Abstract
Introduction
An estimated 2.4 million people in the United States live with hepatitis C. The prevalence of hepatitis C among adults in the United States is approximately 0.9%. 1 However, among adults born between 1945 and 1965, the prevalence of hepatitis C infection is 3.25%, more than 3 times higher. 2 Though there are effective treatments for chronic hepatitis C, many infected individuals remain untreated, likely due to a combination of the limited cost-effectiveness of treatment, particularly for individuals without active liver disease, and because 40% to 50% of individuals with chronic hepatitis C are unaware of their hepatitis C status.3,4 Even among patients with access to primary care, only 16% of those with chronic hepatitis C receive treatment 5
In 2013, the United States Preventive Services Task Force (USPSTF) recommended that adults born between 1945 and 1965 should be offered one-time hepatitis C screening. 2 One-time testing of those born between 1945 and 1965 could identify approximately 800 000 individuals with hepatitis C infection, and with linkage to care and treatment, avert more than 120 000 hepatitis C–related deaths. 2 Newer, direct-acting antiviral hepatitis C treatment regimens, which are better tolerated and have a 90% to 95% success rate means there is even greater potential public health benefit of birth cohort hepatitis C screening and treatment. 1
In 2017, the self-reported rate of hepatitis C screening among US adults born between 1945 and 1965 was only 12% to 13%, 6 suggesting that birth cohort screening has not yet been widely implemented. Primary care providers are well positioned to implement population-based hepatitis C screening guidelines, given their role in offering a wide range of prevention and screening services. Approaches that have demonstrated effectiveness for increasing hepatitis C screening rates in primary care include: systems-based approaches, such as electronic health record–enabled clinician reminders,7-11 and nurse-led linkage to care for patients with positive hepatitis C screening tests. 12 Understanding current provision of recommended birth cohort hepatitis C screening in diverse primary care systems as well as exploration of clinic or health system factors that support increased rates of screening is critical in guiding future efforts to develop and implement interventions to increase birth cohort hepatitis C screening.
Methods
Setting
This study was conducted in the WWAMI region Practice and Research Network (WPRN), a collaborative group of over 80 primary care practices across the 5-state Washington, Wyoming, Alaska, Montana, and Idaho (WWAMI) region that collaborate on research to improve primary care practice. This study was led by the WPRN Coordinating Center, based at the University of Washington Institute of Translational Health Sciences, and conducted in collaboration with a small group of research champions from WPRN sites. The 9 participating sites represented 22 primary care clinics located in diverse health care systems, each site using a unique instance of their respective electronic health record (EHR) platforms.
Approach
The WPRN Coordinating Center gathered data from 2 sources for this study—the EHR systems of participating WPRN sites and a survey of participating sites.
EHR Data Collection
The sites provided EHR data on hepatitis C testing and test results. Because the 9 participating sites had varied types and instances of EHR systems, the Coordinating Center developed methods that could be generalized across sites to gather consistent data. The Coordinating Center created a data collection tool, a description of data extraction parameters and a webinar to share the study procedures with participating sites. Each site designated a practice representative, or practice champion, to facilitate data collection and reporting. Practice champions worked with their site staff to obtain data from their sites’ EHR systems and returned aggregated data to the WPRN Coordinating Center for analysis and interpretation.
Each site reported the number of patients born between January 1, 1945 and December 31, 1965 who attended a primary care office visit with an MD (medical doctor), DO (doctor of osteopathic medicine), NP (nurse practitioner), or PA (physician assistant) between July 1, 2013 and September 30, 2015 (the study period). The study period onset coincided with the June 2013 USPSTF’s publication of birth cohort hepatitis C screening recommendations (USPSTF). Each site then reported the number of these patients who:
had laboratory evidence in the EHR of a completed hepatitis C antibody test result at any time prior to the end of the study period.
had laboratory evidence in the EHR of a positive (abnormal) or negative (normal) hepatitis C antibody test result at any time prior to the end of the study period.
Sites were asked to report data for each measure by patient’s sex (male/female), race (African American/Black, American Indian/Alaska Native, Asian, White/Caucasian, Other) and ethnicity (Hispanic/Latino, non-Hispanic/Latino). For the sites that were not able to report race and ethnicity separately we re-categorized race/ethnicity into race and ethnicity as follows: Using 2014 American Community Survey (ACS) data from the US Census Bureau, we calculated the percent of each race that was Hispanic or Latino for the county in which the site was located. We then used this proportion to reassign the “Hispanic/Latino” race responses into the other races.
Site Survey
The WPRN Coordinating Center developed and administered a brief questionnaire to participating sites to assess the presence of clinic or health system activities that might support hepatitis C screening and whether sites currently offered hepatitis C treatment in primary care. The question assessing presence of supportive clinic or health system activities asked whether the site had “a clinic or organizational policy or procedure that supports hepatitis C screening” and if yes, to report a brief description of that activity. Sites were also asked to self report whether primary care providers at the site offered hepatitis C treatment.
Analysis
We calculated the proportions of eligible patients who had evidence of hepatitis C screening and the proportions of screened patients with positive hepatitis C antibody results. We used the chi-square test to compare proportions across sites for hepatitis C screening and for hepatitis C positivity.
To assess whether a site had an activity that supported hepatitis C screening (yes/no), we reviewed descriptions of activities provided by site champions on the site survey, looking for specific clinic or organizational activities that might target increasing hepatitis C screening (eg, the presence of an EHR reminder for patients due for hepatitis C screening). We categorized sites as providing hepatitis C treatment in primary care (yes/no) based on their survey responses about availability of hepatitis C treatment. We then calculated the mean hepatitis C screening rates of those sites with hepatitis C screening activities and the mean hepatitis C screening rate for those sites without hepatitis C screening activities. We tested for differences between the means using the t test, with a significance at P < .01 We also compared the mean of the hepatitis C screening rates for those sites that offered hepatitis C treatment in primary care with the mean of the screening rates at the sites that did not offer hepatitis C treatment in primary care.
Results
Nine primary care organizations (WPRN sites) representing 22 primary care clinics in the WPRN participated in this study. Seven WPRN sites reported outcomes by sex; 4 WPRN sites reported outcomes by race and ethnicity. Most participating WPRN sites were located in urban or suburban areas (data not shown) and the average number of patient visits per year per site was 26 600 (range 6000-53 000). Six of the participating sites were community health centers or federally qualified health centers and 7 sites reported designation as patient-centered medical homes.
Overall, the 9 sites identified a total of 32 139 patients born between 1945 and 1965 who also had an office visit between July 1, 2013 and September 30, 2015. The proportion with evidence in the EHR of hepatitis C screening completed prior to October 1, 2015 was 10.9%, with a range of 1.2% to 49.1% across sites (Table 1). The proportion of patients tested who had a positive result was 16.1% overall, with a range of 6.2% to 30.0% across sites. Among the 4 WPRN sites that were able to report data by race and ethnicity, the rate of hepatitis C screening was 39.9% among African Americans, 23.2% among American Indians/Alaska Natives, and 10.7% among Caucasians (P < .001; Table 2). The rate of hepatitis C screening was 8.6% for Hispanic/Latino patients and 15.0% for non-Hispanic/Latino patients (P < .001).
Prevalence of Hepatitis C Screening and Hepatitis C Positivity Among Patients Born Between 1945 and 1965 Seen in 9 Participating WPRN Sites Representing 22 Primary Care Clinics.
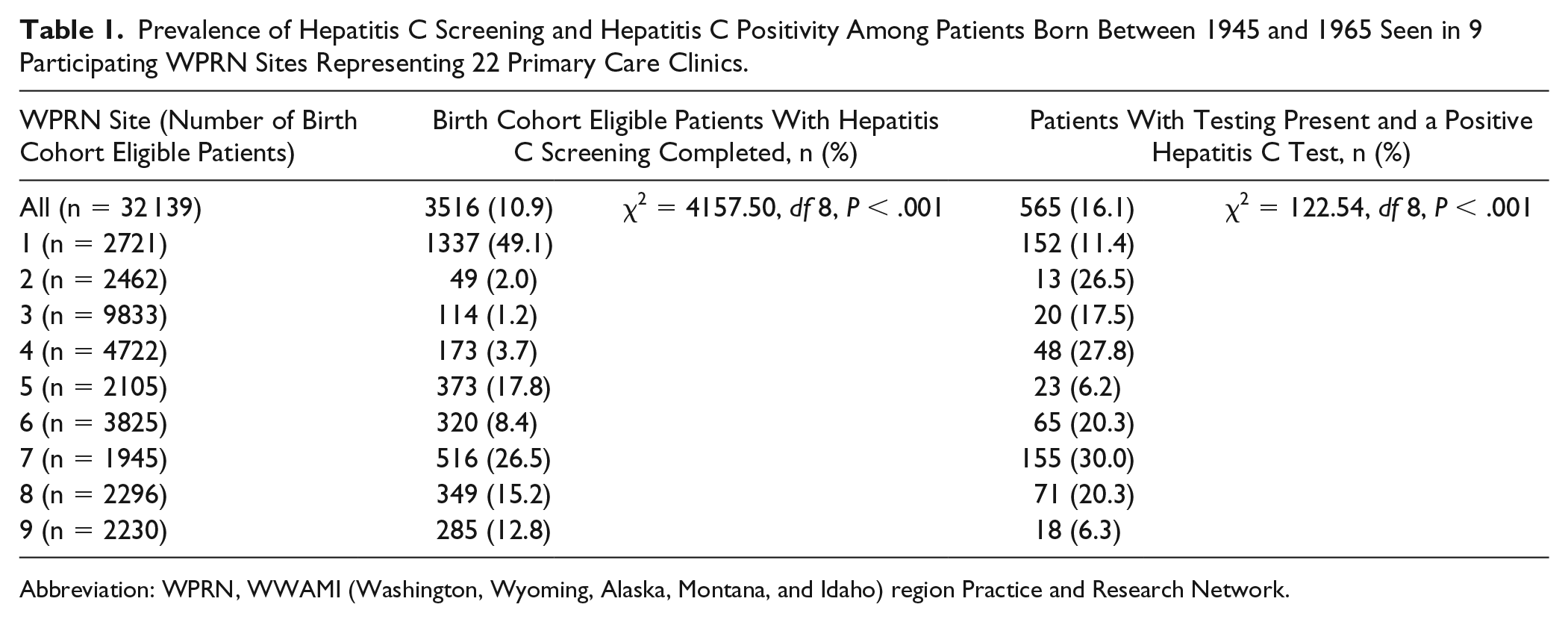
Abbreviation: WPRN, WWAMI (Washington, Wyoming, Alaska, Montana, and Idaho) region Practice and Research Network.
Among WPRN Sites That Reported Data by Race, Ethnicity, and Sex, Rates of Hepatitis C Screening and Rates of Hepatitis C Positivity by Patient Characteristics.

Clinic or Organizational Activities to Support Hepatitis C Screening and Hepatitis C Screening Rates
Two of the 8 sites that completed the site survey (25%) reported having a policy or procedure to support hepatitis C screening (eg, presence of an EHR alert to notify providers of patients in the birth cohort who were eligible for hepatitis C screening). For the 2 sites with a hepatitis C screening policy or procedure in place, the average of these sites’ hepatitis C screening rates was 37.8%, compared to 10.0% for sites without hepatitis C screening policies or procedures.
Offering Hepatitis C Treatment in Primary Care and Hepatitis C Screening Rates
Five of the 8 sites completing the survey (62%) reported offering hepatitis C treatment in primary care. Descriptions of models for offering hepatitis C treatment in primary care included a multidisciplinary hepatitis C treatment clinic within the primary care clinic and training primary care providers to offer hepatitis C treatment to their own patients. For sites that offered hepatitis C treatment in primary care, the average of these sites’ hepatitis C screening rates was 15.7%, compared with 18.2% for sites that did not offer hepatitis C treatment.
Discussion
We found that overall, among participating primary care practices in the WPRN, the prevalence of hepatitis C birth cohort screening for primary care patients seen between 2013 and 2015 was low (10.9%). There was substantial variation across sites, suggesting variable implementation of new hepatitis C screening guidelines. There were also differences in rates of hepatitis C screening based on patients’ race or ethnicity. Our study was not designed to explain why these differences might exist. Patient populations may not be equally distributed across clinic groups, in which case clinic specific factors influencing screening rates may confound interpretation of screening rate differences between populations by race. Similar to what we found, rates of hepatitis C screening in the Veterans Administration are higher among American Indian, Black and Hispanic populations, compared to non-Hispanic Caucasians. 13 Despite higher rates of hepatitis C screening, racial and ethnic minorities are more likely to be excluded from hepatitis C treatment, contributing to poorer hepatitis C health outcomes in these populations. 14 Systematic bias from providers or structural bias from health systems may account for these differences.15,16
WPRN sites with the highest screening rate (49%, 26.5%) both reported clinic or organizational activities that support hepatitis C screening. This reinforces the importance of systems-based approaches to increasing delivery of evidence-based recommendations. Clinician reminders, delivered systematically through electronic health record prompts, have consistently been shown to increase clinician delivery of recommended healthcare services. 8 A large-scale evaluation of an intervention that included both a clinician directed EHR reminder to increase hepatitis C screening and nurse-led linkage to care for patients with positive hepatitis C screening tests found that the effort dramatically increased rates of birth cohort hepatitis C screening across all patient demographics. 12 Systematic clinician reminders have consistently been shown to increasing hepatitis C screening rates.7,9-11 In our study, only two of the participating sites reported offering EHR reminders to clinicians to promote birth cohort hepatitis C screening. Further efforts to support primary care clinic implementation of systematic birth cohort hepatitis C screening interventions are needed to significantly address current gaps in birth cohort hepatitis C screening.
The overall prevalence of hepatitis C positivity among birth cohort WPRN patients screened for hepatitis C was 16.1%. This could be explained by some sites and/or providers still employing risk-based screening strategies (which would result in lower prevalence of screening but higher rates of positivity).
Five of the 8 sites responding to the survey reported providing treatment for hepatitis C in primary care. Integration of hepatitis C treatment in primary care offers an important opportunity to ensure public health benefit from widespread birth cohort screening, ensuring that patients identified through screening receive appropriate treatment. 17 However, primary care providers may lack knowledge about or confidence in providing hepatitis C treatment. 18 Multidisciplinary, team-based approaches for treating hepatitis C in primary care can address provider barriers and increase patient access and improve treatment outcomes. 19 Understanding the elements of effective hepatitis C treatment in primary care and dissemination of effective strategies could provide additional support to primary care practices considering provision of hepatitis C treatment in primary care.
There are several important potential limitations to our results. Our study collected data from a non-random sample of WPRN sites that volunteered to participate in this study. These sites and the results may not be representative of primary care clinics in general. While all queries included only patients with an office visit during the study period, the presence of hepatitis C testing was included if it occurred at any time prior to the end of the study period. Thus, the potential look-back period to detect hepatitis C testing varied across sites, depending on the length of time EHR data were available, which may have contributed to variation in measured hepatitis C screening rates. Variation in rates of hepatitis C screening observed between sites may also be attributable to unmeasured patient or community factors, rather than differences in clinical practice. Despite these limitations, this study collected hepatitis C screening rate data for more than 32 000 patients across 9 WPRN sites representing 22 primary care clinics and demonstrates variation in implementation of birth cohort-based hepatitis C screening across those sites.
The overall rate of hepatitis C screening across WPRN sites, though consistent with national average, was extremely low. This suggests that there are significant opportunities to reduce the burden of chronic hepatitis C through widespread implementation of hepatitis C birth cohort screening in primary care. Only a minority of participating sites reported screening rates well above the national average. Information about organizational policies and procedures at those high performing sites indicate that these sites were successfully using provider EHR reminders and organizational policies to support hepatitis C screening. Further work to engage community partners in increasing awareness of hepatitis C screening recommendations may also be helpful. Effective approaches for dissemination and implementation of strategies for increasing hepatitis C screening and treatment in primary care broadly could significantly decrease the public health burden of chronic hepatitis C.
Footnotes
Authors’ Note
The WWAMI region Practice and Research Network Hepatitis C Champion Group: Ryan Gilles, MD—Family Medicine Coeur d’Alene Residency Program, Kootenai Health, Coeur d’Alene, ID; John Holmes, PharmD—Idaho State University, Department of Family Medicine; Chardonnay Vance, MD—Riverstone Health; Bill Kriesgman, MD—Multicare; Adriana Linares, MD—Family Medicine Residency Program of Southwest Washington; Jaime Hornecker, PharmD—University of Wyoming Family Practice Residency Program; Elizabeth Paddock, MD—Family Medicine Residency Program of Western Montana; Winslow Gerrish, PhD—Family Medicine Residency of Idaho; William Alto, MD—Seattle Indian Health Board; Debra Gould, MD—Yakima Memorial; Jon Neher, MD—Valley Family Medicine Residency Program.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the National Center for Advancing Translational Sciences of the National Institutes of Health under Award Number UL1TR000423. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
