Abstract
Introduction:
Adult hepatitis B vaccination coverage in the United States remains low despite updated national recommendations supporting universal adult immunization. Many adults remain unaware of their susceptibility to hepatitis B virus (HBV) infection. This quality improvement (QI) study evaluated a stepwise “Offer–Offer–Test–Vaccinate” workflow designed to improve hepatitis B vaccine uptake among vaccine-hesitant adults in a primary care setting.
Methods:
This retrospective QI study was conducted at a single primary care clinic in Rochester, Minnesota. Adult patients (≥19 years) seen between October 1, 2022, and October 1, 2024, were routinely offered hepatitis B vaccination during rooming. Two participating physicians implemented an enhanced workflow in which patients who declined vaccination were counseled and then offered serologic screening using the 2023 CDC-recommended triple panel (HBsAg, anti-HBs, and total anti-HBc). Patients with negative serology were notified via the patient portal and reoffered vaccination along with a link to the CDC Vaccine Information Statement. The primary endpoint was initiation of the hepatitis B vaccine series following receipt of negative serology results. Preliminary series-completion data through October 1, 2025, were also assessed.
Results:
Among 501 eligible adults, 104 (20.8%) accepted vaccination at the initial offer. Of the 397 who declined, 106 (26.7%) agreed to serologic testing; 91 (85.8%) were seronegative. Among seronegative patients, 41 (45.1%) subsequently accepted vaccination. Integrating serologic testing increased overall vaccination uptake from 20.8% to 28.9%, an absolute increase of 8.2 percentage points and a relative increase of 39.4%. Approximately 2.6 serologic tests were required to generate 1 additional first-dose vaccination. Preliminary series-completion rates were 74.0% among those vaccinated at the initial offer and 87.8% among those vaccinated following serologic testing.
Discussion:
In this primary care setting, incorporating serologic testing into the workflow was associated with modestly improved hepatitis B vaccination uptake among vaccine-hesitant adults. Serologic results may help re-engage patients by providing personalized immunity information. Implementation considerations include staffing needs, laboratory access, patient portal engagement, and insurance coverage.
Conclusion:
The “Offer–Offer–Test–Vaccinate” strategy was associated with a modest increase in hepatitis B vaccine uptake among hesitant adults. This EMR-integrated, stepwise approach may offer a feasible and scalable strategy to reduce missed vaccination opportunities and support HBV prevention in adult primary care.
Introduction
Background
Chronic hepatitis B virus (HBV) infection is a major global health concern. It is a leading cause of cirrhosis and accounts for an estimated 41% of Hepatocellular Carcinoma (HCC) cases. 1 In 2022 alone, approximately 900 000 cases of liver cancer were reported globally.2,3 In the United States, an estimated 580 000 to 2.4 million people have chronic hepatitis B, with about two-thirds unaware of their infection.4-6 Hepatitis B is effectively preventable through vaccination. After completion of the full series, seroprotection approaches 100% in children and exceeds 90% in adults. Although response may be reduced in some groups, newer formulations have demonstrated enhanced immunogenicity, reinforcing vaccination as a critical strategy to prevent this potentially devastating and vaccine-preventable disease. 7
Historically, the diagnosis of hepatitis B infection relied on serologic testing with hepatitis B surface antigen (HBsAg), which was offered only to patients deemed at risk and to pregnant people. To improve early detection and support treatment efforts, the Centers for Disease and Prevention (CDC) recommended the universal triple-panel HBV screening in 2023 (hepatitis B surface antigen [HbsAg], antibody to hepatitis B surface antigen [anti-HBs], and antibody to hepatitis B core antigen [anti-HBc]) for all adults at least once regardless of vaccination status or risk factors. 8 Utilizing the triple-panel HBV screening offers a comprehensive assessment of a patient’s hepatitis B status by differentiating if a patient is susceptible to infection versus having natural immunity or immunity from previous vaccinations. Multiple factors were considered for this guideline recommendation, including the low prevalence of chronic HBV infection in the U.S.—estimated at around 0.3% overall, though higher in certain subpopulations such as foreign-born individuals, persons who inject drugs (PWID), men who have sex with men (MSM), and persons with HIV 4 —and the low rate of adult hepatitis B vaccination in the U.S. population. 9 Additionally, universal screening is a public health strategy, designed to reduce stigma and disparities surrounding screening for an infection with devastating consequences.
Although safe, effective, and available since 1982, the vaccination rate among adults aged 19 years and older is only 34%, with significant variation depending on factors such as age, race/ethnicity, travel status, and overall health status. 9
Historically, vaccination efforts targeted pediatric patients and high-risk groups. In the United States, the implementation of a universal infant vaccination strategy in 1991 led to a 68% decrease in HBV infection prevalence within the first 10 years. 10 With vaccination uptake exceeding 90%, 12 there were zero reported cases of acute hepatitis B infection among individuals under 19 years of age between 2011 and 2020, according to CDC surveillance data (CDC Viral Hepatitis Surveillance Report—United States, 2021; Figure 2.4). 11
The success of this highly effective pediatric vaccination program has consequently shifted the remaining disease burden toward adults aged 19 years and older, among whom vaccination rates remain substantially lower 11 and acute hepatitis B case rates remain substantially higher (CDC Viral Hepatitis Surveillance Report—United States, 2021; Figure 2.4). 11 In response, in 2022 the CDC recommended universal hepatitis B vaccination for all adults aged 19 to 59 years, and for those aged 60 years and older based on individual risk factors or personal preference. 13 Despite the CDC’s 2022 recommendations, vaccine uptake in adults ages 19 years and older has remained low. 9
Objectives
In our primary care practice, hepatitis B vaccination is embedded within the Health Maintenance tab of the electronic medical record (EMR) and is offered to all adults with incomplete or undocumented vaccination histories. This workflow follows the CDC’s 2022 recommendations, which endorse universal hepatitis B vaccination for adults aged 19 to 59 years and vaccination for adults aged 60 years and older based on individual risk factors or personal preference. During the rooming process, all clinicians in the clinic use the same EMR-based prompts and standardized workflow, in which rooming staff offer the vaccine to any patient identified as overdue and document the patient’s acceptance or decline on the rooming encounter form.
Despite offering vaccination universally to patients, the uptake of vaccination in our clinic remained low, reflecting national trends. For this study, 2 participating physicians piloted an enhanced workflow that extended beyond the standard prompts. Specifically, when patients declined vaccination, these physicians offered the CDC-recommended triple-panel hepatitis B screening (HBsAg, anti-HBs, and total anti-HBc) as a motivational tool to inform vaccine decision-making. We hypothesized that vaccine uptake would increase if patients who initially declined vaccination were offered serologic testing and learned that they were not immune to hepatitis B virus (HBV). Here, we present the results of implementing this “Offer–Offer–Test–Vaccinate” strategy.
Ethics Statement
This project was undertaken as a Quality Improvement (QI) initiative to improve hepatitis B vaccination uptake in our primary care practice. Its purpose was to enhance local processes by implementing an “Offer–Offer–Test–Vaccinate” strategy and to measure its effect within our own clinical setting. This project was reviewed and deemed exempt from review by our institution’s Review Board (IRB) under 45 CFR 46.101, item 2.
Methods
Study Setting
Our institution maintains a presence in both urban and rural settings across 5 U.S. states: Minnesota, Wisconsin, Iowa, Arizona, and Florida. Primary care is a foundational component at each location, with the largest practice situated in the Rochester, Minnesota, region. This regional hub comprises 5 outpatient clinics and 2 express care sites staffed by physicians, nurse practitioners, and physician assistants. Patients for this study were recruited from one of the Rochester-based primary care clinics, which provides care for approximately 275 000 patients annually. The clinic’s multidisciplinary team—comprising family medicine clinicians, community pediatrics and adolescent medicine, and community internal medicine—reflects its significant role in delivering comprehensive primary care to the community. Two family medicine physician investigators from this clinic participated in and adhered to the study protocol throughout the study period.
Study Approach
We retrospectively reviewed charts of adult patients seen in our primary care clinic between October 1, 2022, and October 1, 2024, and obtained corresponding laboratory and vaccination records. During this period, 2 study physicians followed the established clinic workflow for hepatitis B vaccination, beginning with review of encounter rooming forms where rooming staff documented each patient’s decision to accept or decline vaccination. Additionally, a thorough review of each patient’s electronic medical record was conducted, including documentation of past medical history and immunization history.
Eligible adults were defined as patients aged 19 years and older without documented acute or chronic hepatitis B infection, prior resolved infection, or hepatitis B vaccine non-responder status in the electronic medical record (EMR). Additionally, eligibility criteria were consistent with the CDC’s 2022 recommendations for universal hepatitis B vaccination for adults aged 19 to 59 years and risk- or preference-based vaccination for adults aged 60 years and older. Eligibility was determined using the Health Maintenance tab in the EMR, which flags patients as overdue for hepatitis B vaccination based on institutional immunization records and data from the Minnesota Immunization Information Connection (MIIC). Patients who declined vaccination received general and unscripted counseling from study physicians regarding the option of serologic testing with the CDC’s 2023-recommended triple hepatitis B panel (HBsAg, anti-HBs, and total anti-HBc). Physicians explained that the CDC advises 1-time hepatitis B screening for all adults aged ≥18 years, regardless of risk factors. The discussion emphasized that screening can determine whether a patient is immune due to prior vaccination or natural infection, or remains susceptible to hepatitis B. Patients were informed that if they were found to be susceptible, hepatitis B vaccination would again be recommended.
Patients were also informed that while recent updates to national guidelines now support universal 1-time triple panel screening for hepatitis B in all adults—and this alignment is driving expanded insurance coverage beyond traditional risk-based criteria—some insurance limitations and associated costs may still occur. They were encouraged to verify coverage with their insurance provider before proceeding with testing.
Orders for serologic testing were placed during the clinical visit; however, patients could complete the blood draw either immediately afterward or at a separate phlebotomy appointment. This flexibility allowed patients to coordinate testing based on personal preference, timing with other scheduled laboratory work for chronic condition monitoring, or convenience.
Serologic testing for hepatitis B surface antibody (anti-HBs) was interpreted according to laboratory reference standards. Anti-HBs levels <5.0 mIU/mL were classified as negative, levels ≥12.0 mIU/mL indicated protective immunity, and levels between 5.0 and 11.9 mIU/mL were considered indeterminate.
Patients who elected testing were notified of their results via patient-portal messaging. Those with negative serology results were again offered vaccination, accompanied by a link to the CDC’s hepatitis B Vaccine Information Statement. This process reflects the “Offer–Offer–Test–Vaccinate” approach, in which vaccination is offered twice—initially during rooming and again after physician counseling—followed by serologic testing and vaccination as indicated by test results.
The number of patients who initially declined vaccination but subsequently accepted after reviewing negative serology results was recorded. The primary endpoint—the proportion of patients who, after initially declining vaccination, agreed to initiate the hepatitis B vaccine series following receipt of negative serology results—was assessed during the defined study observation period from October 1, 2022, through October 1, 2024. The secondary endpoint—completion of the 2-dose vaccination series—requires longer follow-up. Accordingly, preliminary secondary endpoint data were evaluated through an extended observation period ending October 1, 2025; however, full data collection for this outcome remains ongoing beyond this date to allow assessment of final series completion rates.
Statistical Analysis
All analyses were conducted using R Statistical Software (v4.4.1; R Core Team 2024). Demographic data—including race, gender, primary language, and insurance type—were summarized descriptively. Mean age was calculated along with standard deviation and range, as presented in the attached Demographics Table 1.
Demographics.

Fisher’s Exact Test for Count Data with simulated P-value (based on 2000 replicates).
Linear Model ANOVA.
Wilson-Score confidence intervals (CI) were calculated for proportions, and Fisher’s Exact tests were employed to compare categorical variables. For continuous variables such as age, results were reported as mean (SD) with range, and group comparisons were evaluated using analysis of variance (ANOVA) models. Time between vaccination and serology as well as time between doses was evaluated with Wilcoxon Rank Sum tests due to skewed distributions. This analytic approach ensured robust handling of both categorical and continuous demographic characteristics across study groups.
Results
Patient Characteristics
A total of 501 patients age 19 years and older (mean age = 48.5 years, SD = 14.0; range = 19-91) were evaluated in clinic and offered the hepatitis B vaccine (Figure 1). Demographic characteristics of the 501 patients are summarized in the accompanying table (see Table 1). Of these, 104/501 (20.8%, CI [17.4%, 24.5%]) accepted vaccination at the initial offer by rooming staff and physicians, a rate comparable to national estimates.
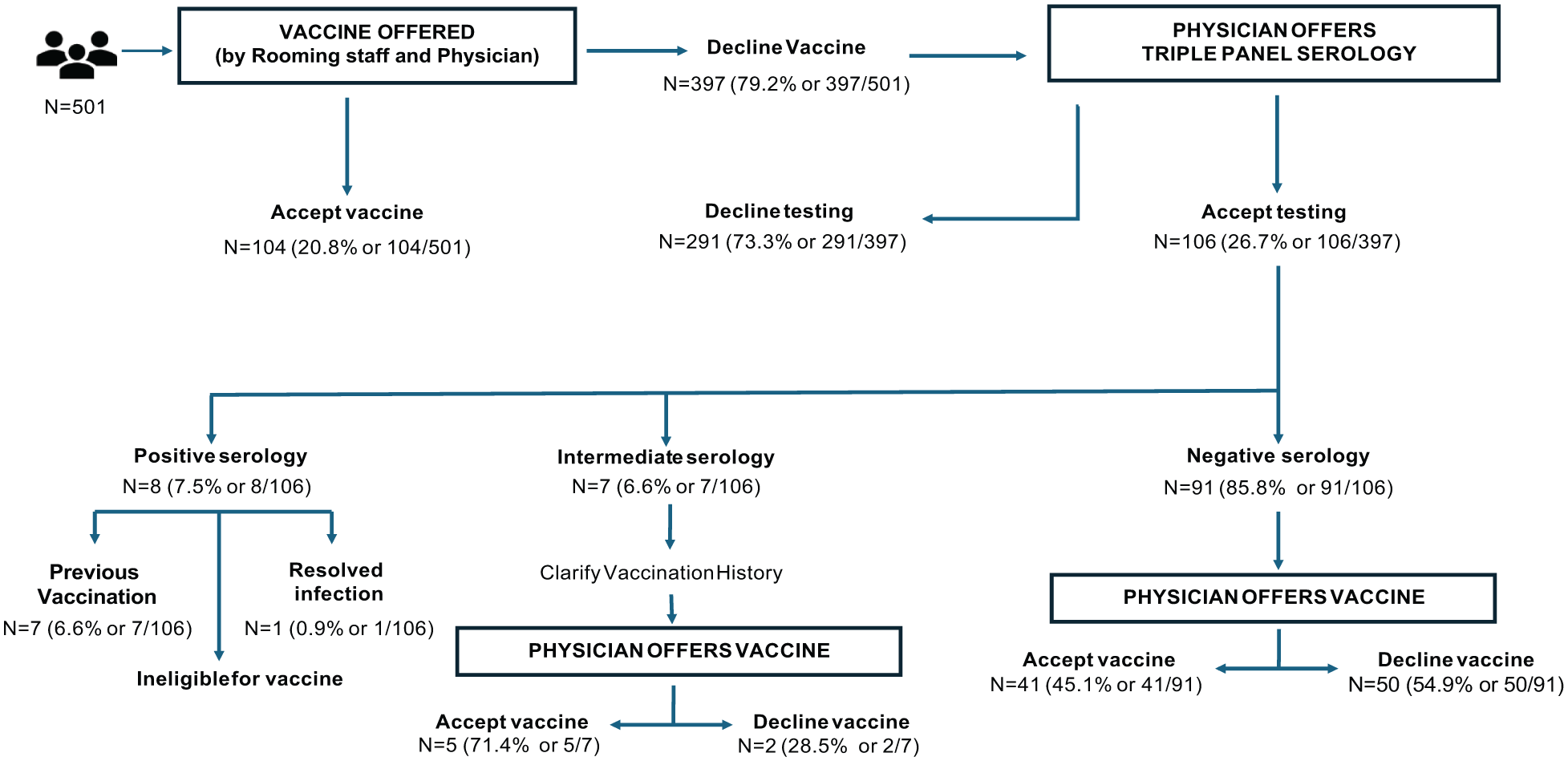
Hepatitis B vaccination and serology workflow.
Serologic Testing Following Initial Decline
Among the 397/501 patients (79.2%) who initially declined vaccination, 106/397 (26.7%, 95% CI [22.6%, 31.2%]) agreed to serologic testing after discussion with the 2 study physicians (Figure 1). The remaining 291/397(73.3%) patients did not undergo testing or receive vaccination during the study period.
Of the 106 patients tested, 8/106 (7.5%) were seropositive for hepatitis B, and 7/106 (6.6%) had indeterminate results. Among the 8 seropositive patients, 7/8 (6.6% or 7/106) demonstrated the pattern HBsAg negative/anti-HBs positive/anti-HBc negative, consistent with prior immunization, and 1/8 (0.9% or 1/106) showed HBsAg negative/anti-HBs positive/anti-HBc positive, consistent with resolved infection. Indeterminate results were defined as HBsAg negative/indeterminate anti-HBs/negative anti-HBc. Seronegative patients (HBsAg negative/anti-HBs negative/anti-HBc negative) accounted for 91/106 (85.8%) of those tested (Figure 2).

Hepatitis B serologic testing results, serology interpretation, and recommended next steps in management among patients undergoing triple-panel screening. Of the 106 patients tested, 8 (7.5%) were seropositive for hepatitis B and 7 (6.6%) had indeterminate results, while 91 (85.8%) were seronegative. Serologic patterns guided interpretation of immunity, prior infection, or indeterminate status and informed subsequent management steps.
Vaccination Uptake After Serologic Testing
Among the 91/106 seronegative patients, 41/91 (45.1%, 95% CI [35.2%, 55.3%]) subsequently accepted vaccination. Incorporating serologic testing and follow-up increased overall vaccination uptake (2-proportion test P = .003) from 104/501 (20.8%, 95% CI [17.4%, 24.5%]) to 145/501 (28.9%, 95% CI [25.1%, 33.1%]), representing an absolute increase of 8.2 percentage points (95% CI [2.9%, 13.5%]) and a relative increase of 39.4% (Figure 3). On average, 2.6 serologic tests were required to achieve 1 additional first-dose vaccination.
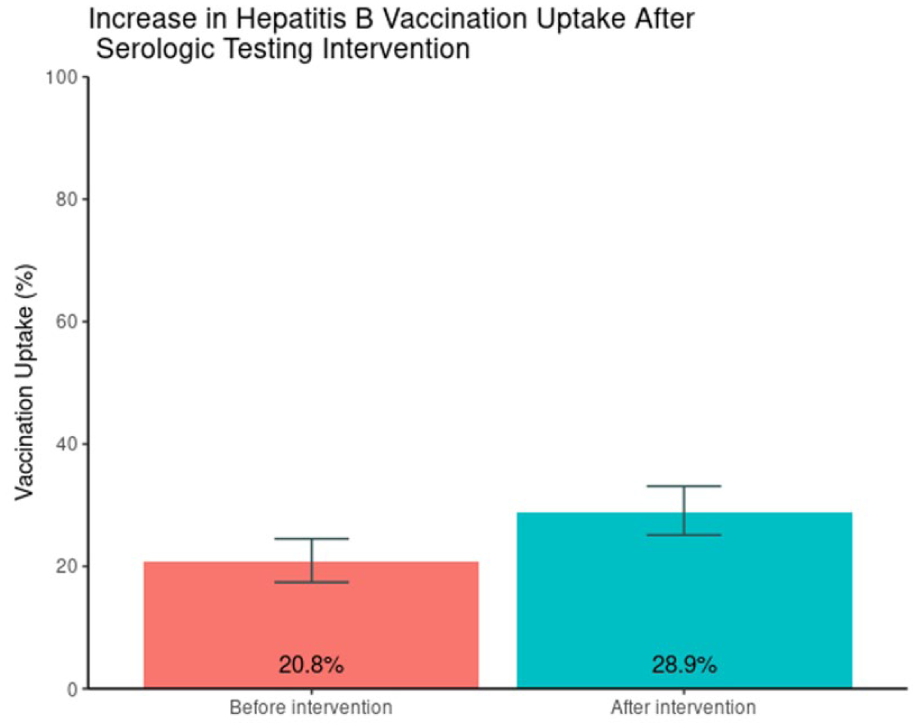
Change in hepatitis B vaccination uptake before and after the serologic testing intervention. Bar chart showing overall vaccination uptake increasing from 20.8% before the intervention to 28.9% after implementation of the “Offer–Offer–Test–Vaccinate” workflow.
Completion of Series
Among the 145/145 patients who initiated vaccination during the study period, 77/104 (74.0%) of those who accepted the vaccine at the initial offering completed the series, compared with 36/41 (87.8%) of those who accepted serologic testing after initially declining. As outlined in the Study Approach section above, completion of the 2-dose series represents a secondary endpoint, and full data collection for this outcome is still ongoing. Preliminary analyses were conducted using data available through the extended observation period ending October 1, 2025, and therefore these completion rates reflect interim findings (current comparison: P = .079). The median time between doses was 104 days for the immediate-uptake group (Q1, Q3: 48, 281) and 66 days for the uptake-following-serologic-testing group (Q1, Q3: 34, 214; P = .068).
Discussion
This study demonstrates that in vaccine-hesitant patients, the stepwise strategy of first offering vaccination, followed by serologic testing with subsequent educational reinforcement through the Electronic Medical Record—what we coined as the “Offer–Offer–Test–Vaccinate” strategy— was associated with modestly increased hepatitis B vaccine uptake. Approximately one-third of vaccine-hesitant patients agreed to undergo serologic testing using the triple panel test. Of those testing negative, approximately 45% of patients subsequently accepted vaccination. The observed 39.4% relative increase in vaccine series initiation highlights the potential of this approach to improve vaccination uptake among adults in a primary care.
A potential source of selection bias must be acknowledged: patients who agreed to serologic testing may systematically differ from those who declined, including in their underlying trust in clinicians, baseline vaccine attitudes, health literacy, or resource availability. These differences could partially account for the higher conversion rate among those who underwent testing. Future work should explore predictors of testing acceptance to better understand which patients are most likely to benefit from this approach and to ensure equitable implementation.
In our clinic, this strategy fit readily within existing workflows. Rooming staff offered vaccination and documented the initial decision, after which the study physicians re-offered vaccination and discussed the option of serologic testing for those who initially declined. Patients who elected testing were notified of their results and re-offered vaccination if non-immune.
While this process was feasible in our resource-rich, integrated EMR environment, implementation in more diverse or resource-limited settings may pose challenges. Practices with limited patient portal use, reduced laboratory access, or high out-of-pocket testing costs may find that uptake of serologic testing is lower, and follow-up efforts more burdensome. Additionally, the scalability of this strategy will depend on practical considerations, including staff time for repeated counseling, laboratory turnaround times, reimbursement structures for serologic testing, and the complexity of EMR workflows required to support reliable patient notification and follow-up.
Overall, while our findings are promising, the strategy’s optimal application will require adaptation to local resources, staffing models, and technological infrastructure.
Limitations
This study has several limitations. It was conducted at a single center in 1 Midwestern state with only 2 participating physicians, which may limit generalizability. Importantly, there was no pre-implementation comparator arm or control group, and only patients seen by 2 physicians experienced the full “Offer–Offer–Test–Vaccinate” workflow. As a result, observed improvements in vaccination uptake cannot be definitively attributed to the intervention and may reflect other contextual or temporal influences.
Selection bias is also possible, as patients who agreed to serologic testing likely differ from those who declined in ways that may influence their subsequent vaccine acceptance.
Vaccination status was obtained from institutional records and the Minnesota Immunization Information Connection (MIIC); therefore, doses received at external or out-of-state facilities may not have been fully captured. The reasons patients declined serologic testing were not systematically collected, limiting insight into financial, logistical, or attitudinal barriers. Additionally, we did not assess the number of patient visits during the observation period; some patients may have been offered vaccination or testing multiple times. Data collection for the secondary endpoint—completion of the vaccination series—is ongoing. Preliminary data suggest that delays in series completion are driven largely by system-wide challenges in patient follow-up and multi-dose vaccine reminders. Efforts are underway to implement automated reminder systems to improve series completion.
Future Direction
These promising findings warrant further investigation through larger, multi-center studies to evaluate the effectiveness, sustainability, and potential cost-effectiveness of the “Offer–Offer–Test–Vaccinate” approach across diverse clinical settings. Building on this work, we plan to expand the initiative by assessing vaccine uptake, serologic testing acceptance, demographic influences, and reasons for hesitancy among vaccine-hesitant patients offered either direct vaccination after counseling or serologic testing first with vaccination recommended if negative. This approach aims to enhance understanding of vaccine acceptance barriers, minimize missed prevention opportunities, and contribute to ongoing efforts toward hepatitis B elimination.
Conclusion
This study demonstrates that the “Offer–Offer–Test–Vaccinate” strategy may modestly improve hepatitis B vaccination uptake among hesitant adults in primary care, with a nearly 40% relative increase compared to initial acceptance rates. Integration of serologic testing with counseling and a structured follow-up effectively re-engaged hesitant patients and reduced missed prevention opportunities. Despite limitations described above, these findings provide the foundation for future research. Larger, multi-center studies and cost-effectiveness evaluations —neither of which were included in the present study—are warranted to confirm its effectiveness, clarify generalizability, and determine this approach’s value across a broader range of practice environments.
Footnotes
Acknowledgements
On March 8, 2025, this research was presented at the Minnesota Academy of Family Physicians (MAFP) Research and Innovations Forum, where it was selected as the Project of Greatest Interest in the Physician/Research category for the 2025 Innovation & Research Forum. An abstract and a non–peer-reviewed summary of this work were subsequently published in Minnesota Family Physician, Fall 2025, Vol. 9, No. 4, with permission from the journal’s Editor-in-Chief. We gratefully acknowledge Dr. Thomas A. Thacher, Consultant, Department of Family Medicine, and Parker D. Sanders & Isabella G. Sanders Professor of Primary Care, Mayo Clinic College of Medicine, for his insightful input during this work and his continual encouragement of our research endeavors.
ORCID iDs
Ethical Considerations
This project was conducted as a Quality Improvement (QI) initiative and, as such, did not constitute human subjects research requiring Institutional Review Board (IRB) review or approval, in accordance with institutional policy and federal regulations. The activity focused on evaluating and improving existing clinical processes using data obtained from the Epic electronic health record system. No interventions outside routine care were implemented, and all data were handled in compliance with institutional privacy and confidentiality standards.
Consent to Participate
Not applicable. This project was a Quality Improvement initiative and did not require IRB review or approval. Patients were provided the option to undergo testing and vaccination in alignment with current CDC guidelines through shared decision-making as part of standard clinical care.
Consent for Publication
I, the corresponding author, confirm that I give consent for publication of this manuscript in the Journal of Primary Care & Community Health, including all data, tables, and figures. No videos are included in this manuscript. Therefore, no specific participant consent for publication was required.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by internal funding from Mayo Clinic through a Career Development Award (Cost Center #30020219, Activity Code: CAREERDEV). The award provided support equivalent to 0.1 FTE for a duration of 6 months.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this project are available from the corresponding author upon reasonable request. The dataset includes information obtained from our institution’s electronic health record system and the associated statistical analyses. Due to institutional privacy regulations and patient confidentiality, these data cannot be shared publicly. Aggregate or deidentified data may be provided to qualified researchers upon request and with appropriate data use agreements in place.
