Abstract
Background:
Diabetes self-management education and support (DSMES) is an evidence-based intervention that improves outcomes for people with diabetes, but uptake is <10%. We conducted a pilot pragmatic trial to increase DSMES in rural primary care clinics that integrated health information technology with a quality improvement (QI) learning collaborative.
Methods:
Two healthcare systems from rural Kentucky participated in the study. Within each system, 2 clinics received the intervention and 1 clinic served as a control (N = 6 clinics). Each intervention clinic identified a QI team (5-6 individuals) who participated in a 9-month QI learning collaborative followed by 5 months of follow-up. Our primary outcome was DSMES referrals; secondary outcomes were DSMES attendance and percentage with A1c >9%.
Results:
The 6 study clinics served 1,803 patients with diabetes; n = 1083 (60.1%) from 4 intervention clinics and n = 720 (39.9%) from 2 control clinics. DSMES referrals significantly increased in the intervention clinics compared to the control clinics (31.7% vs 0.1%, P < .001) and continued to increase throughout 5-month follow-up (40.0% vs 0.1%, P < .001).
Conclusions:
A structured QI intervention can significantly increase DSMES referral rates in primary care clinics. Findings highlight the potential of systems-level approaches to address persistent gaps in diabetes care delivery in rural settings.
Clinical Trial Registry:
Using Practice Facilitation and Operationalizing Referral Information Technology to Increase DSMES Utilization (Clinicaltrials.gov registration number no. NCT05472142). Clinicaltrials.gov URL (https://clinicaltrials.gov/study/NCT05472142).
Keywords
Introduction
Diabetes self-management education and support (DSMES) is an integral part of evidence-based diabetes care. DSMES is a structured course, delivered by a credentialed diabetes educator, that teaches people living with diabetes how to effectively self-manage their disease. Participation in DSMES is associated with improved glycemic control (up to a 1-percentage point reduction in A1c), reduced mortality, and improved quality of life.1-3 In addition to improving patient outcomes, DSMES reduces the overall cost of care for people with diabetes.4,5 However, despite evidence supporting DSMES and guidelines from the American Diabetes Association (ADA) specifying when it should be offered, 6 DSMES remains significantly underutilized with less than 10% of individuals with diabetes having used DSMES,7,8 and even lower rates in rural populations. 9
Multilevel barriers to DSMES utilization exist nationally and in Kentucky. Programmatic-, healthcare provider-, and individual-level barriers to DSMES have been identified in prior studies, consensus reports, and the Centers for Disease Control and Prevention’s DSMES Toolkit.10-12 Notable barriers include limited geographic and temporal availability of DSMES classes, low awareness of DSMES services among patients and providers, and challenges with referral processes. One fundamental barrier identified by the American Association of Diabetes Educators was lack of a DSMES referral from a provider; 13 while DSMES is a covered benefit by most insurance plans, patients must have a written referral from the provider who manages their diabetes. Referring a patient to DSMES necessitates clinician awareness and engagement, patient identification, a referral mechanism, and an available DSMES supplier. Barriers can and do occur throughout this process.
Many of these DSMES barriers exist in Kentucky. Over the past decade, the Kentucky Diabetes Prevention and Control Program through the Kentucky Department for Public Health prioritized increasing access to and utilization of DSMES through the Healthy Living with Diabetes Program. 14 This program involves innovative approaches such as partnerships with local health departments to house the state’s diabetes educators and making telehealth DSMES available to individuals in all counties in Kentucky. As of 2019, Kentucky had 91 nationally recognized DSMES programs with 80 counties offering in-person classes and all 120 of Kentucky’s counties covered by the telehealth program. Despite the increasing availability of DSMES, however, utilization remained relatively low through 2019. 15
In response to low uptake and to more broadly improve diabetes care across the state, the Kentucky Diabetes Prevention and Control Program developed the Kentucky Diabetes Learning Collaborative.14,15 This Collaborative was based on the Institute for Healthcare Improvement’s Breakthrough Series Model and used practice facilitation to engage primary care systems in quality improvement activities related broadly to improving quality of diabetes care;16,17 DSMES referrals was a secondary metric. Kentucky Diabetes Prevention and Control Program reported improvements in A1c levels, increased clinician DSMES referrals, and increased patient referrals to DSMES services at clinic sites that participated in 2 iterations of the Kentucky Diabetes Learning Collaboratives, suggesting the utility of this approach.14,15 While suggesting effectiveness, this programmatic work lacked a control group and a rigorous study design, which we added in this study to better assess the effectiveness of this approach.
In the current study, we adapted the Kentucky Diabetes Learning Collaborative using feedback from previous participants 18 to address clinic and provider-level barriers to DSMES. Then, we conducted a pilot pragmatic cluster randomized trial to test the feasibility and effectiveness of the approach. Here, we describe the study design and findings related to feasibility (clinic staff participation in intervention activities, clinic retention, and completeness of EHR extracts for study data) and preliminary effectiveness (DSMES referrals at end of active intervention and end of follow-up) of the intervention.
Methods
Overview of Study Design
We conducted a pilot 2-arm parallel group pragmatic trial with 2 health systems and a total of 6 primary care clinics (n = 3 per system) with clinics randomized 2:1 to the intervention or control arm within health system. Partnering health systems were identified in collaboration with our practice facilitation partners, the Kentucky Regional Extension Center. The Kentucky Regional Extension Center provides quality improvement, practice facilitation, health IT, and data reporting support to healthcare organizations across Kentucky. To recruit health systems for the current project, the Kentucky Regional Extension Center solicited interest by sharing the opportunity through its professional network and hosting a webinar in which they provided an overview of the project and answered any questions. Of those health systems interested, only 2 had the minimum number of primary care clinics necessary for participation (n = 3 total primary care clinics). We formalized agreements with these 2 health systems and proceeded with study.
Prior to randomization, we completed a preliminary screen to ensure clinics were located in USDA-designated rural locations. 19 Within each healthcare system, we verified all 3 primary care clinics were located in rural settings, that each served a sufficient volume (N > 50) of patients age 18 to 75 with diabetes, and that each had a clear division of providers across clinics to minimize contamination/spillover of intervention into control sites. We, then used cluster randomization to allocate clinics to the intervention or control arm using a 2:1 parallel group design within health system. In health system 1, we randomized using random number generation—2 clinics to intervention, 1 to control. In health system 2, during the prescreen process, it came to light that 1 provider saw patients in 2 of the 3 clinics. Therefore, to avoid contamination we decided to allocate the 2 clinics that this provider served to intervention and the remaining clinic to control. The study team and some intervention clinic staff were aware of their allocation because it was infeasible to blind investigators and clinic staff due to the type of intervention. To minimize bias, the practice facilitation partner (Kentucky Regional Extension Center) managed data collection.
We used the Pragmatic Explanatory Continuum Indicator Summary (PRECIS-2) Assessment tool to guide pragmatic study design. 20 For example, we did not impose patient eligibility criteria beyond carrying a diagnosis of diabetes, which is the same criterion a patient needs for DSMES referral in standard care. As another example, follow up of patients utilized routine care, including clinician-directed A1c measures, and there were no study-specific participant visits. Our assessment of the study’s pragmatic design per the PRECIS-2 domains along with details on study timing appear in Figure 1 with a maximum of 5 dots indicating the most pragmatic design for each element.

Study design with overlay of pragmatic design elements from the PRECIS-2 assessment.
Description of Intervention
The Learning Collaborative model is based on the Institute for Healthcare Improvement’s Model for Improvement and the Chronic Care Model.17,21 The Model for Improvement is a quality improvement approach that uses rapid cycle Plan-Do-Study-Act (PDSA) cycles meant to identify barriers to quality healthcare and rapidly implement and measure change. The learning collaborative is a structured, facilitated process that brings together teams from different organizations to learn from one another. Teams from intervention clinics apply this quality improvement strategy to identify and address quality gaps in diabetes care uptake at their clinics. Quality improvement advisors and practice facilitators on the study team provide technical assistance and individualized coaching (practice facilitation) to clinics in the intervention arm of the study. The Chronic Care Model is a primary care-based approach for providing high quality care to people with chronic diseases, such as diabetes. The Chronic Care Model includes 6 elements essential to quality diabetes care and provides a framework to help primary care clinics identify opportunities to improve care for patients with diabetes. The learning collaborative that our team has pilot tested, integrates components that map to 4 of these 6 CCM elements—Delivery System Design, Decision Support, Clinical Information Systems, Patient Self-Management Support.
Once clinics were randomized, our team began ‘pre-work’, a 3-month period of study start-up activities. These activities include providing technical assistance to all clinics to identify their population of patients with diabetes, extracting EHR data to capture and validate baseline measures (diabetes care metrics and demographics, outlined in Data Collection and Measures below), and helping to setup the bidirectional referral system between clinics and external DSMES educators through the statewide umbrella network, Healthy Living with Diabetes. Both intervention and control clinics received this support. For each intervention clinic, our implementation team assisted in identifying a quality improvement (QI) team of 5 to 6 individuals who would serve as the primary study participants. We encouraged clinics to engage a range of members of the clinical team who contributed to diabetes care, and we did not specify explicit inclusion/exclusion criteria. Members of the quality improvement teams included physicians, registered nurses, hospital administrators, diabetes educators, dieticians, and practice managers. Intervention clinics proceeded to complete a 9-month QI intervention followed by 5 months of follow-up to track outcomes and assess short-term sustainability. Intervention activities are detailed in Table 1. Briefly, they included participation of each intervention clinic’s QI team in 3 learning sessions spaced at 3-month intervals, monthly educational webinars and technical assistance calls with our implementation team, and monthly plan-do-study-act (PDSA) implementation cycles and tracking of study outcomes. Each learning session convened all QI teams from all intervention clinics and centered around a specific topic of relevance to the intervention. The monthly educational webinars also convened all QI teams from the intervention clinics for a 1-h session that included a brief report from each intervention clinic on their progress followed by a 30- to 40-min educational activity on a topic of interest (patient testimonial, optimizing workflow, etc.). Monthly technical assistance calls provided individualized support to each clinic QI team to help them address individual barriers. There were no significant protocol deviations after commencement. There were no stopping rules due to minimal risk. At the end of the active intervention, we conducted preliminary analyses comparing DSMES referrals across intervention and control clinics.
Summary of Study Activities, Hours, and Content for Intervention and Control Clinics.

Ethics Approval
All methods and materials were approved by our Institutional Review Board (protocol #75928) prior to engaging in study activities. All members of the QI teams (primary study participants) provided oral consent to participate in all study activities and data collection procedures. Per IRB, patients were not considered human subjects as our study team did not interact with patients nor collect identifiable patient information. All patient data was aggregated by the clinics and reported to our study team at the clinic-level. This study is registered in ClinicalTrials.gov (NCT#: NCT05472142).
Data Collection and Measures
Baseline characteristics were collected to describe the population of eligible patients with diabetes and attributes of the intervention and control clinics. Baseline data were extracted from the 12 months prior to intervention start. All patients with diabetes aged 18 to 75 who had an encounter in the prior year were included in the clinic’s eligible patient population. Baseline characteristics of the population of patients with diabetes included sex, race/ethnicity, mean A1c at baseline, and % of the clinic’s diabetes patient population with A1c >9%. Attributes of the clinics included the distribution of payer type for all encounters in 2023, rurality based on USDA designation, availability of an onsite DSMES educator, and total number of clinicians at the clinic.
Our primary outcome was DSMES referrals. Secondary outcomes were DSMES attendance and percentage of the study population with A1c >9%. All outcomes were extracted and reported from the baseline period. DSMES referrals and attendance were reported monthly for the duration of the intervention (9 months) and 5-month follow-up period; A1c >9% was reported at 3 timepoints: baseline, study end, and at the end of the 5-month follow-up. If a patient had multiple A1c values measured in a given timeframe, A1c >9% was assessed using the last available value from the corresponding timeframe. Outcomes were aggregated to the clinic-level and were calculated as the percent of patients with the outcome divided by the eligible patient population for the measure. For DSMES referrals and A1c, the eligible patient population was all patients aged 18 to 75 with documented diabetes and an encounter at the given clinic in the prior 12 months. For DSMES attendance, the eligible patient population was all patients aged 18 to 75 with documented diabetes and an encounter at the given clinic in the prior 12 months who had been referred to DSMES. ‘DSMES referral’ was operationalized as an order placed either in the EHR or via the secure messaging platform for external DSMES referring the patient to DSMES services. ‘DSMES attendance’ was operationalized as a patient attending at least 2 h of DSMES. The percentage of patients with A1c >9% was operationalized to align with the Center for Medicare and Medicaid Services Merit-Based Incentive Payment System Quality Measure for diabetes control (NQF0059). 22 The QI team at each clinic reported DSMES referrals and DSMES attendance monthly to our implementation team; as indicated above, A1c >9% was reported at 3 timepoints: baseline, study end, and at the end of the 5-month follow-up. All data was aggregated to the clinic-level prior to reporting to our team.
Sample Size and Power Calculation
Our primary outcome was DSMES referrals. Our power calculations used a 2-sided test with α = .05, power (1 − β) = .80 and made the following assumptions: by the end of follow-up, clinics in the control arm would have referred a total of 7% of eligible patients and clinics in the intervention arm would have referred at least 17% of eligible patients,14,15 exchangeable correlation matrix, intra-cluster correlation <.1, and cluster size to vary from 500 to 200 patients. Based on these parameters, we needed between 3 and 5 clusters to be adequately powered to detect a statistically significant effect of the intervention on DSMES referrals. With 6 clinics across our 2 partner healthcare systems, we were adequately powered to detect a difference in DSMES referrals. Importantly, however, because this is a pilot study it was primarily designed to evaluate aims other than effect size.
Statistical Analysis
We used descriptive statistics to characterize the population of patients with diabetes as well as clinic attributes across the 6 participating clinics. Next, we calculated the percent of patients referred to DSMES from baseline through the end of follow-up monthly by intervention and control group and at the clinic-level. Finally, we compared the percent of patients referred to DSMES in intervention vs. control clinics out of the total eligible study population at 3 pre-specified time points: baseline, end of active intervention, and end of follow-up. We also compared the percent of patients who attended DSMES in intervention vs. control clinics out of the total number of patients who were referred and the percent of patients with A1c >9% out of the total eligible study population at the same 3 time points. Statistical comparisons of DSMES referrals, DSMES attendance and percent of patients with A1c >9% across intervention and control clinics were made using 2-sided Fisher’s exact tests with a P-value <.05 used to determine statistical significance.
Results
A total of 1,803 patients with diabetes were included in the study; n = 1083 (60.1%) from 4 intervention clinics and n = 720 from 2 control clinics (39.9%). Patient demographics were similar across intervention and control clinics (Table 2). Across all clinics, about half of the patients with diabetes were female and >95% were non-Hispanic White. Mean A1c at baseline across the clinics was also comparable (mean A1c 7.0%-8.0% at all clinics). The percent of the patient population with A1c >9% ranged from 5.0% to 20.4% at intervention clinics and 10.8% to 23.7% at control clinics. All clinics were located in USDA-designated rural locations in Kentucky. The number of clinicians at intervention clinics ranged from 4 to 8 and at control clinics 3 to 5. Payer mix at clinics varied, but, at each clinic, the overall patient volume served was comprised of >50% patients on publicly-funded insurance (Medicare or Medicaid).
Characteristics of the Patient Population and Intervention and Control Clinics.

Rural designation from the U.S. Department of Health & Human Services Health Resources & Services Administration. Federal Office of Rural Health Policy.
Insurance categories map to broad categories of insurers in the US reflecting the multi-payer system that exists in the US; Medicaid and Medicare are both publicly-funded insurance, Commercial refers to privately financed, market-based coverage—typically offered through employers or purchased individually; Self-pay refers to patients directly covering the full cost of their healthcare services out-of-pocket, without using insurance, often occurring when individuals are uninsured, seeking elective care, or choosing services outside their insurance coverage.
Figure 2 presents results on our primary outcome, DSMES referral rates. At baseline, the aggregate proportion of DSMES referral across intervention and control clinics was 0%. By the end of the intervention, 31.7% of patients with diabetes at intervention clinics had been referred to DSMES and 0.6% of patients at control clinics had been referred to DSMES (P < .001). At the end of follow-up, 40.0% of patients from intervention clinics and 0.6% of patients at control clinics had been referred to DSMES (P < .001). DSMES referrals increased at all intervention clinics but there was substantial variation in the individual intervention clinics referral rates with 1 intervention clinic referring 94.1% of eligible patients while another referred only 7.2% of eligible patients.

Cumulative DSMES Referrals across rural primary care clinics participating in a Learning Collaborative Intervention. Line graph presents monthly DSMES referral rates aggregated across intervention and control clinics; bar graph presents clinic-level rates within individual intervention clinics at 3 pre-specified timepoints (baseline, end of intervention, and end of follow-up).
Table 3 presents the primary study outcome as well as secondary outcomes across study arms at baseline, the end of the active intervention and the end of follow-up. At baseline, 0% of patients had been referred to DSMES in the prior 12 months in both intervention and control clinics. DSMES referrals at the end of the active intervention and the end of study follow-up were significantly higher in the intervention than control clinics (31.7% vs 0.1%, P < .0001; 40.0% vs 0.1% P < .001; respectively). At baseline, 0 patients had attended DSMES in the prior 12 months. By the end of study follow-up, 61 patients from intervention clinics (14.1%) and 0 patients from control clinics had completed DSMES classes, but this difference was not statistically significant (P > .999). At baseline, the intervention clinics had a higher percentage of patients with A1c >9% (33.2%) as compared to control clinics (16.4%; P < .001). At the end of follow-up, the percentage of patients with A1c >9% had decreased to 16.0% in the intervention clinics and increased to 22.6% in control clinics (P = .003). As noted in Table 3, the number of participants for whom A1c data was shared with the study team changed for one of the 2 control clinics over the course of the study. Because we collected aggregated data, the EHR data extract was linked to each provider’s overall patient population rather than to a fixed cohort of individual patients. As a result, when provider turnover occurred between baseline and the end of the study for this one clinic, the composition of the associated patient panels also changed, leading to shifts in the number of patients included in the control group over time.
Comparison Across Study Arms of DSMES Referrals, DSMES Attendance, and % With A1c >9% From Baseline to End of Intervention and End of Follow-up.
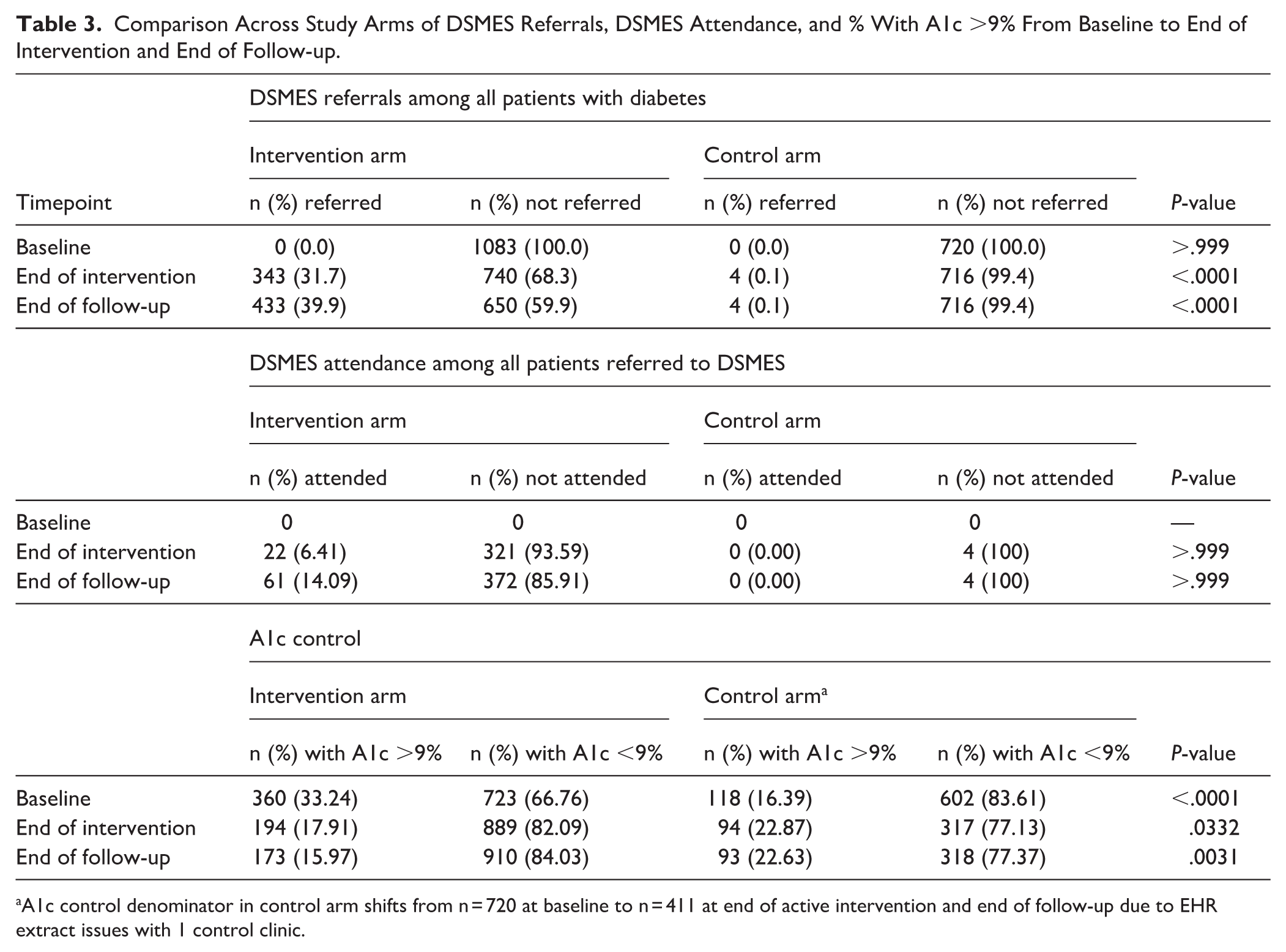
A1c control denominator in control arm shifts from n = 720 at baseline to n = 411 at end of active intervention and end of follow-up due to EHR extract issues with 1 control clinic.
Discussion
Our study demonstrated that a pragmatic, clinic-level intervention with iterative, facilitated QI significantly increased referrals to DSMES services in rural primary care settings. At baseline, DSMES referral rates across both intervention and control clinics were 0%. Following the 9-month intervention and 5-month follow-up, DSMES referrals among intervention clinics surpassed 40.0%. By contrast, DSMES referral rates in control clinics remained flat throughout the study period. This substantial increase highlights the potential of targeted implementation strategies to overcome entrenched system-level barriers such as lack of provider awareness, inefficient referral mechanisms, and limited integration between primary care and DSMES providers. 11
Our findings are consistent with prior studies that successfully used practice facilitation to improve chronic disease management.23,24 This study adds to that literature by demonstrating that such models can be feasibly adapted to rural clinics where resource limitations, workforce shortages, and geographical isolation often amplify barriers to care.
We observed considerable variation in the intervention effect size across intervention clinics. Some clinics achieved near-universal referral rates (94.1% of eligible patients referred), while others saw more modest gains (7.2% of eligible patients referred). This source of this variability as well as strategies to optimize referrals merit further exploration and underscore the importance of contextual adaptation and flexible implementation strategies, particularly in diverse clinical environments.25,26
DSMES attendance, while improved, did not increase in proportion to referrals. Several factors likely contributed to this outcome. First, the short duration of follow-up limited the time available for patients to attend DSMES after referral, especially considering potential scheduling constraints for both patients and educators. Second, while the intervention focused explicitly on provider and system-level barriers, it did not address patient-level factors such as transportation, health literacy, or personal beliefs—all of which are known to affect DSMES uptake. 12 These findings highlight the need for future work that explicitly incorporates strategies to address patient-level barriers, alongside provider- and system-focused efforts.
The study also incorporated A1c outcomes as a secondary metric. Although we did not analyze data at the patient-level, clinic-level A1c control improved significantly from baseline through the end of follow-up for intervention clinics, while A1c control worsened slightly in the control clinics. Given the relatively short follow-up period and the time it typically takes for improved self-management behaviors to translate into measurable clinical improvements, these findings are particularly promising.
Our study had several strengths. We used a cluster randomized design to allocate clinics to intervention and control arms within each health system. This minimized potential confounding due to organizational differences. The intervention was rooted in strong theoretical foundations, drawing on the Chronic Care Model and the Institute for Healthcare Improvement’s Model for Improvement, as well as lessons learned from previous iterations of the Kentucky Diabetes Learning Collaborative.16,18,27,28 Importantly, we partnered with rural clinics, thereby addressing a critical gap in the literature on DSMES implementation in underserved settings. Additionally, the study maintained a high degree of pragmatism throughout with study decisions guided by the PRECIS-2 tool. 17 This included broad inclusion criteria, reliance on routine clinical workflows, and use of existing EHR systems to identify eligible patients and track outcomes.
While our pragmatic approach enhanced generalizability and real-world relevance, it also introduced several limitations. Chief among these was the absence of patient-level data, which precluded analyses of patient-level predictors of referral or attendance (eg, use of regression models) and more rigorous handling and evaluation of missing data. Another key limitation was the resource-intensive nature of the intervention. Sustained engagement from clinic staff, particularly members of the QI teams, required significant time and effort, raising important questions about scalability and long-term sustainability, particularly in resource-constrained settings. While the Learning Collaborative model proved effective in this context, future efforts may need to identify lower-touch or technology-enabled models to support broader dissemination. This intervention approach was not amenable to investigator or clinic blinding, which may have introduced measurement bias. To minimize this potential bias, the practice facilitation partner managed study data collection (and not the investigators). Additionally, while the intervention produced promising short-term gains in DSMES referral, the long-term sustainability of these improvements remains unknown. Continued follow-up beyond the study period is needed to determine whether QI teams maintain their momentum in increasing DSMES referrals. Embedding DSMES referral processes into routine workflows and EHR systems may help support sustainability, as would systems-level interventions such as expanding the types of providers who can refer to DSMES, allowing patients to self-schedule for DSMES classes, expanding the pool of providers who provide DSMES, and altering reimbursement structures to make offering DSMES more financially feasible. 12 While our study sample was representative of the area in which the study was conducted, there was a lack of ethnic and racial diversity in our sample which may limit generalizability. Finally, this was a pilot study conducted within 1 state in the United States (US) with the intent of generating preliminary data for a larger scale trial. Our findings are not necessarily generalizable to other settings in the US or internationally. However, the general approach of engaging clinic staff to identify, design, and test quality improvement strategies to increase DSMES referrals is, by design, transferrable to diverse healthcare systems, as it relies on the highly contextualized knowledge of the clinic workers to identify and implement strategies to improve care.
Conclusion
In conclusion, our study suggests that a structured QI intervention based on Learning Collaborative principles can significantly increase DSMES referral rates in rural primary care settings. While challenges remain around DSMES attendance, patient-level barriers, and long-term sustainability, the study demonstrates the potential of systems-level approaches to address persistent gaps in diabetes care delivery especially in rural settings. Future research should build on these findings to test the model at scale, integrate patient-level components, and explore strategies to ensure lasting impact across diverse clinical environments.
Footnotes
Acknowledgements
We would like to acknowledge the Kentucky Department for Public Health’s Diabetes Prevention and Control Program (KDPCP) for their work leading the Diabetes Learning Collaboratives.
Ethical Considerations
This study was approved by the University of Kentucky Institutional Review Board (protocol #75928).
Consent to Participate
Study participants (QI teams) provided informed consent.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by funding from the National Institute of Diabetes and Digestive and Kidney Diseases, National Institutes of Health, through grant R34DK132548. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Results of the study are reported on clinicaltrials.gov ([NCT05472142]). The data that support the findings of this study are not publicly available but are available from the corresponding author* on reasonable request.
