Abstract
Background:
Colorectal cancer (CRC) screening uptake is low despite availability of population-based screening.
Aim:
To assess the feasibility and acceptability of an Asian-validated risk prediction tool (RPT) in increasing screening uptake.
Design:
Randomised controlled trial; in-depth interviews.
Method and Setting:
Sixty primary care patient participants were recruited in 3 primary care practices in Singapore. Eligible patients were ≥50-year-old, with no recent CRC screening, were randomised (1:1) into control (standard recommendations) or intervention (stratified risk) groups. Follow-ups at 1 and 6 months assessed screening uptake; feasibility were evaluated through surveys. In-depth interviews were conducted with patients and their Primary Care Physicians (PCPs) to assess acceptability.
Results:
Response rate of patients was 56.6%. Median age was 62.5 years. Overall, the intervention group had higher CRC screening uptake (63.3%) than control group (33.3%; P = .022) with use of the RPT. The tool was reported by patients to be easy to use (93.3%), questions were easy to understand (100%), words easy to see (93.3%). Logistic regression associated post-secondary education with reduced need for assistance (P = .042). The RPT was acceptable to patients and PCPs. Patients’ perceived barriers included burdensome procedure, cost and lack of knowledge. Facilitators included intrinsic motivation and perceived early detection benefits. PCPs recognised patients’ pre-determined mindset and fear of receiving bad news as barriers to screening. Education, patient empowerment and integration of RPT into workflow may aid adoption.
Conclusion:
Risk stratification of CRC using a RPT showed increased screening uptake. Future research is needed to understand which components of this complex intervention worked to bring about improved outcomes.
Keywords
How This Fits In
Providing access to cancer screening does not determine success in screening uptake
Uptake of cancer screening is a complex decision, incorporating beliefs, barriers and behavioural factors
Risk prediction tools aim to stratify high-risk patients to colonoscopy compared to non-invasive stool tests for average-risk patients
A systematic approach to educate, empower patients and integrate workflows for PCPs are critical for effective implementation
Introduction
Colorectal cancer (CRC) poses a growing global public health challenge, as it represents the third most common cancer worldwide. 1 In Singapore, colorectal cancer represents the most common cancer in males and the second most common cancer in females. 2 The survival rate for stage I cancer is 84% to 86% but drops steeply to just 10% to 11% for stage IV cancer and late-stage CRC treatment is more expensive and requires a longer duration of treatment.4,5 Regular screening has been known to be effective in preventing and detecting CRC early, yet CRC screening programmes have often been associated with low uptake.3-6
Singapore currently adopts a risk-stratified model for CRC screening, where asymptomatic individuals without a family history of CRC are regarded as average-risk and are recommended annual faecal immunochemical occult blood test (FIT) or 5 to 10 yearly colonoscopies from age 50 years. Individuals with a personal and/or family history of CRC are regarded as high risk; they are recommended to begin colonoscopies 10 years prior to the youngest case of CRC in the family or age 40 years, whichever is earlier.7-10 Despite the presence of a national CRC screening programme in Singapore, only 28% of the population aged 50 to 69 years have undergone FIT, whilst only 14% have undergone a colonoscopy or sigmoidoscopy.11-14 More than half of CRC patients are diagnosed at later stages III and IV. 2 Our current model does not account for other well-established CRC risk factors such as alcohol use, smoking and body mass index (BMI), likely underestimating one’s CRC risk.11,12,15-17
The Asia-Pacific Colorectal Screening (APCS) score represents a tool that can determine CRC risk based on age, gender, family and smoking history in unselected asymptomatic Asians aged above 50 years, flagging high risk patients for colonoscopies as a priority. 18 It was first developed by Yeoh et al 18 in 2011 from a prospective, cross-sectional study in 11 Asian cities and was recently incorporated as a risk prediction tool (RPT) in the updated Asia Pacific consensus recommendations to optimise CRC screening and shown to effectively stratify the advanced colorectal neoplasia risk in the Asia-Pacific population.19,20 Wong et al. modified the APCS score to replace smoking history with body mass index (BMI) and self-reported ischaemic heart disease, 21 as low body weight carried a strong protective effect against colonic neoplasia.22-24
Benefits of risk-stratified screening include early detection and treatment for high-risk individuals, whilst minimising screening-related harms such as false-positive or false-negative test results, physical or psychological harm, overdiagnosis and overtreatment in lower-risk individuals. High risk individuals also have the option to engage in primary prevention and risk management, such as lifestyle changes, risk-reducing medication or prophylactic surgery.25,26
Primary care physicians (PCPs) are the first point-of-contact with patients and are ideally positioned to utilise RPTs to provide personalised cancer screening recommendations for the general population. Singapore has a robust primary care network with 26 island-wide polyclinics and more than 1500 private clinics. Polyclinics are regarded as multi-disciplinary community health centres that manage acute and chronic medical conditions, conduct preventive healthcare, cancer screening and health education. 27 Aligned with Singapore’s healthcare focus for a “HealthierSG” 28 to empower preventative care of its citizens, polyclinics provide subsidised CRC screening for all citizens, as per the recommended national schedule and eligibility criteria.28,29
We chose the modified APCS by Wong et al as the RPT for this study. 21 This is because proximal (right-sided) colon lesions are harder to detect as they are often asymptomatic, or present with non-specific signs such as anaemia. These lesions may also have different and sometimes more aggressive features, so a tool that predicts proximal neoplasia improves the accuracy and completeness of CRC screening.30,31 The primary aim of this study was to assess effect of implementing the RPT in primary care on increasing CRC screening uptake. This is done by comparing objective completion of CRC screening by patients under the usual Ministry of Health (MOH) recommendation (usual arm) vs. the patients who underwent personalised CRC screening advice using the RPT (intervention arm). The secondary aim was to understand the feasibility and acceptability of the RPT amongst patients and PCPs in guiding colorectal cancer screening behaviours. Finally, we explored barriers and facilitators of CRC screening.
Methods
Study Design and Setting
We conducted a pilot cross-over RCT with embedded semi-structured interviews. Study reporting followed the CONSORT checklist for pilot and feasibility trials and COREQ for qualitative research.
The pilot RCT was conducted in 3 primary care practices in Singapore (Outram, Bukit Merah and Ang Mo Kio Polyclinics), from January 2023 to July 2023. PCPs recommended CRC screening, following the usual Ministry Of Health (MOH) recommendations (usual care arm) vs. the RPT (intervention arm).
Randomisation
The PCPs were randomised into “intervention arm” or “usual care” allocations by Microsoft excel random number generator (lowest 50% will be control). Based on the allocation, each PCP would recruit 5 patients. After 1 month, the PCPs crossed over into “usual care” or “intervention arm” allocations where they would recruit another 5 patients. By the end of 2 months, each PCPs would have discussed CRC recommendations with 10 patients. Training of PCPs with the RPT was provided prior to the study implementation.
Study Participants – Patients
Eligible patients were (1) ≥50 years old, (2) has not performed recent CRC screening within the last 10 years, 32 (3) able to read/ speak English. Patients who reported history of colorectal, ovarian, endometrial cancer, colorectal polyps, known Lynch Syndrome, familial adenomatous polyposis or inflammatory bowel disease, were excluded. Recruitment of patients was done by convenience sampling, with the research team approaching participants in the waiting area after being screened for eligibility by the PCPs. Patients were reimbursed for their time in the study.
Study Participants – PCPs
PCPs were eligible if they work in a primary care setting and were recruited to provide feedback on the RPT tool through an individual in-depth interview. The interviews were conducted by trained facilitators, utilising a semi-structured interview guide (Supplemental Box 1) developed from literature reviews/discussions with the research team with qualitative interview experience (RF, JY, ZYC).
Usual Care Arm: Conventional CRC Recommendations
Patient participants enrolled under the usual care arm were stratified based on current MOH recommendations for CRC screening which is based on age, personal and family history.7,10
Intervention Arm: Addition of a Risk Prediction Tool
The RPT uses 5 variables including age, gender, family history, body mass index and self-reported ischaemic heart disease to compute a score which was then stratified into “average-risk”: 0 to 2 and “high-risk”: 3 to 5. Patient participants in the high-risk group had 1.51-fold (P < .001) of higher risk of proximal CRC than the average-risk group, and were recommended to undergo colonoscopy instead of FIT. This RPT was validated in an Asian Hong Kong population, and the scoring system was useful in predicting the risk of proximal CRC to prioritise patients for colonoscopy. 21
Patient participants enrolled under the intervention arm were asked to self-complete the RPT. This was done before their clinic consult, in a private waiting room. If needed, the research team assisted the patient.
The final score of the RPT was a sum of the points assigned to each risk factor. Based on their score, patients were informed of their CRC risk and screening recommendation during their consultation with their PCPs.
Study Outcomes of the RCT
Primary outcome was to compare the uptake rate of CRC screening by participants under current MOH recommendations (usual care arm) vs. participants using the RPT (intervention arm).
Secondary outcome was to assess the feasibility and acceptability of the RPT. This included quantitative data of the number of eligible patients recruited into the study, the proportion of enrolled patients who self-completed the RPT, the number of patients who needed assistance and a self-reported “ease of-use” 5-point Likert scale.
Data Collection
Eligible patient participants, who gave informed consent to participate in this study, completed a baseline questionnaire (Supplemental Box 2), which collected their biodata including age, ethnicity, personal medical and family history. Patients were informed of their enrolment arm (usual vs. intervention arm) based on the allotment given to their PCP.
Adherence to screening recommendations were assessed at 1- and 6-month follow-ups. At the 1-month mark, patient participants in both the usual care and intervention arms were contacted via telephone or video-conference to verify their adherence to the recommended screening. The call served as a reminder to schedule their screening appointment if they had not done so. At the 6-month mark, patient participants were contacted again to verify adherence to screening for those who did not do so at the 1-month mark.
All patient participants from the intervention group provided feedback on the usability of the RPT tool through a survey 33 (Supplemental Table 1). Patient participants from the intervention group were invited to participate in an individual in-depth interviews.
Interview Guide (PCPs and Patients)
We utilised a semi-structured topic guide adopting a constructivist paradigm, 34 which was pilot-tested and underwent minor revisions before being finalised for use in the study. The interview probes explored experiences, perceptions, attitudes, adoption, motivations, barriers/ motivators of CRC screening uptake, views on feasibility and acceptability of the RPT (Supplemental Box 3). The interviews were conducted face to face or via online platforms (Zoom), depending on the participants’ preferences. Each session lasted 45 to 60 min, was audio-recorded and transcribed verbatim.
Data Analysis
Quantitative Data
The demographic and clinical characteristics were compared using Fisher’s exact test for categorical variables and Kruskal-Wallis test for continuous variables among patients from 3 different polyclinics as well as between usual care and intervention groups. Feasibility of patients self-completing the RPT was assessed using univariable logistic regression. Univariable logistic regression was used to evaluate the effectiveness of RPT by comparing the screening uptake between control and intervention groups. Patients’ experiences regarding the RPT were analysed through feedback surveys. All P-values were two-sided and those less than .05 were considered statistically significant. Statistical analyses were performed using R software (version 4.2.0). We conservatively estimated a medium effect size of 0.5 for this pilot. To achieve 90% power with a 95% upper confidence limit in future trials, the required sample size was 52, which was rounded up to 60 patient participants to factor in attrition rate. 35
Qualitative Data
Recorded interviews were transcribed verbatim. The transcripts were coded by 2 independent coders (GT and DI), and any discordance reconciled upon discussion with a third coder (ZYC). Each coder developed a codebook which was iteratively used to code remaining transcripts. Thematic analysis, according to Braun and Clarke, 36 was used to analyse the data and the coding process was facilitated by the QSR NVivo 14. Recruitment of both patient and PCP participants for interviews were stopped when data saturation was reached where no new themes emerged. 37
We adopted the Health Belief Model as a conceptual framework. 38 The core constructs include perceived susceptibility, perceived severity, perceived benefit, perceived barriers, modifying variables, cues to action and self-efficacy.
The researchers (RF, JY and ZYC) were experienced health service researchers by training with substantial experience in qualitative research and did not have any prior relationship with the participants. The recordings, transcripts, coding, field notes were maintained in organised secure archives, to establish a clear audit trail. 39
Results
Patient Participants
As shown in Figure 1, the CONSORT flow diagram summarises participant recruitment, randomisation and retention across the trial arms. A total of 152 potential patients were screen for eligibility, of which 46 were not eligible. Of the remaining, 106 eligible patients, 60 agreed to participate in the study (response rate 56.6%). The number of participants was equally distributed with 30 patient participants each in both the intervention and control groups.

CONSORT diagram summarising the flow of participants in the pilot trial.
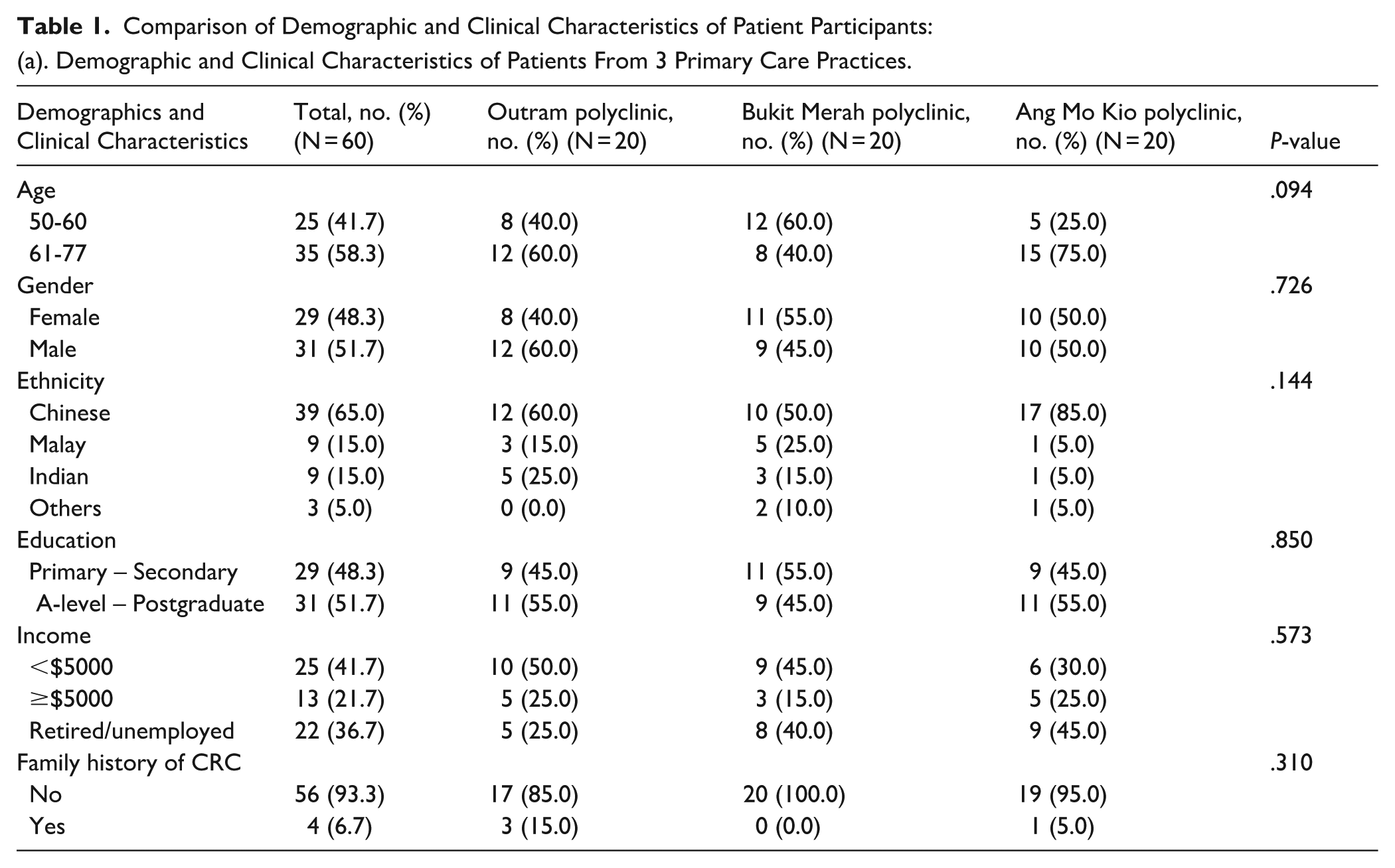
The demographic and clinical characteristics were similar within the cohorts from both usual care and intervention groups, except for income. The proportion of ethnicities were representative of the ethnic compositions previously reported in Singapore. 40 The retired or unemployed control group had significantly higher income than those in the intervention group (P = .026; Table 1).
Comparison of Demographic and Clinical Characteristics of Patient Participants: (a). Demographic and Clinical Characteristics of Patients From 3 Primary Care Practices.
(b) Demographic and Clinical Characteristics of Patients From Control and Intervention Groups.

Abbreviation. CRC, colorectal cancer.
Participants’ ages ranged from 50 to 77 years, with a median age of 62.5 years (IQR = 57.8-67.0 years). The majority were Chinese (65.0%), male (51.7%), education level of A-level to postgraduate (51.7%), with an income of less than $5000 (51.7%) and without a family history of CRC (93.3%).
Primary Outcome: Preliminary effectiveness, Compliance and Risk scores
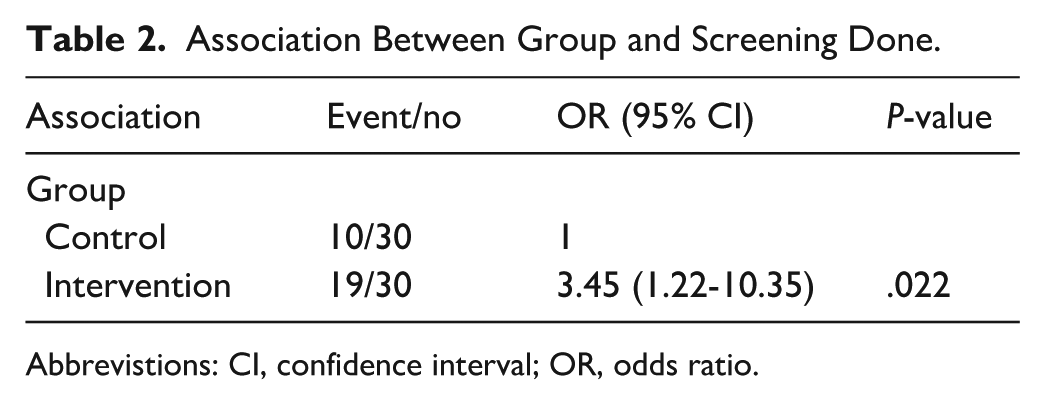
The odds of the intervention group taking up CRC screening was significantly higher than that of the control group (P = .022) as shown in Table 2.
Association Between Group and Screening Done.
Abbrevistions: CI, confidence interval; OR, odds ratio.
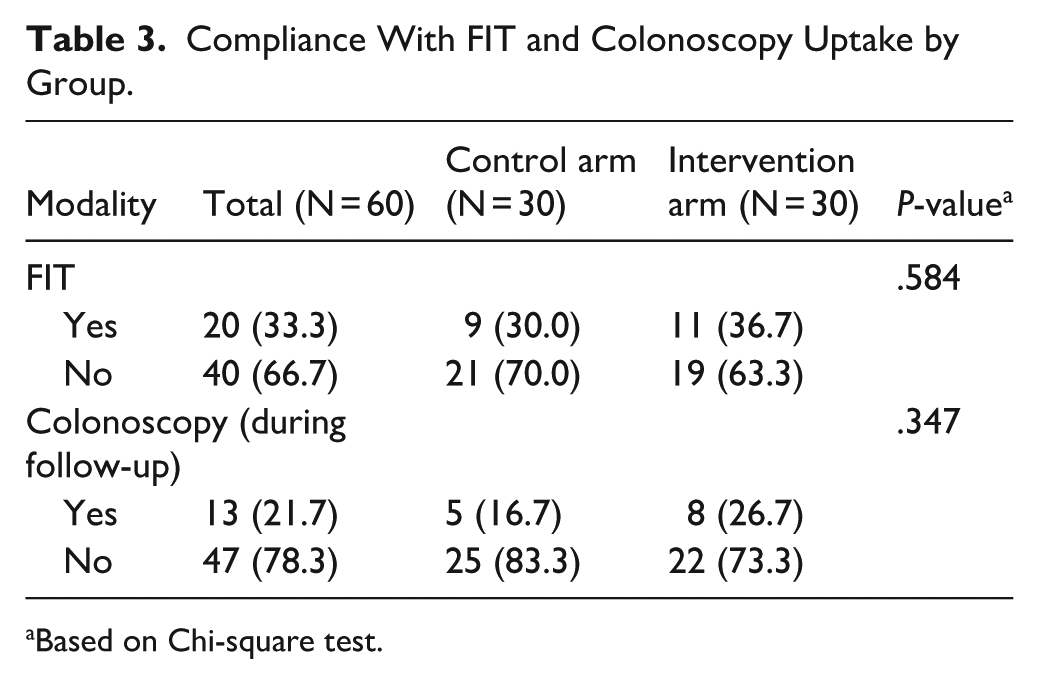
Compliance with FIT and colonoscopy uptake by control and intervention groups are listed in Table 3. More patients in the intervention arm underwent colonoscopy.
Compliance With FIT and Colonoscopy Uptake by Group.
Based on Chi-square test.
In both the control and intervention arms, all patients had average risk at the time of enrolment. Out of the 30 patients in the intervention arm, 8 were recategorised by the RPT to be high risk, as shown in Table 4. Actual completion of CRC screening according to recategorised risk scores are illustrated in Figure 2.
Summary of Risk Score and Risk Levels of Patients in the Intervention Arm.

Abbreviations: AR, average risk; HR, high risk; IQR, interquartile range; SD, standard deviation.

Recategorised risk scores and compliance with CRC screening in intervention arm.
Secondary Outcome: Feasibility and Acceptability of Risk Prediction Tool
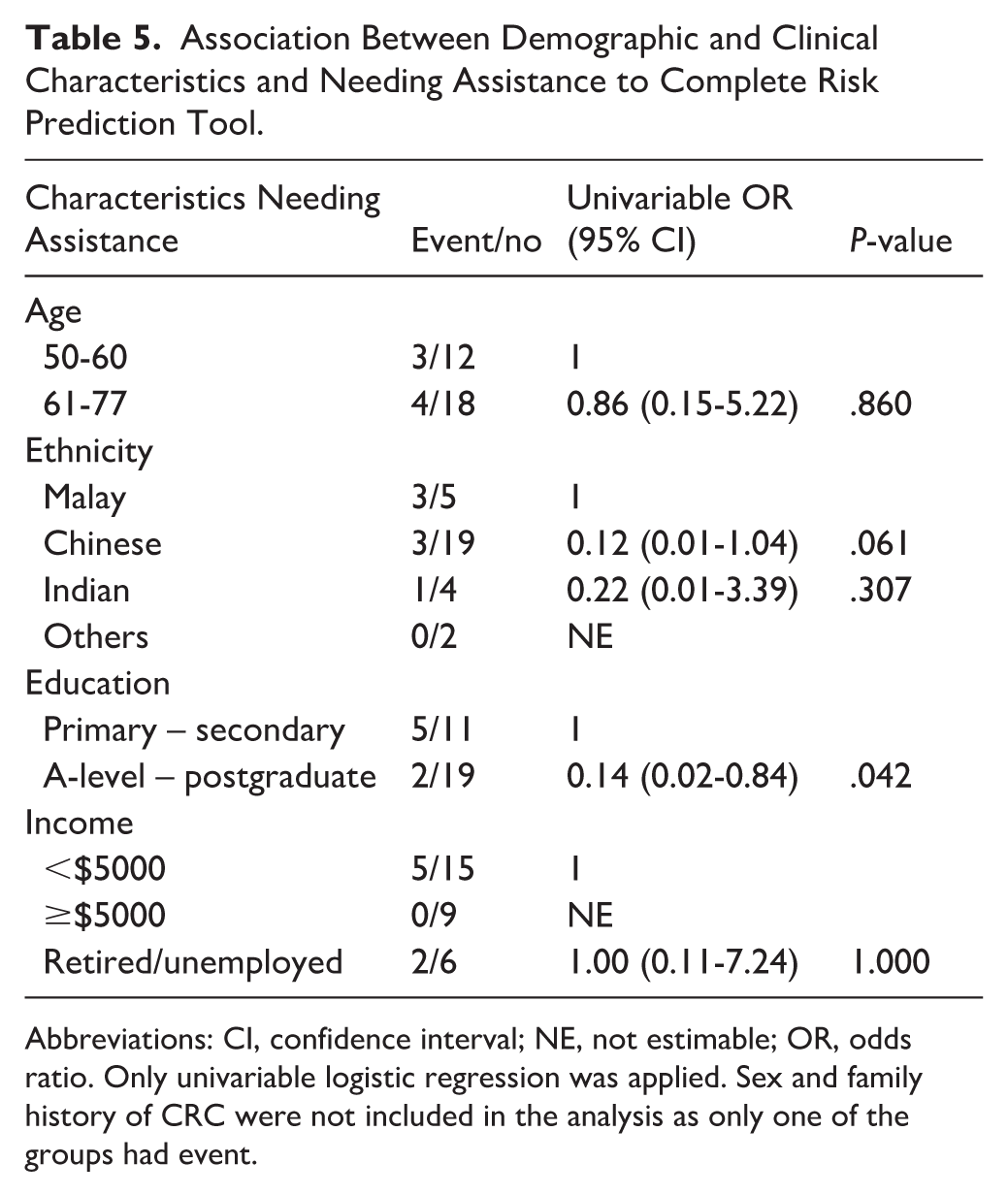
Out of 30 patient participants in the intervention group, 7 patients (23.3%) required assistance to complete the RPT. Among the demographic and clinical characteristics, only education level was associated significantly with needing assistance to complete the RPT (Table 5). The patients who reached “A-level – postgraduate level” had lower odds of requiring assistance to complete the tool, compared to those with primary or secondary education level (P = .042). No significant difference in completion ability by age or income was observed.
Association Between Demographic and Clinical Characteristics and Needing Assistance to Complete Risk Prediction Tool.
Abbreviations: CI, confidence interval; NE, not estimable; OR, odds ratio. Only univariable logistic regression was applied. Sex and family history of CRC were not included in the analysis as only one of the groups had event.
Self-reported ease-of-use was high with 93.3%, 100% and 93.3% of patients who reported that the RPT tool was easy to use, the questions were easy to understand, and the words were easy to see, respectively (Supplemental Table 2). Ninety percent of patients agreed that they had enough information to complete the tool.
In-Depth Interviews: Themes and Sub-Themes on CRC Screening
As shown in Figure 3, the themes and subthemes illustrate the continuum and dichotomy of barriers, recommendations and facilitators guided by the Health Belief Model. 38 Exemplar quote can be found in Supplemental Box 4.
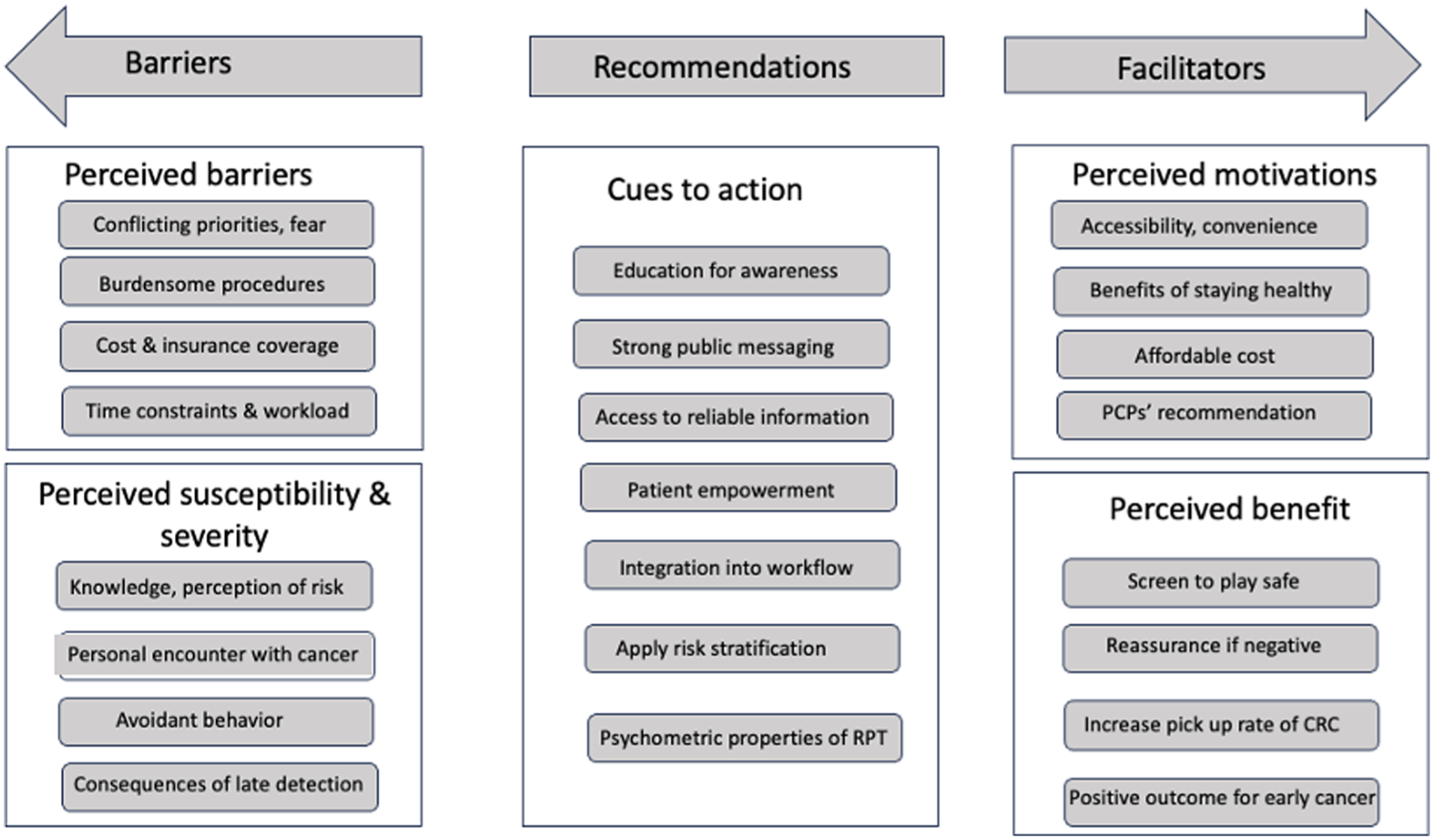
Applying the Health Belief Model to understand perceptions on CRC screening from patients and PCPs.
Perceived Barriers
For patients, conflicting priorities included allocating time for screening. Barriers included fear/worry of procedural risk, financial concerns and insurance coverage.
PCPs perceived that incorporating a RPT was time-consuming and contribute additional workload.
But it is very onerous . . . I feel that it is time-consuming . . . we cannot manage it (incorporating RPT) in a ten-minute consult. PCP06
Perceived Susceptibility
Patient participants perceived increase age as a risk factor for CRC.
PCPs perceived that patients may have a pre-determined mindset and adopt an avoidant behaviour rather than inviting trouble by going for screening.
(Patients) might think that . . . cancer won’t happen to them . . . because they don’t have the signs and symptoms. PCP04
Perceived Severity
Patients were aware of the limited treatment options and psychological distress if cancer is detected late.
Perceived Benefits
Patients perceived benefits of screening to provide reassurance.
Perceived Motivations
Motivators include accessibility, affordable cost, convenience, benefits of staying healthy and PCPs recommendations.
PCPs recommendations were perceived as a motivator for screening uptake.
I (patient) am a layman I need (the) doctor to tell me that…, then I will follow. Otherwise, I’m clueless. ID04, Female, 66-year-old
Cues to Action
Enhancing patients’ education, easy access to reliable sources of information would empower patients and increase awareness of the need for screening.
If there’s . . . very strong signalling and public messaging . . . then definitely there would be more people who would be inclined to go for screening. PCP02
PCPs perceived that integration of RPT into the clinical workflow would ease adoption. Risk stratification would aid resource allocation for those at higher risk.
Themes on Perceptions of RPT
The RPT was acceptable to patients and PCPs, and capable of self-administering.
PCPs agreed that the tool was helpful in stratifying risk, potentially improving their practice. Knowledge of psychometric properties of the RPT would boost confidence in adoption.
It’s very easy to understand this RPT, . . . (but it still requires) . . . an extra step that we need to do. PCP01
Discussion
Study Findings
This study of a multicentre, randomised controlled trial showed that the intervention group which utilised the RPT had higher CRC screening uptake (63.3%) than the control group (34.4%; P = .029). Our study provided evidence that the use of a tailored CRC screening advice with a validated RPT enhanced screening uptake in primary care settings in Singapore. Studies on barriers and facilitators of CRC screening in Singapore are well established.41-43 Our study focussed on exploring barriers and facilitators in using RPT for CRC screening in the intervention group.
Patient participants in our study found the RPT to be acceptable and comprehensible and those with post-secondary education were able to complete the tool independently. PCPs acknowledged the RPT’s utility in stratifying CRC risk, with majority endorsing it. Most PCPs found that the RPT eased their practice; majority noted an improvement in their workflow. However, they were cautious about the impact of RPT on screening uptake and further recommended patient education and empowerment to promote screening.
Our findings concurred with a UK study which showed that PCPs and patients, considered risk-stratified screening acceptable as it could facilitate early diagnosis, maximise diagnostic yields, identification of higher risk individuals towards chemoprevention/lifestyle changes, enabling more efficient use of resources and reduce healthcare costs. 44 Although a systematic review broadly considered risk stratification acceptable, healthcare professionals noted that system support, staff training and clear communication are essential to facilitate implementation. 26 An Australian study found self-completed CRC RPT to be feasible but emphasised the importance of co-design inputs to make an intervention sustainable for clinical use.45,46 An area of priority is to strengthen the evidence base for risk stratification, particularly recommending less intensive screening for low-risk groups incorporating non-invasive screening modalities. 47
Studies have showed that PCPs are supportive of incorporating RPT into routine care but advocate for accessible evidence-based guidelines, professional education, risk calculators and targeted public messages to increase the feasibility in general practice.48,49 However, various online CRC risk calculators that have been developed are not generalisable to other settings. Jennifer et al found that the National Cancer Institute’s CRC risk assessment tool is a rigorously-developed calculator that provides the most accurate estimate for the US patient population to provide 5-year and lifetime risk estimates but has limited applicability to non-US populations.50,51 Similar findings apply for QCancer tool that was developed in the United Kingdom population.52,53 Given the perceived benefit of a RPT, it is essential to develop a RPT that is specific and useful to the local population.
Barriers about CRC screening from our patient participants included procedural discomfort, lack of knowledge and myths that hindered screening uptake. PCPs participants perceived patients’ low healthcare literacy. fear of receiving bad news of CRC as barriers to screening. Similarly studies showed that lack of awareness and knowledge were predominant barriers to CRC screening, especially among low-income, unmarried and ethnic minority groups.54-56 Other barriers included procrastination, forgetting, fear of the test result, screening anxiety, role of physician, access to care, low self-efficacy and misconceptions of not having symptoms and being healthy.13,57-59
Facilitators from our patient participants included accessibility of screening locations, affordable cost, simple/less troublesome screening modalities and recommendations from PCPs. Enhancing education, patient empowerment and integration of RPT into PCPs’ clinical workflow has the potential to aid adoption. Our findings concurred with studies that understood the value of early detection of CRC, privacy and high self-efficacy, provided solutions to overcome barriers.58,60
Implications for Practice
Risk-stratified CRC screening has potential to increase screening uptake as shown in our study where the intervention group using the RPT had higher CRC screening uptake. Modelling studies show this is cost-effective, and there are calls to move away from population-based to risk-based approaches to CRC screening.61-63
Out of the 30 patients in the intervention arm, 8 were recategorised by the RPT to be high risk. However, only 1 out of 8 recategorised patients completed CRC screening. We postulate that applying RPT did increase CRC awareness but is not the only factor to determine if high risk patients would complete CRC screening. Thus we hope explored perspectives through patient interviews to shed light into other determinants of CRC screening. This aids in incorporating behavioural interventions like motivational interviewing to effect change. Potentially, there are 3 groups of people who would benefit from risk-stratification models. Firstly, patients would better understand their personal CRC risk, likely increase uptake of screening and provide motivations for lifestyle modification. 52 Secondly, healthcare practitioners would be able to identify patients at greater risk of developing advanced CRC, thereby improving diagnostic yield. 44 Lastly, this model would benefit service providers as they would be able to improve on existing services to provide efficient allocation of resources.64,65
Strengths and Limitations
Strengths of our study include broad sampling and representation from a diverse group of community patients from multiple study sites. The data collection was complete with no missing data. We have a diverse group of research team members providing perspectives from primary care, genetics, oncology and public health. Our qualitative study was done with much rigour from the robust coding process yielding important comparative data.
Our study is limited by a small sample size, which reduces its power to generalise the findings. However, it will help to provide a good estimate of the sample size needed for future, larger trials. We also acknowledge that there may be potential for selection bias as patients willing to participate in this risk stratification process may be more likely to follow recommendations. Another limitation is the primarily urban healthcare structure not shared by all primary care systems. A limitation of our study would be the lack of previous data on patients’ experiences with CRC screening, pertaining to preparation, tolerability and results, as these may influence the uptake of CRC screening. However, we believe this is pilot data that may be useful for larger trials in the future.
As this is a multi-component tool, there may be uncertainty whether the increased screening uptake is due to the implementation of the RPT into the consultation workflow or whether the added intervention increased awareness or enhanced education. Other potential confounders include patient’s healthcare literacy and screening advice that was given by PCPs.
Conclusion
Our study provided evidence that the use of a validated RPT increased screening uptake among primary care patients in Singapore. Both patient participants and PCP found the RPT acceptable and feasible for use. Patients found the RPT comprehensible and those with post-secondary education were able to complete the tool independently. Majority of PCPs found the RPT eased their practice and improved their workflows. Barriers and facilitators about CRC screening were prevalent and strategies to tackle these should include approaches at the individual, provider and health care system levels. As the RPT is a complex intervention with multiple interacting components, future research is needed to understand which components worked to bring about improved outcomes.
Supplemental Material
sj-docx-1-jpc-10.1177_21501319251397205 – Supplemental material for Assessing the Feasibility and Acceptability of a Colorectal Cancer Risk Prediction Tool in Primary Care to Increase Screening Uptake in Singapore: A Multi-Methods Pilot Study
Supplemental material, sj-docx-1-jpc-10.1177_21501319251397205 for Assessing the Feasibility and Acceptability of a Colorectal Cancer Risk Prediction Tool in Primary Care to Increase Screening Uptake in Singapore: A Multi-Methods Pilot Study by Rose Wai-Yee Fok, Sze Wern Lim, Dhana Letchimy, Jianbang Chiang, Jeanette Yuen, Tiffany Qing Lim, Zi Yang Chua, Shun Zi Liong, Grace Meijuan Yang, Ngiap Chuan Tan, Eng Sing Lee and Joanne Ngeow in Journal of Primary Care & Community Health
Footnotes
Acknowledgements
Our research team would like to thank: Michelle Ch’ng Wan Xing, Cheryl Lam Shi En, Aminath Shiwaza Moosa, Patricia Chia Ching Yen, Benjamin Lee and Rajni Gupta for their assistance in patient recruitment and participation in the interviews for PCPs. Gretchen Tan (GT) and Nur Diana Binte Ishak (DI) for their assistance in transcribing and coding the interviews.
Ethical Considerations
Ethical Approval This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of SingHealth Central Institutional Review Board (ECOS Ref No.: 2022-2128). Informed Consent participants included in the study.
Author Contributions
JN, RF, GMY, NCT and ESL: conceptualisation, methodology and supervision. SWL, DL and ESL: patient recruitment and resources. TQL, ZYC, JBC and JY: obtaining ethical approval, collecting and analysing data. SZL: biostatistician for data analysis. RF: drafting of the manuscript. All authors reviewed the manuscript and approved the final manuscript for publication.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by National Cancer Centre Singapore (NCCS) Cancer Fund. NCCSCF-R-YR2021-APR PG1.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
