a distinct impairment in physical activity, debilitating fatigue accompanied by impairments in memory, cognition, and concentration, enhanced experience of pain, and dysregulation of gastrointestinal, cardiovascular, and immune systems.23–35 No diagnostic or screening test currently exists for this illness.
We previously identified single nucleotide polymorphisms (SNPs) in genes encoding receptors that require Ca2+ as an important component of their function. These include genes encoding TRP ion channels and AChRs, namely TRPM3, TRPA1, TRPC4, 36 mAChRM3, nAChRs alpha 10, alpha 5, and alpha 2 in peripheral blood mononuclear cells (PBMCs) and isolated natural killer (NK) cells from CFS/ME patients.36,37,45 Additionally, we documented changes in intracellular Ca2+ mobilisation for TRPM3 from NK cells and B lymphocytes. 38 These SNPs and their genotypes may alter the structures and functions of TRP ion channels and AChRs. A recent study reported that a subgroup of CFS/ME patients had muscarinic antibodies, and observed a modest positive response with reduced symptom presentation following anti-CD20 intervention. 39 Given the important roles of Ca2+ and ACh signalling in B cell activation as well as the potential for antibody development, the present study aimed to identify SNPs and their genotypes for TRPs and AChRs from isolated B cells of CFS/ME patients.
Patients and methods
Recruitment
CFS/ME patients were defined in accordance with the 1994 CDC criteria for CFS/ME. 40 A total of 11 CFS/ME patients and 11 non-fatigued controls with no medical history or symptoms of prolonged fatigue or illness of any kind were recruited from the National Centre for Neuroimmunology and Emerging Diseases (NCNED) research database. 40 All participants attended NCNED to participate in research. Participants were excluded if they were pregnant or breastfeeding, or had a history of smoking or substance use. All participants provided signed consent and this study was approved by the Griffith University Research Ethics Committee (MSC22/12HREC). This study was approved by the Griffith University Human Research Ethics Committee (MSC22/12HREC).
Sample preparation and measurements
A total of 40 ml of blood was collected from the antecubital vein of participants into lithium heparinised and EDTA collection tubes between 09:00 h and 11:00 h. Routine blood samples were analysed within 6 h of collection for red blood cell counts, lymphocytes, granulocytes, and monocytes using an automated cell counter (ACT Differential Analyzer, Beckman Coulter, Miami, FL).
B cell isolation
PBMCs were isolated from 40 mL of whole blood using a previously described method.46 Briefly, PBMCs were isolated using a Ficoll-Paque density gradient (GE Healthcare, Uppsala, Sweden), then washed twice with phosphate-buffered saline (PBS) (Gibco-BRL, Gaithersburg, MD).
Cells were then resuspended in autoMACs separation buffer (PBS with bovine serum albumin, EDTA, and 0.09% azide) (Miltenyi Biotec, Auburn, CA). Immunomagnetic negative selection of B cells was performed with B-cell isolation kit II (Miltenyi Biotec) according to the manufacturer’s instructions. Briefly, non-B cells such as T cells, NK cells, dendritic cells, monocytes, granulocytes, and erythroid cells were indirectly magnetically labelled using a cocktail of biotin-conjugated antibodies against CD2, CD14, CD16, CD36, CD43, and CD235a (glycophorin A). B cell isolation was then achieved following the depletion of magnetically labelled cells.
Untouched B cells fluorescently stained with anti-CD19-BV421 and anti-CD3-PerCP antibodies were measured using LSR Fortessa X-20 flow cytometry. Cell debris and dead cells were excluded from the analysis based on scatter signals. The mean purity was 85.66%± 9.6% for non-fatigued controls and 76.5% ± 13.1% for CFS/ME patients, representing no significant difference between groups for levels of B lymphocytes.
DNA extraction
A total of 40 mL blood was collected into EDTA tubes for SNP analysis. Genomic DNA was extracted from whole blood samples using the Qiagen DNA blood mini-kit according to the manufacturer’s instructions (Qiagen, Venlo, Netherlands). SNP genotyping studies were performed as previously described.36,42
SNP analysis
A total of 661 SNPs were examined from 21 mammalian TRP ion channel genes (TRPA1, TRPC1, TRPC2, TRPC3, TRPC4, TRPC6, TRPC7, TRPM1, TRPM2, TRPM3, TRPM4, TRPM5, TRPM6, TRPM7, TRPM8, TRPV1, TRPV2, TRPV3, TRPV4, TRPV5, and TRPV6) and nine mammalian ACh receptor genes (muscarinic M1, M2, M3, M4, and M5, nicotinic alpha 2, 3, 5, 7, 9, and 10, beta 1, and 4, and epsilon) in B cell DNA using the MassARRAY iPLEX Gold Assay (a Matrix Assisted Laser Desorption Ionisation Time-of-Flight mass spectrometry platform; Sequenom Inc.).
Genomic DNA quality and quantity were assessed as previously described,44,48 including quantification using a Nanodrop spectrophotometer (Nanodrop). Approximately 2 µg of genomic DNA was then used to perform SNP analysis. MassARRAY was employed to discriminate alleles based on single-base extensions of an extension primer of known mass designed to attach next to the SNP site of interest. Custom multiplexed wells were designed in silico using Agena Bioscience's Assay Design Suite, and built using custom synthesised oligonucleotides that were pooled for sample processing. The iPLEX Gold chemistry utilised two multiplexed oligo pools for each genotyping well. A multiplexed PCR pool was used to generate short amplicons that included all genomic markers of interest in that particular well. Following PCR and clean-up steps, a secondary PCR ‘extension’ step was undertaken with pools of extension primers that were designed to attach next to the SNP sites of interest. During the extension phase, a termination mix was added that enabled these extension primers to be extended by a single base only. Given the known molecular weight of the extension primer, allele discrimination could be measured using the peak heights of the unextended primer plus single-base extension possibilities for the SNP.
TRP ion channel and AChR SNP assays
Primers and extension primers were designed for each of the SNPs using Assay Designer software (Sequenom Inc.) according to the manufacturer’s instructions and as previously described.44,45 PCR conditions were as follows: 94℃ for 2 min, 94℃ for 30 s, 56℃ for 30 s, and 72℃ for 1 min. Amplification products were treated with shrimp alkaline phosphatase at 37℃ for 40 min then 85℃ for 5 min, and stored at 4℃. Extension primers were optimised to control the signal-to-noise ratio. This involved examining unextended primers (UEPs) on the spectroCHIP array and evaluating them using Typer 4.0 software as low-mass UEP, medium-mass UEP, and high-mass UEP. iPLEX extension was performed using iPLEX Gold Buffer Plus, iPLEX termination mix, iPLEX enzyme, and primer mix at an initial denaturation of 94℃ for 30 s, annealing at 52℃ for 5 min, extension at 80℃ for 5 min (five cycles of annealing and extension were performed, but the entire reaction involved 40 cycles), and an extension at 72℃ for 3 min. Resin beads were used to rinse iPLEX Gold reaction products. MassARRAY was performed using the MassARRAY mass spectrometer, and generated data were analysed using TyperAnalyzer software.
Statistical analysis
Statistical analysis was performed using SPSS software version 22 (IBM). Experimental data were reported as means ± standard error of the mean (±SEM), while clinical data were reported as means ± standard deviation (±SD). Comparative assessments among participants (CFS/ME and non-fatigued controls) were performed using the analysis of variance test and the criterion for significance was set at P < 0.05.
The PLINK v1.07 (http://pngu.mgh.harvard.edu/purcell/plink/) whole genome analysis tool set was used to determine associations between CFS/ME patients and the non-fatigued control group. A two-column χ2 test was used to examine differences, with P < 0.05 taken to be significant. Further genotype analysis for differences between CFS/ME and the non-fatigued group was also completed according to a two-column χ 2 test, and Yate’s correction factor was applied with significance at P < 0.05. Analyses were performed at the Australian Genome Research Facility Ltd. (Victoria, Australia).
Results
Participants
Characteristics of chronic fatigue syndrome/myalgic encephalomyelitis patients and non-fatigued controls.

CFS/ME, chronic fatigue syndrome/myalgic encephalomyelitis
SNP analysis
Frequency and distribution of SNPs in B cell genes for TRP ion channels and AChRs in chronic fatigue syndrome/myalgic encephalomyelitis patients and non-fatigued controls in rank order of significance.

CHR, chromosome location; Ref SNP, reference SNP identification; χ2, chi-squared for basic allelic test (one degree of freedom); OR, odds ratio
Genotype analysis
Genotype and odds ratio of SNPs in B cell genes for TRP ion channels and AChRs in chronic fatigue syndrome/myalgic encephalomyelitis patients and non-fatigued controls in rank order of significance.
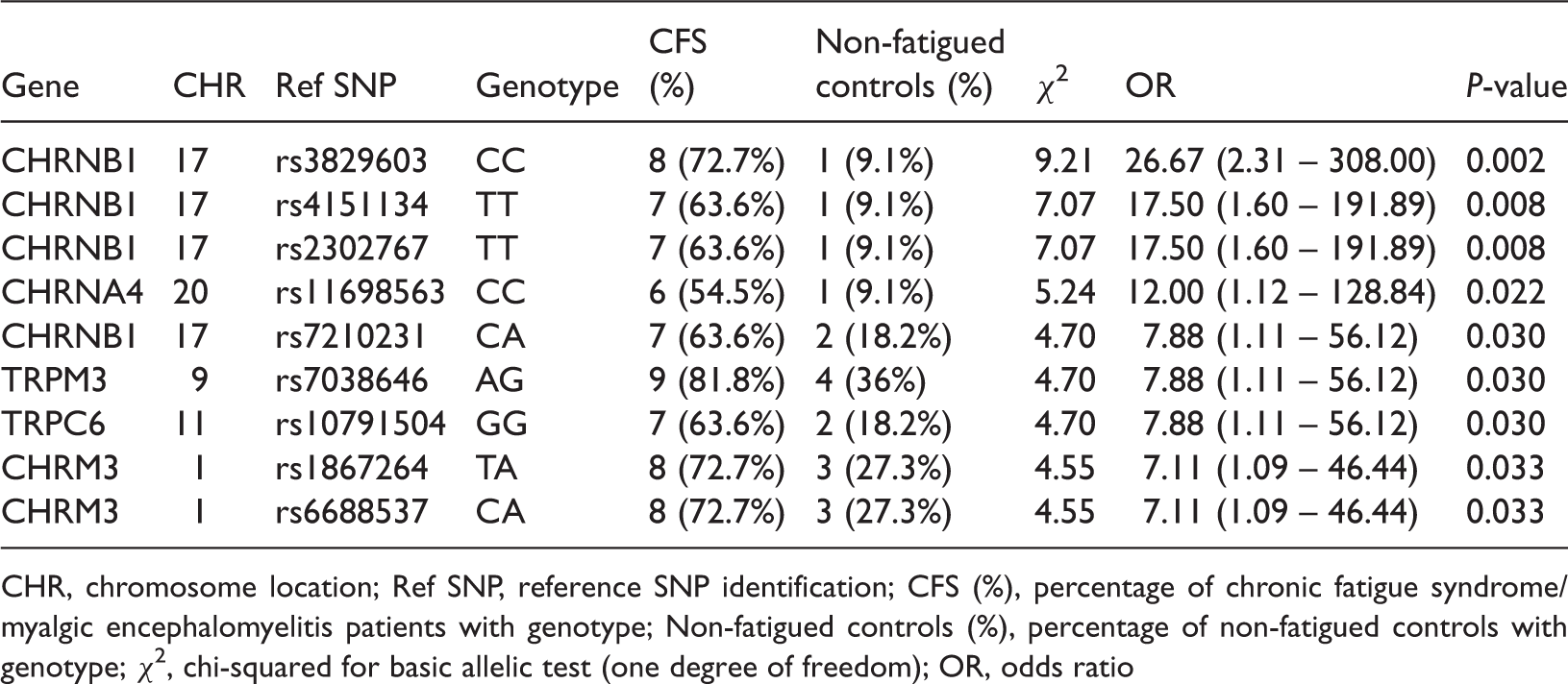
CHR, chromosome location; Ref SNP, reference SNP identification; CFS (%), percentage of chronic fatigue syndrome/myalgic encephalomyelitis patients with genotype; Non-fatigued controls (%), percentage of non-fatigued controls with genotype; χ2, chi-squared for basic allelic test (one degree of freedom); OR, odds ratio
Discussion
The current investigation reports novel findings for AChR and TRP variants and genotypes in B cells from CFS/ME patients. These data are consistent with our previous findings for PBMCs and NK cells in CFS/ME patients.36,37,41
Intracellular Ca2+ levels are substantially modulated by receptor-induced alterations, and are regulated by plasma membrane channels, intracellular receptor channels, non-selective cation channels, specific membrane transporters, and the cell membrane potential.20,46,47 Ca2+ is critical for lymphocyte differentiation and function, as well as the regulation of antigen receptors, co-receptors, signal transduction, mitochondrial function, transcriptional factors, and gene expression.43–46
The immune system is dependent on cholinergic signalling because B and T cells express cholinergic receptors and regulate cytokines during inflammation48,49 and the immune response. 50 Indeed, cholinergic signalling influences both B cell 9 and T cell 51 responses and has been found to initiate B cell autoimmunity. 52 Of the cholinergic receptor SNPs identified, mAChM3R featured prominently (45%) which is consistent with our previous findings for SNPs and their genotypes in NK cells. 37 In our current investigation, we identified two SNP genotypes for mAChM3R. However, given the small sample number as well as taking into account our previous results for SNP genotypes from isolated NK cells and PBMCs, other genotypes for this receptor may be present in B cells of CFS/ME patients. A recent study reported a subgroup of CFS/ME patients with muscarinic antibodies (against mAChM3R), and in whom a modest positive response occurred with reduced symptom presentation following anti-CD20 intervention. 39 Although patient genotypes were not reported in this subgroup analysis, our current findings, together with our previous genotype findings in isolated NK cells from a larger CFS/ME cohort, suggest that these genotype changes may play a role in B cell function. Moreover, the ubiquitous distribution of cholinergic receptors throughout the body suggests that anomalies in SNP genotypes and their heterodimer configuration and pattern may contribute to the various clinical symptoms of CFS/ME.
Our identification of SNPs in mAChRs and nAChRs from a diverse range of blood cells, such as PBMCs and isolated NK cells in larger cohorts of CFS/ME patients, suggests that cholinergic signalling may be impeded in this disorder. Muscarinic signalling was previously shown to play a role in gastrointestinal function, 53 and antibodies to mAChM3Rs have been found to inhibit gastrointestinal motility and cholinergic neurotransmission. 54 mAChM3Rs are present in the heart where they regulate intracellular phosphoinositide hydrolysis to improve cardiac contraction, haemodynamic function, 55 and provide a protective effect against ischaemia. 56 They are also located in the pancreas where they mediate acetylcholine control over insulin secretion and have other important regulatory functions.57–59
Nicotinic signalling via nAChRs is widely distributed in organisms demonstrating the universal nature of cholinergic signalling. Muscle-type nAChRs, such as β1, are similar throughout the body. 7 Our present study identified a high number of SNPs and genotypes in nAChRs, suggesting that the extent of SNP genotypes in cholinergic receptors may play a role in B cell function. Indeed, ACh functions as a paracrine/autocrine regulator of immune and other physiological functions. 60 We demonstrate significant ORs for the genotypes of nAChR β1 SNPs rs3829603 (C/C) and rs4151134 (T/T), located in the 3′ untranslated region (UTR), ranging from17.50 to 26.67 for the CFS/ME group. This is a binding site for regulatory proteins involved in the post-transcriptional control of gene expression. 61 Binding to specific sites within the 3′ UTR may decrease the expression of various mRNAs either by inhibiting translation or directly causing degradation of the transcript. The agonist-binding site of nAChRs is located at the interface between adjacent subunits. An α subunit (α1, α2, α3, α4, α6, α7, or α9) comprises the positive side of the binding site, while the negative side is composed of α10, β2, β4, δ, γ, or ɛ subunits. α5, β1, and β3 subunits assemble in the receptor complex as a fifth subunit, not in the direct formation of the agonist-binding site, but forming an integral configuration for binding agonists and ligand selectivity. 62 Given the number of SNP genotypes for nAChR β1 that were located in the 3′ UTR, the fifth subunit might be expected to alter ligand selectivity.37
Various subunit combinations have previously been shown to give different nAChR subtypes that vary in their kinetic parameters, ion channel selectivity, ligand specificity, signalling pathways, and tissue functions. 63 The high density of AChR distribution throughout the body means that many tissues are likely to be affected following changes in AChR expression, with a potential loss of function of neuronal and non-neuronal cholinergic signalling pathways. This may explain the changes in B cell phenotypes of CFS/ME patients previously observed by us and others;26,64 we also separately reported a reduction in Ca2+ mobilisation into B cells via TRPM3 in association with receptor 3′ UTR SNP genotypes. 38
Cholinergic signalling in the brain is primarily focused on two main loci: the basal forebrain and the pedunculopontine area of the hindbrain. 65 Acute vasoconstriction occurs after removal of the cholinergic parasympathetic input to forebrain cerebral arteries, 66 indicating the critical importance of intact cholinergic signalling in the brain. Both nicotinic and muscarinic cholinergic signalling influence hippocampal synaptic plasticity and the processing of cholinergic-dependent higher cognitive functions. 67 Cholinergic and glutamatergic signalling previously demonstrated interdependence in cortical glial cell function during sleep/wake studies. 68 Moreover, the key CNS function of memory formation is Ca2+-dependent through its association with cholinergic signaling, 69 and is associated with long term potentiation in hippocampal synapses.
Although the present study is only a preliminary investigation, its small sample size is a limitation. Moreover, we only examined changes in patients with moderate CFS/ME, so future work should examine patients with severe CFS/ME who are housebound or bedridden. Nevertheless, our findings suggest that further investigations of mAChR and nAChR SNPs in a larger cohort of CFS/ME patients and healthy controls are warranted. We further suggest a comparison between a group meeting the diagnostic criteria for chronic fatigue alone, compared with those meeting criteria for CFS/ME, as an additional validation of the identified markers.
Conclusion
Our identification of SNP genotypes in cholinergic and TRP receptor genes in B cells, and previously in PBMCs and isolated NK cells, of CFS/ME patients suggests a potential contribution to systemic disease pathology including the immune, CNS, heart, gastrointestinal, and hormonal systems. The effects of these SNP genotypes on cholinergic signalling are likely to be particularly important in the CNS, peripheral nervous system, and autonomic nervous system. The functional effects of these genotypes and their combinations indicate that they may be contributing factors in the aetiology and clinical phenotypes of CFS/ME.
Footnotes
Acknowledgements
The authors would like to thank Dr Lavinia Gordon (Australian Genome Research Facility, Melbourne, Australia) for the bioinformatic SNP analysis.
Authors’ contributions
SMG and DRS designed and developed all experiments, and made final changes to the manuscript. PS, SJ, AC, and TH assisted in data analysis, and revised the manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This study was supported by funding from the Stafford Fox Medical Research Foundation, the Alison Hunter Memorial Foundation, the Mason Foundation, and the Queensland Co-Investment Program. This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
