Abstract
Introduction
Certain titanium alloy stems have been shown to be susceptible to failure at the neck with catastrophic trunnion failure. Failure has been particularly noted in the single wedge Accolade 1 stem design. Other stems also used this alloy including the anatomic designed Citation stem.
Methods
This case series details 3 catastrophic failures of the TMZF version of the Citation femoral stem.
Results
Each of these failures appear to be attributed to cyclical wear of the TMZF trunnion against the cobalt chromium femoral head. Wear resulted in ultimate implant failure and significant metal debris in the joint capsule at the time of revision surgery.
Discussion
While surgeons are aware of the risk of catastrophic failure for the Accolade 1 stem, failure may similarly happen in the TMZF Citation stem. Surgeons should monitor these implants with care and discuss the potential for trunnion failure with their patients.
Introduction
Advancements in materials and modern implant design has produced implant survivorship exceeding 25 years, and many older patients can expect their hip replacement to remain functional throughout their lifetime. 1 The risk of requiring a revision surgery is higher if the patient is younger and 1 goal for continued improvement and refinement of arthroplasty devices is to increase the time to failure in the younger, active patient. 2 Design changes have been made in the past in an attempt to improve stem survivorship. Titanium and its alloys are useful as biomaterials because they have desirable corrosion, mechanical, biocompatibility, and osteointegration properties. TMZF is a β-titanium alloy (titanium–molybdenum–zirconium–iron) that better approximates bone, providing increased flexibility and greater tensile strength compared to other titanium alloys such as Ti6Al4V. 3 TMZF has greater tensile strength, greater flexibility, greater ductility, and greater yield strength than the most widely used titanium for medical and surgical implant applications (Ti-6Al4V ELI). 4
However, femoral stems manufactured from TMZF have been susceptible to trunnion failure. The majority of reports involving trunnion failure involve the single wedge Stryker Accolade I TMZF stem design.5-7 Failure has also been reported in the Meridian stem. 8 Trunnion failure is thought to be due to fretting when paired with a Cobalt-Chromium head. 9 We present 3 cases of trunnion fracture with the TMZF version of the Citation femoral implant, which to our knowledge, has not previously been reported in the literature.
Case # 1
A 57 year old male underwent a left total hip replacement for primary osteoarthritis with a 60 mm Stryker Trident (Mahwah NJ, USA) acetabular component with 32 mm inner diameter polyethylene liner and a size 6 Stryker Citation TMZF femoral stem with 32 mm cobalt chromium +8 femoral head. Twelve years later he felt a pop in the hip with immediate instability, pain and inability to walk. He was found to have failure of the prosthetic femoral neck (Figure 1). He was admitted through the emergency department and underwent revision of the left total hip replacement using a posterolateral approach. A large amount of thick black fluid was encountered upon entry into the capsule that was consistent with metal debris and mechanical prosthetic failure. The acetabular component was assessed and found to be well fixed and in appropriate position. The polyethylene was exchanged to a 36 mm inner diameter liner. The femoral stem was ingrown and required an extended trochanteric osteotomy for removal. The stem was revised to a Zimmer Biomet Arcos (Warsaw, IN) modular implant and a Ceramtec Biolox Delta (Plochingen, Germany) ceramic femoral head was utilized. He did well postoperatively and initially mobilized without difficulty. He was last seen at 3 months postoperatively with callus formation and partial healing of the osteotomy site along with initial integration of the femoral stem. Anteroposterior left hip radiograph of a 57 year old male (Case #1) with acute failure of Stryker Citation TMZF stem 12 years after implantation.
Case #2
A 71 year old patient underwent right total hip using a Stryker Citation TMZF femoral stem with 32 mm cobalt chromium +8 femoral head with a Stryker Trident (Mahwah NJ, USA) acetabular component with 32 mm inner diameter polyethylene liner. At 14 years after total hip replacement, he developed intermittent clicking in the right hip with occasional moderate pain prior to the injury. Despite this, he was active and routinely walked over a mile daily and was living independently. A year later, he presented to the emergency department with severe right hip pain after he felt a pop. He was found to have failure of the trunnion and was admitted for revision surgery (Figure 2). At the time of surgery, heterotopic ossification was encountered on approach to the femur that was excised. A large amount of black, tarry fluid was expressed upon entry into the capsule consistent with metal debris from mechanical implant failure with significant damage and blackened synovial tissue. The acetabular component was evaluated. There was damage to the retained polyethylene due to mechanical abrasion with the broken stem so the polyethylene was changed to a 40 mm inner diameter bearing and the acetabular component was well fixed in an appropriate position and retained. The stem was able to be removed from the proximal femur with the use of high speed router and flexible osteotomes to break the bone-implant interface. Inspection of the implant after removal revealed significant damage to the trunnion prior to fracture with a large amount of material loss (Figures 3 and 4). After stem explantation, a Stryker Restoration modular stem was placed followed by a 40 mm neutral offset Ceramtec Biolox Delta (Plochingen, Germany) ceramic femoral head. The patient was seen at 6 weeks postoperatively with no immediate postoperative complications and was subsequently lost to follow up. Anteroposterior pelvis radiograph of a 71 year old male (Case #2) with acute failure of right hip Stryker Citation TMZF stem 14 years after implantation. Broken neck fragment from Case #2 placed into the femoral head after explantation demonstrating the significant loss of material from the femoral neck. Broken neck fragment from Case #2 placed onto the femoral stem after explantation demonstrating the wear pattern leading to failure with the femoral head falling into varus with erosion into the inferior femoral neck.

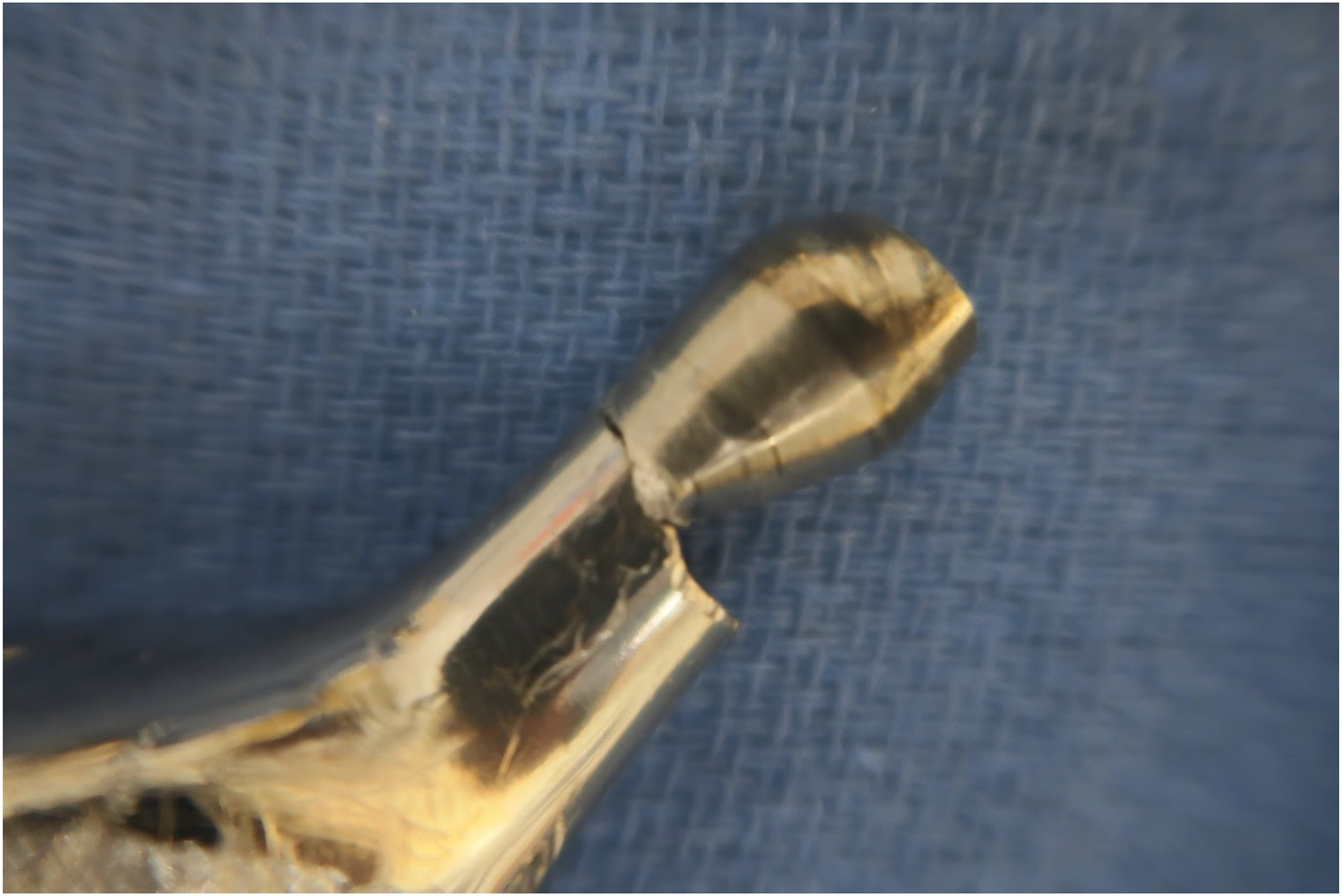
Case # 3
The patient is a 65 year old male who initially underwent left total hip arthroplasty in 2003 with a Stryker Trident acetabular component and Citation femoral stem with a 36 cobalt chromium femoral head with +10 offset. Seventeen years later, the patient felt a pop with immediate pain in his left hip. He was ambulatory with crutches until presentation for care. Radiographs of the left hip revealed catastrophic failure of the femoral implant due to trunnion fracture (Figure 5). Preoperative cobalt and chromium serum ion analysis was performed with mild elevation of serum cobalt level (1.5 μg/L with upper normal threshold of 1.0 μg/L) and chromium level within normal limits (4.8 μg/L with upper normal threshold of 5.0 μg/L). The patient underwent revision surgery via a posterolateral approach. The hip fluid was blackened from metallosis. The broken trunnion appeared to have chronic wear with failure and fracture. Both the acetabular component and the Citation femoral stem were well fixed. The liner had significant polyethylene wear. The stem was revised to an Exactech Alteon (Gainesville FL, USA), monobloc revision stem with a length of 245 mm. A Ceramtec Biolox Delta (Plochingen, Germany) 36 mm ceramic head with neutral offset was installed. A Stryker Trident 10 degree-lipped 36 mm polyethylene liner was placed into the retained acetabular component. He did well with no complications and was seen at 3 months postoperatively. He had significant improvement in his Hip disability and Osteoarthritis Outcomes Score for Joint Replacement (HOOS JR) patient-reported outcome scores during the course of his recovery with an initial 2 week postoperative score of 76.8 that improved to a score of 100 at his 3 month follow up visit. Anteroposterior pelvis radiograph of a 65 year old male (Case #3) with acute failure of left hip Stryker Citation TMZF stem 17 years after implantation.
Discussion
This case series reports 3 patients with catastrophic trunnion failure of the TMZF anatomic Citation stem. Each of the failures presented in this study experienced total mechanical failure suddenly with no preceding pain or dysfunction. We suspect that each of the implants underwent an extended process of chronic fretting to the point of failure due to the intra-operative findings consistent with chronic accumulation of metal particulate within the joint and findings consistent with cyclical wear. Concerns have been raised about increased risk of fretting with the use of high offset cobalt chromium femoral heads 10 and all 3 patients in this case series had high offset (8-10 mm), skirted cobalt chromium femoral heads. There have been other reports of TMZF femoral stem designs failing, most notably Rejuvenate stems, ABG II modular stems,11,12 the Accolade I TMZF stems13,14 and the Meridian TMZF stem. 8 Both the Rejuvenate and ABG II modular stems were voluntarily recalled by Stryker beginning in June of 2012 due to the potential risk of fretting and corrosion of the modular neck junction. 15 Both the Accolade I TMZF and Citation TMZF femoral stems have recalls on particular sizes or particular lots,16,17 but a total recall has not been issued for either device. The given reason for some of the largest recalls of the Accolade I TMZF and Citation TMZF stems was “Specific lots of sprayed HA hip stems did not meet Stryker’s Internal Material Specification for tensile bond strength and crystallinity.”
Several case reports and case series have been published on failures of the Accolade I TMZF femoral stems, especially when used in conjunction with CoCr femoral heads;13,14,18 but no such article on Citation TMZF femoral stems has been reported to our knowledge. As of this writing, there are 314 reported Citation TMZF failures that can be found on the Food and Drug Administration’s Manufacturer and User Facility device experience (MAUDE) database, several of which are described as similar to the cases outlined in this article. 19 This gap in the literature, along with the evidence that similar failures have been occurring elsewhere, is the reason for this case series. It is possible that other patients who received Citation TMZF implants may suddenly have a failure such as those presented in this series, and it may be of benefit for patients with previously placed Citation TMZF stems to be informed of that potential risk.
The 3 patients presented in this case series all had catastrophic fracture of the trunnion that appears to have been preceded by a long process of trunnion damage from the cobalt chromium head, which is a much stiffer material in comparison to TMZF. Previous retrieval analysis of cobalt chromium heads paired with the Stryker Accolade I TMZF stem found significantly higher corrosion and wear of the TMZF trunnion in comparison to trunnions manufactured from titanium alloy (Ti6Al4V), stainless steel (SS-316L) and cobalt chromium alloy. 5 There currently is no specific published guidance for recommended surveillance, or threshold for revision, of retained TMZF stems. In a study of 2635 patients with Accolade I TMZF stems, trunnion failure was overall a rare complication. 20 At 11.5 years average follow-up this involved only 3 (.1%) of patients. This makes monitoring and determination of which patient is at risk for failure difficult. Measurement of metal ions may be 1 method of surveillance. The 1 patient with available preoperative cobalt and chromium serum ions levels had results that typically would not raise concern for implant failure despite the concurrent trunnion fracture and intraoperative findings consistent with metallosis. However, there is continued debate regarding an acceptable threshold for cobalt and chromium ions in the setting of surveillance for trunnion wear in metal on polyethylene hip replacement. Several studies have found any elevation in cobalt or chromium ion levels above reference thresholds to raise concern for adverse local tissue reaction due to head-neck corrosion.21-23 This is well summarized in the review article by Cooper et al. 24 Metal suppression MRI may be another way to assess for metallosis resulting from early trunnion failure. As all 3 of our patients had acute failures, MRI was not performed. Only 1 of these 3 patients had mechanical symptoms for a year preceding the implant fracture while the other 2 patients were asymptomatic until trunnion fracture. These findings in our patient cohort are concerning that the mechanical damage leading to ultimate implant failure can be a silent problem without symptoms or abnormal lab tests. Due to this, we have a high index of suspicion for mechanical failure potential in patients with retained TMZF stems. All 3 of our patients presented in the setting of stem fracture and so surveillance data, such as metal suppression MRI or cobalt chromium serum ion levels, was not routinely obtained. We recommend monitoring patients with established guidelines for the monitoring of retained metal on metal THA implants as there is currently not enough available information to provide guidance on the specific monitoring of this patient population with TMZF stems.
Conclusions
This case series details 3 cases in which the Stryker Citation TMZF femoral component catastrophically failed due to trunnion fracture in the setting of high offset, skirted cobalt chromium femoral heads. This failure mechanism has been previously described in several other stem designs that utilized TMZF but to our knowledge has not been described in the Citation. We found 2 of the 3 patients were asymptomatic prior to failure and the 1 patient with cobalt and chromium serum ion levels with values that typically would not raise concern for imminent implant failure. We recommend a high index of suspicion for potential failure and patient counseling in patients with retained TMZF implants as further data to guide surveillance is obtained.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
