Abstract
Human islets can be transplanted into the portal vein for T1 diabetes, and a similar procedure is being used in a clinical trial for stem cell–derived beta-like cells. Efforts have been underway to find an alternative transplant site that will foster better islet cell survival and function. Although conceptually attractive, the subcutaneous (SC) site has yielded disappointing results, in spite of some improvements resulting from more attention paid to vascularization and differentiation factors, including collagen. We developed a method to transplant rat islets in a disk of type 1 collagen gel and found improved efficacy of these transplants. Survival of islets following transplantation (tx) was determined by comparing insulin content of the graft to that of the pre-transplant islets from the same isolation. At 14 days after transplantation, grafts of the disks had more than double the recovered insulin than islets transplanted in ungelled collagen. SC grafts of disks had similar insulin content to grafts in a kidney site and in epididymal fat pads. In vivo disks underwent contraction to 10% of initial volume within 24 h but the islets remained healthy and well distributed. Whole mount imaging showed that residual donor vascular cells within the islets expanded and connected to ingrowing host blood vessels. Islets (400 rat islet equivalents (IEQ)) in the collagen disks transplanted into an SC site of NOD scid IL2R gammanull (NSG) mice reversed streptozotocin (STZ)-induced diabetes within 10 days as effectively as transplants in the kidney site. Thus, a simple change of placing islets into a gel of collagen 1 prior to transplantation allowed a prompt reversal of STZ-induced diabetes using SC site.
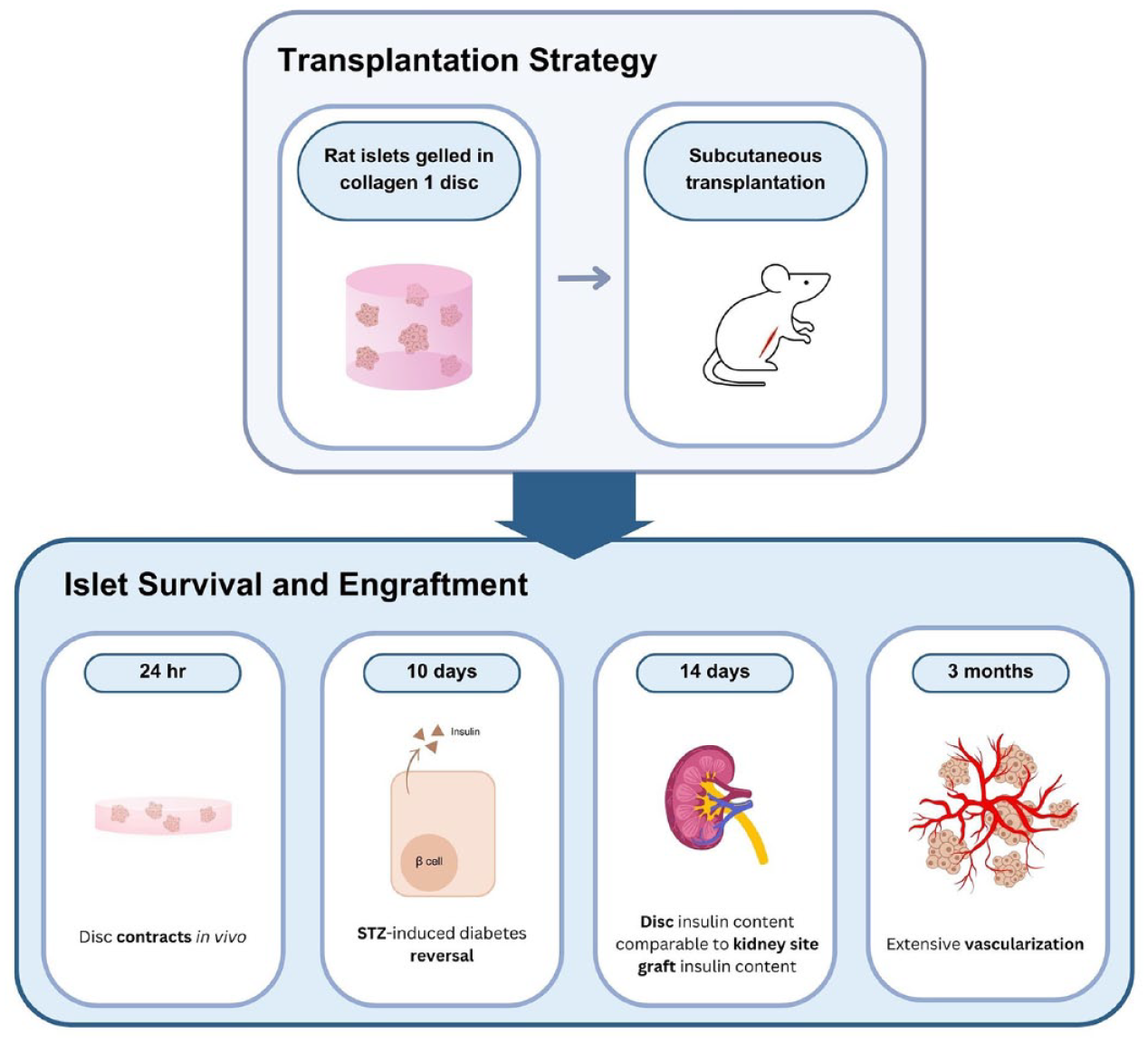
Introduction
Beta cell replacement with islet transplantation should provide enormous benefit to people with diabetes. Currently, human cadaveric islets are transplanted into the portal vein as an FDA-approved therapy, while a similar procedure using beta-like cells generated from pluripotent stem cells is in clinical trial. Considerable progress has been made in recent clinical trials1,2, but the future will require finding new sources of beta cells or beta-like cells likely to come from advances in stem cell biology 3 . Despite these advances, the liver has worrisome inefficiencies resulting in marked loss of beta cells thought to be due to anoxia and immune reactions in the peri-transplant period4,5 in addition to being an invasive procedure limiting graft retrieval. Finding a transplant site superior to the liver is a high research priority.
The possibility of using muscle has received a lot of attention because of encouraging results found in a variety of transplant models6–9. Another possibility with potential is the peritoneal cavity, which has led to plans for creating a microporous scaffold that can be placed in the omentum 10 .
Subcutaneous (SC) sites have been attractive in concept, but for many years the results have showed little promise11–13. Yet the large amount of available SC space may be what is required to accommodate the needed volume of islet tissue to manage diabetes. Unlike the liver, the accessibility of SC sites would facilitate monitoring for the potential development of teratomas and assessment of immune reactions and provide easy retrieval of the graft in case of such events. Work on islets in the SC site over the past few decades has been impressive and strategies influencing vascularization and differentiation are important.
A recent study examined the possibility that ready-made microvessels isolated from adipose tissue can support connection to host vasculature, thus promoting engraftment and function of either human embryonic stem cell (hESC)-derived pancreatic progenitors or human islets 14 . The importance of vascularization on function was seen when islets were placed on a vascularizing bed that developed when methacrylic acid-polyethylene glycol was provided in a hydrogel 15 . In another study, the implantation of recombinant basic fibroblast growth factor into an SC scaffold increased the number of vascular cells and gave higher cure rates for diabetes 16 . Other strategies that enhanced vascularization of transplanted islets without the use of growth factors or scaffolds relied on prevascularization of the transplant site17–20.
The effects of different matrices on graft survival and function have also received particular attention. The function of secretory and support cells can be greatly influenced by the different matrix components, of which collagen has been of particular interest. Considerable attention has been paid to fibrillar type1 collagen, a component of extracellular matrix that has complex effects on islet development and function21,22, and has been found to improve islet transplant outcomes in a number of circumstances23,24. These data support the concept that mechanisms responsible for differentiation may create opportunities to enhance function. With a transplant, there are multiple factors such as islet cells, angiogenesis, and different kinds of support cells such as mesenchymal stromal cells, which can all contribute to better secretory function24–26.
Because we were attracted to the reported benefits of collagen 121–24,27, our current study focuses on the transplant efficacy of islets contained in disks of gelled collagen 1 that can be transplanted into SC spaces or other sites. We used the approach of measuring the insulin content of islets, which provided insights into how the preservation of beta-cell mass and function are associated with improved transplant outcomes.
Methods
Animals
Male 7- to 8-week-old Sprague-Dawley rats (Taconic Biosciences, Germantown, NY) were used for isolating islets for transplantation. NOD scid IL2R gammanull (NSG) mice (catalog number 000557, Jackson Laboratory, Bar Harbor, ME, USA) were used as transplant recipients. Diabetes was induced using streptozotocin (STZ; Sigma-Aldrich, St. Louis, MO, USA; either a single 140 mg/kg dose intraperitoneal (IP) or five 40 mg/kg doses IP over 5 days) and confirmed by blood glucose readings of over 340 mg/dl. Morning-fed blood glucose levels were measured weekly by tail snip with an Ascensia Contour Next EZ glucometer (Ascensia, Parsippany, NJ, USA). Animals were kept on a 12-h light/dark cycle with food and water ad libitum. All animal procedures were approved by the Joslin Institutional Animal Use and Care Committee.
Islet Isolation
Rat islets were isolated by collagenase digestion followed by Ficoll density gradient centrifugation as previously described 28 . Islets were hand-picked and cultured overnight (o/n) in RPMI 1640 containing 11 mM glucose, 10% fetal calf serum, 100 units/ml penicillin, and 100 μg/ml streptomycin at 37°C with 5% CO2. After o/n culture, the islets were passed through a 200-µm filter to remove large or clumped islets and to increase consistency among aliquots.
Islet Viability Matrix (IVM) Preparation
IVM was prepared as described by Yu et al.
23
using Human Type 1 collagen (Advanced Biomatrix, Carlsbad, CA, USA), 10× Medium 199 (M199), fetal bovine serum, NaHCO3 (7.5%), and
Disk Preparation
High concentration type 1 rat tail collagen (Catalog number IKD119261001 Advanced Biomatrix, Carlsbad, CA, USA) was diluted to 3 mg/ml using a 0.1% acetic acid solution. A working solution was then prepared using 1 part M199 to 9 parts 3 mg/ml collagen, and then adjusting the pH to 7.0–7.5 using 0.1M NaOH. Rat islets were suspended in 150 μl of the 3 mg/ml collagen working solution, then set into an 8 × 8-mm cloning ring lightly attached to a coverslip with silicone. The collagen was then allowed to polymerize for 1 h at 37°C. For in vitro analyses, disks were gently removed from cloning rings and cultured on a 6-well ultra-low attachment plate (Corning Inc, Corning, NY, USA) with RPMI 1640.
Transplantation
Using a Kent Scientific anesthesia machine, 3–4% isoflurane (Pivetal NDC #46066-755-04, Patterson Veterinary Supply, Loveland, CO, USA) were used to keep the mice asleep for the procedure. Anesthesia was confirmed by toe pinching. The incision site was shaved and then washed with 70% ethanol and Betadine. The o/n (1 or 2 nights)-cultured islets were sized (by diameter) so that the transplant aliquots each contained a set number of islet equivalents (IEQ). Aliquots of islets were made shortly before transplantation, with 6–8 aliquots taken for pre-transplant samples for insulin and DNA content. Pre-transplant samples remained on ice throughout the transplantations and were then immediately processed following completion of the procedure.
For rat islet transplantation into an SC pocket, a BD Insyte Autoguard 18 g 1.16 in shielded IV catheter (BD, Franklin Lakes, NJ, USA) with the needle removed was placed tightly on a 1-ml syringe. To create a pocket in the SC space for the tissue to be expelled into, 0.02 ml of air was added to the tip of the syringe. Either 250 IEQ or 100 hand-counted islets (sized between 50 and 200 µm in diameter) were spun down in an Eppendorf tube and supernatant removed. Then, 100-μl IVM or rat tail type 1 collagen or 50-μl RPMI 1640 was added to the tube containing the islets and mixed gently using the tip of the catheter. All the tissue and liquid were drawn into the syringe. A small incision large enough to accommodate the catheter was cut in the skin on the right side of the midline of the ventral abdomen midway between the ribs and the pelvis. The catheter was inserted under the skin about 1 inch toward the ribs of the animal. Using fingers to hold the skin tightly around the catheter, the contents of the syringe were expelled quickly into the SC space. The catheter was removed and the opening was stapled closed.
For SC disk implantation, a 1-cm incision was made in the skin on the right side of the midline of the ventral abdomen midway between the ribs and the pelvis. A small pocket in the SC space across the midline was formed by gently inserting and opening scissors. The cloning ring with the gelled collagen disk was removed from the coverslip, placed through the incision and the disk was then gently removed with forceps. The cloning ring was then removed from the animal and the incision stapled close.
Fat pad disk implantation was done by placing a tiny amount of Vetbond tissue adhesive (3M, Saint Paul, MN, USA) on the epididymal fat pad, placing the gelled collagen disk containing 100 hand-counted islets (sized between 50 and 200 µm in diameter) on top, folding the epididymis over it and using more Vetbond to encase the collagen disk within the fat pad; the incision was stapled shut.
For kidney capsule transplantation, 400 IEQ in 100-µl RPMI 1640 were pelleted into PE50 tubing attached to a Hamilton syringe and injected under the kidney capsule as previously described 29 .
Graft Removal and Insulin Extraction
Graft and surrounding tissue were removed under terminal anesthesia. Some grafts were taken for histology or whole mounts (see below); others for insulin content. Dithizone (diphenylthiocarbozone), which stains insulin-containing cells bright red, was used to assess islets within freshly isolated grafts. To measure insulin content, the removed tissue was placed into 3-ml phosphate buffered saline (PBS) and homogenized (Ultra-Turrax T-25, Ika Works, Wilmington, NC, USA) with 7-ml acid ethanol (EtOH). The mixture was incubated o/n at 4°C on an orbital shaker and then centrifuged at 1,800 ×g to obtain the supernatant used for the insulin enzyme-linked immunosorbent assay (ELISA).
DNA Content and Insulin ELISA
DNA content was measured from pre-transplant samples using the CyQUANT Cell Proliferation Kit (Thermo Fisher, Waltham, MA, USA). For both pre-transplant samples and grafts, insulin content was measured using the ALPCO Stellux Chemiluminescent ELISA for rodent insulin (ALPCO, Salem, NH, USA).
Percent of Pre-Tx Insulin Calculation
For ease of comparison across the different islet isolations, times of engraftment, and the different numbers of transplanted islets, we normalized the graft islet insulin content to be a percentage of the measured pre-transplant insulin content (ng/IEQ). The DNA content for each pre-transplant sample was divided by 9.4 ng [the estimated DNA content in a cultured 150-µm islet (IEQ) assuming 1,560 cells per islet and 6 pg of DNA per cell]30,31 to determine the number of IEQs per pre-transplant sample. The average of all pre-Tx samples (n = 6–8) from each islet isolation was used to normalize the graft insulin content of that experiment [measured graft insulin content divided by its average pre-transplant IEQ number = normalized graft insulin content (ng/IEQ) value].
Histology
Tissue samples, removed grafts, and collagen disks taken for immunohistochemistry or whole mounts were fixed in fresh 4% paraformaldehyde (Sigma-Aldrich, St. Louis, MO, USA) for 2–6 h at room temperature (RT), then moved to PBS and stored at 4°C. Samples were processed, embedded in paraffin, and sectioned by the Joslin Advanced Microscopy Core. For immunostaining, paraffin sections were deparaffinized, rehydrated through graded ethanol, and placed in PBS with 0.5–2% lamb serum before incubation in primary antibody (see Supplemental Table 1) either at 4°C o/n or for 2.5 h at RT. Slides were washed for 5 min ×2 in PBS with lamb serum and then incubated in secondary antibody (Supplemental Table 2) for 1 h at RT. For stains with multiple antibodies, all incubations were done sequentially. In cases where amplification was needed, slides were blocked for 30 min each with streptavidin and biotin from a Blocking Kit (SP-2002 Vector Laboratories, Newark, CA, USA) before the primary antibody incubation, and a biotinylated secondary antibody to the primary host species was used followed by a fluorophore-conjugated streptavidin step. Slides were mounted with VECTASHIELD® either with or without 4’,6-diamidino-2-phenylindole (DAPI) (Vector Laboratories). Slides were imaged using either a Carl Zeiss AxioSkop 2 MOT with a Carl Zeiss Axiocam 305 color camera or Zeiss LSM-710 confocal microscope. Images were analyzed with Carl Zeiss Zen Black Lite and Blue Lite.
For whole mounts, fixed samples were stored in PBS at 4°C until undergoing an immunostaining and clearing process adapted from the Visikol® HISTO™ Protocol. Samples were first washed in PBS- calcium and magnesium free (PBS-CMF, Corning Inc) for 30 min ×2 while rocking at RT before undergoing a series of permeabilization steps for 1.5 h each: 50% methanol (MeOH) in PBS, 80% MeOH in distilled water (dH2O), and 100% MeOH. Samples were then incubated o/n at 4°C rocking in Dent’s Bleach (30% H2O2/100% dimethyl sulfoxide (DMSO)/100% MeOH at 1:1:4, respectively). Samples were next rehydrated by undergoing 1 h rocking at RT in each of the following: 20% DMSO in MeOH, 80% MeOH in dH2O, 50% MeOH in PBS-CMF, and 100% PBS-CMF. Next, samples were incubated in Permeabilization Buffer (Visikol, Hampton, NJ, USA) for 2 h at RT rocking. Samples were washed in Blocking Buffer [PBS-CMF with 0.05% Triton® X-100 (Sigma-Aldrich, St. Louis, MO, USA)] and 1% bovine serum albumin (Sigma-Aldrich, St. Louis, MO, USA) for 2 h at RT and then incubated in the first primary antibody, Guinea pig anti-Insulin (Thermo Fisher, Waltham, MA, USA) at 1:50 o/n at 37°C shaking. Samples were then washed in Blocking Buffer for 1 h ×5 at RT rocking and were then incubated in the first secondary antibody, Alexa Fluor® 594 conjugated anti-Guinea Pig (Jackson ImmunoResearch, West Grove, PA, USA) at 1:100 o/n at 37°C shaking. Samples were then washed in Blocking Buffer again for 1 h ×5 at RT rocking and then o/n at 4°C for the last wash. The next day, samples were incubated in the second primary antibodies, a cocktail of mouse anti-CD34 (BioGenex, Fremont, CA, USA) and Mouse anti-smooth muscle actin (SMA; Sigma-Aldrich, St. Louis, MO, USA) at 1:100 and 1:800, respectively, o/n at 37°C shaking. Samples were then washed in Blocking Buffer for 1 h ×5 at RT rocking and were then incubated in the second secondary antibody, Alexa Fluor® 488 32 conjugated anti-Mouse (Jackson ImmunoResearch, West Grove, PA, USA) at 1:100 o/n at 37°C shaking. Samples were then washed in Blocking Buffer again for 1 h ×5 at RT rocking and then o/n at 4°C for the last wash. Finally, samples were dehydrated to prepare for the clearing process by rocking at 4°C for 1.5 h in each of the following: 50% EtOH in PBS-CMF, 80% EtOH in dH2O, and 100% EtOH. Tissue samples were then removed from the EtOH and gently wiped with a Kimwipe to ensure removal of all the EtOH before being put in Visikol® HISTO-1™ (Visikol, Hampton, NJ, USA) for 4 h at 4°C rocking. Samples were then transferred into a CoverWell™ Imaging Chamber (Electron Microscopy Sciences, Hatfield, PA, USA) in Visikol® HISTO-2™ (Visikol, Hampton, NJ, USA) and mounted directly onto a slide (Thermo Fisher, Waltham, MA, USA) before being imaged on a Zeiss LSM-710 confocal microscope as z-stacks. Images were analyzed in Carl Zeiss Zen Black Lite and Blue Lite. The original z-stacks ranged from 55 to 221 images, with the thickness of the areas covered ranging from 356 to 838 µm. For projection images, screen capture of subsets of images from the original z-stack was taken to create a 2-dimensional representation of a 3-dimensional (3D) image. These subsets ranged from 34 to 171 images and thickness of 136 to 646 µm, which allowed for analysis of different planes of focus of the same z-stack. Movies were created by viewing the entire z-stacks as a 3D image rotating in space or as a progression.
Statistical Analysis
Data are shown as mean ± standard error of the mean. Statistical significance (P < 0.05) was calculated using Graphpad Prism version 9.0.2. An unpaired Student’s t test was used to compare differences between two groups. When comparing differences among 3 or more groups, one-way analysis of variance was used followed by Tukey’s multiple comparison tests.
Results
Evaluation of IVM
We were impressed by a report describing increased survival of murine, porcine, and human islets transplanted into SC pockets of mice using IVM
23
, which consisted of islets mixed with human collagen 1,
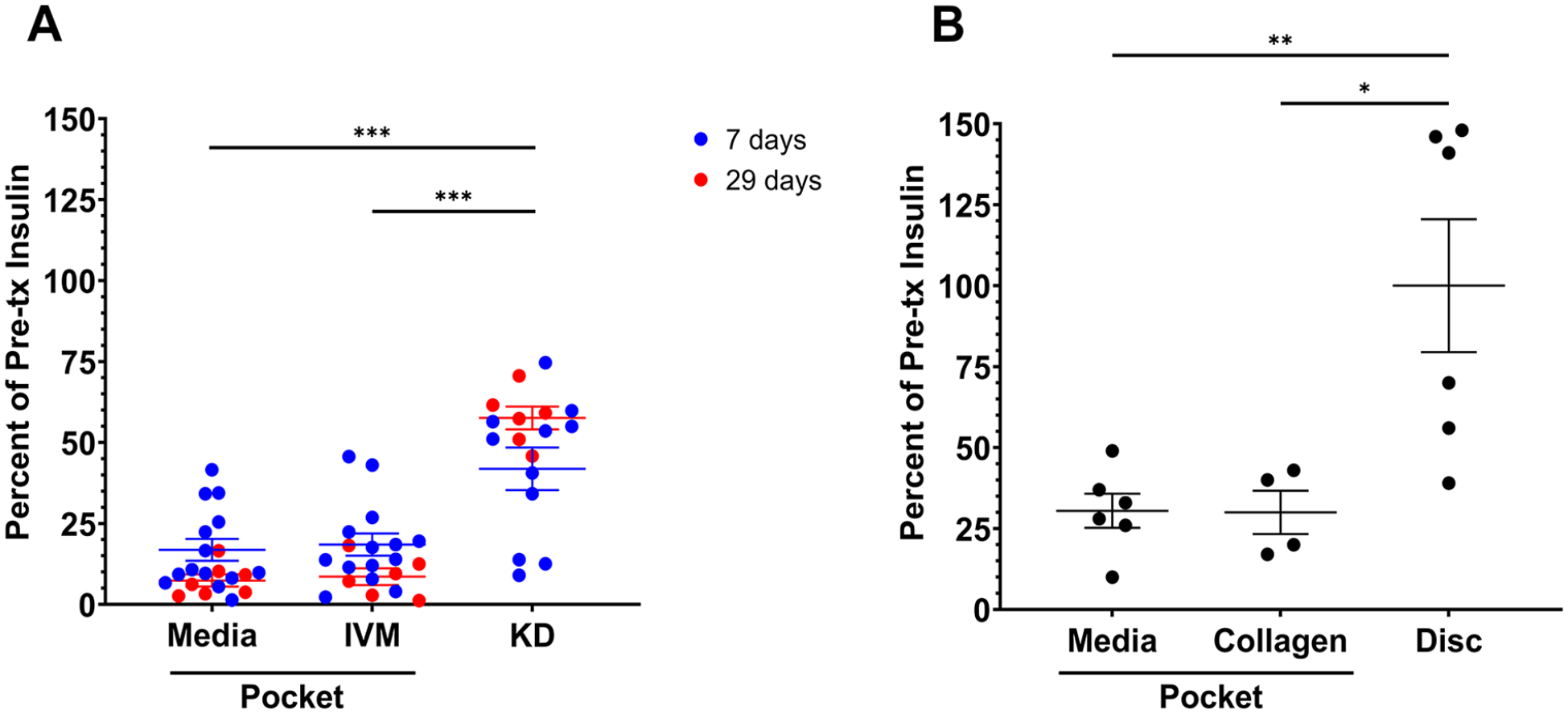
Comparison of rat islets transplanted in the subcutaneous space on ventral abdomen and under the kidney capsule in nondiabetic NSG mice. Insulin recovered from the graft as a percent of that of pre-transplant samples was used as a readout. (A) There was no difference in the insulin recovered from grafts of rat islets in pressure-induced pockets using IVM or RPMI media at either 7 (blue) or 29 (red) days after transplantation. Both had reduced insulin compared to grafts under the kidney capsule. Data shown for individual animals (n = 21 media, 21 IVM, 17 kidney capsule). (B) Comparison of insulin recovery at 14 days from rat islets transplanted in the pressure-induced pockets and collagen disks. Hundred hand-counted rat islets transplanted subcutaneously as either collagen disks, collagen pockets, or media pockets (collagen for disk = 150-μl 3 mg/ml rat tail type 1 collagen; collagen for pocket = 100-μl 3 mg/ml rat tail type 1 collagen; media = 50-μl RPMI). Pre-Tx samples (n = 8) had 22.9 ± 1.2 ng/islet. At 14 days after transplantation, the grafts of the disks had more than double the recovered insulin than either of the pocket conditions (n = 6 collagen disk; 4 collagen pocket; 6 media pocket). IVM: islet viability matrix; KD: kidney capsule; NSG: NOD scid IL2R gammanull; Tx: transplantation; P-values are denoted as follows: *P = 0.03; **P = 0.002; ***P < 0.001.
Enhanced Function of Islets Contained in a Collagen Gel
In previous studies, we found that islets transplanted into the kidney capsule site were clumped, and had disrupted architecture and often areas of necrosis, which fits with a modest reduction in insulin content 4 . We therefore posited that immobilizing islets in gelled hydrogel might be beneficial. Since others had shown that the addition of collagen 1 was beneficial for transplanted islets22–24,27, we devised a way to make islet-containing disks of collagen 1 that were easy to handle and could be transplanted into SC sites or the epididymal fat pad. The disks were made by filling 8 × 8-mm cloning rings attached to a coverslip with a suspension of rat islets in 150 ml of rat tail collagen 1 (3 mg/ml) and allowing it to gel. When removed from the cloning rings, the disks had a diameter of 6 mm, a height of 5 mm, and an average volume of 9.1 mm3.
A pilot experiment using these gelled disks compared to pockets with media or the same amount of un-gelled collagen encouragingly showed increased insulin recovery after 14 days (Fig. 1B). In follow-up experiments (Fig. 2), it was clear that islets in pre-gelled disks placed into an SC site had comparable beta-cell survival to those transplanted under the kidney capsule or islets in disks placed into the epididymal fat pad. As seen in the pre-transplant insulin content data (Fig. 2A), islets from different isolations (batches) differed in their insulin content even though the general techniques and personnel were the same. To facilitate comparison across transplant sites, engraftment times, and islet doses, the simple measurement of insulin recovery was used as a surrogate for beta-cell mass. For each islet isolation, insulin content was measured from aliquots of pre-transplant islets and lysates from the grafted tissue from that isolation to determine the insulin recovery as percent of pre-transplant insulin content. As seen in Fig. 2B, islets in SC site disks had insulin recovery comparable to that of the “gold standard” kidney capsule site. Surprisingly, it was found that some grafts had more than 100% recovery of pre-transplant insulin. However, as noted before and as is shown in Supplemental Fig. 1, with o/n culture there can be a 30% loss of insulin without a change in DNA content. Thus, the observed variation in the insulin content of batches (isolations) may reflect differential loss of insulin content with o/n culture.
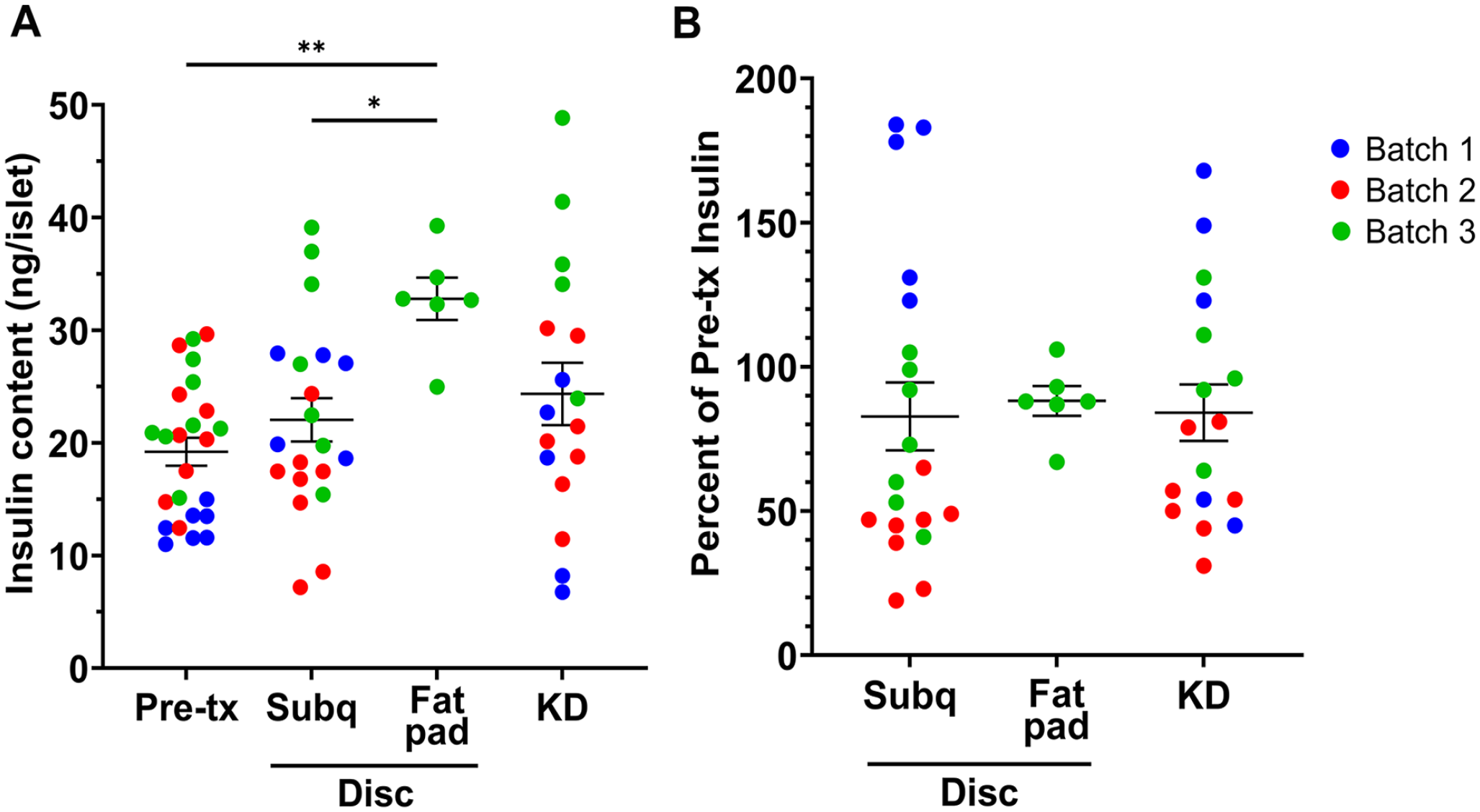
Collagen disks in the subcutaneous space had comparable insulin recovery to disks in the epididymal fat pad and the kidney capsule at 14 days after surgery. Hundred hand-counted rat islets. Data points are from individual animals. Three independent experiments (separate colors) with n = 27 pre-transplant samples; 20 subcutaneous disks; 6 fat pad disks; 17 KD. (A) Results given as insulin content per initially transplanted islet (ng insulin/islet). There were differences in insulin content for the overnight-cultured pre-transplant samples in the experiments: batch 1 = 12.7 ± 0.5 ng insulin/islet; batch 2 = 21.2 ± 1.9 ng insulin/islet; batch 3 = 22.7 ± 1.6 ng insulin/islet. (B) To account for the differences in insulin content of the separate islet batches, the data are graphed as percent of pre-transplant insulin. The three sites had comparable insulin recovery in each experiment (batch). KD: kidney capsule. P-values are denoted as follows: *P = 0.03; **P = 0.002; ***P < 0.001.
To learn about their potential packing density, disks containing 100, 300, 400, or 600 IEQ were engrafted in SC sites for 14 days (Fig. 3). At 14 days, the insulin recovery was proportionally lower when the number of IEQ was increased to 600, suggesting a crowding effect that limited survival of beta cells.
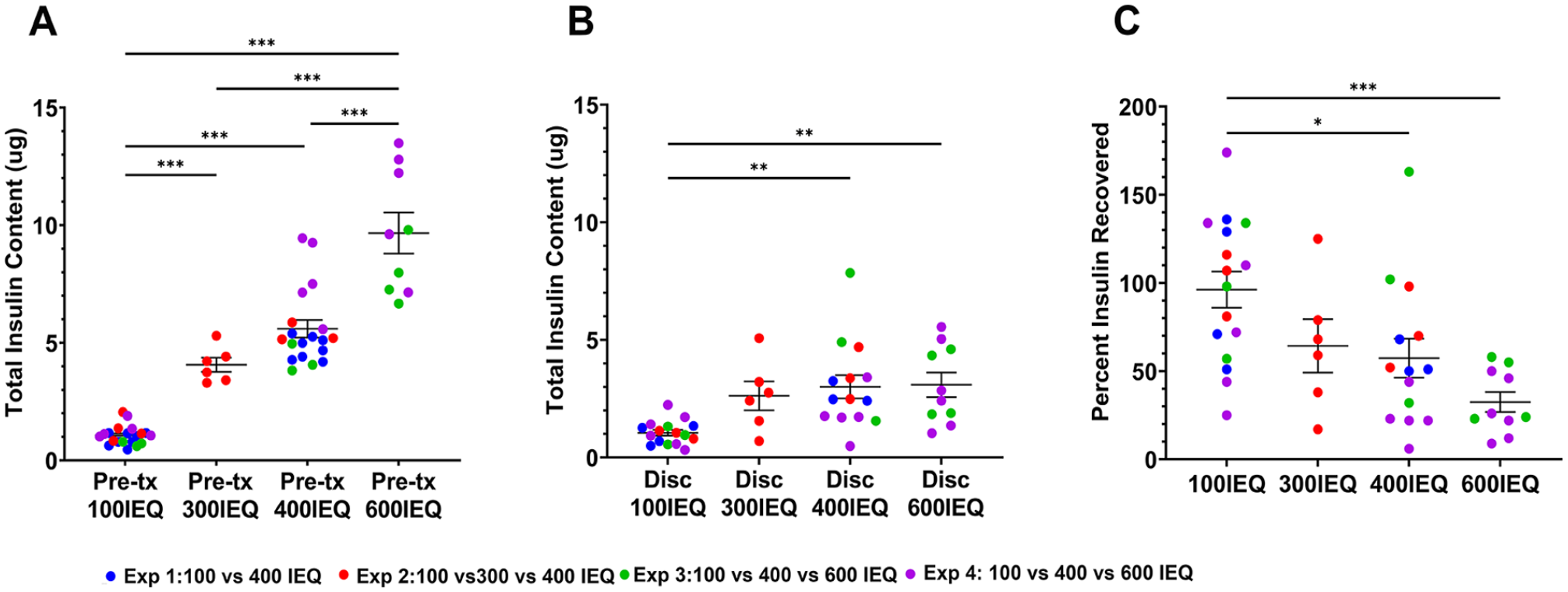
Increased islet density led to lower insulin recovery at 14 days. (A) The pre-Tx samples had insulin content proportional to the number of islets. Experiment 1 (blue) compared 100 versus 400 IEQ; experiment 2 (red) compared 100, 300, and 400 IEQ; experiment 3 (green) and experiment 4 (purple) compared 100, 400, and 600 IEQ. (B) The total insulin recovered at 14 days after transplantation from 300, 400, and 600 IEQ did not differ from each other but were higher than that of 100 IEQ. (C) The percent of recovered insulin compared to the pre-transplant insulin decreased with higher islet density. Pre-Tx: 100 IEQ, n = 20; 300 IEQ, n = 6; 400 IEQ, n = 19; 600 IEQ, n = 9; disk: 100 IEQ, n = 16; 300 IEQ, n = 6; 400 IEQ, n = 14; 600 IEQ, n = 10. IEQ: islet equivalents; Tx: transplantation. P-values are denoted as follows: *P = 0.03; **P = 0.002.
Contraction of Gel Disk Volume In Vivo
A remarkable contraction of the volume of the disks was seen during in vivo experiments. The volume of the transplanted disk fell to about 7% of the original value (Fig. 4), which agrees with the well-known contraction of collagen 1 33 . In an in vivo time-course experiment, most of the contraction occurred within the first 24 h (Fig. 4A; day 0: 229 ± 35 mm3, n = 3; day 2: 14.4 ± 2 mm3, 6.9% of initial volume, n = 4; day 5: 9.1 ± 0.6 mm3, 4.0% of initial volume, n = 4). However, in vitro there was no contraction over a week (Fig. 4B; day 0: 157 ± 18 mm3; day 7: 171 ± 8, 109% of initial volume; n = 6). A separate experiment comparing day 0 and day 1 for both in vivo and in vitro disks (Fig. 4C) showed similar selective volume reduction only in the in vivo setting (day 0: 197 ± 5.7 mm3, n = 6; day 1 in vivo: 14.4 ± 1.5 mm3, 7.3% of initial volume, n = 6; day 1 in vitro: 221 ± 12 mm3, 112% of initial volume, n=6). The presence or absence of islets within the disks did not change these results (data not shown). Disks removed at day 14 (ie, after contraction) and stained with dithizone showed good spacing between the islets suggesting that clumping might not be a problem (Fig. 4D).
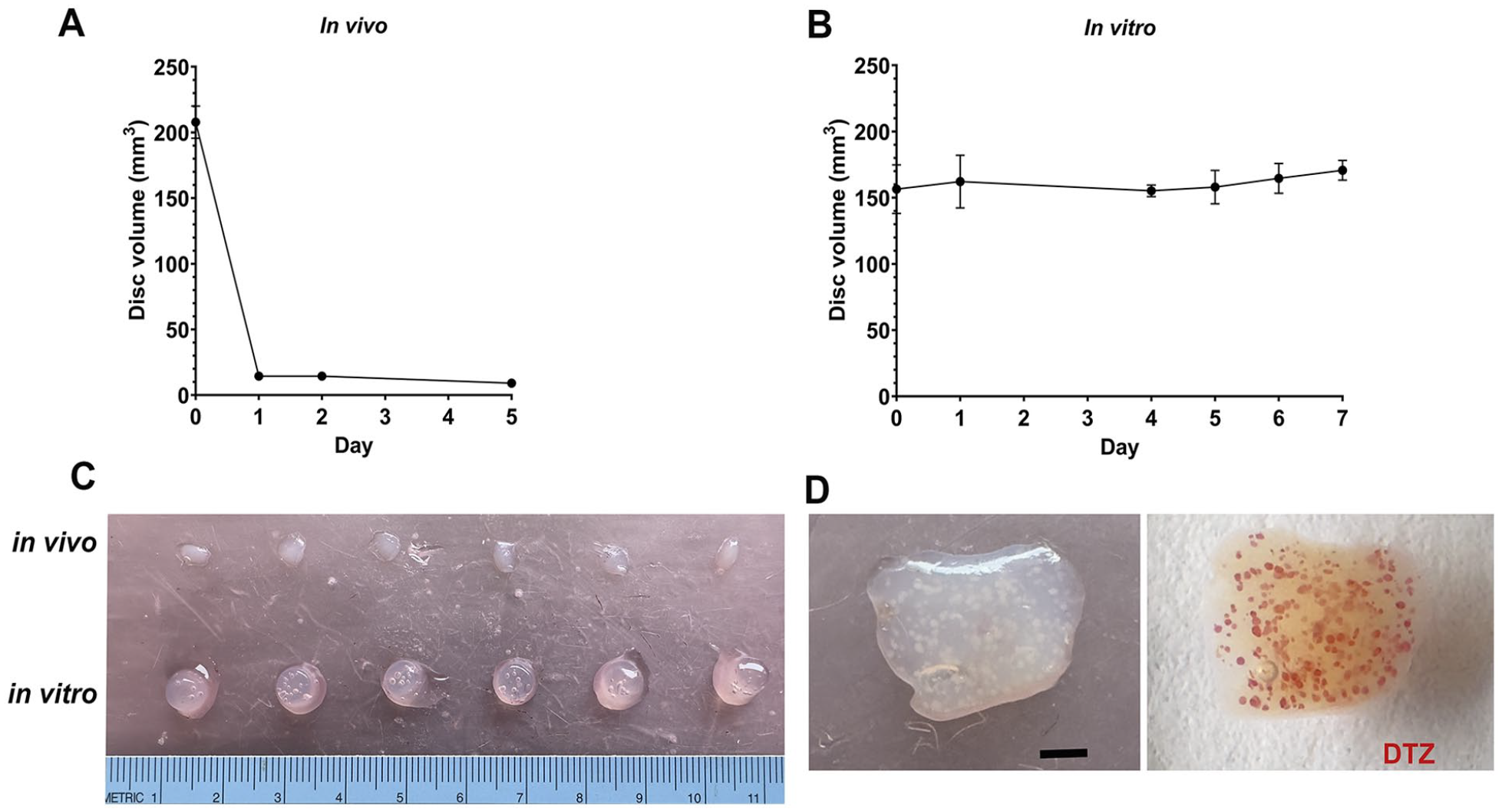
Collagen disks in vivo rapidly contracted in volume whereas those in vitro did not. (A) Even without embedded islets, the collagen disks in vivo contracted within 24 h to 7.3 ± 0.7% (n = 6) of their original volume with no further significant contraction. (B) In contrast, in vitro disks with islets cultured in media showed no contraction (P = 0.11). (C) In vivo day 0, n = 9; day 1, n = 6; day 2, n = 4; day 5, n = 4; in vitro, n = 6. The difference in contraction between in vitro and in vivo is easily seen at 24 h. (D) In vivo collagen disk removed at 7 days shows islets are well distributed even with contraction. White dots (left) on unstained image are the islets as shown with DTZ staining (red; right). Scale bar = 1 mm. DTZ: dithizone.
Whole-Mount Analysis of Islets Contained in Collagen Gels
To further understand the potential benefit of these collagen disks for islet transplantation into an SC site, we collected islet-loaded disks for whole-mount analysis of the islets and their revascularization (Fig. 5). Of note was the presence of vascular cells (CD34+/SMA+) in the islets cultured o/n before transplantation and after 1 day engraftment (Fig. 5A). In disks maintained in vitro for 7 days, these vascular cells appeared both within the islets and scattered in the collagen (Fig. 5B), suggesting their migration. In disks in vivo for 7 days, these vascular cells were seen enveloping islets and extending across the collagen (Fig. 5C, Supplemental Videos 1 and 2). These data support the notion that the collagen facilitates the revascularization of the transplanted islets by promoting the survival and outgrowth of the donor vascular cells. By 14 days in vivo large vessels from the skin or the underlying muscle could be seen at the disks (Supplemental Fig. 2) with some vessels clearly penetrating the engrafted disk (Fig. 5D). In a cured diabetic mouse 3 months after transplantation the vasculature was more extensive with clear connections from the host to the engrafted islets (Fig. 5E–H). As seen in the sequential projection images (Fig. 5H) and in Supplemental Video 3, small vessels extend throughout some of the larger islets.
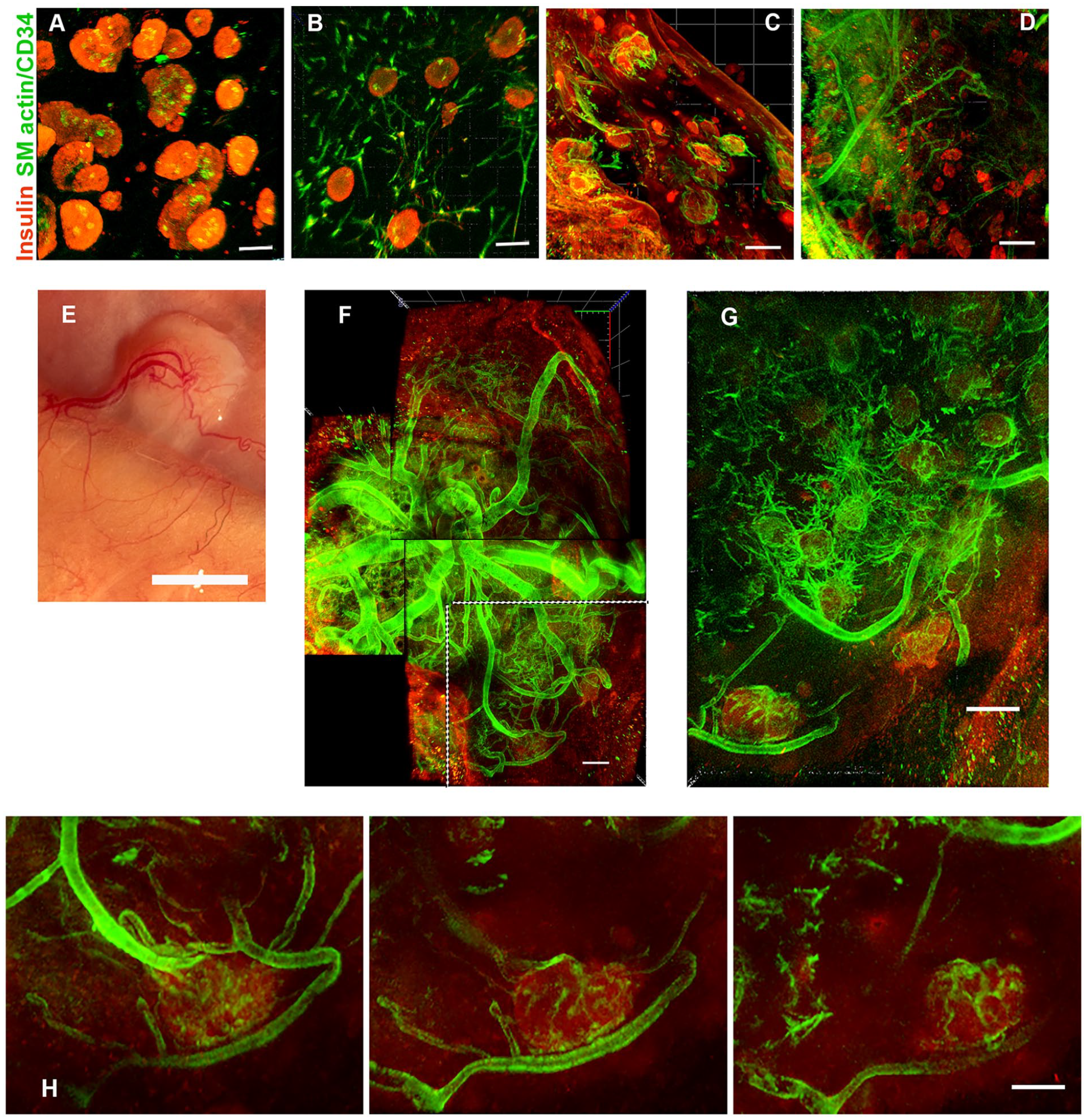
Vascularization of the disk grafts over time. (A) At day 1 after transplantation in the subcutaneous site, the islets within the disk are well distributed as shown in Fig. 4D and shown here in projection (290 μm) from whole mount. In most of the islets (red; insulin), residual vascular cells (green; CD34/SMA+) remain in clumps. (B) After 1 week in vitro, CD34/SMA+ cells have migrated through the collagen (projection, 405 μm). (C) At 1 week in vivo, the blood vessel cells have coalesced around the islets (projection, 506 μm). (D) At 2 weeks in vivo, large blood vessels from the host can be seen penetrating the collagen disk and surrounding the islets (projection, 648 μm). In (C) and (D), host tissue is seen in the lower left corner. (E) After 3 months in a cured diabetic NSG mouse, the islet-containing disk had extensive vascularization. (F) Projection image (495 μm) of whole mount of the same disk removed from the mouse. (G) Projection image (136 μm) of optical sections deeper into the whole mount of the area delineated (dotted lines) in (F) showing vessels surrounding and entering islets. (H) Sequential projection images (each 38 μm) showing penetration of blood vessels into an islet. Insulin (red); blood vessels combined SMA and CD34 (green). Scale bars: (A and B) 100 μm; (C, D, F, and G) 200 μm; (E) 5 mm; and (H) 150 μm. Videos are available as Supplemental Material. NSG: NOD scid IL2R gammanull; SMA: smooth muscle actin.
Reversing STZ-Induced Diabetes in NSG Mice With SC Islet Transplants
We then asked if islets in the collagen disks transplanted into an SC site of NSG mice could reverse STZ-induced diabetes as effectively as transplants in the kidney capsule site. As seen with islets in the kidney site (Fig. 6A), 400 rat IEQ in gelled disks in the SC site corrected the diabetes within 10 days in all but 1 mouse (Fig. 6B). The disk in this mouse was found within the stapled incision, leading us to call it a technical failure; it was deleted from the following analyses.
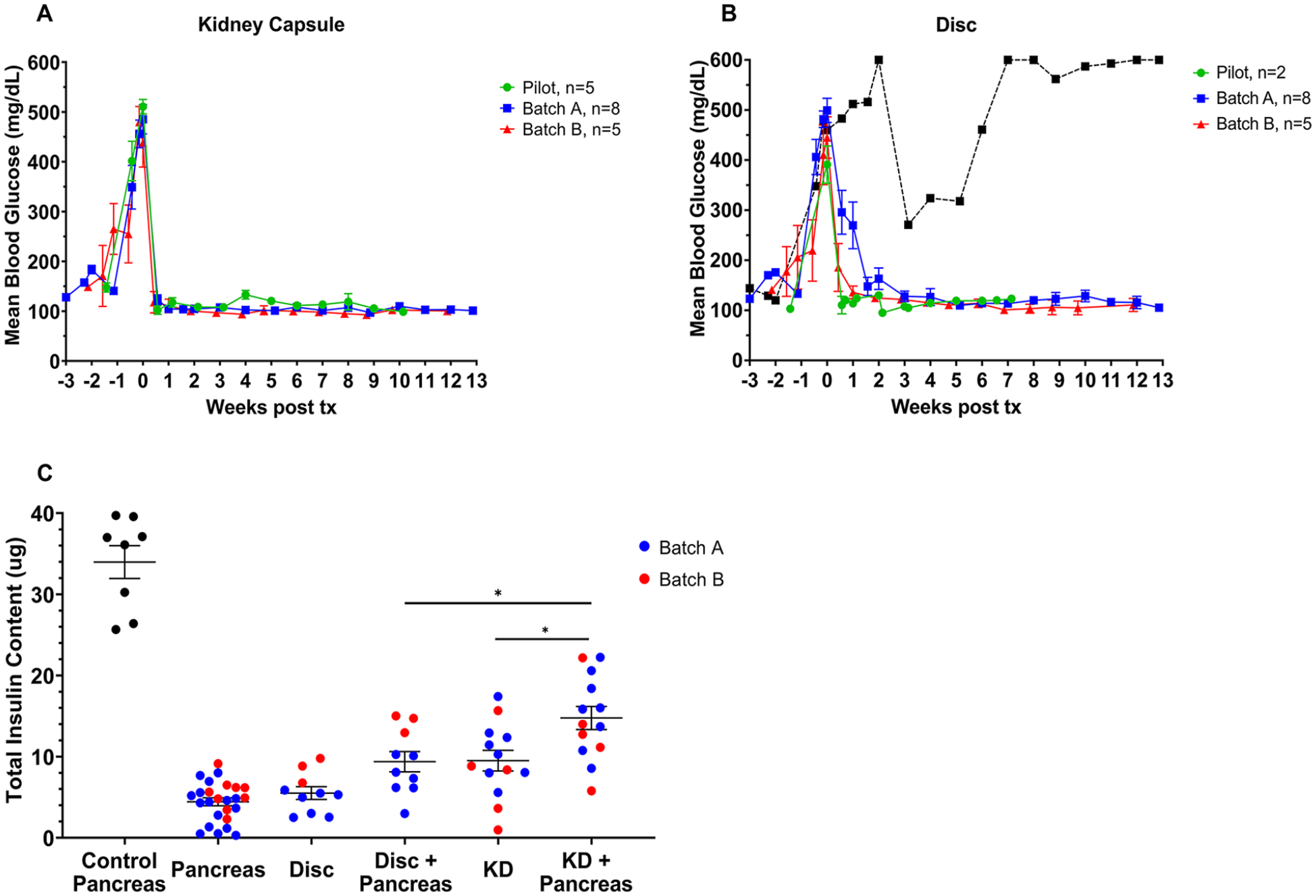
Islets transplanted subcutaneously when contained in collagen disks are as effective as islets transplanted into the kidney subcapsular space in STZ-induced diabetic NSG mice. Transplantation of 400 IEQ rapidly corrected STZ diabetes when transplanted into kidneys (A) or in a subcutaneous site embedded in collagen disk (B). Three independent experiments: pilot (green) n = 5 kidney, 2 disk; batch A (blue) n = 8 kidney, 8 disk; batch B (red) n = 5 kidney, 5 disk. Islets of batch A were cultured for 2 days whereas those of the pilot and batch B were only cultured for 1 day. Grafts were removed at the following weeks: week 7 for pilot disks, week 10 for pilot kidney, and week 12–13 for batches A and B. One animal transplanted with a disk (black line) did not achieve normoglycemia. At graft removal, its disk was found to have migrated into the area of skin that was stapled. We thought it best to call the transplant with this mouse a technical failure. Pre-Tx mean insulin content (ng/IEQ) = 26.7 ± 6.5 for batch A; and 17.6 ± 1.8 for batch B. (C) Even with successful restoration of normoglycemia, the total insulin content (graft plus residual pancreas) of the restored mice (shown in A and B) was only 30–50% of that of the pancreas of nondiabetic mice. All sample groups significantly differed from the nondiabetic pancreas (P < 0.001); among the transplant groups, the total insulin content was significantly lower (P = 0.021) for the mice with disks compared to those with kidney site. Batch A (blue); batch B (red). Tx: transplantation. P-values are denoted as follows: *P = 0.03
To determine if the grafts were solely responsible for the lowered glucose levels, we measured the insulin content of the islet grafts and that of the pancreases (Fig. 6C) at 12–13 weeks following transplantation. The average amount of insulin in the adult pancreas of the strain of mice used was 34.0 ± 2.0 μg (n = 8). For the animals transplanted at the kidney site, the mean insulin content of the grafts was 9.5 ± 1.3 μg and of the pancreas 5.3 ± 0.7 μg (n = 13). The mean total insulin of combined graft and pancreas was 14.8 ± 1.4 μg, which was 43.5% of that of nondiabetic mice. For the 10 mice transplanted with islets in a gel disk, the insulin content of the graft was 5.5 ± 0.8 μg and of the pancreas was 3.9 ± 0.6 μg. The combined insulin content of the two sites was 9.4 ± 1.3 μg, which represents 27.6% of the normal pancreatic insulin content. While there was no statistical difference in the insulin content of the kidney grafts and the SC disks nor in their pancreases, the total insulin recovered (pancreas plus graft) was higher (P > 0.03) for those mice with the kidney graft. The values of graft and pancreas paired for individual mice are shown in Supplemental Fig. 3.
We and others have assumed that STZ efficiently kills beta cells, but we were surprised by the amount of residual insulin remaining in the pancreas in mice that had over 340 mg/dl morning-fed blood glucose levels before transplantation (mean 471.3 ± 16.2 mg/dl, n = 25). The mean insulin content of the 23 pancreases from cured STZ mice was 4.7± 0.5 μg, or 13.8% of pancreas of nondiabetic control mice. While in some of the pancreases, the insulin content was negligible (4 were below 2 μg), in others it could be as high as 20% of the normal pancreatic insulin content. While monitoring a return to hyperglycemia after the removal of a graft is the gold standard for graft dependency, we found in pilot experiments that the mice did not survive more than a day on the removal of their grafted disk, so we could not rely on this procedure. Our assumption is that normalization of glucose levels was usually accomplished by combined insulin production from both the transplanted islets and the residual insulin–containing cells in the pancreas. It seems likely that the residual insulin–containing beta cells in the pancreas secrete insulin in a manner similar to transplanted islets, but we should be cautious about making that assumption.
Discussion
The SC space is an attractive site for clinical transplantation due to its accessibility, yet for many years experimental transplantation of islets into the SC site has provided very poor results. Improvements started to appear as several groups began focusing on variables such as changes in blood flow and cellular differentiation. Our present report shows that the simple change of placing islets into a gel of collagen 1 at an optimal density makes it possible for them to promptly reverse STZ-induced diabetes following transplantation in an SC site. This study adds to others showing that collagen 1 can provide beneficial effects on SC transplants2,21–24,27,34. We postulate that the beneficial effects of the gelled disks are two-fold. First, the collagen gel maintains a more even distribution of the islets thus preventing increased hypoxia from their clumping, as we saw in the kidney capsule site 4 . Second, within the collagen gel, the donor islet vascular cells not only survived, but expanded to eventually connect with the invading host blood vessels.
The function of transplanted islets must depend on the complex environment of the transplant site, and dramatic changes occur over the 2- to 3-week period following the transplant. When transplanted in the subcapsular space of the kidney, the gold standard site in rodents, many beta cells die and the normal interactions between the different islet cell types are disrupted, which leads to obvious remodeling of the cellular architecture 4 . In addition to the interactions between endocrine cells, islets contain a variety of other cell types that determine their structure and function. There is active angiogenesis, which must exert some influence. Jansson and Carlsson35,36 described active growth of host blood vessels seen in the first 3–5 days leading to re-establishment of blood flow within 7–14 days but with a lower density of vessels in the engrafted islets than endogenous ones. In our study, branches from host blood vessels were seen penetrating the collagen disks with islets by 7–14 days. While others 37 have shown some of the blood vessels in the transplanted graft originated from the donor, the collagen gels seemed to facilitate the expansion of the donor vascular cells as seen clearly in the in vitro cultured disks. In our whole mounts of grafts transplanted subcutaneously we saw exuberant angiogenesis, but we have yet to define the relative contributions between host and donor. However, the density of vessels in some islets was striking and approached that of endogenous islets.
Our favorable result in the SC space was similar to that obtained with islets transplanted under the kidney capsule of diabetic mice. However, we cannot yet conclude that the islets contained in disks provided better results than islets transplanted into kidneys because that comparison was not studied in detail. Regardless, the collagen effects are seemingly enough to render the SC space a comparable site to under the kidney capsule, and future studies would be helpful to further elucidate its effects on islet secretion and survival mechanisms in the SC space and in comparison with the gold standard kidney subcapsular space.
The disk gels containing islets are easy to make and transplant. They also undergo remarkable contraction when placed into an in vivo environment as the volume falls to less than 10% of where it started. Questions can be raised about possible benefit or harm caused by this phenomenon. On one hand, it might be helpful to have insulin output maintained by only about 10% as much volume. An obvious question is whether the contraction of the disks creates space that could provide room for the addition of more disks. Another possibility is that more effective shapes other than disks might be developed. These questions can be addressed by future measurements of insulin secretion and content.
The variability of islet size is also turning out to be an important issue. The Sefton group noted that preparations of isolated islets always have significant numbers of large islets that have problematic hypoxia of the islet centers. Using dispersion and reaggregation, it was possible to obtain a population of pseudoislets that had fewer of these large islets 38 and improved transplant results. The benefits of optimally sized islets for transplantation efficiency have been seen by us in other islet transplant systems 39 . Added collagen 1 seemed to stabilize the population of the pseudoislets in one study 38 .
One potential issue with the SC space is temperature control, which is normally an important function of skin. In hot weather, blood flow increases to enhance heat loss and with cold temperature, blood vessels clamp down to conserve heat. Further complexity comes from regional differences, such that change in blood flow of the hands will be different from that in more protected areas. If the SC space proves to be a potential site for clinical transplantation, it may be important to examine whether temperature changes might cause problems with insulin delivery from SC-transplanted islets.
Providing a stable environment for islet survival and vascularization following transplantation is key to maintaining successful graft function. Suspending islets in a gelled disk of collagen 1 appears to do just that and is able to cure diabetic mice with transplants in the SC space as well as in the gold standard kidney site. The clinical potential of this is exciting, as it allows the possibility of transplant into an easily accessible and monitored location. Future studies should be done to understand the host versus donor relationship in revascularization and how it is enhanced by collagen 1. Also, further measurements to determine the ideal packing density, size, and shape of the collagen disks will be essential in optimizing this method to realize its full potential as a therapeutic treatment of diabetes.
Supplemental Material
sj-docx-1-cll-10.1177_09636897241283728 – Supplemental material for Enhancement of Subcutaneous Islet Transplant Performance by Collagen 1 Gel
Supplemental material, sj-docx-1-cll-10.1177_09636897241283728 for Enhancement of Subcutaneous Islet Transplant Performance by Collagen 1 Gel by Anna French, Jennifer Hollister-Lock, Brooke A. Sullivan, Eline Stas, Albert J. Hwa, Gordon C. Weir and Susan Bonner-Weir in Cell Transplantation
Supplemental Material
sj-tif-2-cll-10.1177_09636897241283728 – Supplemental material for Enhancement of Subcutaneous Islet Transplant Performance by Collagen 1 Gel
Supplemental material, sj-tif-2-cll-10.1177_09636897241283728 for Enhancement of Subcutaneous Islet Transplant Performance by Collagen 1 Gel by Anna French, Jennifer Hollister-Lock, Brooke A. Sullivan, Eline Stas, Albert J. Hwa, Gordon C. Weir and Susan Bonner-Weir in Cell Transplantation
Supplemental Material
sj-tif-3-cll-10.1177_09636897241283728 – Supplemental material for Enhancement of Subcutaneous Islet Transplant Performance by Collagen 1 Gel
Supplemental material, sj-tif-3-cll-10.1177_09636897241283728 for Enhancement of Subcutaneous Islet Transplant Performance by Collagen 1 Gel by Anna French, Jennifer Hollister-Lock, Brooke A. Sullivan, Eline Stas, Albert J. Hwa, Gordon C. Weir and Susan Bonner-Weir in Cell Transplantation
Supplemental Material
sj-tif-4-cll-10.1177_09636897241283728 – Supplemental material for Enhancement of Subcutaneous Islet Transplant Performance by Collagen 1 Gel
Supplemental material, sj-tif-4-cll-10.1177_09636897241283728 for Enhancement of Subcutaneous Islet Transplant Performance by Collagen 1 Gel by Anna French, Jennifer Hollister-Lock, Brooke A. Sullivan, Eline Stas, Albert J. Hwa, Gordon C. Weir and Susan Bonner-Weir in Cell Transplantation
Footnotes
Author Contributions
GCW and AJH conceived the project. JH-L, BAS, AF, ES, and SB-W collected data. GCW and SB-W wrote the manuscript. All authors edited and approved the manuscript. SB-W is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the accuracy of the data analysis.
Availability of Data and Material
All data are available from the corresponding author on request. Most relevant data are already included.
Ethical Approval
All procedures with the rodents were approved by the Joslin Institutional Animal Care and Use Committee.
Statement of Human and Animal Rights
This article does not contain any studies with human subjects. All procedures with the rodents were approved by the Joslin Institutional Animal Care and Use Committee.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Support for this study was provided by JDRF (2-SRA-2021-1082-S-B [SB-W]), the NIH-funded Joslin Diabetes Research Center (PO DK036836), the Diabetes Research and Wellness Foundation, and an important group of private donors.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
