Abstract
Background
There are multiple recommended protocols for Vitamin D (VitD) supplementation in elderly; however, only a few studies achieved to examine the role of VitD supplements before intertrochanteric fracture surgery on mortality and complications after surgery.
Methods
This single-center block-randomized double-blinded trial was conducted on 80 patients with intertrochanteric fractures and a sufficient level of 25 (OH) VitD. The intervention group received an intramuscular 300,000 IU VitD ampule before surgery. The primary outcome was a 6-month mortality rate, and the secondary outcomes were 1- and 2-year mortality rates and Harris Hip Score (HHS) in 6, 12, and 24 months after surgery. Chi-square, t-test, repeated measure ANOVA, and Cox regression survival model was used for statistical analysis.
Results
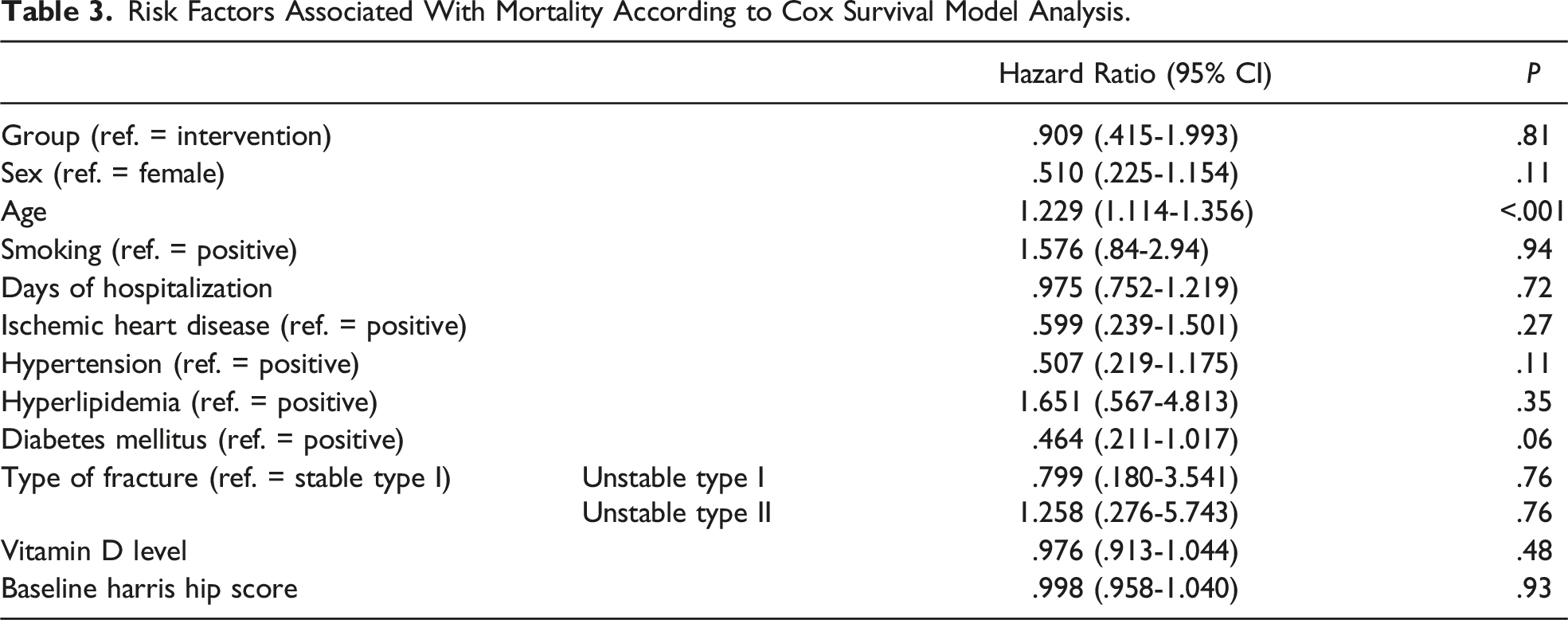
40 patients were allocated to each group. Demographic, clinical characteristics, and preoperative evaluations were not significantly different between the groups. Mortality rate 6-month after the surgery was 7.5% and 10% for the intervention and placebo groups respectively (P value = .71), 15% and 12.5% at 1-year (P value = .83), and 25% and 27.5% at 2-year (P value = .98). Based on the Cox regression model, only age was significantly associated with mortality (HR = 1.229, P value <.001). Significant HHS changes from baseline through 24 months after surgery were observed within both groups; however, mean differences were not significantly different between groups.
Conclusions
A single preoperative 300,000 IU VitD did not significantly impact 2-year survival and HHS in patients with intertrochanteric fractures and sufficient serum VitD level.
Introduction
Hip fractures are more likely to occur in the elderly by reducing bone mineral density and an increase in the rate of falls. 1 The worldwide number of hip fractures is estimated to reach 6.26 million by the year 2050. 2 Hip fractures are the most crucial type of osteoporotic fracture, 3 with an adverse impact on physical, mental, psychosocial, and other aspects of a patient’s life. 4 The 1-year mortality after hip fracture ranges between 20 and 40% and is an age-related phenomenon.5-10 Intertrochanteric fractures (ITFs) are one of the most common types of hip fractures and include approximately 50% of all hip fractures. 11 ITFs usually occur in a patient with osteoporosis, a history of falls, impaired vision, malnutrition, and female gender. 1 Approximately 50% of women and 20% of men over 50 years will have an osteoporotic fracture during the rest of their lifetimes.12,13 The incidence of ITFs has increased as life expectancy, and the older population increased during the last years. 14 A mortality rate of 10.2% after 1 year of ITFs surgery has been reported. 15 Several studies have found that mortality after intertrochanteric fracture is associated with the patient’s age, type of fracture, delay in performing the surgery, and treatment type.16,17
A sufficient level of serum Vitamin D (VitD) is critical for bone mineralization, calcium homeostasis, and proper bone and muscle functioning, and performs an essential role in preventing fractures in the old age population.18,19 It was revealed that hypovitaminosis D at hospital visits in patients with hip fractures is associated with delirium, a new hip fracture, and medical readmissions, but not orthopedic complications. 20 Although VitD supplements are recommended for older patients to prevent fractures, especially menopausal women, 21 the association between the use of VitD supplements and fracture risk is still controversial.21,22 According to the high prevalence of VitD deficiency worldwide, the use of 50,000 IU VitD at admission to all elderly patients with hip fractures has been recommended in the textbook. 23 Besides its preventive role for fracture, it was shown that VitD deficiency may result in pain, proximal muscle weakness, dynamic imbalance, and decreased gait speed, which has an impact on mobilization during the postoperative rehabilitation periods, hip functionality, and postoperative mortality.24-26 Therefore, VitD level may be an important modifiable factor that may reduce the associated postoperative complications of hip fractures such as the severity of postoperative disability, falling, and mortality. Thus, providing a loading dose at the time of surgery may provide prominent outcomes in the near future.
However, Seng et al have shown that serum VitD level was not related to postoperative outcomes and the 1-year mortality rate of ITF surgery in the elderly. 27 On the other hand, there are multiple recommended protocols for VitD supplementation; however, only a few studies have examined the role of a high-loading dose of VitD supplements on the outcomes of ITF patients.25-28 As more patients are being diagnosed as VitD deficient and received appropriate treatment, we will have more elderly with VitD sufficiency; therefore, adjuvant supplementation in patients with sufficient levels of VitD has to be evaluated. Therefore, this study aimed to evaluate the effects of injecting an adjuvant 300,000 IU VitD ampule before ITF surgery in elderly patients with a sufficient serum VitD on mortality rate and Harris Hip Score (HHS) during the 2 years after the surgery.
Materials and Methods
Trial Design
The study was conducted as a randomised double-blinded placebo-controlled parallel clinical trial after receiving ethical approval from the institutional review board and registering in the clinical trials databases. All procedures performed in this study complied with the Consolidated Standards of Reporting Trials (CONSORT) 2010 guidelines (Figure 1),
29
and in accordance with the ethical standards of institutional and national research committees and 1964 Helsinki declarations and their later amendments. Written informed consent was obtained from all of the participants before the enrollment (IRCT20180729040636N2). Consort flow diagram. Eligibility of 158 patients were assessed at the baseline and 80 patients were enrolled and randomly allocated to the intervention group (n = 40), who received a 300,000 IU vitamin D ampule before intertrochanteric fractures and placebo group (n = 40). During the 2-year follow-up, no patients were excluded; however, 10%, 12.5%, and 27.5% of them from the intervention group and 7.5%, 15%, and 25% of them form them placebo group were expired after 6 months, 1, and 2 years after the surgery respectively. According to intention-to-treat principle, all enrolled patients were analyzed at the end of the trial.
Participants and Eligibility Criteria
The participants of this trial were selected from our trauma referrals between October 2019 and January 2020. Inclusion criteria were patients aged 50-80 years old with ITF who were willing to participate in this study and had serum 25 (OH) VitD level of 30-80 nanograms per milliliter (ng/ml), able to provide informed consent, either directly or via the “person responsible,” and deemed suitable by the treating medical team. Patients with a severe systemic illness involving the musculoskeletal system, systemic diseases related to the VitD and calcium supplements such as hypercalcemia (serum calcium >2.65 mmol/L), a history of renal stones, thyrotoxicosis, and Paget disease, unable to read, speak, or write, metastatic cancers or history of chemotherapy/radiotherapy, debilitating underlying neurologic diseases, history of adverse reactions to injection of VitD, received 300,000 IU VitD ampule in the last 30 days, simultaneous fracture at other parts of the body, and participation in other studies were not included.
Patients’ demographic data and baseline characteristics were documented in a checklist. Each comorbid disease was considered positive based on previously prescribed medications, history of blood tests, and visiting specialists for that reason. ITF was diagnosed and categorized according to preoperative anterior-posterior and lateral X-ray radiography of the hip and femur. Patients were followed-up every 3 months in the first postoperative year and every 6 months in the second year. Patients were visited either in the clinic or by the principal investigator who had a home visit. All pre and postoperative assessments were conducted by both the principal investigator and his assistant.
Intervention
The same surgical protocol was performed for all patients in both groups. The intervention group received a 300,000 IU VitD ampule (Osvah Pharmaceutical Co. IRAN) intramuscularly at the surgery’s due date. Considering oral VitD compliance due to difficult swallowing and gestational side effects is low and considering our study population consists of elder patients who may receive rather oral pharmacological treatments, using oral supplements of VitD may not be efficient enough to rely on for results, hence we decided to administer 300,000 IU VitD via bolus injection. The placebo group received normal saline from an exact ampule with the same color, odor, and volume (designed and manufactured in the Osvah Pharmaceutical Co. IRAN).
Outcomes
The primary outcome was a 6-month mortality rate after ITF surgery, which was retrieved by asking on the telephone and searching the national identification number of each patient on our national registration regulatory.
The secondary outcomes were 1- and 2-year mortality rates and HHS in 6, 12, and 24 months after the surgery. The HHS is a physician-completed questionnaire that consists of subscales for the severity of pain (1 item, 0-44 points), level of function (7 items, 0-47 points), limb deformity (1 item, 0-4 points), and hip range of motion (2 items, 0-5 points). Scores range from 0 (worse disability) to 100 (less disability). Baseline HHS was calculated based on examination of our patients in our clinic when they were healthy.
Sample Size
Considering alpha error (type 1) of .05, beta error (type 2) of .2, and based on previously published mortality rate after ITF surgery, a total of 80 patients were calculated as a sufficient sample size.
Randomization
The patients were allocated into either the intervention or placebo group using a non-stratified block randomization method. At the beginning of the study, our expert analytics created the sequence and groups of patients with Random Allocation Software. He was blinded to the patients’ confidential information, the selection process, outcome assessments, and the surgery. The software was set to generate a sequential case number in blocks of 4 patients. The randomization mechanism and block sizes were not revealed to anyone involved in this study until the end of the trial. Then, the output was converted into a concealed opaque tamperproof envelope. The unique case number was written at the top of each envelope, and a piece of paper with either “Group A” or “Group B” written on it was put inside the envelope. All envelopes were put sequentially in a box with a locker that had one key and was kept in a safe room. Patients were enrolled and operated on according to their turn. The analytic gave the tamperproof envelope of each patient to a specific nurse on their surgery due date. She opened the envelope in a private room, read the assigned group, grabbed the specific ampule from the study drug shelf, and passed it to the principal investigator for injection. She had no access to patients’ confidential information or contact number and was not involved in the patient selection process, randomization, and pre and postoperative evaluations.
Blinding
This study was conducted using the double-blinded method. Neither the patients included in this study nor the leading surgeon, operating room staff, outcome assessors, and principal investigator of this study were aware of the participants’ assignments to either the intervention or placebo group. The active drug and its exact designed placebo were ordered confidentially to the pharmaceutical company and were sent to the non-involved pharmacist of our center. He just erase the old labels, grouped the ampules into two distinct packages and named them “A” as an active drug and “B” as a placebo, and gave each package secretly to our nurse to put them in her private room for use during the study. The allocation of active drug and placebo to their assigned group were kept confidential until the end of the trial.
Statistical Analysis
The statistical analysis was performed using IBM SPSS software 22.0 (USA). All data analysis was carried out according to a pre-established intention-to-treat analysis plan. Continuous and categorical data were presented as mean ± standard deviation and frequency (percentage), respectively. The normality of continuous data was evaluated using the Kolmogorov-Smirnov test. Categorical data were compared between groups using the chi-square test. Bonferroni correction was used and reported where appropriate. Fisher’s exact test was used when the minimum expected count was less than five. T-test was used to compare each numerical variable between groups. Mann-Whitney U test was used where appropriate. Repeated measure ANOVA was used to compare means between different time points within a group. The Cox regression model was performed for survival analysis. A hazard ratio (HR) with a 95% confidence interval (CI) is reported. P-value (2-tailed) <.05 was considered as a significant level. For nonsignificant results, the GPower 3.1 software was used to calculate the power of the study.
Results
Demographics, Comorbidities, and Baseline Evaluations Between Study Groups.

Mortality Rates Six Months, One Year, and Two Years After Intertrochanteric Fracture Surgery in Each Study Group.

Risk Factors Associated With Mortality According to Cox Survival Model Analysis.
Harris Hip Scores (HHS) at Six Months, One Year, and Two Years After Intertrochanteric Fracture Surgery in Each Study Group.

Harris Hip Score Mean Differences Between Two Groups After Comparing Two Measurements.

Discussion
The most important findings of this study are that adjuvant 300,000 IU VitD injection has no significant effect on the 2-year survival rate and the HHS during the 2 years after ITF surgery of elderly with sufficient levels of preoperative VitD. Previous studies have demonstrated that pre-operative hypovitaminosis D in osteoporotic hip fracture population is significantly decrease 2 years mortality. 30 Also, a study by Sim et al revealed that prior VitD deficiency was significantly related to poorer Parker Mobility Score (PMS) and individual domains of 36-Item Short Form Health Survey (SF36). 31 Mentioned results bring the hypothesis about the effect of VitD supplement even in patients with normal VitD, however, in their study like our presenting work paper, HHS was not related to VitD supplementation. Our findings are relatively supported by others in the subject. Lee et al. found no significant correlation between serum VitD level and mortality after hip fracture. 32 A recent large cohort study with long-term follow-up demonstrated that VitD supplement use (≥800 IU daily) did not significantly reduce the mortality rate for 5 years after hip fractures in men, unlike in women. 33 Trivedi et al. revealed that the use of 100,000 IU oral VitD every 4 months for 5 years does not significantly influence older people’s mortality. 34 In contrast, some studies discovered the positive effects of prescribing VitD and calcium supplements on survival after hip fracture. Degli Esposti et al found that the use of calcium/VitD supplements combined with anti-osteoporotic medication was related to a decrease in all-cause mortality and re-fracture during 3-year follow-up compared to osteoporosis treatment only in the patient above 50 years with osteoporosis and previous hip or vertebral fracture. 35 A study conducted by Nurmi-Lüthje et al showed an association between post-hip fracture use of VitD + Calcium supplements in males and anti-osteoporotic medications in addition to calcium and VitD in females and lower mortality rate. 36 However, most of the patients involved in that study had hypovitaminosis D before fracture. Therefore, this difference may be due to a difference in pre-fracture serum VitD level. However, similar to what Nurmi-Lüthje et al have found, preoperative VitD level was not a significant risk factor for mortality of ITFs patients. 36
The Replenishment of Vitamin D in Patients with Hip Fracture (REVITAHIP) trial has shown that a loading dose of 250,000 IU VitD in addition to a daily supplement with 800 IU VitD + 500 mg Ca was associated with reduced rates of falls and severity of the pain; however, it had no significant effect on gait velocity and refracture rate due to falls. Finally, although their findings require further evidence, they recommended that a high-loading dose of VitD within 7 days is inexpensive and could be beneficial after hip surgery.29,37 In contrast to what we have found for HHS, a meta-analysis by Zhang et al showed a correlation between hypovitaminosis D and lower HHS in patients after hip surgery. 38 In another study by Nawby et al, a positive association was found between VitD and postoperative HHS in patients with osteoarthritis and undergoing total hip replacement. 39 However, the patients studied in that article had a lower HHS before surgery, and it is known that postoperative outcome is affected by the preoperative function. Therefore, their results may simply reflect the relation between VitD level and the preoperative status of the function.
Age was a major predictor of mortality in our study. This finding is consistent with the results of previous research.16,38,40 Moreover, sex was not a significant independent predictor of mortality in multivariate analysis, which is similar to other studies.41,42 In contrast, White et al determined that men with hip fractures had a higher mortality rate than women, however, their conclusions were biased with a higher male/female ratio in the high-risk group. 43 Consequently, the association between sex and mortality rate after hip fracture is still controversial. This study had a few limitations. At first, we did not measure other factors affecting bone status such as calcium, phosphorous, and parathyroid hormone in the follow-up evaluations. The study terminated at a 2-year follow-up. Literature needs long-term evaluations of other types of hip fractures with other modes of VitD administration and regimens of supplementation. Further investigation on the relation between VitD administration in patients with normal VitD level and other dysfunction evaluation methods like Parker Mobility Score (PMS) and individual domains of 36-Item Short Form Health Survey (SF36) can be beneficial.
Conclusions
A single preoperative injection of 300,000 IU VitD did not significantly alter the postoperative survival rate and HHS of patients with intertrochanteric fractures and sufficient serum VitD.
Footnotes
Author Contributions
Conceptualization: PM, HA.; Data curation: PM, HA.; Formal Analysis: AAS.; Funding acquisition: HA; Investigation: MR, GG.; Methodology: AAS, HA.; Project administration: PM, HA.; Resources: PM, HA.; Software: AAS.; Supervision: PM, HA.; Validation: AAS, PM, HA.; Visualization: PM, HA.; Writing – original draft: MR, GG; Writing – review & editing: MR, GG, HA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
