Abstract
Introduction
Limited evidence exists on health system characteristics associated with initial and long-term prescribing of opioids and nonsteroidal anti-inflammatory drugs (NSAIDs) following total hip and knee arthroplasty (THA/TKA), and if these characteristics differ among individuals based on preoperative NSAID exposure. We identified orthopedic surgeon opioid prescribing practices, hospital characteristics, and regional factors associated with initial and long-term prescribing of opioids and NSAIDs among older adults receiving THA/TKA.
Materials and Methods
This observational study included opioid-naïve Medicare beneficiaries aged ≥65 years receiving elective THA/TKA between January 1, 2014 and July 4, 2017. We examined initial (days 1-30 following THA/TKA) and long-term (days 90-180) opioid or NSAID prescribing, stratified by preoperative NSAID exposure. Risk ratios (RRs) for the associations between 10 health system characteristics and case-mix adjusted outcomes were estimated using multivariable Poisson regression models.
Results
The study population included 23,351 NSAID-naïve and 10,127 NSAID-prevalent individuals. Increases in standardized measures of orthopedic surgeon opioid prescribing generally decreased the risk of initial NSAID prescribing but increased the risk of long-term opioid prescribing. For example, among NSAID-naïve individuals, the RRs (95% confidence intervals [CIs]) for initial NSAID prescribing were 0.95 (0.93-0.97) for 1-2 orthopedic surgeon opioid prescriptions per THA/TKA procedure, 0.94 (0.92-0.97) for 3-4 prescriptions per procedure, and 0.91 (0.89-0.93) for 5+ opioid prescriptions per procedure (reference: <1 opioid prescription per procedure), while the RRs (95% CIs) for long-term opioid prescribing were 1.06 (1.04-1.08), 1.08 (1.06-1.11), and 1.13 (1.11-1.16), respectively. Variation in postoperative analgesic prescribing was observed across U.S. regions. For example, among NSAID-naïve individuals, the RR (95% CIs) for initial opioid prescribing were 0.98 (0.96-1.00) for Region 2 (New York), 1.09 (1.07-1.11) for Region 3 (Philadelphia), 1.07 (1.05-1.10) for Region 4 (Atlanta), 1.03 (1.01-1.05) for Region 5 (Chicago), 1.16 (1.13-1.18) for Region 6 (Dallas), 1.10 (1.08-1.12) for Region 7 (Kansas City), 1.09 (1.06-1.12) for Region 8 (Denver), 1.09 (1.07-1.12) for Region 9 (San Francisco), and 1.11 (1.08-1.13) for Region 10 (Seattle) (reference: Region 1 [Boston]). Hospital characteristics were not meaningfully associated with postoperative analgesic prescribing. The relationships between health system characteristics and postoperative analgesic prescribing were similar for NSAID-naïve and NSAID-prevalent participants.
Discussion
Future efforts aiming to improve the use of multimodal analgesia through increased NSAID prescribing and reduced long-term opioid prescribing following THA/TKA could consider targeting orthopedic surgeons with higher standardized opioid prescribing measures.
Conclusions
Orthopedic surgeon opioid prescribing measures and U.S. region were the greatest health system level predictors of initial, and long-term, prescribing of opioids and prescription NSAIDs among older Medicare beneficiaries following THA/TKA. These results can inform future studies that examine why variation in analgesic prescribing exists across geographic regions and levels of orthopedic surgeon opioid prescribing.
Introduction
Analgesic prescribing has changed dramatically in the last decade, particularly for postoperative pain management following high-volume procedures such as total hip and knee arthroplasty (THA/TKA). Prescribing patterns have shifted to reduce opioid exposure and increase the use of multimodal analgesia, where a variety of analgesics are incorporated into treatment regimens. Older adults have an increased risk of serious adverse events related to opioids (e.g., hospitalizations, falls and fracture), but they are often susceptible to medication-related adverse effects or have contraindications to therapy with nonopioid analgesics, particularly nonsteroidal anti-inflammatory drugs (NSAIDs).1,2 Evidence-based guidelines do not make specific recommendations on which opioid and nonopioid combinations are preferred for older adults following THA/TKA.3-8 This is likely due to a dearth of information on postoperative analgesic prescribing and use post-THA/TKA as well as the factors that influence analgesic prescribing practices. This foundational knowledge is essential to inform future work examining which combinations of multimodal analgesia are safest and most effective among older adults receiving THA/TKA. Optimizing postoperative pain management is especially important for older adults following THA/TKA because these procedures often result in severe pain, which may interfere with functional recovery if treated inadequately.
Further investigation is needed to understand how health system characteristics, such as orthopedic surgeon prescribing patterns, hospital characteristics, and regional factors impact postoperative analgesic prescribing following THA/TKA. Prior observational studies have examined surgeries that are heterogeneous in their pain etiology and course of recovery,9-14 have considered younger, commercially-insured populations,10-13,15-17 or have focused on patient-level characteristics (e.g., use of benzodiazepines, back pain) associated with postoperative opioid use after THA/TKA.14,15,17-20 Understanding the relationships between health system level factors and postoperative analgesic prescribing following THA/TKA can guide future interventions aiming to improve postoperative pain management. For example, if patients treated by orthopedic surgeons who prescribe opioids to a greater extent have an increased risk of long-term opioid use, then interventions aiming to reduce long-term opioid use and dependence could potentially target orthopedic surgeons with such opioid prescribing patterns. Additionally, prior literature only considered use of opioids before THA/TKA and has not examined how preoperative analgesia with nonopioids impacts postoperative pain management.15,17,21,22 Examining individuals separately based on exposure to preoperative nonopioid analgesics could reveal distinct patterns of postoperative analgesic prescribing that could inform future research efforts.
The objective of this study was to identify health system characteristics, including orthopedic surgeon, hospital, and regional characteristics, associated with initial and long-term prescribing of opioids and prescription NSAIDs among Medicare beneficiaries ≥65 years old receiving elective THA/TKA. We chose to examine NSAIDs specifically as they are the most commonly used prescription nonopioid analgesic following THA/TKA.23-26 Additionally, we examined how the use of prescription NSAIDs prior to THA/TKA impacted the relationships between health system characteristics and postoperative analgesic prescribing. We hypothesized that the risk of initial and long-term opioid prescribing would be higher among patients with orthopedic surgeons who prescribe opioids to a greater extent, while the risk of initial and long-term prescription NSAID prescribing would be lower.
Materials and Methods
Study Design and Data Sources
This observational study linked data from Medicare fee-for-service (i.e., Traditional Medicare) insurance claims to several publicly available data sources. Medicare claims data included the Master Beneficiary Summary File (MBSF), which included beneficiary plan enrollment information, demographics, and information on conditions through the Chronic Conditions Warehouse; Medicare Provider Analysis and Review (MedPAR) claims for inpatient (i.e., hospital) services; and a 20% random sample of Medicare Part B claims for outpatient services and Part D claims for prescription drug dispensings. The validated Residential History File, which leverages Medicare claims data, provided information on the timing and location of healthcare services. 27 Publicly available data included the Medicare Provider Payment and Utilization Data, which was used to construct standardized measures of orthopedic surgeon characteristics; the Dartmouth Health Atlas Hospital Tracking File for hospital characteristics; and the Prescription Drug Abuse Policy System (PDAPS), which contained data on state policies for prescription drug monitoring program (PDMP) utilization.28,29 All data sources included data from January 1, 2013 to December 31, 2017 to allow for a 12-month covariate ascertainment period prior to THA/TKA procedures. This study was approved by the Brown University Institutional Review Board (Protocol Number: 2010002823). The need for informed consent was waived. The STROBE checklist was used for this cohort study.
Study Population
The study population included community-dwelling Medicare Fee-for-Service beneficiaries aged ≥65 years on the date of elective THA/TKA procedure occurring between January 1, 2014 to July 4, 2017. THA/TKA procedures were identified using Medicare Severity Diagnosis Related Group codes 469 and 470 on inpatient claims. Eligible participants had 12 months of continuous Part A and B coverage and 6 months of continuous Part D coverage prior to THA/TKA. We restricted the study population to individuals without a prescription for opioids in the 6 months prior to THA/TKA (i.e., opioid naïve) because we aimed to estimate the risk of first-time opioid exposure and subsequent risk of long-term opioid use or dependence. A 6-month time frame without opioid use is commonly used to identify opioid naïve individuals.30-34 Individuals were excluded if they were enrolled in Medicare Advantage or used hospice in the 12 months prior to THA/TKA, were admitted for fracture or non-arthroplasty procedures, or were missing data on orthopedic surgeon characteristics or other covariates (eTable 1). For individuals with multiple procedures, we sampled the first THA/TKA during the study period.
Initial and Long-Term Analgesic Prescribing
The outcomes of interest were initial and long-term prescribing of opioids or prescription NSAIDs following THA/TKA. Initial prescribing was defined as a claim for an opioid or NSAID during days 1-30 following THA/TKA, while claims during days 90-180 after THA/TKA were considered long-term prescribing. Systemic opioids and NSAIDs of interest are listed in eTable 2. Claims for analgesics were captured regardless of prescriber type (e.g., orthopedic surgeon, advanced practice professional).
Follow-Up
Eligible individuals were followed from the date of the THA/TKA procedure until disenrollment from Medicare Parts A, B, or D; enrollment in Medicare Advantage; administrative end of follow-up (180 days post-THA/TKA); or death, whichever occurred first.
Health System Characteristics
We ascertained health system characteristics related to the orthopedic surgeon, hospital, and region. We linked Medicare Part B claims from the THA/TKA to public Part B and D Provider Payment and Utilization Data and created several measures related to orthopedic surgeon characteristics (eTable 3). These included annual THA/TKA volume, opioid prescription volume, proportion of prescriptions for opioids relative to all medications (i.e., “opioid share”), ratio of opioid prescriptions to beneficiaries prescribed opioids (i.e., “refill ratio”), and opioid prescriptions per THA/TKA. Hospital characteristics, including critical access designation, total beds, and academic affiliation were obtained from the Dartmouth Atlas Hospital Tracking File. 28 The PDAPS was used to classify the rigor of the PDMP program. 29 Categories included: (1) high rigor: prescribers were required to check the PDMP database each time prior to prescribing opioids to all patients or chronic pain patients; (2) low rigor: there were no requirements for checking the PDMP or checking the PDMP was only required prior to prescribing opioids for the first time; and (3) no PDMP was in place. Finally, region was categorized based on the Department of Health and Human Services regions.
Patient Characteristics
We ascertained 127 person-level covariates for case-mix adjustment (eTable 4). Medicare dual eligibility, demographics (age, sex, race/ethnicity 35 ), and comorbidities (e.g., heart failure, chronic kidney disease) were derived from the Medicare Beneficiary Summary File and Chronic Conditions Data Warehouse. Preoperative medication use in the 180 days prior to THA/TKA was ascertained from Part D claims. MedPAR and Medicare Part B claims were used to estimate healthcare utilization in the 12 months prior to THA/TKA (e.g., hospitalizations, annual wellness visit), comorbidity score, 36 and a claims-based frailty index. 37
Statistical Analysis
We used a two-step process to understand how health system characteristics were associated with the primary outcomes. First, we fit logistic regression models with 127 person-level covariates as independent variables to obtain case-mix adjusted primary outcomes. Second, we estimated risk ratios (RRs) for the potential relationships between health system characteristics and case-mix adjusted outcomes using modified Poisson regression models with robust standard errors.38,39 Ten orthopedic surgeon, hospital, and regional characteristics were assessed while adjusting for the other health system characteristics. Since prior NSAID use before THA/TKA likely influences medications received in the postoperative period, all analyses were stratified based on the presence of a claim for NSAIDs in the 6 months prior to surgery. Individuals were categorized as NSAID-naïve or NSAID-prevalent.
Intermediate Analyses
Although not the primary focus of this analysis, we reported the results from our intermediate analyses for completeness. These included patterns of analgesic prescribing following THA/TKA stratified by preoperative prescription NSAID exposure, including opioid or NSAID prescribing during days 1-180 following THA/TKA; the top 10 combinations of treatments within 30 days following THA/TKA; prescriptions for short-acting or long-acting opioids; opioid dosage and days’ supply; and sustained opioid use based on proportion of days covered (PDC) ≥80% in the 90 and 180 days following THA/TKA. 40 Opioid dosages were standardized using the Centers for Disease Control and Prevention conversion factors for morphine milligram equivalents (MME) per day. 41
We also fit modified Poisson regression models with robust standard errors to estimate risk ratios for the potential relationships between 127 person-level characteristics and the primary outcomes, without adjusting for health system characteristics.
Software
Data were analyzed using SAS, version 9.4 (SAS Institute, Inc., Cary, NC) and Stata, version 16 (StataCorp LLC, College Station, TX).
Results
Study Population
Demographic and Clinical Characteristics of Medicare Beneficiaries following total Knee and Hip Arthroplasty Stratified by Pre-surgery Prescription NSAID Use, 2014-2017.

aCombined Comorbidity Score values <0 possible due to component value −1 in algorithm for HIV/AIDS and Hypertension. 36
bMeasured using the Claims-based Frailty Index and categorized as: <0.15 (robust), 0.15-0.24 (prefrail), and ≥0.25 (mild or moderately-to-severely frail). 37
Initial and Long-Term Analgesic Prescribing Following THA/TKA
Observed Postoperative Analgesic Prescribing Outcomes Among Medicare Beneficiaries With Total Hip and Knee Arthroplasty Stratified by Pre-surgery NSAID Use, 2014-2017.
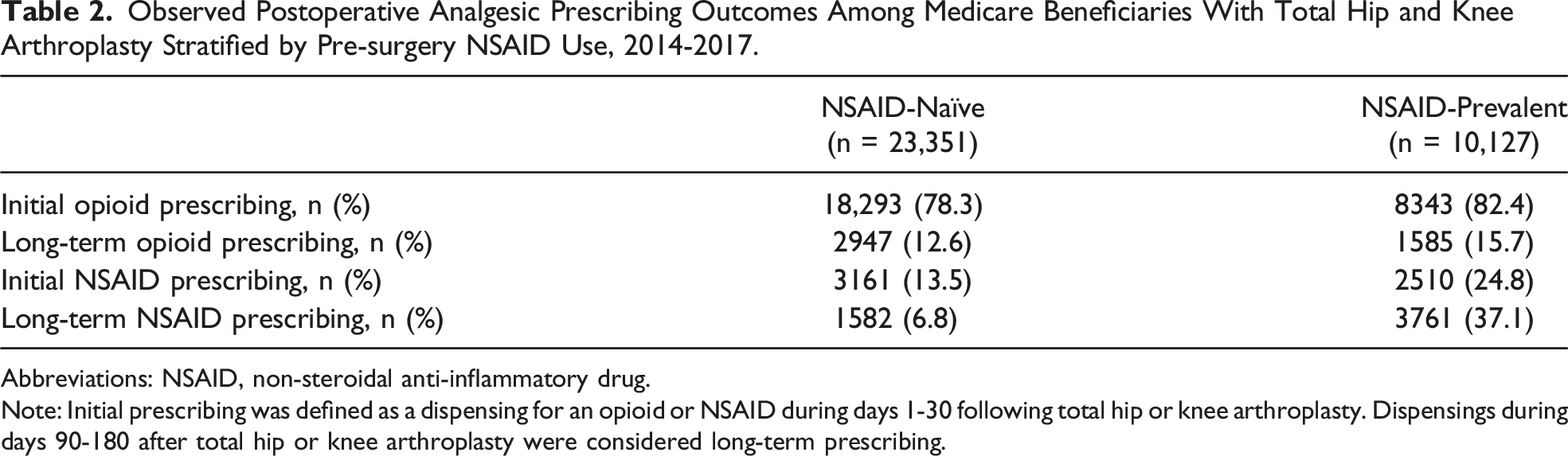
Abbreviations: NSAID, non-steroidal anti-inflammatory drug.
Note: Initial prescribing was defined as a dispensing for an opioid or NSAID during days 1-30 following total hip or knee arthroplasty. Dispensings during days 90-180 after total hip or knee arthroplasty were considered long-term prescribing.
Intermediate Analyses
The mean MME/day, opioid days’ supply, proportion of individuals with opioid PDC ≥80%, prescriptions for long-acting opioids, and the top 10 most common initial postoperative analgesic treatment patterns were similar for NSAID-naïve and NSAID-prevalent participants (eTable 5). Risk ratios for the associations between person-level characteristics and primary outcomes without adjustment for health system characteristics are presented in eTables 6-7.
Health System Characteristics Associated with Initial and Long-Term Opioid Prescribing
Adjusted associations between health system level characteristics and opioid prescribing. Notes: Presents risk ratios for the potential relationships between health system characteristics and case-mix adjusted outcomes, which accounted for 127 person-level covariates. Reference categories are omitted and can be found in eTable 8. Abbreviations: NSAID, non-steroidal anti-inflammatory drug; THA/TKA, total hip and knee arthroplasty; Rx, prescription; PDMP, Prescription Drug Monitoring Program.
An increase in the opioid share was associated with an increased risk of long-term opioid prescribing among NSAID-prevalent participants only (RR [95% CIs] 1.05 [1.01-1.08] for opioid share 25%–49%, 1.07 [1.03-1.11] for opioid share 50%–74%, and 1.05 [1.01-1.10] for opioid share 75%–100% [reference: opioid share 0%–24%]) (eTable 8).
An increase in the number of orthopedic surgeon opioid prescriptions per THA/TKA procedure generally decreased the risk of initial opioid prescribing and increased the risk for long-term opioid prescribing among NSAID-naïve and NSAID-prevalent beneficiaries (Figure 1, eTable 8). For NSAID-naïve individuals, the RRs (95% CIs) for initial opioid prescribing were 0.97 (0.96-0.98) for 1-2 opioid prescriptions per procedure, 0.97 (0.96-0.98) for 3-4 prescriptions per procedure, and 0.95 (0.93-0.96) for 5+ opioid prescriptions per procedure (reference: <1 opioid prescription per procedure). The RRs (95% CIs) for long-term opioid prescribing were 1.06 (1.04-1.08), 1.08 (1.06-1.11), and 1.13 (1.11-1.16), respectively. For NSAID-prevalent individuals, the RRs (95% CIs) for initial opioid prescribing were 0.98 (0.96-0.99) for 1-2 opioid prescriptions per procedure, 0.98 (0.96-1.00) for 3-4 prescriptions per procedure, and 0.95 (0.93-0.97) for 5+ opioid prescriptions per procedure (reference: <1 opioid prescription per procedure). The RRs (95% CIs) for long-term opioid prescribing were 1.02 (0.99-1.05), 1.06 (1.02-1.09), and 1.08 (1.05-1.12) respectively.
Health System Characteristics Associated with Initial and Long-Term NSAID Prescribing
Adjusted associations between health system level characteristics and NSAID prescribing. Notes: Presents risk ratios for the potential relationships between health system characteristics and case-mix adjusted outcomes, which accounted for 127 person-level covariates. Reference categories are omitted and can be found in eTable 9. Abbreviations: NSAID, non-steroidal anti-inflammatory drug; THA/TKA, total hip and knee arthroplasty; Rx, prescription; PDMP, Prescription Drug Monitoring Program.
Higher opioid prescription volume was generally associated with an increased risk of initial, but not long-term NSAID prescribing (Figure 2, eTable 9). The RRs (95% CIs) for initial NSAID prescribing among NSAID-naïve individuals were 1.02 (1.00-1.03) for 100-199 opioid prescriptions, 1.04 (1.02-1.06) for 200-299 opioid prescriptions, 1.05 (1.02-1.09) for 300-399 opioid prescriptions, and 1.05 (1.01-1.09) for 400+ opioid prescriptions (reference: 11-99 opioid prescriptions). The RRs (95% CIs) for initial NSAID prescribing were 1.03 (1.01-1.04), 1.04 (1.02-1.06), 1.05 (1.02-1.09), and 1.06 (1.01-1.11), respectively, among NSAID-prevalent individuals.
Increases in the opioid share and refill ratio were associated with a slightly decreased risk of initial NSAID prescribing, but no statistically significant associations were observed for long-term NSAID prescribing (Figure 2, eTable 9). For NSAID-naïve individuals, the RRs (95% CIs) for initial NSAID prescribing were 0.96 (0.93-0.98) for 25%–49% opioid share, 0.96 (0.94-0.99) for 50%–74%, and 0.97 (0.94-1.00) for 75%–100% (reference: 0%–24%). The RRs (95% CIs) were 0.96 (0.95-0.98) for an opioid refill ratio of 2-2.9 and 0.93 (0.90-0.97) for 3+ (reference: 1-1.9). Among NSAID-prevalent individuals, the RRs (95% CIs) for initial NSAID prescribing were 0.95 (0.93-0.98) for 25%–49% opioid share, 0.96 (0.93-0.98) for 50%–74%, and 0.96 (0.93-0.99) for 75%–100% (reference: 0%–24%). The RRs (95% CIs) were 0.97 (0.95-0.98) for an opioid refill ratio of 2-2.9 and 0.97 (0.93-1.02) for 3+ (reference: 1-1.9).
In general, increases in the number of orthopedic surgeon opioid prescriptions per THA/TKA procedure were associated with a decreased risk of initial NSAID prescribing and increased risk of long-term NSAID prescribing (Figure 2, eTable 9). For NSAID-naïve individuals, the RRs (95% CIs) for initial NSAID prescribing were 0.95 (0.93-0.97) for 1-2 opioid prescriptions per procedure, 0.94 (0.92-0.97) for 3-4 prescriptions per procedure, and 0.91 (0.89-0.93) for 5+ opioid prescriptions per procedure (reference: <1 opioid prescription per procedure). The RRs (95% CIs) for long-term NSAID prescribing were 1.01 (1.00-1.03), 1.04 (1.02-1.07), and 1.06 (1.03-1.08), respectively. For NSAID-prevalent individuals, the RRs (95% CIs) for initial NSAID prescribing were 0.99 (0.97-1.01) for 1-2 opioid prescriptions per procedure, 0.97 (0.95-1.00) for 3-4 prescriptions per procedure, and 0.96 (0.93-0.98) for 5+ opioid prescriptions per procedure (reference: <1 opioid prescription per procedure). The RRs (95% CIs) for long-term NSAID prescribing were 1.04 (1.02-1.06), 1.04 (1.02-1.07), and 1.06 (1.04-1.09), respectively.
Discussion
In this large observational study of opioid-naïve Medicare beneficiaries receiving THA/TKA, we found that approximately 30% of individuals used prescription NSAIDs prior to THA/TKA and NSAID-prevalent individuals were more likely to have prescriptions for postoperative opioids and NSAIDs. Orthopedic surgeon opioid prescribing measures and U.S. region were the greatest predictors of initial, and long-term, opioid and NSAID prescribing following THA/TKA. We also found that the relationships between most health system characteristics and postoperative analgesic prescribing were similar among NSAID-naïve and NSAID-prevalent participants.
Clinical guidance recommends prescribing the lowest dose of opioids and the fewest number of opioid pills that is clinically effective following THA/TKA to help prevent opioid-related adverse events and diversion of unused pills.42,43 Multimodal analgesia with different oral analgesic classes is also recommended to provide pain relief while limiting the use of opioids. Routine use of NSAIDs is recommended for patients without contraindications following THA/TKA, although risk factors for adverse events, such as history of bleeding, gastroduodenal ulcer, cardiovascular morbidity, and kidney and liver disease should be taken into consideration prior to prescribing postoperative NSAIDs. 5 Multiple studies have focused on person level characteristics associated with postoperative analgesic use,14,15,17-20,44-48 reporting that factors like benzodiazepine use, TKA (versus THA), and tobacco use were predictors of long-term opioid use. Very few studies have considered the relationships between health system level characteristics and postoperative analgesic prescribing following THA/TKA. We demonstrated that several measures of orthopedic surgeon opioid prescribing had consistent associations with initial and long-term opioid and NSAID prescribing following THA/TKA. Variation in postoperative opioid and NSAID prescribing was also observed across Department of Health and Human Services regions. However, there were no meaningful relationships between hospital characteristics and postoperative analgesic prescribing. These results can inform future studies that examine why variation in analgesic prescribing exists across geographic regions and levels of orthopedic surgeon opioid prescribing.
Prior literature suggests that high-intensity opioid prescribers provide higher MMEs, greater days’ supply, and are more likely to prescribe opioids long-term vs low-intensity prescribers.49,50 We found that participants who had orthopedic surgeons with higher standardized opioid prescribing measures (opioid share, opioid refill ratio, opioid prescriptions per THA/TKA procedure) were generally less likely to initiate NSAIDs, but were at an increased risk of long-term opioid prescribing following THA/TKA. It may be beneficial for future efforts aiming to improve the use of multimodal analgesia through increased NSAID prescribing and reduce long-term opioid prescribing following THA/TKA to target orthopedic surgeons with higher standardized opioid prescribing measures. Creating standardized evidence-based guidelines and institutional protocols are potential strategies to combat variability in postoperative prescribing and minimize the risk of unintended long-term analgesic use.44,51-53
Notably, we found that standardized measures of orthopedic surgeon opioid prescribing created using publicly available provider-level data had opposite associations with postoperative analgesic prescribing compared to orthopedic surgeon opioid prescription volume. For example, higher orthopedic surgeon opioid prescription volume was associated with a lower risk of long-term opioid prescribing, but a greater number of opioid prescriptions per THA/TKA procedure was associated with a higher risk of long-term opioid prescribing for both NSAID-naïve and NSAID-prevalent participants. These results suggest that it is important for future work to consider orthopedic surgeon opioid prescribing in the context of their case load and overall medication prescribing, rather than solely considering the number of opioid prescriptions.
Limitations
There are several potential limitations to this analysis. First, our results may not generalize well to older adults without Medicare Fee-for-Service (i.e., Medicare Advantage), those who were not opioid naïve at the time of THA/TKA, or to those residing outside of the United States.
Second, due to the nature of our prescription drug dispensing data, we could not capture medication administrations (i.e., patterns of medication use). Additionally, we could not identify use of non-prescription (over-the-counter) analgesics. Thus, we could not ascertain use of non-prescription NSAIDs (e.g., ibuprofen, naproxen) or other analgesics that are used as part of multimodal analgesia (e.g., acetaminophen). However, to our knowledge, there is currently no data source with information on the use of non-prescription medications that could have been applied to this national population of Medicare beneficiaries to fill this gap in knowledge.
Third, potentially influential predictors of opioid and NSAID prescribing following THA/TKA, such as beneficiary preoperative and postoperative pain scores, hospital prescribing protocols, and enhanced recovery pathways were not available in our data. Thus, we could not assess the relationships between these potentially important predictors and postoperative analgesic prescribing.
Finally, standardized opioid prescribing measures calculated for orthopedic surgeons should be interpreted in light of several considerations. Since Medicare Part B and D Provider Utilization and Payment data relate to the procedures performed and prescriptions written by each provider in aggregate, these measures do not consider providers’ case mix, including patient health status or prior medication use, which may impact postoperative prescribing practices. Additionally, although we attempted to characterize orthopedic surgeon prescribing, it is possible that another clinician may have been responsible for managing postoperative pain (e.g., advanced practitioner working with the surgeon). In this case, our inferences may be incorrect when attributing postoperative prescribing to the surgeon. However, if the Part D dispensings were captured under the surgeon’s National Provider Identifier through collaborative agreements, our approach would still be valid. Finally, we assumed that the orthopedic surgeon would be responsible for prescribing analgesics in most THA/TKAs performed each year. In our study, 58.6% of beneficiaries overall had a prescription written by an orthopedic surgeon during days 1-89 after THA/TKA, and 70.6% of beneficiaries had a corresponding prescription when restricting to those with a Part D claim. Our finding that greater opioid prescribing intensity was significantly associated with a higher risk of long-term opioid prescribing and lower risk of initial NSAID prescribing could suggest that the relationships were strong enough for people whose pain was managed by the orthopedic surgeon that the results were still significant even when applied to the whole cohort (which included participants who did not receive a prescription from the orthopedic surgeon in the initial postoperative period). Further research is needed to validate these standardized opioid prescribing measures, understand the impact of orthopedic surgeon opioid prescribing intensity on clinical outcomes, examine the clinical appropriateness of opioid and NSAID prescribing, and elucidate the pathways through which these relationships operate.
Conclusion
Overall, we found that orthopedic surgeon opioid prescribing measures and U.S. region were the greatest health system level predictors of initial, and long-term, prescribing of opioids and prescription NSAIDs among older Medicare beneficiaries following THA/TKA. The relationships between health system characteristics and postoperative analgesic prescribing were similar among NSAID-naïve and NSAID-prevalent individuals. Future work aiming to improve the use of multimodal analgesia through increased NSAID prescribing and reduce long-term opioid use following THA/TKA could consider targeting orthopedic surgeons with higher standardized opioid prescribing practices.
Supplemental Material
Supplemental Material - Initial and Long-Term Prescribing of Opioids and Non-steroidal Anti-inflammatory Drugs Following Total Hip and Knee Arthroplasty
Supplemental Material for Initial and Long-Term Prescribing of Opioids and Non-steroidal Anti-inflammatory Drugs Following Total Hip and Knee Arthroplasty by Melissa R. Riester, Elliott Bosco, Francesca L. Beaudoin, Stefan Gravenstein, Andrew J. Schoenfeld, Vincent Mor, and Andrew R. Zullo in Geriatric Orthopaedic Surgery & Rehabilitation.
Footnotes
Declaration of Conflicting Interests
F.L.B. works as a Senior Medical Advisor for the Institute for Clinical and Economic Review with funds paid directly to Brown University. S.G. reports grants from Seqirus, Sanofi; and consulting or speaker fees from Sanofi, Seqirus, Merck, and the Gerontological Society of America related to vaccines or nursing home care quality. A.J.S. reports royalties from Wolters-Kluwer and Springer-Nature. V.M. is a paid consultant and Scientific Advisory Board chair to NaviHealth, Inc. Which offers post-acute care management and services to health plans and health systems. A.R.Z. reports prior grants from Sanofi paid directly to Brown University for research on the epidemiology of infections and vaccinations in older nursing home residents as well as respiratory syncytial virus in infants.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the National Institute on Aging [R01AG065722 and R01AG077620 to A.R.Z]. Some authors are VA employees (S.G., V.M., A.R.Z.). The content and views expressed in this article are those of the authors and do not necessarily reflect the position or policy of the Department of Veterans Affairs or the United States government.
Ethical Statement
Data Availability Statement
The data that support the findings of this study are available from the Centers for Medicare and Medicaid Services (CMS) but restrictions apply to the availability of these data, which were used under data use agreements for the current study, and so are not publicly available. However, other researchers can establish their own data use agreement and obtain the datasets employed through the Research Data Assistance Center (ResDAC), a CMS contractor that provides free assistance to researchers interested in CMS data.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
