Abstract
Molybdenum is a trace element that functions as a cofactor for at least 4 enzymes: sulfite oxidase, xanthine oxidase, aldehyde oxidase, and mitochondrial amidoxime reducing component. In each case, molybdenum is bound to a complex, multiring organic component called molybdopterin, forming the entity molybdenum cofactor. The best sources of dietary molybdenum are legumes, grains, and nuts. Bioavailability of molybdenum is fairly high but depends on form, with molybdenum preparations having greater bioavailability than food-bound molybdenum. Molybdenum deficiency and toxicity are rare, possibly because of the body’s ability to adapt to a wide range of molybdenum intake levels. At low intakes of molybdenum, the fractional transfer of molybdenum from plasma to urine is lower and a greater fraction is deposited into tissues, and at high intakes of molybdenum, the opposite occurs. Molybdenum has proven to be an interesting trace mineral that is essential for life.
Molybdenum is a trace element that was discovered in 1778 by Swedish chemist Karl Scheele. Initially mistaking the substance for lead, he later realized that he had encountered a new element, which he named molybdenum after the mineral molybdenite, which had acquired its name from the Greek work “molybdos,” meaning “lead-like.” Molybdenum was known to be essential to plant life long before its essentiality to animals was realized. In 1953, molybdenum’s role as a cofactor for the enzyme xanthine oxidase was discovered,1,2 establishing the essentiality of molybdenum in the diet.
Molybdenum Form and Function
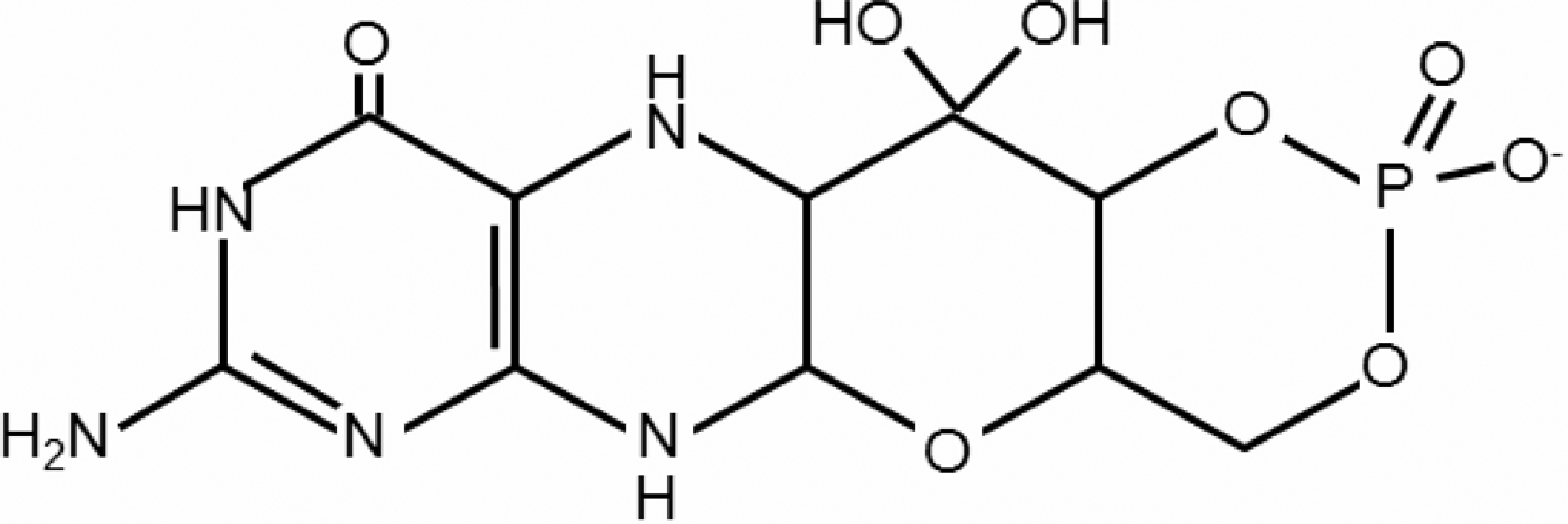
For humans, molybdenum functions as a cofactor for at least 4 enzymes: sulfite oxidase, xanthine oxidase, aldehyde oxidase, and mitochondrial amidoxime reducing component. In each case, molybdenum is bound to a complex, multiring organic component called molybdopterin, forming the entity molybdenum cofactor (Figure 1). Xanthine oxidase catalyzes the oxidation of purines to uric acid. 3 The aldehyde oxidase ortholog in humans is involved in the metabolism of various endogenous and exogenous N-heterocyclic compounds. 4 Sulfite oxidase catalyzes the terminal reaction in the degradation of sulfur amino acids cysteine and methionine. 5 Mitochondrial amidoxime reducing component (mARC) was just recently discovered, 6 and it is presumed to form the catalytic portion of a 3-component enzyme system with heme/cytochrome b5 and reduced nicotinamide adenine dinucleotide/flavin adenine dinucleotide–dependent cytochrome b5 reductase. The structure of mARC along with its high abundance in liver and kidney suggests that mARC could play a role in detoxification of N-hydroxylated substrates.

Structure of molybdenum cofactor
Dietary Sources of Molybdenum
Molybdenum is widespread in the diet. In plant foods, molybdenum content is primarily determined by the regional richness of the soil, and the content in meat depends on forage of the animals. The main dietary contributors of molybdenum are legumes, grain products, and nuts.7–9 Tsongas et al 10 measured molybdenum content of a wide range of foods to find the following molybdenum contents (per 100 g wet weight): vegetables, 0.1 to 0.8 mg; legumes, 2 to 87 mg; eggs, milk, and cheese, ˜0.5 to 1.0 mg; chicken, beef, and pork, ˜0.2 to 0.5 mg; bread, pasta, and rice, ˜3.0 to 4.0 mg; and fruit, 0.1 to 0.9 mg. Legumes are the richest sources of molybdenum (Table 1), with lima beans providing 87 mg molybdenum per 100 g. 10
Molybdenum Content of Legumes

Estimates of molybdenum intake among different countries vary, because of both dietary differences and also analytical methodology differences. In the United States, one study reported intakes ranging from 120 to 240 μg/d. 10 Another study in the United States suggested intakes of 76 μg/d for women and 109 μg/d for men. 8 In Japan, intake has been estimated to be 225 μg/d, with primary sources being rice and soy products. 7 In Korea, molybdenum intake was estimated to be 136 μg/d for men and 123 μg/d for women. 11 In France, mean molybdenum intake was estimated to be 275 μg/d. 12 The International Atomic Energy Agency sponsored a study of the molybdenum content of representative total diets from 11 countries, 13 and from these data the World Health Organization estimated that adults consume about 100 μg molybdenum per day. 14
Dietary Requirement
The Food and Nutrition Board of the United States Institute of Medicine established that the average minimum molybdenum requirement is 22 μg/d plus an additional 3 μg/d for miscellaneous losses for a minimum requirement of 25 μg/d. Further accounting for bioavailability (assumed to be 75%) and interindividual variability in molybdenum metabolism (additional 30%, or 2 standard deviations), the recommended daily allowance for molybdenum in the United States was set at 45 μg/d for both men and women. 15 The World Health Organization (WHO) has established a slightly higher daily molybdenum requirement of 100 to 300 μg/d for adults, 14 although it has acknowledged that lower intake levels could be adequate. The Food and Nutrition Board established a tolerable upper intake level of 2 mg/d for molybdenum. 15 Agencies outside the United States have set upper limit values for molybdenum intake that are much lower, including the Health and Consumer Protection Directorate General of the European Commission, which recommended an upper limit of 600 μg/d 16 and The Expert Group on Vitamins and Minerals of the United Kingdom, which recommended an upper limit of 230 μg/d. 17
Bioavailability
A few studies conducted with isotopically labeled molybdenum have provided information about molybdenum bioavailability. Small amounts of molybdenum preparations (1 mg or less) are almost completely absorbed across the gastrointestinal tract, with absorption efficiencies of 90% to 100%.18–20 Molybdenum in food is less bioavailable than molybdenum preparations. Werner et al 20 studied bioavailability of molybdenum from cress using both an intrinsic label (in which the labeled molybdenum was directly incorporated into the plant tissue) and an extrinsic label (in which the labeled molybdenum was simply mixed with the cress before serving). For the intrinsically labeled cress, bioavailability values ranged from approximately 50% to 80%, and for the extrinsically labeled cress, bioavailability was 70% to 90%, as would be expected since an extrinsic tag would not be expected to bind to plant material as completely as an intrinsic tag. Similarly, Turnlund et al 21 found the bioavailability of molybdenum from 97Mo-labeled kale to be approximately 86%. Turnlund et al 21 also examined the bioavailability of molybdenum from soy to find the bioavailability to be around 57% to 58%. In good accord with those studies, compartmental modeling studies have suggested that molybdenum from a mixed diet exhibited bioavailability of around 76% to 83%.18,19 A study of molybdenum balance in Japanese women suggested that more than 90% of dietary molybdenum is absorbed from a mixed Japanese diet. 22 The bioavailability of molybdenum from an extrinsically labeled mixed meal (chicken, vegetables, and noodles) 20 was found to be lower (35% to 50%) than bioavailability values found in other studies. When labeled molybdenum was added to black tea, bioavailability was dramatically reduced to less than 15%. 20 This could be because of the high polyphenol content of tea, which is also known to reduce iron absorption. 23
In low dose ranges, size of ingested dose does not seem to inhibit gastrointestinal absorption. Novotny and Turnlund 19 reported the bioavailability of molybdenum was similar over doses ranging from 22 μg/d (0.23 μmol/d) to 1490 μg/d (15.5 μmol/d). However, at large doses (5 mg), a slight reduction in bioavailability was observed. 20
Tissue Distribution and Excretion
Schroeder et al 9 analyzed tissues from autopsies on 381 individuals to determine molybdenum levels. Highest concentrations were detected in liver, kidney, adrenal, and omentum. Lowest concentrations (when detectable) were found in skin, esophagus, trachea, aorta, uterus, and bladder. Generally, molybdenum was consistently detected in fat, blood, liver, and kidneys, and the average molybdenum content detected in these tissues was 2286 μg. Molybdenum was inconsistently found in other body tissues, and accounting for these tissues, Schroeder et al 9 suggested that the maximum potential molybdenum store would be 9000 μg. When Rosoff and Spencer 24 administered 99Mo to mice, the liver took up the largest amount of molybdenum (18%) followed by kidney (9%) and pancreas (3%).
Compartmental analysis has provided estimates of total body molybdenum stores in healthy adults. For adults males consuming 121 μg/d of molybdenum (which is close to estimates of free-living molybdenum intake),8,10 total body molybdenum stores were calculated to be approximately 2224 μg (23.1 μmol). This estimate is in good accord with the findings of Schroeder et al. 9
Significant amounts of molybdenum are excreted through both feces and urine, with urine being the primary route of elimination.19,22,25 Moreover, urinary excretion is responsive to increasing levels of molybdenum intake. A study of men who received 5 different levels of dietary molybdenum for 24 days each showed that urinary output was responsive to increasing molybdenum intake. 19 Increasing dietary molybdenum intake from 22 to 72 μg/d resulted in a 3-fold increase in fractional transfer of molybdenum from plasma to urine, and an additional 2-fold increase was observed when subjects transitioned from 121 to 467 μg/d (fractional transfer refers to the fraction of plasma molybdenum transferred to the urine per unit time). The percentage of plasma molybdenum deposited into tissues was also sensitive to molybdenum intake, with fractional tissue deposition decreasing at increasing molybdenum levels. The ability for the body to adapt to different molybdenum levels could explain the low incidence of deficiency and toxicity.
Deficiency
Observation of molybdenum deficiency in humans is very rare. One case of molybdenum deficiency in a patient on prolonged total parenteral nutrition for Crohn’s disease was reported by Abumrad et al. 26 Clinical symptoms included irritability, tachycardia, tachypnea, and night blindness. Tissue sulfite oxidase activity was low, whereas sulfate output was depressed and thiosulfate excretion was elevated; these symptoms were alleviated by reducing intake of protein and sulfur-containing amino acids. Clinical symptoms were completely ameliorated by provision of 300 μg ammonium molybdate per day.
Molybdenum deficiency symptoms are more likely to occur because of a rare genetic disorder in the production of molybdopterin. This condition is called molybdenum cofactor deficiency. Several mutations have been observed in genes encoding for molybdopterin.27–29 Molybdenum cofactor deficiency leads to accumulation of toxic metabolites, particularly sulfite, which in turn causes neurological damage, seizures, and early childhood death. Until recently, therapy has been symptomatic, with no effective cure available. However, very recently it was reported that one type of molybdenum cofactor deficiency could be treated with substitution therapy. In this case study, purified cyclic pyranopterin monophosphate (Figure 2) was administered intravenously, resulting in normalization of clinical markers. 30 This represents the first real potential therapy for this devastating disorder.
Structure of cyclic pyranopterin monophosphate
Toxicity
Although molybdenum can be very toxic to some animals, particularly ruminants, it does not seem to present a severe toxicity risk to humans. 31 In rodent studies, very high doses of molybdenum have been associated with reduced growth,32–34 kidney histological changes and renal failure,32,35 liver histological changes, 32 reproductive abnormalities,36,37 bone deformities,33,38 and anemia. 33
Little toxicity data exist specifically for humans. One study has reported the effect of high dietary molybdenum intake on individuals in areas of Armenia where dietary molybdenum is unusually high 39 because of high molybdenum levels in soil, resulting in estimated daily intake levels of 10 to 15 mg/d. In this population, elevated serum uric acid levels and high tissue xanthine oxidase levels were observed. Individuals in this population displayed aching joints, symptoms resembling gout, hyperuricosuria, and elevation of blood molybdenum. Another study reported the effect of molybdenum exposure via air on humans. Workers exposed to high air concentrations in a molybdenum production plant had increased blood uric acid concentrations, aching joints, headaches, and gout-like symptoms. 40
In 2005, a case study was reported of an individual with high molybdenum exposure. 41 An electrician with high occupational molybdenum exposure presented with hyperuricemia and gouty arthritis, which was ascribed to the molybdenum exposure. During an exposure-free period, his symptoms lessened, but then again worsened when he was once again exposed to high levels of environmental molybdenum. The authors noted that the association could be circumstantial.
In 2008, Meeker et al 42 reported an association between male infertility (impaired sperm motility) and blood molybdenum levels, based on volunteers attending infertility clinics. In a follow-up study published 2 years later, it was reported that circulating testosterone levels were inversely associated with blood molybdenum levels in the same study population. 43
Conclusion
Although the silvery metal known as molybdenum receives less attention than other micronutrients for its role in human health, neither causing great deficiency symptoms nor toxicity disorders in humans, molybdenum remains an essential part of the human diet. The keen ability of the body to adapt to a wide range of molybdenum intakes could be the reason that high or low molybdenum intakes have minimal impact on health. Nonetheless, decades of research have shown that molybdenum displays very interesting chemistry in its role as part of an essential cofactor for several very important enzymes, and without molybdenum, life could not be sustained.
Footnotes
The author declared no conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the United States Department of Agriculture.
